Comparative Evaluation of Preoperative Short Course Radiotherapy Versus Long Course Chemoradiotherapy for the Treatment of Rectal Cancer
Bora Uysal*, Hakan Gamsiz and Murat Beyzadeoglu
University of Health Sciences, Gulhane Medical Faculty, Department of Radiation Oncology, Turkey
Submission: July 28, 2022; Published: September 01, 2022
*Corresponding Address: Bora Uysal, MD, Asc. Professor of Radiation Oncology, University of Health Sciences, Gulhane Medical Faculty, Department of Radiation Oncology, 06010, Ankara, Turkey
How to cite this article: Bora U, Hakan G, Murat B. Comparative Evaluation of Preoperative Short Course Radiotherapy Versus Long Course Chemoradiotherapy for the Treatment of Rectal Cancer. Canc Therapy & Oncol Int J. 2022; 22(2): 556084. DOI:10.19080/CTOIJ.2022.22.556084
Abstract
Objectıve: Our goal is to comparatively evaluate the short term radiotherapy with long course chemoradiotherapy in the management of rectal cancer patients.
Methods: Consecutive patients with rectal cancer referred to our department for RT between January 2011 and December 2020 were evaluated. All 425 patients had rectal adenocancer and had undergone surgery (abdominoperineal resection or low anterior resection) after RT. The patients in short term group were treated with a dose of 25 Gy in 5 fractions and long term group with a dose of 45-50.4 Gy in a 25-28 fractions with 825 mg/m2 bid ( twice in a day)oral capecitabine chemotherapy.
Results: At a median follow-up of 48 (24-60) months, local control rate was 10.1 % for short term and 9.8 % for long term (p=0.054). Overall survival rates were 7.4 (5-9.5) years for short term radiotherapy and 7.7 (5.5-9) years for long term chemoradiotherapy (p=0.063). 25 % percent of short term group had distant metastases whereas 23.7 % of long term had 22.8 % distant metastases rate (p=0.066). No ≥ grade 3 acute toxicity requiring treatment interruption was encountered. Acute grade 1 gastrointestinal toxicity was observed in 65 patients (15 %). Grade 2 acute gastrointestinal toxicity was observed in 120 patients (28.2 %). Grade 1 acute urinary toxicity (urgency/ frequency) was observed in 61 patients (14.3 %) and grade 2 acute urinary toxicity (urgency/frequency) was observed in 17 patients (4 %) whereas no urinary late toxicity was observed in all patients.
Conclusion: Preoperative neoadjuvant short term radiotherapy is noninferior to conventional long term chemoradiotherapy because of similar results in local control, disease free survival, overall survival and adverse effects.
Keyword: Short course; Rectal cancer; Chemoradiotherapy; Noninferiority
Introduction
Rectum is a part of colorectum consisting of cecum, ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, sigmoid colon. This last part of system extends from rectosigmoid junction to the puborectalis ring with an approximately 15 cm in length. There are upper, mid and lower segments of rectum draining in different lymph nodes. 1.2 million colorectal cancer cases are diagnosed and 600 000 deaths occur yearly worldwide. Early cancer diagnosis, prevention and up-to-date treatment regimens declined 2-3 percent of the incidence associated with colorectal cancer. Male sex, hereditary syndormes, family history, inflammatory bowel disease, obesity, processed meat, refined diet, alcohol consumption and smoking are the genetic or enviromental factors of developing colorectal cancer.
Management of rectal cancer is considered with surgery as a primary treatment while local recurrence rates vary between 20-50 percent. So dramatic changes were released in recent 20 years. Depth of penetration and regional node involvement are the associated factors for recurrence. Neoadjuvant therapy including chemoradiotherapy, hypofractionated radiotherapy or chemotherapy plus surgery results with better outcomes compared to surgery alone. Surgery methods consist of local excision, low anterior resection, abdominoperineal resection and total mesorectal excision. Potential problem after surgery is local recurrence. Postoperative radiotherapy is delivered for high risk group including T3-4 tumor or involved regional nodes. Hypoxic surgical bed, increased bowel in the radiation area are the maor disadvantages of radiotherapy. Disease free survival or distant metastases are not improved but only local failure reduces with adjuvant radiotherapy. Chemotherapy and radiotherapy combination presents better local control and overall survival tendency compared to only radiotherapy. Favorable T3 patients with low recurrence rates might be omitted for adjuvant therapy but this is still not clear. Neoadjuvant therapy is widely accepted as chemoradiotherapy whereas short-term radiotherapy or long course chemoradiotherapy are the different modalities. Total neoadjuvant therapy is considered nowadays. These include 3 months chemotherapy following chemoradiotherapy plus surgery and 3 months adjuvant chemotherapy.
Methods
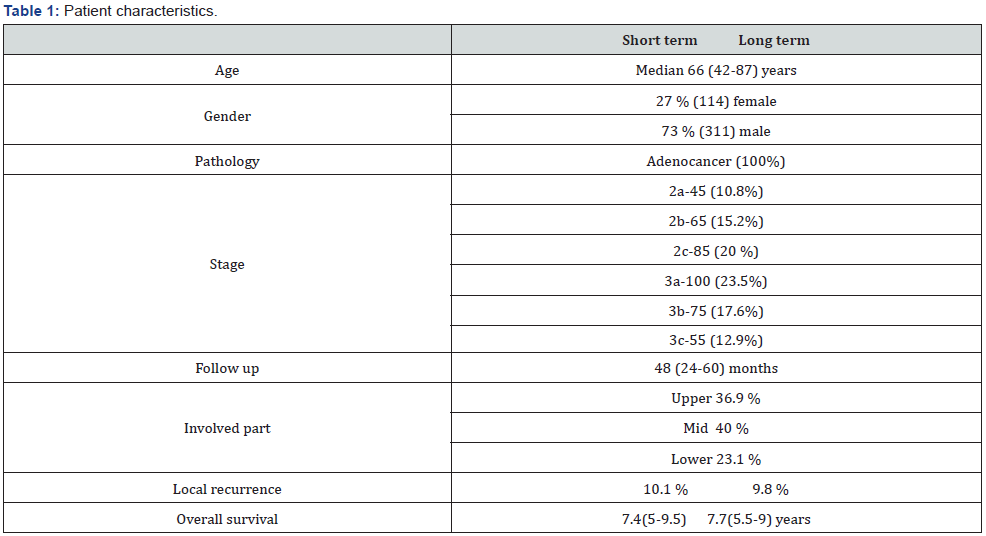
Consecutive patients with rectal cancer referred to our department for RT between January 2011 and December 2020 were evaluated. All 425 patients had rectal adenocancer and had undergone surgery (abdominoperineal resection or low anterior resection) after RT. The patients in short term group were treated with a dose of 25 Gy in 5 fractions and long term group with a dose of 45-50.4 Gy in a 25-28 fractions with chemotherapy. Concomitant chemotherapy dose is 825 mg/m2 of bid oral capecitabine. Median age was 66 (42-87) years. Histopathological diagnosis was adenocarcinoma in all 425 patients. 36.9 % of adenocarcinoma patients were in middle part, 40 % of all in lower section and 23.1 % of remaining were in upper part of rectum. Out of the total 425 patients, 45 patients (10.8 %) had stage 2a, 65 patients (15.2 %) had stage 2b, 85 patients (20 %) had stage 2c, 100 patients (23.5 %) had stage 3a, 75 patients ( 17.6 %) had stage 3b and 55 patients (12.9 %) had stage 3c rectal cancer according to American Joint Committee on Cancer (AJCC 2017) staging. Patient and tumor characteristics are summarized in Table 1.
All patients initially received pelvic external RT to planning target volume (pelvic lymph nodes, primary, mesorectum) with a 4-field box technique in the supine position. All fields were treated daily, 5 days a week, with 6-18 MV X-rays. The boost treatment to planning target boost volume (primary tumor) was delivered using with a commercially available multileaf collimator-based LINAC treatment machine (Elekta Synergy, UK). Quality Assurance Algorithm was used to optimize treatment planning with 6-18 MV X-rays.
For treatment planning, the rectum was emptied the night before and 1 to 2 hours before simulation using sodium phosphate enemas. No special preparation was mandatory to reproduce bladder filling during treatment. All patients were positioned in supine position with arms on the chest. Knee support and head pillow was used for immobilization of the patient on treatment table. Contrast enhanced CT simulation was applied for all patients (CT Lightspeed, GE Healthcare, Chalfont St. Giles, UK). After all, cross-sectional images with 2.5 mm slice thickness were acquired. The acquired images from CT Simulator were sent to the contouring Workstation via network for IGRT (image guided radiotherapy planning). Advantage SimMD simulation and localization software (Advantage SimMD, GE, UK) was used to contour the CTV and organs-at-risk (OARs). Pretreatment MRI of patients were used to evaluate disease extent but not for treatment planning purposes since the main tool for planning was CT images. Surrounding critical structures including the bladder, small bowel and femoral heads were contoured as OARs in planning CT images according to the RTOG guidelines. GTV was the primary tumor of rectum. CTV was set as rectum, mesentery, perirectal, presacral and internal iliac nodes. PTV was created after adding 0.7-1 cm. margin expansion to the CTV.
All contouring procedures were performed by an experienced radiation oncologist of gastrointestinal oncology. Dose volume histograms and isodose curves were individually generated for each patient. Dose to the PTV, OARs were recorded. Cone beam CT and XVI (X-ray Volume Imaging, Elekta, UK) were used for set-up verification. After treatment completion, local control assessment was done with pelvic MRI and endoscobic examination at 3-month intervals for the first year and at 6-month intervals for the second year. CTCAE (common toxicity criteria for adverse events) version 5.0 was used for evaluation of gastrointestinal and genitourinary toxicity. Informed consent was provided for each patient in the study along with Helsinki Declaration.
Statıstıcal Analysis
All statistical analyses were performed using standard software (SPSS version 22; SPSS Inc. Chicago, IL, USA). In descriptive statistics, mean and standard deviation were used for normally distributed variables, which were analyzed using the paired t-test, and median (minimum–maximum) was used for nonnormally distributed variables, which were analyzed using the Wilcoxon signed rank test. Statistical Package for the Social Sciences, version 15.0 (SPSS, Inc. Chicago, IL) software was used for analysis and the level of significance was set at P < 0.05.
Results
Between January 2011 and December 2020, 425 patients with stage 2-3 rectal adenocancer were treated with conformal pelvic RT using LINAC. 212 of all were received short-term RT and remaining 213 patients were delivered long-term RT. The short term prescribed dose was 25 Gy in 5 fractions and long term was 45-50. 4 Gy in 25-28 Gy fractions. PTV coverage with the 95-107 % isodose line was achieved in all patients. 38 % of long term group was received 50.4 Gy and remaining 62 % of all received 45 Gy. 27 % (114) of all the patients were female and 73 % (311) of remaining were male. At a median follow-up of 48 (24-60) months, local control rate was 10.1 % for short term and 9.8 % for long term (p=0.054). Overall survival rates were 7.4 (5- 9.5) years for short term radiotherapy and 7.7 (5.5-9) years for long term chemoraditoherapy (p=0.063). All patients completed the prescribed radiotherapy. 25 % percent of short term group had distant metastases whereas 23.7 % of long term had 22.8 % distant metastases rate(p=0.066). No ≥ grade 3 acute toxicity requiring treatment interruption was encountered. The most common acute toxicities included diarrhea, constipation, nausea and urinary urgency/frequency. Acute grade 1 gastrointestinal toxicity was observed in 65 patients (15 %). Grade 2 acute gastrointestinal toxicity was observed in 120 patients (28.2 %). Grade 1 acute urinary toxicity (urgency/ frequency) was observed in 61 patients (14.3 %) and grade 2 acute urinary toxicity (urgency/frequency) was observed in 17 patients (4 %) whereas no urinary late toxicity was observed in all patients.
Discussion
Neoadjuvant radiotherapy and chemoradiotherapy were compared in a study by FFCD 9203. 45 Gy radiotherapy or 5-FU plus leucovorine and radiotherapy combination were randomized. 4 adjuvant chemotherapy regimens were given after surgery in this study. Complete response rates were higher and local recurrence were lower with chemoradiotherapy whereas toxicity scores were slightly higher [1]. Four arm study was designed by EORTC 22921. 45 Gy alone or 5-FU and leucovorine plus radiotherapy following surgery further randomized to adjuvant chemotherapy or not. 5 year overall survival was not improved but local reccurence rate were lower and better downstaging was done with combined treatment [2].
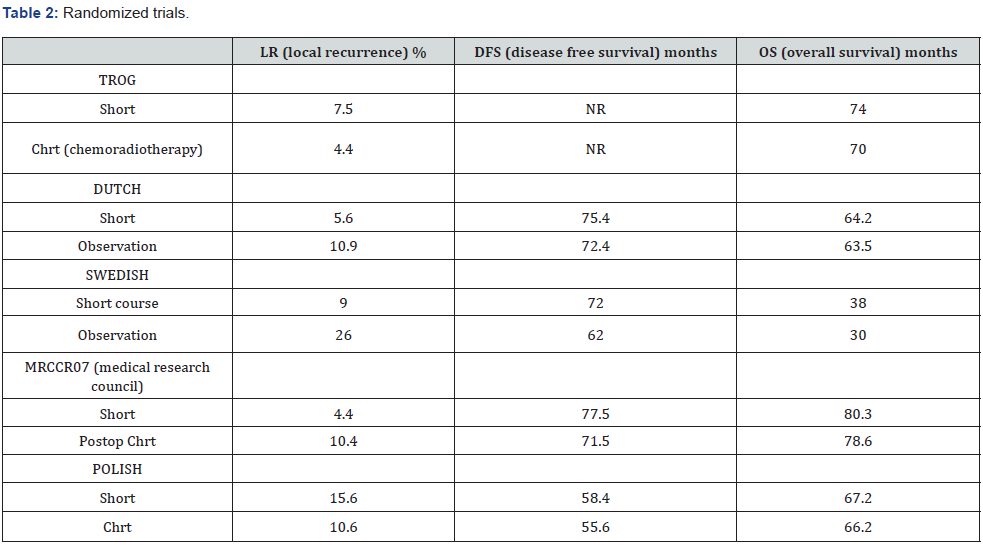
Hypofractionated preoperative short term radiotherapy was randomized with long term 50.4 Gy chemoradiotherapy by Polish rectal study group (Table 2). Complete response was higher and positive radial margin was reduced with chemoradiotherapy but no difference was seen in sphincter preservation, local control or overall survival between groups. In an Australian group study, short term was compared with long term chemoradiotherapy plus surgery and adjuvant chemotherapy. Local recurrence, sphincter preservation rate, overall survival was not significantly different but only downstaging was better with chemoradiotherapy [3].
Optimal fractionation and interval between surgery is still under debate among researchers. In a study by Erlandsson et al, short term radiotherapy following one week and 4-8 weeks surgery were randomized in 18-hospital Sweden study. Postoperative adverse effects were reduced with short course radiotherapy. Delay in surgery after radiotherapy was an viable alternative to immediate surgery [4]. Recent STAR-TREC trial asks the question of saving rectum by watchful waiting or surgery following chemoradiotherapy versus total mesorectal excision for early rectal cancer. This prospective phase 2/3 trial will give shed light on short course radiotherapy versus long term chemoradiotherapy evaluation on organ preservation approaches [5].
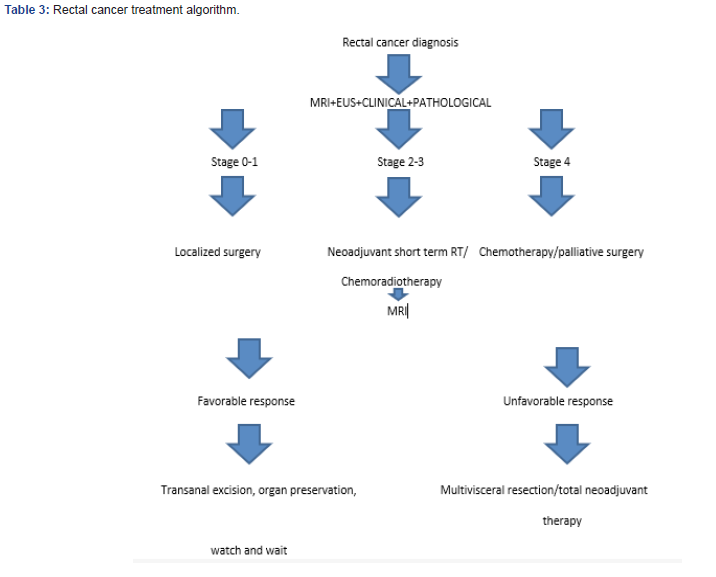
The past, present and the future of rectal cancer treatment are based on the neoadjuvant and adjuvant strategies of clinical trials but novel therapeutic approaches will focus on organ sparing management [6] (Table 3). Recent ASCO 2020 updates showed that modification of the surveillance scheme was required to detect early recurrences in locally advanced rectal cancer and the importance of total neoadjuvant therapy [7]. Short course radiotherapy is noninferior on compliance and oncological outcomes compared to long course treatment. It may be standard in developing countries for reduced treatment time and resources [8]. Rectal cancer treatment should be treated and followed by multidisciplinary team including gastroenterologist, oncologic surgeon, pathologist, medical and radiation oncologist [9].
NAR (neoadjuvant rectal) score were analysed in a randomized study by Huang et al. Higher NAR score was associated with higher distant metastasis and lesser overall survival and disease free survival rates between short term and long term schemes [10]. Neoadjuvant therapy is probably better than adjuvant therapy for stage 2-3 rectal cancer. Radiotherapy is a viable option for locally advanced, recurrent, oligometastatic and also palliative patients [11]. Recent advances in radiotherapy such as online adaptive planning proved its benefit in reducing doses for OAR (organ at risk) in rectal cancer patients [12]. The optimal treatment in rectal cancer patients with synchronous liver metastases is still debatable but neoadjuvant therapy and surgery plus adjuvant chemotherapy should be proposed for oligometastatic group [13]. Wait and see project was found with dismal overall survival, local recurrence and distant metastases compared to surgery group after neoadjuvant therapy [14]. Although fewer studies showed local recurrence and survival after short course was worse compared to chemoradiotherapy, recent studies demonstrated similar local control and long term survival with both therapy [15-17]. Disease free survival and overall survival rates were not significantly different in short term and long term group of rectal cancer patients by Wang et al. [18,19].
Lımitations of the Study
Our study is single center experience and a retrospective comparison trial. We had limited number of patients although the 10 years database. The study results including adverse effects might be better with the recent contemporary technological advances in radiotherapy if they would be applied before 10 years.
Conclusion
Preoperative neoadjuvant short term radiotherapy is noninferior to conventional long term chemoradiotherapy because of similar results in local control, disease free survival, overall survival and adverse effects.
References
- Gerard J, Conroy T, Bonnetain F, Olivier Bouche, Olivier Chapet, et al. (2006) Preoperative radiotherapy with or without concurrent fluorouracil and leucovorin in T3-4 rectal cancers: results of FFCD 9203. J Clin Oncol 24(28): 4620-4625.
- Bosset J, Calais G, Daban A, C Berger, L Radosevic-Jelic, et al. (2004) Preoperative chemoradiotherapy versus preoperative radiotherapy in rectal cancer patients: assesment of acute toxicity and treatment compliance. Report of the 22921 randomised trial conducted by the EORTC Radiotherapy Group. Eur J Cancer 40(2): 219-224.
- Ngan S, Burmeister B, Fisher R, Michael Solomon, David Goldstein, et al. (2012) Randomized trial of short course radiotherapy versus long course chemoradiation comparing rates of local recurrence in patients with T3 rectal cancer: Trans-Tasman Radiation Oncology Group trial 01.04. J Clin Oncol 30(31): 3827-3833.
- Erlandsson J, Holm T, Pettersson D, Ake Berglund, Bjorn Cedermark et al. (2017) O, Optimal fractionation of preoperative radiotherapy and timing to surgery for rectal cancer (Stockholm 3): a multicentre, randomised, non-blinded, phase 3, non-inferiority trial. Lancet Oncol 18(3): 336-346.
- Rombouts A, Al-Najami I, Abboth N, Ane Appelt, Gunnar Baatrup et al. (2017) Can we save the rectum by watchful waiting or transanal surgery following chemoradiotherapy versus total mesorectal excision for early rectal cancer (STAR-TREC)? Protocol for a multicentre, randomised feasibility study. BMJ Open 7(12): e019474.
- Oronsky B, Reid T, Larson C, Knox S (2020) Locally advanced rectal cancer: the past, present, and future. Semin Oncol 47(1): 85-92.
- Alawawdeh A, Krishnan T, Roy A, Christos Karapetis, Rohit Joshi et al. (2021) Curative therapy for rectal cancer. Expert Rev Anticancer Ther 21(2): 193-203.
- Chakrabarti D, Rajan S, Akhtar N, S Qayoom, S Gupta, et al. (2021) Short course radiotherapy with consolidation chemotherapy versus conventionally fractionated long course chemoradiotherapy for locally advanced rectal cancer. Randomized clinical trial. Br J Surg 108(5): 511-520.
- Goldenberg B, Holliday E, Helewa R, Harminder Singh, et al. (2018) Rectal cancer in 2018: a primer for the gastroenterologist. Am J Gastroenterol 113(12): 1763-1771.
- Huang W, Kuan F, Lin M, Miao-Fen Chen, Wen-Cheng Chen, et al. (2020) Prognostic significance of neoadjuvant rectal scores in preoperative short course radiotherapy and long course concurrent chemoradiotherapy for patients with locally advanced rectal cancer. Ann Surg Oncol 27(11): 4309-4318.
- Hafner M, Debus J (2016) Radiotherapy for colorectal cancer: current standards and future perspectives. Visc Med 32(3): 172-177.
- Jong R, Crama K, Visser J, N van Wieringen, J Wiersma et al. (2020) Online adaptive radiotherapy compared to plan selection for rectal cancer: quantifying the benefit. Radiat Oncol 15(1): 162.
- Moslim M, Bastawrous A, Jeyarajah D (2021) Neoadjuvant pelvic radiotherapy in the management of rectal cancer with synchronous liver metastases: is it worth it? J Gastrointest Surg 25(9): 2411-2422.
- Lin G, Lee K, Wang J, Jinn-Shiun Chen, Chun-Ju Chiang et al. (2018) Status for clinically complete remission rectal cancer after concomitant chemoradiotherapy in Taiwan. Asian J Surg 41(3): 203-209.
- Wawok P, Polkowski W, Richter P, Marek Szczepkowski, Janusz Oledzki et al. (2018) Preoperative radiotherapy and local excision of rectal cancer: long term results of a randomised study. Radiother Oncol 127(3): 396-403.
- Zhu J, Zhang Z (2014) Timing of radiotherapy for locally advanced rectal cancer. Zhonghua Wei Chang Wai Ke Za Zhi 17(11): 1072-1075.
- Cisel B, Pietrzak L, Michalski W, L Wyrwicz, A Rutkowski et al. (2019) Long course preoperative chemoradiation versus 5x5 Gy and consolidation chemotherapy for clinical T4 and fixed clinical T3 rectal cancer: long term results of the randomized Polish 2 study. Ann Oncol 30(8): 1298-1303.
- Rodel C, Trojan J, Bechstein W, Woeste G (2012) Neoadjuvant short or long term radio (chemo) therapy for rectal cancer. How and who should be treated? Dig Dis 30(2): 102-108.
- Wang J, Long Y, Liu K, Pei Q, Zhu H (2021) Comparing neoadjuvant long course chemoradiotherapy with short course radiotherapy in rectal cancer. BMC Gastroenterol 21(1): 277.






























