A Phase II Trial of Moderate Hypofractionated Chemoradiation In Postoperative Cases of Carcinoma Oral Cavity
Arpit Dwivedi1, Mohd Waseem Raza2*, Archana Singh2 and Suraiya K Ansari3
1Junior Resident J K Cancer Institute, India
2Associate Professor J K Cancer Institute, India
3Associate Professor GSVM Medical College, India
Submission: June 12, 2023; Published: June 23, 2023
*Corresponding Address:Mohd Waseem Raza, Associate Professor, J K Cancer Institute, Kanpur, India
How to cite this article: Arpit D, Mohd Waseem R, Archana S, Suraiya K A. A Phase II Trial of Moderate Hypofractionated Chemoradiation In Postoperative Cases of Carcinoma Oral Cavity. Canc Therapy & Oncol Int J. 2023; 24(3): 556137. DOI:10.19080/CTOIJ.2023.24.556137
Abstract
Introduction: Conventional fractionation in post-operative adjuvant radiotherapy (usually 60 Gy in 2 Gy per fraction delivered over six weeks) remains the standard in carcinoma oral cavity. Post-operative hypofractionated radiotherapy is less explored especially in post-operative oral cavity cancers. The aim of this study is to evaluate postoperative moderate hypofractionation as the adjuvant treatment in post-operative oral cavity cancer.
Method: Patients of oral cavity cancers meriting adjuvant postoperative irradiation were inducted in the study. Hypofractionation consisted of a total dose of 52 Gy in 20 fractions over 4 weeks with concurrent chemotherapy. The primary endpoint was acute toxicity and late toxicity according to CTCAE V4.03 classification. The secondary endpoints included progression free survival at 1 year. Acute toxicity within 3 months of treatment and late toxicity at 1year follow up.
Result: Thirty patients were recruited over 18 months. All patients completed a minimum of 1 year of follow-up. Twenty-Nine patients completed planned radiotherapy doses within a median treatment time of 30 days (27-33). Grade 2, 3and 4 skin reactions were seen in 86.2%, 10.34%, and 3.4% respectively. Grade 2,3,4 mucositis was seen in 55.17%, 41.3%, 3.4% patients respectively during treatment (acute toxicity). Recurrence was seen in 17% of patients at 3 months. Progression free survival at 1 year was 51%. Overall survival in 1 year was 79%.
Conclusion: The schedule of 52 Gy in 20 fractions with concomitant chemotherapy is tolerable in patients with better compliance and equivalent PFS to conventional fractionation postoperative radiotherapy. Phase 3 trial is required for further validation of results.
Keywords: Hypofractionation; Radiotherapy; Post operative; Oral cavity cancer
Introduction
Surgery followed by postop adjuvant radiotherapy is the primary therapeutic option for locally advanced squamous cell carcinoma of oral cavity. There is a high risk of treatment failure in patients with high risk features such as extra capsular spreading (ECS) of involved lymph nodes, positive surgical margins and lymph node staging ≥N2. In high-risk postoperative squamous cell carcinoma of head and neck, concurrent chemoradiotherapy is the standard treatment improving overall survival (OS), disease free survival and locoregional control [1,2]. During the COVID-19 pandemic in the scarcity of various health and human resources across the world, there has been renewed interest in hypofractionated chemoradiation and hypofractionated radiation for oral cavity cancer [3-6]. Hypofractionation may be advantageous in reducing the risk of contracting and spreading an infective agent by reducing the number of visits to hospital. In addition, it may permit radical treatment when there is a shortage of radiotherapy facilities due to lockdown, staff illness or quarantine. Furthermore, shortened radiotherapy schedules may be less vulnerable to treatment breaks [7]. In the ASTRO-ESTRO consensus statement for the COVID-19 pandemic, while acknowledging a shortage of evidence, there was strong agreement among panelists on the use of hypofractionated radiation alone in locally advanced disease [4]. There was also an agreement to reserve the use of synchronous chemotherapy to standard fractionationed or moderately hypofractionated radiotherapy. In our study patients with postoperative squamous cell carcinoma of the oral cavity received definitive postoperative chemoradiation with moderate hypofractionated doses of 52 Gy in 20 # with concurrent chemotherapy .The compliance, toxicity and survival outcomes were compared with that of conventional chemoradiotherapy . Fractionation schemes were permitted to be administered with synchronous chemotherapy.
Aim
The primary aim was to evaluate the acute and late skin and mucosal toxicity and progression free survival. The secondary end point was to evaluate overall survival at 1 year in the postoperative oral cavity cancer cases treated with moderately hypofractionated chemoradiation.
Methodology
Study design and eligibility
Thirty patients of histologically proven squamous cell carcinoma of oral cavity requiring postoperative radiotherapy registered in the OPD were recruited for the study. Patients included in the study had age >= 18 to <=70, Karnofsky performance scale > 70, with adequate bone marrow reserve (Hb>10gm/dl; TLC>4000/cmm, platelet count >1,00,000/mm3), creatinine clearance >50 ml/min. Patient’s having evidence of distant metastasis, prior history of radiation or chemotherapy for the disease or having any comorbid medical condition were excluded from the study. Acute toxicity and Late toxicity according to CTCAE V4.03 classification was recorded at 3 months and 1 year respectively. Progression free survival and overall survival was recorded at 1 year.
Treatment
All the patients underwent complete evaluation of medical history, physical examination, complete blood count, blood chemistry, CT or MRI of head and neck, chest radiograph. Patients were treated with cisplatin at 100mg/m2 once in every 3 weeks depending on the indication. Radiotherapy was administered using at a moderately hypofractionated regimen of 52 Gy/20#/@2.6 Gy/fraction /day /5 days per week by EBRT Co60(Bhabhatron) using Dosisoft planning system by 2D Technique.
Assessment and outcomes
Follow up visits were analysed till January 2023. OS time was calculated as the period between the date of start of treatment and patient alive on last analysis of data. Progression free survival was calculated as the period between the start of treatment and date of disease relapse. Acute toxicity was defined as toxicity noted during RT or within 3months after completion of RT.
Statistical Analysis
This was a single-stage study of 30 patients to evaluate the feasibility of the 4-week dose-escalated chemoradiotherapy schedule. Statistical sample size was not calculated in this study. The primary outcome is reported with 95% confidence intervals. Categorical data are reported descriptively as percentages with 95% confidence intervals. Continuous data are reported as median and interquartile range.
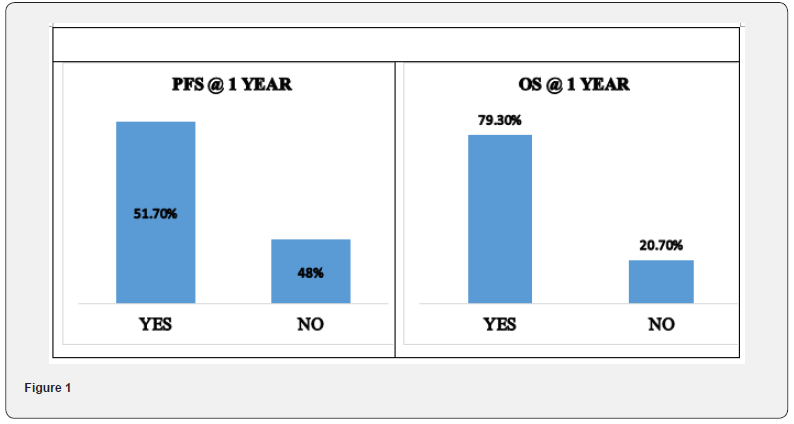
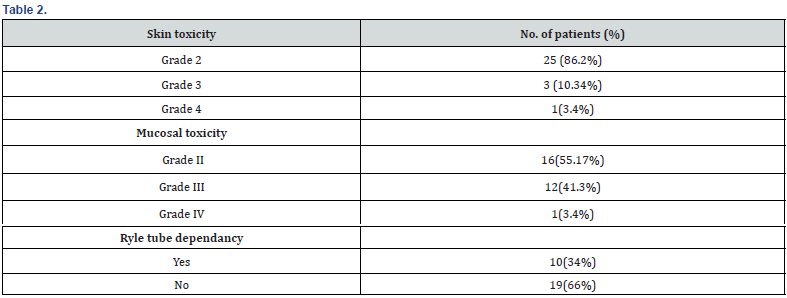
Thirty patients were recruited over 18 months. Out of which one patient defaulted treatment. Twenty-Nine patients completed planned dose radiotherapy with median treatment time of 30 days (27-33) (Table 1). All patients completed a minimum of 1 year of follow-up. Grade II, III and IV skin reactions were seen in 25(86.2%), 3(10.34%), 1(3.4%) patient respectively. Grade III skin and mucosal toxicity was recorded only in one patient. Mucosal reactions of Grade II, III, IV toxicity in was seen in 16(55.17%), 12(41.3%), 1(3.4%) patient respectively during treatment (acute toxicity) (Table 2). Maximum mucositis range lies between grade II and grade III toxicity which was manageable. During follow up we got recurrence was seen in 5 (17%) patients at 3 months. During treatment Ryles Tube dependency noticed in 10 patients(34%) Progression free survival at 1 year was 15(51%) patients. Overall survival in 1 year was 23(79.3%) patients (Figure 1).

Discussion
The 5-year rates of local control with buccal mucosa and tongue carcinoma are 85%, disease free survival rate 70% and distant metastasis around 8-15%. 3-year OS and recurrence free survival were 47% and 50% respectively. Cisplatin 100mg/m2 3 weekly regimens with conventional radiotherapy (60 Gy/30#) is the standard regimen recommended for adjuvant CCRT for SCCHN. The lack of difference in efficacy and quality of life endpoints at baseline (acute effects) and at 1 years (late effects) between hypofractionated and conventional radiotherapy provides some support to moderate hypofractionation. ASTRO-ESTRO consensus statement and Royal College of Radiologists also advocate the use of hypofractionation [4-6]. Various single center series and the data from this study would appear to justify the use of this moderate hypofractionation in the absence of direct phase 3 comparisons [8-11].
Use of a 4-week schedule results in a moderate decrease in the number of visits for patients by saving two weeks per patient. Single center data from The Princess Margaret Hospital hypofractionated radiotherapy alone regime 60Gy/25# over 5 weeks has been suggested for T1-T4 N0-N3b oropharyngeal tumors [5]. This regime may be more advantageous in term of reduced patient visits and lower utilization of available radiotherapy facilities. The radiobiological advantage with shorter schedules as that of hypofractionation, is that less dose is required to overcome accelerated repopulation. However, when accelerating, both acute mucosal toxicity and late effects have to be considered. Using the linear quadratic model, Fowler predicted that optimum schedules completed treatment between 4 and 6 weeks [12]. In Developing countries like ours, cancer incidence is on the rise with limited radiotherapy facilities. A 4-week dose escalated hypofractionated radiation schedule (52 Gy in 20# over 20 days) with concurrent cisplatin (as per postoperative chemotherapy indication) has been tested and is being planned for further evaluation in a randomized clinical trial. This regime employs a fraction size of 2.6 Gy per fraction.
The 4-week regime 55 Gy in 20# over 25 days has historically been used with several single agents including methotrexate, cetuximab, carboplatin and capecitabine for locally advanced diseases [13-18]. More recent IMRT series have used 55 Gy in 20 fractions with synchronous single agent cisplatin, carboplatin and cetuximab [8,19,20]. The International Atomic Energy Agency is currently examining 4 weeks accelerated hypofractionated chemoradiation or radiation versus conventional fractionationed chemoradiation or radiation in a large international randomized trial [20]. This study adds to the available data on the use of six and to a lesser extent four week hypofractionated chemoradiation. The results of the ongoing randomized studies discussed above are awaited. In our study we have noticed patient compliance with hypofractionated regimen is 96% and acute toxicity(maximum grade II/III) was manageable, and progression free survival and overall survival was comparable to conventional chemoradiotherapy . Hyporactionated schedule was seen to be offering lesser patient burden to institute with higher compliance. Further randomized study is being planned for further analysis of this regimen.
Conclusion
The schedule of 52 Gy in 20 fractions with concomitant chemotherapy is tolerable in patients with better compliance and equivalent PFS to standard postoperative radiotherapy. In a country like ours where cancer incidence is rising at a higher pace than the available radiotherapy facilities it may prove to be a better option. Phase 3 trial is required for further validation of results.
References
- Cooper JS, Pajak TF, Forastiere AA, Jacobs J, Campbell BH, et al. (2004) Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N Engl J Med 350(19): 1937-1944.
- Bernier J, Cooper JS, Pajak TF, van Glabbeke M, Bourhis J, et al. (2005) Defining risk levels in locally advanced head and neck cancers: a comparative analysis of concurrent postoperative radiation plus chemotherapy trials of the EORTC (#22931) and RTOG (# 9501). Head Neck 27(10): 843–850.
- Gupta T, Ghosh-Laskar S, Agarwal J (2020) Resource-sparing curative-intent hypofractionated-accelerated radiotherapy in head and neck cancer: More relevant than ever before in the COVID era. Oral Oncol 111: 105045.
- Thomson DJ, Palma D, Guckenberger M, Balermpas P, Beitler JJ, et al. (2020) Practice Recommendations for Risk-Adapted Head and Neck Cancer Radiation Therapy During the COVID-19 Pandemic: An ASTRO-ESTRO Consensus Statement. Int J Radiat Oncol Biol Phys 107(4): 618-627.
- Huang SH, O'Sullivan B, Su J, Ringash J, Bratman SV, et al. (2020) Hypofractionated radiotherapy alone with 2.4 Gy per fraction for head and neck cancer during the COVID-19 pandemic: The Princess Margaret experience and proposal. Cancer 126(15): 3426-3437.
- Roques T, Prestwich R (2020) Head and Neck Cancer and COVID 19.
- Sanghera P, McConkey C, Ho KF, Glaholm J, Hartley A (2007) Hypofractionated accelerated radiotherapy with concurrent chemotherapy for locally advanced squamous cell carcinoma of the head and neck. Int J Radiat Oncol Biol Phys 67(5): 1342-1351.
- Fong C, Boon I, Boon C, Benghiat H, Hickman M, et al. (2017) Hypofractionated accelerated chemoradiation for oropharyngeal cancer and the 2016 Royal College of Radiologists' Fractionation guidelines. ClinOncol (R CollRadiol)29(7).
- Bhide SA, Ahmed M, Barbachano Y, Newbold K, Harrington KJ, et al. (2008) Sequential induction chemotherapy followed by radical chemo-radiation in the treatment of locoregionally advanced head-and-neck cancer. Br J Cancer 99(1): 57-62.
- Loo SW, Geropantas K, Wilson P, Martin WM, Roques TW (2013) Target volume definition for intensity-modulated radiotherapy after induction chemotherapy and patterns of treatment failure after sequential chemoradiotherapy in locoregionally advanced oropharyngeal squamous cell carcinoma. Clin Oncol (R CollRadiol) 25(3): 162–170.
- Miah A, Schick U, Bhide S, Guerrero-Urbano M, Clark C, et al. (2015) A phase II trial of induction chemotherapy and chemo-IMRT for head and neck squamous cell cancers at risk of bilateral nodal spread: the application of a bilateral superficial lobe parotid-sparing IMRT technique and treatment outcomes. Br J Cancer 112(1): 32-38.
- Fowler JF (2007) Is there an optimum overall time for head and neck radiotherapy? A review with new modelling. Clin Oncol 19: 8e22.
- Chan A, Teoh D, Sanghera P, Hartley A (2009) Radiotherapy compliance is maintained with hypofractionation and concurrent cetuximab in locally advanced head and neck cancer. Radiother Oncol 93(3): 654.
- Chan AK, Sanghera P, Choo BA, McConkey C, Mehanna H, et al. (2011) Hypofractionated accelerated radiotherapy with concurrent carboplatin for locally advanced squamous cell carcinoma of the head and neck. Clin Oncol (R Coll Radiol) 23(1): 34-39.
- Sanghera P, McConkey C, Ho KF, Glaholm J, Hartley A (2007) Hypofractionated accelerated radiotherapy with concurrent chemotherapy for locally advanced squamous cell carcinoma of the head and neck. Int J Radiat Oncol Biol Phys 67(5): 1342-1351.
- Teoh DC, Rodger S, Say J, Hartley A (2008) Hypofractionated radiotherapy plus cetuximab in locally advanced head and neck cancer. Clin Oncol (R Coll Radiol) 20(9): 717.
- Tobias JS, Monson K, Gupta N, Macdougall H, Glaholm J, et al. (2009) Chemoradiotherapy for locally advanced head and neck cancer: 10-year follow-up of the UK Head and Neck (UKHAN1) trial. Lancet Oncol 11(1): 66-74.
- Jegannathen A, Mais K, Sykes A, Lee L, Yap B, et al. (2011) Synchronous chemoradiotherapy in patients with locally advanced squamous cell carcinoma of the head and neck using capecitabine: a single-centre, open-label, single-group phase II study. Clin Oncol (R Coll Radiol) 23(2): 149-158.
- Benghiat H, Sanghera P, Cashmore J, Hodson J, Mehanna H, et al. (2014) Four week hypofractionated accelerated intensity modulated radiotherapy and synchronous carboplatin or cetuximab in biologically staged oropharyngeal carcinoma. Cancer and Clin Onc 3(2).
- Jacinto AA, Batalha Filho ES, Viana LS, De Marchi P, Capuzzo RC, et al. (2018) Feasibility of concomitant cisplatin with hypofractionated radiotherapy for locally advanced head and neck squamous cell carcinoma. BMC Cancer 18(1): 1026.






























