ALK Positive Non-Neural Granular Cell Tumour in Two Paediatric Patients- A Case Report
Kirti Chadha1, Shaikhali Barodawala2 and Shibani Ramchandran3*
1Chief Scientific Officer, Metropolis Healthcare Ltd, India
2HOD Surgical Pathology & Integrated Oncopathology, Metropolis Healthcare Ltd, India
3Medical Writer, Academics science cell, Metropolis Healthcare Ltd, India
Submission: February 16, 2023; Published: March 03, 2023
*Corresponding Address: Dr. Shibani Ramchandran, Medical Writer, Academics science cell, Metropolis Healthcare Ltd, Mumbai, India
How to cite this article: Kirti Chadha, Shaikhali Barodawala and Shibani Ramchandran. ALK Positive Non-Neural Granular Cell Tumour in Two Paediatric Patients- A Case Report. Canc Therapy & Oncol Int J. 2023; 23(3): 556111. DOI:10.19080/CTOIJ.2023.23.556111
Abstract
Non-neural granular cell tumour (PNNGCT) is a rare mesenchymal neoplasm of unknown lineage with prominent cytoplasmic granularity [1]. These tumors are more common in children and young adults, and commonly involve the back and extremities. A panel of immunostains is important to rule out other differential diagnoses. In contrast to conventional granular cell tumor (GCT), NNGCT lacks S100 expression, hence, NKI-C3 and anaplastic lymphoma kinase (ALK) stains can be particularly helpful in confirming the diagnosis. In addition, an underlying ALK gene rearrangement has been reported in a small subset of cases [2]. In general, most of these tumors appear to behave in a benign fashion, and conservative management is recommended. Our results demonstrate that NNGCT harbour ALK fusions, which suggests that NNGCT are molecularly diverse, and further substantiate NNGCT as distinct from GCT [3].
Keywords: Non neural Granular cell tumour (NNGCT); Anaplastic lymphoma kinase (ALK); Transcription Factor E3 (TFE3)
Introduction
Non neural granular cell tumor (NNGCT) also known as (primitive polypoid GCT) is a rare neoplasm first described by LeBoit in 1991 [1]. As compared to classic GCT, NNGCT are immunohistochemically distinct lacking expression of markers of neural differentiation like SOX10 and S100. Recent studies have shown ALK gene fusion in these types of tumors by FISH or IHC [2,3]. We report 2 cases of S100 and SOX10 negative granular cell tumor, both in paediatric patients.
Case Presentation
Case 1
An 11-year old male presented with a subglottic mass. CT Scan revealed a small elongated soft tissue lesion in cervical trachea arising from its wall and projecting into its lumen measuring 8 x 7 x 10 mm without luminal encroachment or stenosis. PET-CT-Hypermetabolic ill-defined nodular lesion was identified in the left upper tracheoesophageal space which was seen to indent upon the upper tracheal wall extrinsically with spur like projection into the tracheal lumen? lymphnode? parathyroid in origin. No other remarkable findings were observed.
Case 2
A 4-year-old female presented with a lump arising from the left lateral chest wall. Histopathologic examination: Microscopic examination in both cases revealed ovoid, polygonal cells with abundant eosinophilic cytoplasm. The cells had epithelioid morphology with enlarged nuclei with one or more prominent eosinophilic nucleoli. Mitotic figures, were in the range of 0–10 mitoses (median 1–2) per 10 high power fields. No necrosis or vascular invasion were noted. The cytoplasmic granules were positive for periodic acid-Schiff (PAS) (Figures 1 & 2).
Discussion
Since first reported by LeBoit in the 1990s, non-neural granular cell tumors have been described in patients between 5 and 83years [1]. Both our cases were paediatric patients. Shahabi et al described PNGCTs to be typically small in size ranging from 0.2 to 2.8cm (median: 0.5–0.8cm), similar to our cases [4]. PNGCTs are often seen on the trunk; but Rawal et al had reported two cases in the buccal mucosa. and hard palate [5]. As reported by Shahabi et al and Rawal et al, the tongue, vermilion of the lower lip, hard palate, alveolar ridge over tooth extraction site, and buccal mucosa are the most commonly affected anatomic areas [4,5]. In our case one was located in the subglottis whilst the other case was seen in the trunk. Furthermore, as described by Habeeb AA et al, nonneural granular cell tumors are usually well-circumscribed lesions with vesicular nuclei, pleomorphism, and averaging 5-6/10 HPF (high-power field) mitosis, while conventional granular cell tumors typically have poorly circumscribed margins and no mitotic figures [6]. Both our cases had similar morphological observations.
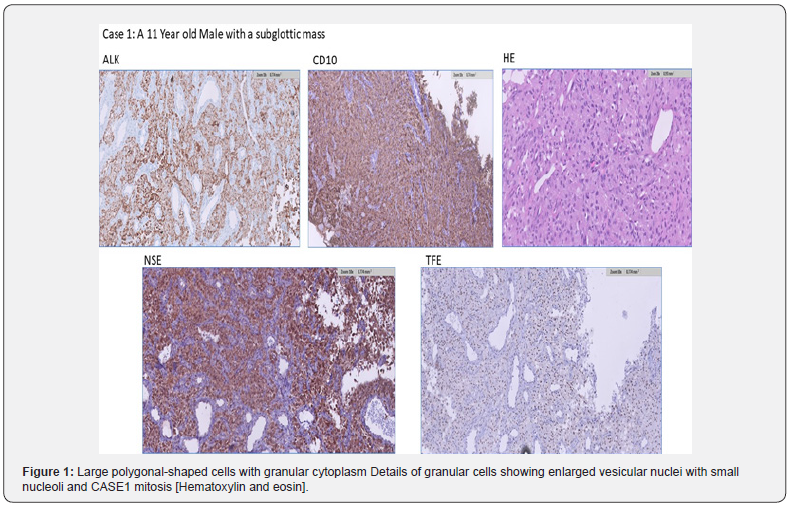
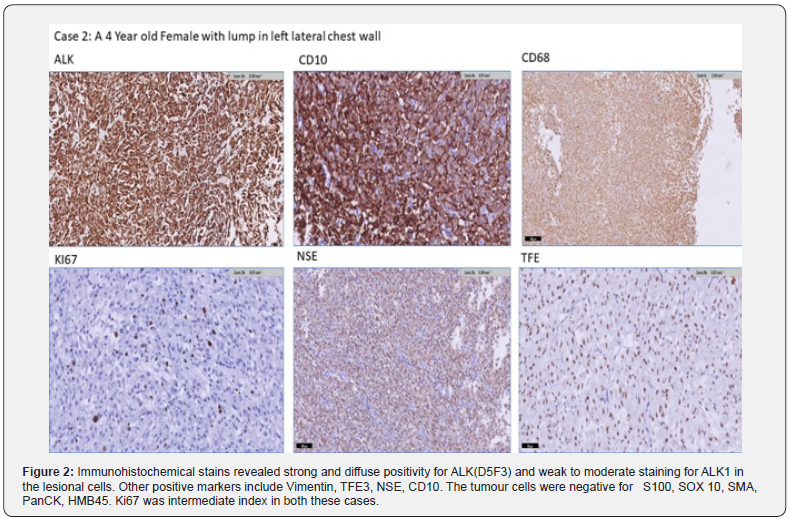
As reported by Habeeb AA et al, the tumors do not show clear evidence of neural differentiation with absence of S100 and Sox 10 staining by immunohistochemistry. El Ochi et al further observed that in contrast to conventional granular cell tumours, NNGCT lacks S100 expression. The diagnosis of this entity is one of exclusion. Further histological examination and appropriate immunohistochemical panel would be required prior to a definitive diagnosis [7]. The granular cells are usually positive for NKI-C3 and CD68, both stains reflecting a non-specific reaction to lysosomes. Immunohistochemistry for ALK demonstrate positive weak-to-moderate staining. The tumor cells were also negative for Cytokeratins, Epithelial membrane antigen (EMA) and Melanoma markers. The tumor was also negative for CD34 [6]. As seen in the case reported by Feci L et al, the tumor cells were positive for CD68 and PGP9.5, as well as CD10, vimentin, and inhibin [8].
In our cases the tumours were negative for S100, SOX10 Cytokeratin, EMA, Desmin, Myogenin and SMA, CD34, CD1a & CD30. Both our cases showed immunoreactivity for ALK(D5F3), ALK1, NSE, CD10, Vimentin and TFE3. ALK gene rearrangement by FISH was also positive in both cases. It is noteworthy that TFE3 is usually reported in granular cell tumours. This association with ALK positivity seems interesting. To our knowledge this association has not been explored with respect to granular cell tumours especially the non-neural ones. The ALK (D5F3) as well as ALK1 positivity by IHC and FISH would make targeted therapy an important option in patient management in the future. NNGCT are molecularly diverse, which further substantiate them as distinct from GCT [2]. In our both cases the morphology showed vesicular nuclei and prominent nucleoli, which are less common in conventional GCT. Despite the atypia and frequent mitotic figures, the studies show excision is curative [4,6]. The prognosis is usually good in most cases [7]. In our case, the patient with the subglottic mass is free of disease at 1-year follow-up [8-12].
Conclusion
Non-Neural granular cell tumours are thus rare tumours with uncertain histogenesis. Appropriate immunohistochemical panels are required to differentiate them from conventional granular cell tumours. The majority of the tumors appear to behave in a benign fashion, and conservative management is recommended. Being ALK positive, targeted treatment with ALK inhibitors can be attempted.
References
- Boit L, Barr PE, Burall RJ, J S Metcalf, T S Yen, et al. (1991) Primitive polypoid granular-cell tumor and other cutaneous granular-cell neoplasms of apparent nonneural origin. Am J Surg Pathol 15(1): 48–58.
- Cohen JN, Yeh I, Jordan RC, Wolsky RJ, Horvai AE, et al. (2018) Cutaneous non-neural granular cell tumors harbor recurrent ALK gene fusions. Am J Surg Pathol 42(9): 1133-1142.
- Di J (2022) Qasem Primitive non-neural granular cell tumor: Literature review. Human Pathology Reports 28.
- Shahabi A, Hastings H, Winfield A, Amer Khiyami (2020) A rare case of a non-neural granular cell tumor presenting as an enlarging right chest wall nodule in a 4-year-old male and review of the literature SAGE. Open Medical Case Reports 8: 2050313X20938246.
- Rawal YB, Dodson TB (2017) S-100 negative granular cell tumor (so-called primitive polypoid non-neural granular cell tumor) of the oral cavity. Head Neck Pathol 11(3): 404-412.
- Habeeb AA, Salama S (2008) Primitive nonneural granular cell tumor (so-called atypical polypoid granular cell tumor). Report of 2 cases with immunohistochemical and ultrastructural correlation. Am J Dermatopathol 30(2): 156-159.
- El Ochi MR, Essaoudi A, Allaoui M, Abrid JE, Touri S, et al. (2022) Dermal nonneural granular cell tumor: a case report. J Surg Case Rep 2022(7): rjac317.
- Feci L, Miracco C, Fimiani M, Rubegni P (2014) A case of primitive non-neural granular cell tumor presenting as a single painless bleeding nodule. Dermatol Pract Concept 4(2): 59–61.
- Mathew JG, Carvalho M, El-Shinnawy I, Salama S (2019) Non-Neural Granular Cell Tumor of the Eyelid: Case Report with Immunohistochemical and Ultrastructural Correlation. Int J Ophthalmic Pathol 8(1).
- Lazar AJF, Fletcher CDM (2005) Primitive nonneural granular cell tumors of skin: clinicopathologic analysis of 13 cases. Am J Surg Pathol 29(7): 927-934.
- Yeh I, Tran DT, Davis TL, Argenyi ZB (2009) An infiltrative variant of non-neural granular cell tumor: a case report. J Cutan Pathol 1: 46–51.
- Hobbs LK, Kozak MD, Gradeki SE, Mcgahren ED (2022) Raghavan SS A rapidly growing nodule on the leg of an adolescent: A unique presentation of a non-neural granular cell tumor Journal of cutaneous. Journal of cutaneous 49(12): 1040-1043.






























