Trastuzumab induce Cardiomyopathy in Breast Cancer Patient: A Case report; What Cardio- Oncology Guideline in this Case?
Dr. Amani Saleh Hadi Saeed*
Specialist of Clinical Oncology and Nuclear Medicine, National Oncology Center-Aden, Yemen
Submission: January 15, 2023; Published: February 16, 2023
*Corresponding Address: Dr. Amani Saleh Hadi Saeed, Specialist of Clinical Oncology and Nuclear Medicine, National Oncology Center-Aden, Yemen
How to cite this article: Amani Saleh Hadi Saeed. Trastuzumab induce Cardiomyopathy in Breast Cancer Patient: A Case report; What Cardio-Oncology Guideline in this Case?. Canc Therapy & Oncol Int J. 2023; 23(2): 556109. DOI:10.19080/CTOIJ.2023.23.556109
Abstract
Trastuzumab is a monoclonal antibody highly effective in the treatment of several cancer, are crucial part of the treatment of patients with HER-2 positive invasive breast cancer in both early and metastatic setting. but its use is associated with cardiac toxicity which usually responds cassation of the drug and /or medical therapy. we present an unusual case of acute cardiac toxicity temporally related to administration of Trastuzumab in which clinical presentation suggested cardiac failure after 8cycles. The patient did not respond to withdrawal of drug or medical therapy for heart failure. LV function surveillance based on left ventricular ejection fraction (LVEF) and global longitudinal strain (GLS) is recommended prior to and every 3months duringHER2-targeted therapies treatment surveillance. The overarching goal of the cardio-oncology discipline is to allow patients with cancer to receive the best possible cancer treatment safely.
Keywords: Trastuzumab; Cardiac toxicity; Cardio-oncology guideline
Introduction
Trastuzumab is humanized monoclonal antibody that links the exteracullar domain of the HER-2 protein (HER-2/neuor erbB2) overexpressed in about 20% to 25% of breast cancers. This antibody mediates signaling pathways leading to increase proliferation .it is approved for treatment of early and metastatic breast cancer [1-3]. Several trials have described potiental risk factors such as:
Age, Weight, High body index (BMI). History of coronary artery disease and hypertension Cumulative doxorubicin dose, HER2 expression level Previous treatment radiation of chest and negative hormonal receptor status [4] However, among these, only age and concomitant doxorubicin therapy result to correlate with an increased risk of cardiotoxicity [3]. Case Report: In September 2020, A49 year-old women under went right modified mastectomy for stage IIA invasive lobular carcinoma T3N0Mx, negative hormone receptors and HER2 overexpressed (score 3+ at immunochemistry).
In her medical history there were no cardio vascular comorbidities and she had no family history of cardiovascular disease. She received 4cycles AC (Adriamycin+ cyclophosphamide) and 4 cycles Taxoter (docetaxel) end in 7-2-2021. Post-operative radiotherapy 3D comprehensive 400cGy/15/5week ended 15-3-2021(in Cairo) recommendation for adjuvant Herceptin (Trastuzumab) if possible, for one year.
Patient come to our center in Aden, she received 8 cycles of Herceptin 360mg every 21days according to body weight (60 kg) – 6mg /kg. In 26/5/2021 ,patient complain bone pain, bone densimetery done showed high risk osteoporosis. A supportive therapy with bisphosphonates (Zoledronic acid 4mg i.v. inj) Every 28 days in 13/12/2021 at nine cycle of Herceptin, LEVF 43% dose will be decrease 50% No complain of dyspnea or chest pain continue to 11 cycles with dose of 200mg in cycle 12: 3/3/2022 ,LEVF 40% give only one cycle, than stope Herceptin with given supportive treatment for heart failure pharmacological Treatment was introducing orally. 21/6/2022: evaluation done by CT-scan and lab investigation with normal level. Echocardiography done: LEVF 52%. Patient put on follow up every 3months for first year (Figure 1).

Discussion
In this case report we described the case of patient treated with trastuzumab who developed a severe.
Congestive heart failure during treatment and who resumed trastuzumab therapy after proper cardiac management and left ventricular ejection fraction recovery.
unlike anthracycline –induce cardiotoxicity, trastuzuamb cardiotoxicity has some peculiar feature:
i. The risk does not seem to be dose related.
ii. There is no evidence that cardiac damage is associated with ultra-structural changes in myocytes.
iii. It is fully reversible after treatment suspension.
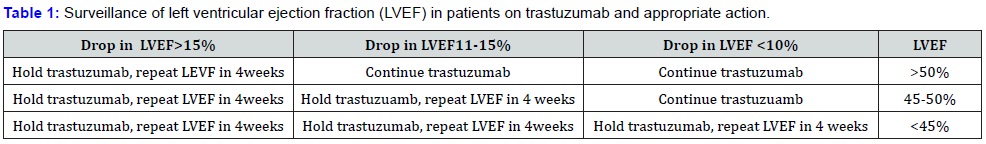
Baseline cardiac function should be assessed with echocardiography or MUGA scanning prior to chemotherapy exposure. LVEF is the most variable Table 1. Idenfecication of pre-existing risk factors and assessment of LVEF are vital before trastuzumab is started. LVEF should be assessed at least every 3months during treatment. there is evidence suggesting that trastuzumab-related cardiotocxity is reversible [5]. The main time to recovery is about 6 weeks. However, if the LVEF fails to recover sufficiently after a second consecutive measurement, trastuzumab should be discontinued. Other than study by cardinal et al., there is little data from the literature on the utility of cardiac biomarker for trastuzumab, such as TnI (I troponin) [6].
Role of cardio-oncology services
The overarching goal of the cardio-oncology discipline is to allow patients with cancer to receive the best possible cancer treatment safely, minimizing CTR-CVT (cancer therapy –related cardiovascular toxicity). Across the entire continuum of cancer care [7]. Recommendation for baseline risk assessment and monitoring during trastuzumab therapy. In the first 12 months after therapy [8,9].
i. Baseline echocardiography is recommended before HER-2targeted therapy in all patients.
ii. In patients receiving neoadjuvant or adjuvant HER2- targeted therapies, echocardiography is recommended every3months and within 12 months after completing treatment.
iii. In Lowe –risk HER+EBC (early breast cancer) patients who are a symptomatic and with normal assessment after 3months, reducing.
iv. Monitoring to every 4 months may be considered. In high and very high-risk HER2+EBC patients. More frequent echocardiography monitoring should be considered during treatment.
v. In metastatic HER2+ disease, echocardiography is recommended every 3months during the first year; if patient remains asymptomatic without CV toxicity, the surveillance can be reduced to every 6 months during future treatment.
vi. In metastatic HER2+ disease patient at high and very high –risk more frequent echocardiography monitoring may be considered.
vii. Martin and colleagues reported on a patient with a left ventricular ejection fraction drop from 76% to 55% during adjuvant sequential anthracycline -containing chemotherapy followed by trastuzmab -based therapy; ejection fraction was restored after starting therapy with the ACE-inhibitor captopril and trastuzumab was resumed without complications [10]. A recently published ESC-CCO document describes appropriate criteria for the organization and implement of cardio-oncology services [7].
Conclusion
Trastuzumab can lead to a sever cardiac toxicity, including in patients never previously Exposed to anthracyclines and irrespective of other risk factors.it is usually reversible and when recognized early, responds to the discontinuation of the antibody and to standard heart failure treatment, opportunity for resumption of therapy if necessary.
References
- Marinko, Dolenc J, Bilban-Jakopin Cvetka (2014) cardiotoxicity of concomitant radiotherapy and trastuzumab for early breast cancer. Radio.Onclogia 48(2): 105-112.
- Telli Melinda L, Hunt Sharon A, Carlson Robert W, Guardino AE (2007) Trastuzumab-realted cardiotoxicity calling in to question the concept of reversibility J. Clin Oncol 25(23): 3525-3533.
- Keefe Deborah L (2002) Trastuzumab -associated cardictoxcity. Cancer 95(7).
- Huzno J, Les D, Sarzyczny-Solota D, Nowara (2013) Cardiac side effect of trastuzamb in breast cancer patients -single centers experiences. Wspolczenena onclo 17(2): 190-195.
- Ewer MS, Vooletich MT, Durand JB, Woods ML, Davis JR, et al. (2005) Reversibility of trasutzumb -related cardiotoxicity : new insights based on clinical course and response to medical treatment. J Clin Oncol 23: 7820-7826.
- Cardinal D, Colombo A, Torrise R, Sandri MT, Civelli M, et al. (2010) Trastuzumab -induced cardiotoxicity. Clinical and prognostic implications of tropoin l evaluation. J clin oncol 28(25): 3910-3916.
- Lancellotti P, Suter TM, Lopez-fernandaez T, Galderisi M, Lyon AR, et al. (2019) Cardio-oncology services rational, organization, and implementation Heart J 40(22): 1756-1763.
- Cameron D, Piccarrrrrrt-gebhart MJ, Gebler RD, Porcter M, Goldhirsh A, et al. (2017) 11years follow-up of trastuzamb after adjuvant chemotherapy in HER-2postive early breast cancer. Final analysis of Herceptin Adjuvant (HERA) trial. Lanct 389: 1195-1205.
- Tan TC, Bouras S, Sawaya H, sebag IA, Cohen V Picard MH, et al. (2015) Time trend of left ventricular ejection fraction and myocardial deformation inices in cohort of women with breast cancer treated with anthracyclines ,taxanes, and trastuzumab. j Am Soc Echocardiogr 28(5): 509-514.
- Martins JS, Dos Santos VM, Thommen Teles L, Alves Leite V (2012) Reversible cardiotoxicity in a54-year-old woman treated with trastuzumab therapy . RevMed Chile 140(6): 763-766.






























