Combined Endovascular and Surgical Treatment of High-Grade Meningioma - Case Report
L Lazovic1*, P Bosnjakovic1, W Yousef2, A Chelghoum2, T Khan2, G A Hamden3, M Almurshed4 and A Andrejevic5
1Medical Imaging Department, Ibn Sina Hospital, Ministry of Health, Kuwait
2Neurosurgery Department, Ibn Sina Hospital, Ministry of Health, Kuwait
3Anesthesia and Intensive Care Department, Ibn Sina Hospital, Ministry of Health, Kuwait
4Pathology Department, Alsabah Hospital, Ministry of Health, Kuwait
5Medical Faculty, University of Nis, Nis, Serbia
Submission: June 12, 2023; Published: June 20, 2023
*Corresponding Address:Lazar Lazovic, Interventional Neuroradiologist, Medical Imaging Department Ibn Sina Hospital, PO box 25427 Safat 13115, Kuwait, Email: lazovic_lazar@yahoo.com
How to cite this article: L Lazovic, P Bosnjakovic, W Yousef, A Chelghoum, T Khan, et al. Combined Endovascular and Surgical Treatment of High- Grade Meningioma - Case Report. Canc Therapy & Oncol Int J. 2023; 24(3): 556136. DOI:10.19080/CTOIJ.2023.24.556136
Abstract
Surgical resection of hypervascular meningiomas is often complex and may be accompanied by blood loss, followed by incomplete resection and tumor recurrence. We present a case of successful combined endovascular and surgical treatment for a large, complex, transcalvarial meningioma that is successfully resected. A 52-year-old male is admitted to our hospital with large left fronto-parietal tumefaction. Magnetic resonance imaging revealed extra-axial, transcalvarial fronto-temporal-parietal space occupying lesion with vivid contrast enhancement. Preoperative embolization was done using microspheres and coils. Surgical resection of tumor was done and pathohistology report showed Grade III meningioma. The patient recovers completely after the surgery and at first clinical follow-up 30 days later mRS was 0. Preoperative embolization of grade III meningioma reduces the surgery time, intraoperative blood loss and allows better resection without significant increase of perioperative complications. Reports on the association between embolization and tumor recurrence are insubstantial and require more research in future.
Introduction
Surgical resection of hypervascular meningiomas is often complex and may be accompanied by blood loss, followed by incomplete resection and tumor recurrence [1]. Even though preoperative embolization has generally been accepted as an adjuvant therapy that makes resection easier, the endovascular treatment is still controversial [2]. We present a case of successful combined endovascular and surgical treatment for a large, complex, transcalvarial meningioma that is successfully resected.
Case Presentation
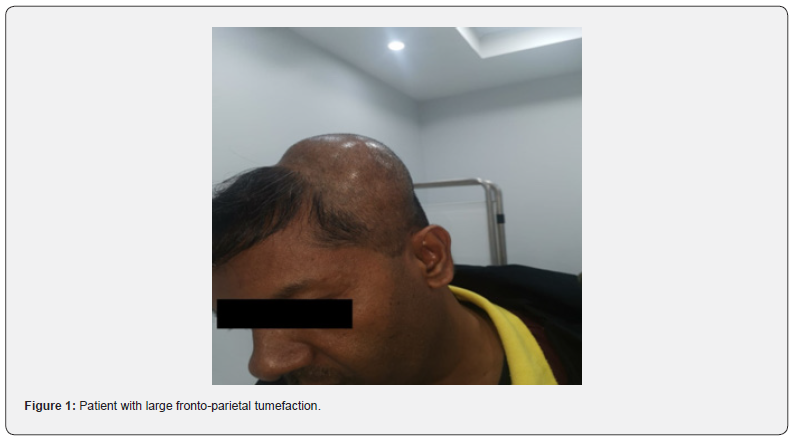
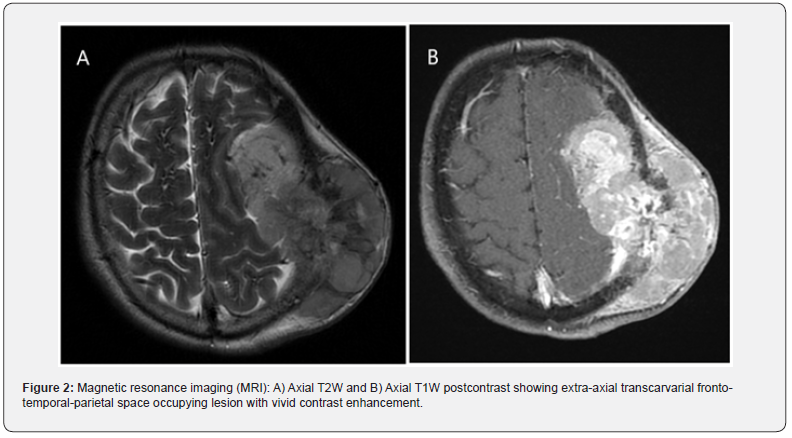
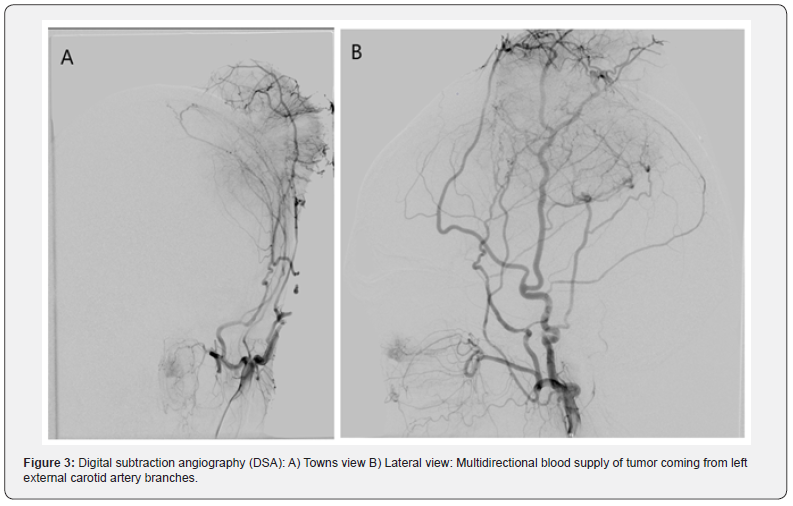
A 52 years old male is admitted to our hospital with large left fronto-parietal tumefaction (Figure 1). Magnetic resonance imaging (MRI) with application of contrast media was done. Imaging revealed extra-axial, transcalvarial fronto-temporal-parietal space occupying lesion with vivid contrast enhancement (Figure 2). Mass was measured 7x10x8 cm. There is mild perilesional T2W hyperintensity in favor of perilesional edema. Midline shift to contralateral side measuring 6 mm. Using Seldinger technique right femoral approach was made, and six vessels selective digital subtraction angiography (DSA) was done. DSA revealed multidirectional blood supply of tumor coming from left external carotid artery (Figure 3). No blood supply coming from the left MCA was identified. Guiding catheter Envoi 5F (CERENOVUS, Johnson & Johnson Medical Co, US) was selectively introduced within left ECA.
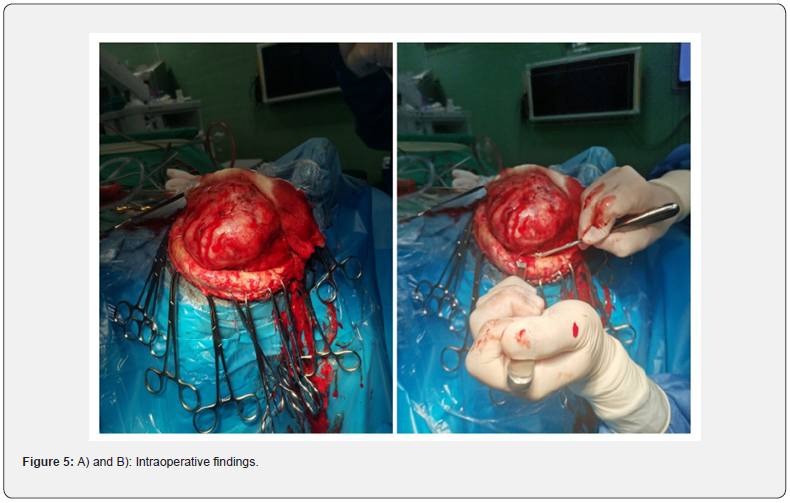
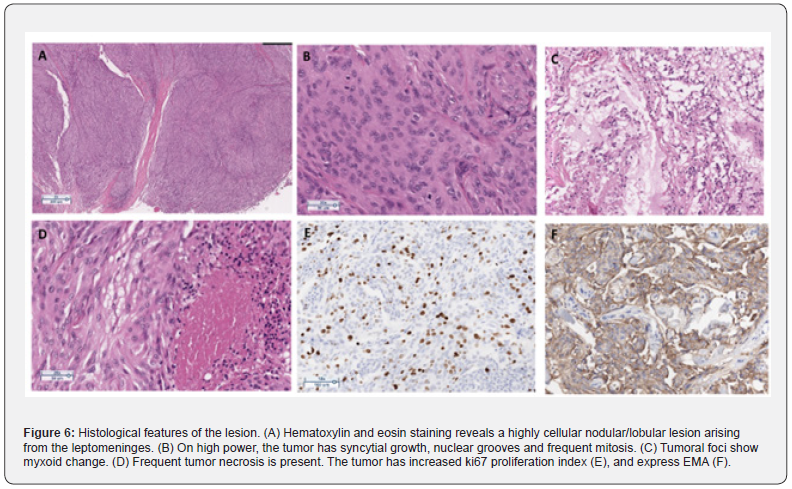
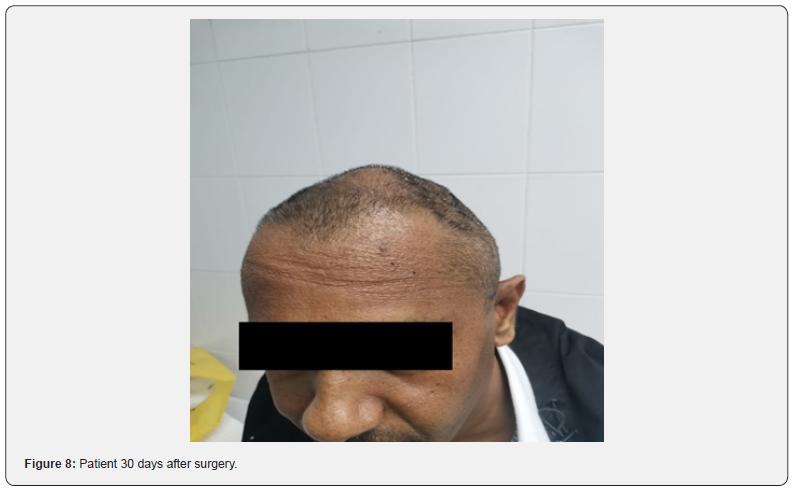
Supraselective catheterization of feeding vessels was done using Prograde microcatheter system 2,8F (TERUMO Medical Co, Japan). Embolization done with Embosphere® (MeritMadical Co, USA) size 500-700 μm and AzurTM peripheral HydroCoil embolization system (TERUMO Medical Co, Japan) size 4x10 mm, 3x10 mm, and 2x4 mm. Follow up DSA revealed devascularization of tumor for more than 90% (Figure 4). Surgical resection of tumor was done 24 hours later (Figure 5). Duration of surgery was 3 hours and blood loss during the surgery was 800 ml. Pathohistological report revealed Grade III meningioma (Figure 6). The case was sent for further molecular testing by Oncomine Comprehensive assay (DNA and RNA), that revealed NF2: c.1396C>T, p.(R466*) non-sense mutation, which is likely oncogenic. Follow up MRI revealed complete tumor resection without residual tumor (Figure 7). The patient recovers completely after the surgery and at first clinical follow up 30 days later mRS was 0 (Figure 8). Three months after the surgery, the patient started radiation therapy 60 Gy/30Fx.


Discussion
Meningiomas are among the most common primary brain tumors, accounting for one third of all central nervous system tumors. It is estimated that more than 90% are WHO Grade, I, 5% are Grade II and 1-3% are Grade III [3]. Higher grade tumors commonly have higher vascularity and surgery may lead to intraoperative blood loss followed by incomplete resection and tumor recurrence [4]. Meningiomas mainly arise from the dura, and blood supply comes from meningeal arteries branching from external carotid artery. Although large and invasive Grade III meningioma can recruit blood supply from nearby arteries and even cerebral and cerebellar arteries [5]. Blood loss during the surgery of hypervascular meningioma depends on size and location and can range from 200 ml to 2,2 L. Our patient had large convexity grade III meningioma with multidirectional blood supply in which embolization has been shown to be most beneficial [6]. Blood loss during the surgery was 800 ml. There is much controversy regarding timing of embolization with respect to resection surgery [7]. Mostly the average time for surgery after embolization ranges from 1.9 to 6.3 days [8]. Considering that waiting too long may increase the risk of tumor hemorrhage and may increase the chance of recanalization of embolized vessels, surgery in our patient was done 24 hours after embolization procedure. Moreover, delayed resection after embolization of large tumors may cause tumor mass effect and lead to herniation and hydrocephalus which can affect the outcome [9,10]. Furthermore, studies analyzing duration of surgical resection of meningioma with and without preoperative embolization showed shorter operation time in embolized group with no significant difference in perioperative complication [11]. Duration of surgical resection in our patient was 3 hours and mRS at first follow up was 0. Moreover, some of the studies suggest that preoperative embolization might prolong the Grade I meningioma recurrence [12]. However, the evidence of combined endovascular and surgical treatment of Grade III meningioma recurrence has not been confirmed.
Conclusion
Preoperative embolization of Grade III meningioma reduces the surgery time, intraoperative blood loss and allows better resection without significant increase of perioperative complications. Reports on the association between embolization and tumor recurrence are insubstantial and require more research in future.
Author contributions
The first draft of the manuscript was written by Lazar Lazovic and all authors commented on previous versions of the manuscript.

Consent to participate
All authors read and approved the final manuscript.
Code availability
Code availability is not applicable to this article.
Conflict of Interest
The authors declare that they have no conflict of interest.
Consent to Publish
The patient provided written informed consent to publish this case report and any accompanying images.
References
- Alvarez-Cubero MJ, Martinez-Gonzalez LJ, Saiz M, Pedro Carmona-Saez , Juan Carlos Alvarez, et al. (2015) Prognostic role of genetic biomarkers in clinical progression of prostate cancer. Exp Mol Med 47(8): e176.
- Canfield SE, Kibel AS, Kemeter MJ, Febbo G, H Jeffrey Lawrence, Judd W Moul, et al. (2014) A guide for clinicians in the evaluation of emerging molecular diagnostics for newly diagnosed prostate cancer. Rev Urol 16(4): 172-180.
- Wein AJ, Kavoussi LR, Partin AW, Peters CA (2012) Urology Book (Tenth edn). PA, Elsevier, Philadelphia, USA.
- Heindenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, et al. (2013) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent-update. Eur Urol 65(1): 124-137.
- Castillejos-Molina R, Rodriguez-Covarrubias F, Sotomayor M, Gomez Alvarado MO, Miguel Villalobos-Gollas, et al. (2011) Impact of metabolic syndrome on biochemical recurrence of prostate cancer after radical prostatectomy. Urol Int 87(3): 270-275.
- Klein EA, Thompson IA Jr, Tangen CM, Crowley JJ, Lucia MS, et al. (2011) Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 306(14): 549-556.
- Lin PH, Aronson W, Freedland SJ (2015) Nutrition, dietary interventions and prostate cancer: the last evidence. BMC Medicine 13: 3.
- Ma RW, Chapman K (2009) A systematic review of the effect of diet in prostate cancer prevention and treatment. J Hum Nutr Diet 22(3): 187-199.
- Bristow SM, Bolland MJ, MacLennan GS, Avenell A, Grey A, Gamble GD, et al. (2013) Calcium supplements and cancer risk: a meta-analysis of randomised controlled trials. Br J Nutr 110(8): 1384-1393.
- Castro E, Goh C, Olmos D, Ed Saunders, Daniel Leongamornlert,et al. (2013) Germline BRCA Mutations Are Associated with Higher Risk of Nodal Involvement, Distant Metastasis, and Poor Survival Outcomes in Prostate Cancer. J Clin Oncol 31(14): 1748-1757.
- Goh CL, Eeles RA (2014) Germline genetic variants associated with prostate cancer and potential relevance to clinical practice. Recent Results Cancer Res 202: 9–26.
- Karlsson R, Aly M, Clements M, Lilly Zheng, Jan Adolfsson, et al. (2014) A Population-based Assessment of Germline HOXB13 G84E Mutation and Prostate Cancer Risk. Eur Urol 65(1): 169-176.
- Amoretti A, Laydner H, Bergfeld W (2013) Androgenetic alopecia and risk of prostate cancer: A systematic review and meta-analysis. J Am Acad Dermatol 68(6): 937-943.
- Rahman AA, Lophatananon A, Stewart-Brown S, D Harriss, J Anderson, et al. (2011) Hand pattern indicates prostate cancer risk. Br J Cancer 104(1): 175–177.
- Myles P, Evans S, Lophatananon A, P Dimitropoulou, D Easton, et al. (2008) Diagnostic radiation procedures and risk of prostate cancer. Br J Cancer 98(11): 1852–1856.
- Nair-Shalliker V, Smith DP, Egger S, Ann Maree Hughes, John M Kaldor, et al. (2012) Sun exposure may increase risk of prostate cancer in the high UV environment of New South Wales, Australia: a case-control study. Int J Cancer 131(5): E726–732.
- Sutcliffe S, Platz EA (2008) Inflammation and prostate cancer: a focus on infections. Curr Urol Rep 9(3): 243–249.
- Sutcliffe S, Neace C, Magnuson NS, Reeves R, Alderete JF (2012) Trichomonosis, a common curable STI, and prostate carcinogenesis--a proposed molecular mechanism. PLoS pathogens 8(8): e1002801.
- De Marzo AM, Platz EA, Sutcliffe S, Jianfeng Xu, Henrik Gronberg, et al. (2007) Inflammation in prostate carcinogenesis. Nature reviews Cancer 7(4): 256–269.
- Huncharek M, Haddock KS, Reid R, Kupelnick B (2010) Smoking as a risk factor for prostate cancer: a meta-analysis of 24 prospective cohort studies. American journal of public health 100(4): 693–701.
- Zu K, Giovannucci E (2009) Smoking and aggressive prostate cancer: a review of the epidemiologic evidence. Cancer Causes Control 20(10): 1799–1810.
- Discacciati A, Wolk A (2014) Lifestyle and dietary factors in prostate cancer prevention. Recent Results Cancer Res 202: 27–37.
- Loprinzi PD, Kohli M (2013) Effect of physical activity and sedentary behavior on serum prostate-specific antigen concentrations: results from the National Health and Nutrition Examination Survey (NHANES), 2003–2006. Mayo Clin Proc 88(1): 11–21.






























