Prognostic Value of Cancer-Associated Fibroblasts in Pancreatic Cancer: A Meta-Analysis
Huaqing Shi1, Zhou Chen1, Shi Dong1, Ru He1 and Wence Zhou2*
1The First Clinical Medical College, Lanzhou University, Lanzhou, China
2Department of General Surgery, Lanzhou University Second Hospital, Lanzhou, China
Submission: June 15, 2022; Published: July 01, 2022
*Corresponding Address: Wence Zhou, Department of General Surgery, the Lanzhou University Second Hospital, No. 82, Zuiyingmen, Lanzhou 730030, Gansu Province, China
How to cite this article: Huaqing S, Zhou C, Shi D, Ru H, Wence Z. Prognostic Value of Cancer-Associated Fibroblasts in Pancreatic Cancer: A Meta- Analysis. Canc Therapy & Oncol Int J. 2022; 21(5): 556073. DOI:10.19080/CTOIJ.2022.21.556073
Abstract
Background: Numerous studies have investigated components of the tumor microenvironment, such as cancer associated fibroblasts (CAF), and their role in pancreatic cancer prognosis. Nonetheless, the prognostic value of CAF in cancer is still controversial.
Aim: The current meta-analysis sought to determine the impact of CAF in pancreatic cancer prognosis.
Methods: A literature screening of articles involving the prognostic significance of CAF in pancreatic cancer was done on PubMed, EMBASE, as well as Web of Science covering the period from databases establishment to Oct 31, 2021. Overall survival (OS) along with disease-free survival (DFS) were explored through data pooling. We adopted the pooled HRs (hazard ratios with 95% CI (confidence intervals) to determine the relationship of CAF with survival of individuals with pancreatic cancer. Overall, 10 studies involving 1308 pancreatic cancer patients were enrolled in our meta-analysis.
Results: Elevated invasion with CAF were remarkably linked to dismal OS (HR=2.20, 95% CI: 1.68-2.87, p=<0.05). Subgroup analysis revealed that being older than 65 years correlated with poor pancreatic cancer prognosis (HR=2.04, p=<.05, 95% CI: 1.5-2.78).
Conclusion: Our findings indicate that CAF is a negative prognostic factor in pancreatic cancer.
Keywords: Cancer associated fibroblasts; Prognosis; Pancreatic cancer
Abbreviations: CAF: Cancer Associated Fibroblasts; OS: Overall Survival; DFS: Disease-Free Survival; HRs: Hazard Ratios; 95% Cis: 95% Confidence Intervals; PDAC: Pancreatic Ductal Adeno-Carcinoma; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; MOOSE: Meta-analysis of Observational Studies in Epidemiology; TME: Tumor Microenvironment; myCAFs: Myofibroblastic Cancer-Associated Fibroblast; α-SMA Alpha Smooth Muscle Actin; iCAFs: Inflammatory Cancer-Associated Fibroblasts; CXCL1: C-X-C Chemokine Ligand 1; CXCL2: C- X-C Chemokine Ligand 2; rCAFs: Cancer-restraining CAFs; MMT: Monocyte-to-Myofibroblast Trans-Differentiation; FDAL Food and Drug Administration; NA: Not Applicable; IHC: Immunohistochemistry; qRT-PCR: quantitative Real-Time Polymerase Chain Reaction; WB: Western Blot; M: Male; F: Female
Introduction
Pancreatic cancer, notably pancreatic ductal adenocarcinoma (PDAC), is characterized by very poor prognosis and has a five-year survival of <8% [1,2]. Despite extensive research, there are no effective predictors of pancreatic cancer prognosis [3]. The tumor microenvironment, a complex stromal network that supports cancer cells, may contribute to treatment resistance by pancreatic cancer [4]. Pancreatic cancer is characterized by an abundant desmoplastic stroma, dominated by CAFs, which influence the behavior of cancer cells [5]. Recent studies show that CAFs are highly heterogeneous, although their role and clinical significance are not well established [6]. Moreover, the prognostic importance of CAF in pancreatic cancer is still controversial. Here, we conducted a meta-analysis to determine the prognostic significance of CAF in PDAC.
Material and Methods
Search strategy and selection of articles
A literature search for studies involving CAFs in the prediction of the prognosis of diverse cancers was done on PubMed, EMBASE, as well as Web of Science (last update by Oct 31, 2021) using the following search terms: Cancer-associated Fibroblasts, Tumor-associated Fibroblasts, Fibroblasts, Tumor-associated or Fibroblasts, Cancer-associated and Pancreatic Cancer, Pancreatic Neoplasms or Cancer of Pancreatic. We utilized free text along with MeSH search of the keywords. The function of ‘‘related information’’ was adopted for broadening of the screening. We reviewed all abstracts, references along with full texts. The study was as per PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) [7], as well as the MOOSE (Metaanalysis Of Observational Studies in Epidemiology) guidelines [8]. First, screening of the titles along with the abstracts of searched articles was done and retrieved those that could not be categorized on the basis of the title coupled with the abstract alone for screening of the full text. The main search was done by the first author (Huaqing Shi) and any discrepancy resolved by consensus or consultation with another investigator (Wence Zhou).
Selection criteria
Two authors independently selected and evaluated the studies. Articles that met the the following criteria were enrolled: (a) study subjects were humans, (b) study involved association between CAF and pancreatic cancer survival outcomes, (c) CAF expression levels were determined using immunohistochemistry, (d) the reported data was sufficient to determine hazard ratio (HR) and 95% CI. We excluded the following types of articles: (a) review articles, conference abstracts, letters, as well as case report articles; (b) duplicate articles; (c) studies lacking key information for further analysis; (d) studies that did not involve pancreatic cancer and CAF, and other indicators were mainly observed, so data could not be obtained. Disagreement was resolved through discussion amongst the investigators.
Data extraction
Two investigators independently abstracted data from included articles. For each article, the following data was abstracted: first author, ethnicity, year of publication, country, overall number of cases, examination methods, tumor stage, grade, gender, threshold value, follow up and HR of CAF for OS, DFS (with 95% CI), and number of lymph node metastases.
Statistical analysis
We employed pooled HR along with its 95% CI to assess the influence of CAF on OS and DFS for individuals with pancreatic cancer with regards to time-to-event outcomes. A remarkable number of enrolled articles had HR along with 95% CIs, which were directly abstracted from the text. In case of the provision of Kaplan-Meier curves instead of HR, HR was determined indirectly from the curves via the Engauge Digitizer v4.1 (http:// digitizer. Source forge. net/) as documented before [9]. The HR from every article presented an estimate of the ratio of the HR for CAF expression a high level over a low-level or positive over negative, CAF expression depicted a dismal prognosis if the HR>1, whilst an HR<1 depicted a good prognosis.
Across the articles, heterogeneity was evaluated with the chisquare- dependent Q statistical test and quantification done via the I2 statistic, with I2 = >50% signifying remarkable heterogeneity. If heterogeneity was not remarkably different (I2 = <50%), we computed pooled effect via the fixed-effects approach. Else, pooled statistics were computed using the random-effects approach. Subgroup analyses were conducted according to country, ethnicity, examination methods, survival analysis outcome, sample size, age, and rate of lymph node positivity. Publication bias was explored via visual inspection of funnel plots and quantitation done with the Egger’s or Begg’s regression approach, with p=<0.10 signifying statistical significance. Meta-regression assessment was done to explore the possible risk factors for heterogeneity. Sensitivity assessment was implemented to assess the stability of the pooled data. Statistical analyses were implemented in STATA (v16.0), with p=<0.05 signifying statistical significance.
Results
Study selection and characteristics
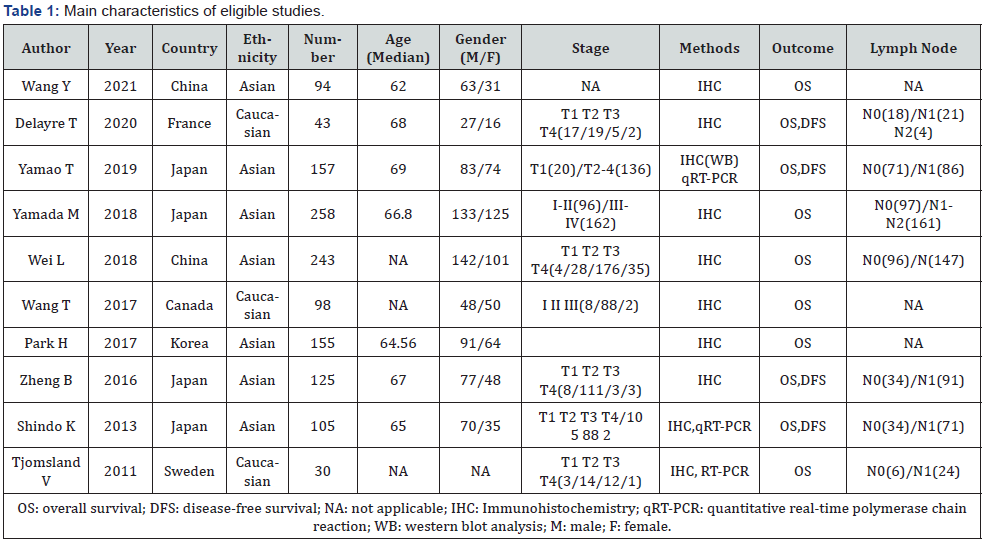
Of the 3454 studies retrieved from the literature screening (Figure 1), 10 met the inclusion criteria (Table 1). These studies involved 1308 individuals with pancreatic cancer. The individual sample size ranged from 30-258 (mean 237.818). These studies were conducted in France, Korea, Canada, Sweden, China, and Japan. Survival data were abstracted from Kaplan-Meier curves in 3 studies [4,10,11]. Four studies had sample sizes of >150. Seven studies involved patients of Asian ethnicity, while three involved Caucasians. Ten articles had OS as a prognostic indicator to explored CAF in pancreatic cancer tissues. Four studies had OS along with DFS as indicators.
CAF and prognosis of pancreatic cancer
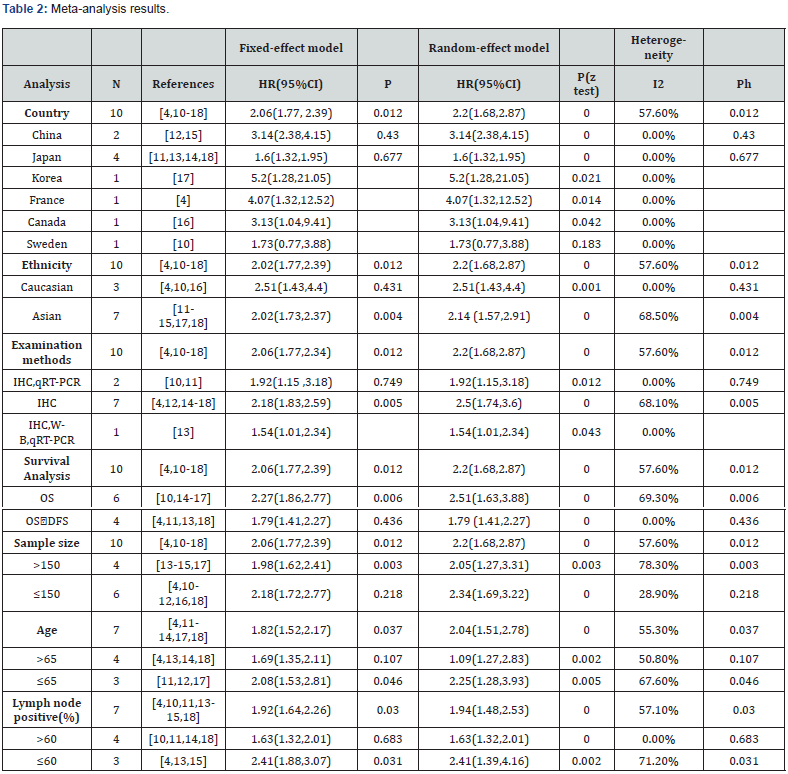
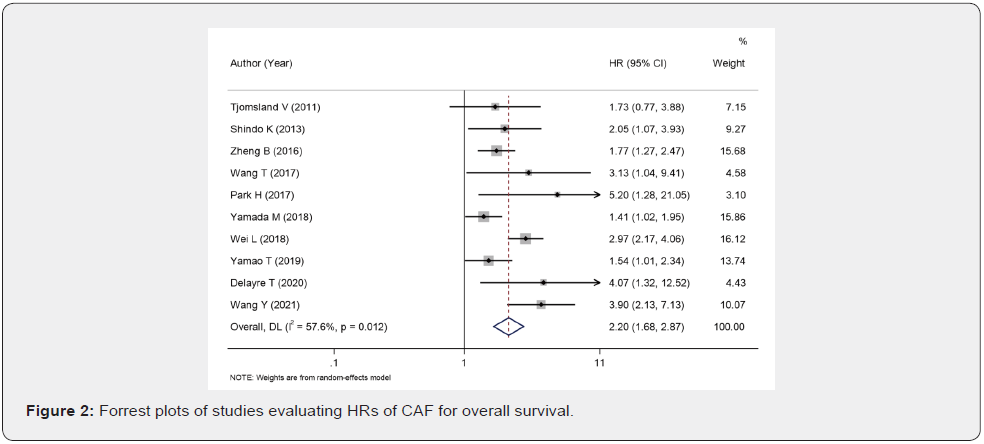
We pooled OS along with DFS to explore the influence of CAF on pancreatic cancer prognosis. The main findings of this meta-analysis are given in Table 2. We established that CAF predicts worse poor OS (combined HR=2.2, 95% CI: 1.68-2.87, Pheterogeneity = 0.012, Figure 2). Sub-group assessment via ethnicity exhibited that negative predictor of CAF for OS was depicted both in Asian cases (HR = 2.14, 95% CI: 1.57-2.91, Pheterogeneity, 0.004) and in Caucasian populations (HR = 2.51, 95% CI: 1.43-4.4). Subgroup analyses of sample size revealed excess risk of pancreatic cancer in sample sizes >150 (HR=2.05, p=0.003, 95% CI: 1.27-3.31) and sample sizes ≤150 (HR=2.34, 95% CI: 1.69-3.22). When country (HR=2.2, 95% CI: 1.68-2.87; P=0.012), examination methods (HR=2.2, 95% CI: 1.68-2.87; P=0.012), survival analysis outcome (HR=2.2, P=0.012, 95% CI: 1.68-2.87), age and lymph node positive were considered, lymph node positive rate >60 (HR=1.63, 95% CI: 1.32-2.01) and lymph node positive rate ≤60 (HR=2.41, 95% CI: 1.39-4.16; P=0.031), age >65 (HR=1.09, 95% CI: 1.27-2.83) and age ≤65 (HR=2.25, P=0.046, 95% CI: 1.28-3.93).
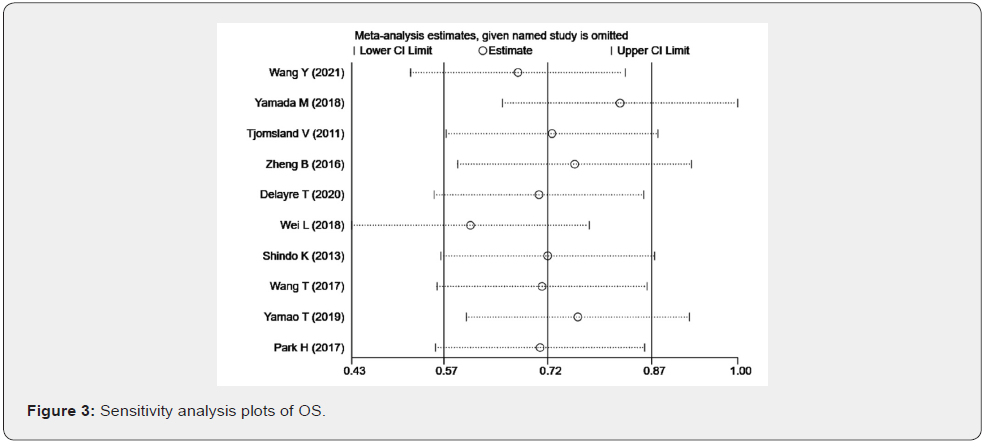
Sensitivity analysis
Leave-one-out sensitivity assessment, in which one study is excluded at a time, was used to check if individual studies influenced the data. Sensitivity assessment results exhibited that HR did not change remarkably after removing any article (Figure 3).
Meta-regression analysis
Meta-regression analyses indicated that age (p=0.082, R2=87.68%), sample size (p=0.512, R2=-17.11%), ethnicity (p=0.618, R2=-8.96%), survival analysis outcome (p=0.391, R2=-3.79%, examination methods (p=0.257, R2=-7.7%, and lymph node positivity rate (p=0.742, R2=-27.52%) did not cause study heterogeneity. Adjusted R2 = -27.52 to -87.68% indicated that these regressors slightly contributed to the explanation of response variables.
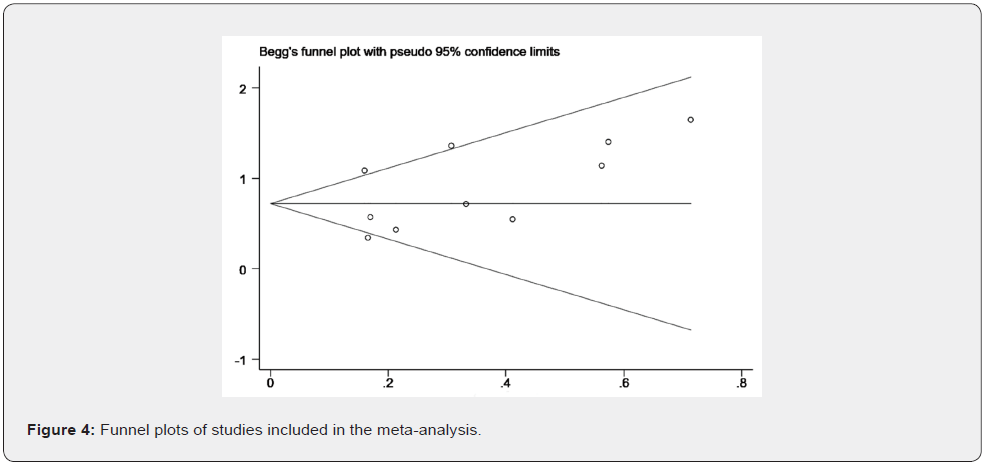
Publication bias
Begg’s test revealed no publication bias after examining the funnel plot for the articles enrolled in this meta-analysis (Figure 4). The funnel plots did not reveal obvious asymmetry (p=0.283), indicating that there was no remarkable publication bias in the articles that utilized OS to assess survival outcome.
Discussion
Pancreatic cancer is the primary cause of cancer morbidity along with mortality and PDAC is responsible for >90% of pancreatic cancers [12,13]. Pancreatic cancer has a dense interstitial composition (such as CAFs) and is not efficiently penetrated by drugs, which contributes to resistance. Hence, novel, effective therapies and early diagnostic strategies for pancreatic cancer are urgently needed. CAFs are an important component of canassociated stromal cells and are also a primary component of the TME (tumor microenvironment). CAFs influence tumorigenesis, invasion, metastasis, immunosuppression, and chemoresistance through various pathways [14]. There are several CAF subtypes, including myCAFs (myofibroblastic cancer-associated fibroblast) that are characteristically expressed by α-SMA (alpha smooth muscle actin) and transgelin. It plays an indispensable role in cancer proliferation, migration and infiltration [15,16]. Along with iCAFs (inflammatory cancer-associated fibroblasts), its characteristic expression of CXCL1 (C-X-C chemokine ligand 1), CXCL2 (C- X-C chemokine ligand 2). Its main function is Immune promotion [17]. Although CAFs generally exhibit cancerpromoting effects, cancer-restraining CAFs (rCAFs) have been identified [18]. Several factors have been shown to promote CAFs, including TGF-β, ROS (reactive oxygen species), as well as FGF- 2 that convert tissue-resident fibroblasts along with quiescent stellate cells into CAFs [19-21], as well as drive monocyte-tomyofibroblast trans-differentiation (MMT) [22]. Drugs targeting CAF-linked effector biomolecules, for instance tumor stromal hyaluronan-targeted depletion in combination with gemcitabine along with nab-paclitaxel enhance survival with fewer systematic side effects [23].
In this meta-analysis, we analyzed 10 studies involving 1308 patients. The HRs for the association between CAF and OS was 2.2. Subgroup analysis revealed that the HRs for sample sizes >150, age >65, lymph node positive rate, and ethnicity, were 2.2, 2.04, 1.94, and 2.2, respectively. These pooled results showed that CAFs are associated with poor pancreatic cancer survival. Numerous CAF subtypes have been identified. In addition to myfibroblast myCAF, which is highly expressed by a-SMe A, the TME also contains inflammatory cytokines like interleukin (IL-6), and iCAF [24], and its related functions and effects need further study. These results provide crucial information on the prognosis and treatment of pancreatic cancer. Thus, further studies involving large sample sizes are needed to validate our findings.
However, this study has some limitations. First, because this study was limited to studies published in English, important studies in other languages may be missed. Second, the patients included in the 10 studies did not essentially have consistent treatments and the TNM classification and histological typing varied. However, the sensitivity analysis exhibited that individual articles had little influence on the overall outcome. Thirdly, some clinicopathological data were missing. Finally, HR and 95% CIs abstracted from survival curves but not directly obtained from the studies may be less reliable.
Conclusion
This meta-analysis revealed that CAF predicts poor pancreatic cancer survival and has potential therapeutic value in pancreatic cancer
Declarations
Consent for publication
Not applicable.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing Interests
The authors declare that they have no competing interests.
Funding
The authors received no funding for this research.
Authors’ contributions
Wence Zhou designed the study; Huaqing Shi wrote the manuscript; Huaqing Shi, Zhou Chen, Shi Dong, Ru He collected and analyzed the data. All authors read and approved the final manuscript, declare no potential conflict of interest, and agree to publish and sign.
Acknowledgement
Thanks to all the team members for their efforts in writing and publishing this article.
References
- Siegel R, Ma J, Zou Z, Jemal A (2014) Cancer statistics, 2014. CA Cancer J Clin 64(1): 9-29.
- Miller KD, Nogueira L, Mariotto AB, Rowland JH, Yabroff KR, et al. (2019) Cancer treatment and survivorship statistics, 2019. CA Cancer J Clin 69(5): 363-385.
- Hidalgo M (2010) Pancreatic cancer. The New England journal of medicine 362: 1605-1617.
- Delayre T, Guilbaud T, Resseguier N, Mamessier E, Rubis M, et al. (2020) Prognostic impact of tumour-infiltrating lymphocytes and cancer-associated fibroblasts in patients with pancreatic adenocarcinoma of the body and tail undergoing resection. Br J Surg 107(6): 720-733.
- Erkan M, Hausmann S, Michalski CW, Fingerle AA, Dobritz M, et al. (2012) The role of stroma in pancreatic cancer: diagnostic and therapeutic implications. Nature reviews Gastroenterology & hepatology 9(8): 454-467.
- Neuzillet C, Tijeras-Raballand A, Ragulan C, Cros J, Patil Y, et al. (2019) Inter- and intra-tumoural heterogeneity in cancer-associated fibroblasts of human pancreatic ductal adenocarcinoma. Nat Rev Gastroenterol Hepatol 248(1): 51-65.
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, et al. (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Journal of clinical epidemiology 6(7): e1-34.
- Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, et al. (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of Observational Studies in Epidemiology (MOOSE) group. JAMA 283(15): 2008-2012.
- Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR (2013) Response to: Practical methods for incorporating summary time-to-event data into meta. Authors' reply. Trials 14: 391.
- Tjomsland V, Spangeus A, Valila J, Sandstrom P, Borch K, et al. (2011) Interleukin 1 alpha Sustains the Expression of Inflammatory Factors in Human Pancreatic Cancer Microenvironment by Targeting Cancer-Associated Fibroblasts. Neoplasia 13(8): 664-675.
- Shindo K, Aishima S, Ohuchida K, Fujiwara K, Fujino M, et al. Podoplanin expression in cancer-associated fibroblasts enhances tumor progression of invasive ductal carcinoma of the pancreas. Mol Cancer 12(1): 168.
- Orth M, Metzger P, Gerum S, Mayerle J, Schneider G, et al. (2019) Pancreatic ductal adenocarcinoma: biological hallmarks, current status, and future perspectives of combined modality treatment approaches. Radiation oncology (London, England) 14: 141.
- Chen H, Kong Y, Yao Q, Zhang X, Fu Y, et al. (2019) Three hypomethylated genes were associated with poor overall survival in pancreatic cancer patients. Aging 11(3): 885-897.
- Mao X, Xu J, Wang W, Liang C, Hua J, et al. (2021) Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Molecular cancer 20: 131.
- Hosein AN, Huang H, Wang Z, Parmar K, Du W, et al. (2019) Cellular heterogeneity during mouse pancreatic ductal adenocarcinoma progression at single-cell resolution. JCI insight 5(16): e129212.
- Lakins MA, Ghorani E, Munir H, Martins CP, Shields JD (2018) Cancer-associated fibroblasts induce antigen-specific deletion of CD8 T Cells to protect tumour cells. Nature communications 9: 948.
- Jiang H, Hegde S, DeNardo DG (2017) Tumor-associated fibrosis as a regulator of tumor immunity and response to immunotherapy. Cancer immunology, immunotherapy : CII 66(8): 1037-1048.
- Mizutani Y, Kobayashi H, Iida T, Asai N, Masamune A, et al. (2019) Meflin-Positive Cancer-Associated Fibroblasts Inhibit Pancreatic Carcinogenesis. Cancer Res 79(20): 5367-5381.
- Hawinkels LJAC, Paauwe M, Verspaget HW, Wiercinska E, van der Zon JM, et al. (2014) Interaction with colon cancer cells hyperactivates TGF-β signaling in cancer-associated fibroblasts. Oncogene 33(1): 97-107.
- Elenbaas B, Weinberg RA (2001) Heterotypic signaling between epithelial tumor cells and fibroblasts in carcinoma formation. Experimental cell research 264(1): 169-184.
- Kuzet S-E, Gaggioli C (2016) Fibroblast activation in cancer: when seed fertilizes soil. Cell and tissue research 365(3): 607-619.
- Wei M, Yang T, Chen X, Wu Y, Deng X, et al. (2017) Malignant ascites-derived exosomes promote proliferation and induce carcinoma-associated fibroblasts transition in peritoneal mesothelial cells. Oncotarget 8(26): 42262-42271.
- Doherty GJ, Tempero M, Corrie PG (2018) HALO-109-301: a Phase III trial of PEGPH20 (with gemcitabine and nab-paclitaxel) in hyaluronic acid-high stage IV pancreatic cancer. Future oncology (London, England)14(1): 13-22.
- Elyada E, Bolisetty M, Laise P, Flynn WF, Courtois ET, et al. (2019) Cross-Species Single-Cell Analysis of Pancreatic Ductal Adenocarcinoma Reveals Antigen-Presenting Cancer-Associated Fibroblasts. Cancer discovery 9(8): 1102-1123.






























