The Exuberant Stratum-Focal Epithelial Hyperplasia
Anubha Bajaj*
Histopathologist in A B Diagnostics, New Delhi, India
Submission: March 23, 2022; Published: April 19, 2022
*Corresponding Address: Anubha Bajaj, Histopathologist in A B Diagnostics, New Delhi, India
How to cite this article: Anubha B. The Exuberant Stratum-Focal Epithelial Hyperplasia. Canc Therapy & Oncol Int J. 2022; 21(2): 556058. DOI:10.19080/CTOIJ.2022.21.556058
Preface
Focal epithelial hyperplasia is an exceptional, benign condition occurring due to infection with various strains of human papilloma virus (HPV). Additionally, designated as “Heck’s disease” or “multifocal epithelial hyperplasia (MEH)”, the benign focal epithelial hyperplasia (FEH) incriminating oral mucosa arises due to infection with human papillomavirus type 13 (HPV13), human papillomavirus type 32(HPV32) or a concurrence of dual viruses [1]. Besides, emergence of diverse HPV variants within oral lesions may be challenging to ascertain. “Superinfection exclusion” is defined by infection of a singular cell by a pertinent viral variant wherein the cell appears incapable of infection with a secondary virus of identical subtype. Infection with multiple HPV genotypes may concur with superinfection exclusion, a feature which may curtail secondary co-infection. Hence, coinfection with multiple HPV genotypes can be infrequently discerned. The benign, selflimiting condition requires therapeutic intervention for cosmetic concerns and for lesions obstructing mastication. Generally, focal epithelial hyperplasia is devoid of malignant metamorphosis.
Disease Characteristics
Human papilloma virus 13(HPV13) and human papilloma virus 32(HPV32) are non-oncogenic viruses commonly discerned in focal epithelial hyperplasia and the lesion exhibits minimal possible neoplastic transformation. However, infection or coinfection with human papilloma virus subtypes 6, 11, 16, 18, 31, 39, 40, 51, 52, 55, 58, 66, 68, 69, 71, 74 or 90 may engender focal epithelial hyperplasia [2,3]. Besides, the condition may occur due high-risk variants of human papilloma virus. Although human papilloma virus 32 (HPV32) is genetically concurrent to human papilloma virus 16, the subtype is devoid of possible induction of malignant metamorphosis [2,3]. Persistent, asymptomatic, oral infection with human papilloma virus 16 (HPV16) is associated with genomic integration of HPV16 with consequent predisposition of infected individuals towards disease progression [2,3]. Nevertheless, focal epithelial hyperplasia depicting coinfection with high-risk HPV genotypes such as HPV16 or HPV18 can be associated with obscure outcome upon possible malignant transformation [2,3]. Although focal epithelial hyperplasia may be co-infected with multiple HPV genotypes, certain degree of superinfection exclusion occurs with high-risk HPV genotypes such as HPV16 and HPV18 [2,3]. Also, pre-existing infection with high-risk HPV16 may be competent in prohibiting secondary infection with high-risk HPV18 upon cellular surface engendered during viral attachment phase. Consequently, a significantly decimated infectivity with HPV18 may ensue following cellular infection with HPV16. Nevertheless, an existing HPV infection may prohibit secondary HPV infection with similar genotypes [2,3].
Viruses incriminated in occurrence of focal epithelial hyperplasia are transmitted through saliva. A female predominance is observed with a female to male proportion of 4:3 [2,3]. Commonly, the condition is observed in childhood or adolescents. Mean age of lesion discernment is 23.1 years although the condition may arise from around ~3 years to up to ninth decade [3,4]. Lesions emerging due to infection with HPV1, HPV13 and HPV32 manifest as discrete, repetitive, papillary plaques confined to the oral mucosa and may especially be discerned in Eskimos and Native American children. Immunosuppressed individuals depict an enhanced disease incidence [3,4]. Focal epithelial hyperplasia may implicate multiple, related individuals. The condition is common in subjects of low socioeconomic status, family members co-habiting or in proximity or within the rural population. Individuals sharing food, personal items as toothbrushes, utensils and subjects exposed to poor hygiene may delineate focal epithelial hyperplasia [3,4].
Clinical Elucidation
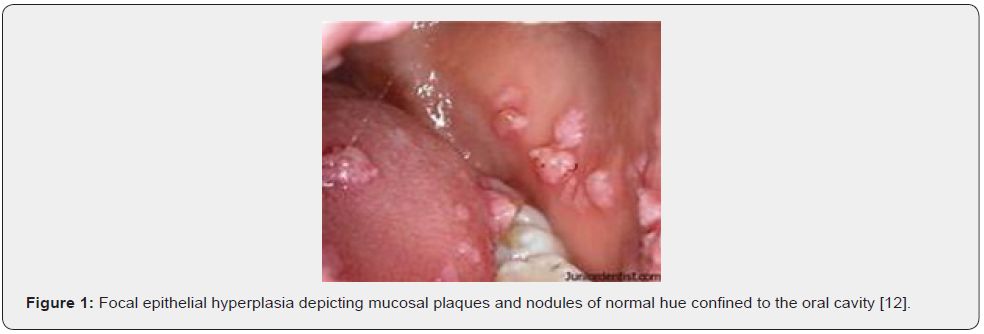
Focal epithelial hyperplasia represents as multiple, asymptomatic, whitish, or mucosa-hued, soft papules or nodular, elevated, oral lesions ranging from 0.2 centimetres to 3 centimetres diameter. The condition is associated with a benign clinical course and may disappear upon stretching of mucosa. However, enlarged lesions necessitate therapeutic intervention [5,6]. Characteristically, focal epithelial hyperplasia may manifest as spontaneously disappearing nodules which are devoid of residual, functional or aesthetic defects. Lesions may be asymptomatic, remain for weeks or extend up to > 30 years and may not warrant pertinent therapy [5,6]. Engendered by HPV 13, HPV32 or HPV31, focal epithelial hyperplasia typically represents as asymptomatic papules confined to the oral mucosa. Asymptomatic, elevated, cutaneous eruptions can appear upon the lower lip. Magnitude and quantification of lesions may gradually enhance. Cosmetic concerns can occur [5,6]. Upon examination, soft papules may be discerned, especially upon mucosa of lower lip. Lesions demonstrate a hue akin to normal buccal mucosa although whitish surface striations may occur. Mucosal surface of upper lip, buccal mucosa and lingual surface may not be incriminated. Focal epithelial hyperplasia may appear as a comorbid condition associated with diverse diseases. Particularly, diseases with immunosuppression and decimated values of naïve CD4+ T lymphocytes such as infection with human immunodeficiency virus (HIV) and intestinal lymphangiectasis may concur with focal epithelial hyperplasia [5,6]. Focal epithelial hyperplasia emerges in individuals with immunodeficiency as observed with lymphopenia, hypo-gammaglobulinemia, leukocyte adhesion deficiency, lung transplant, chronic graft versus host disease (GVH) and rheumatoid arthritis [5,6].
Histological Elucidation
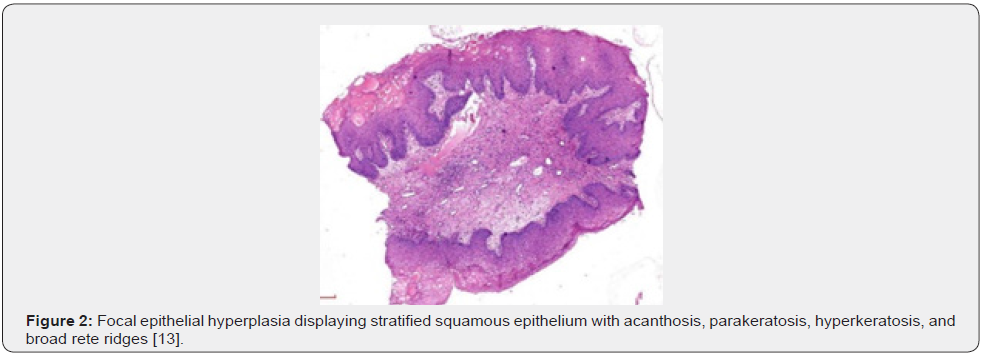
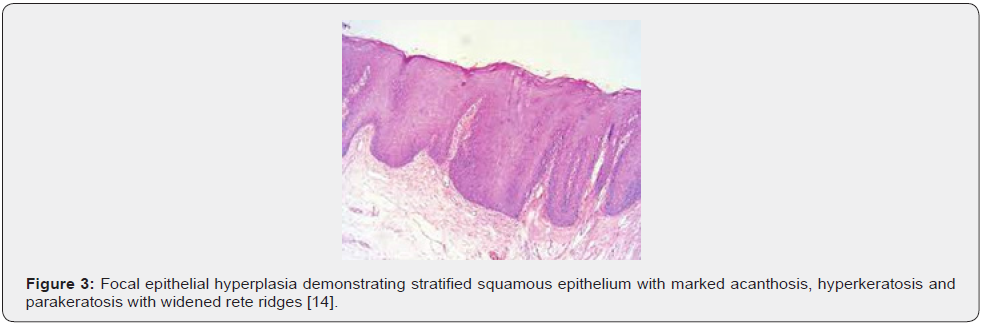
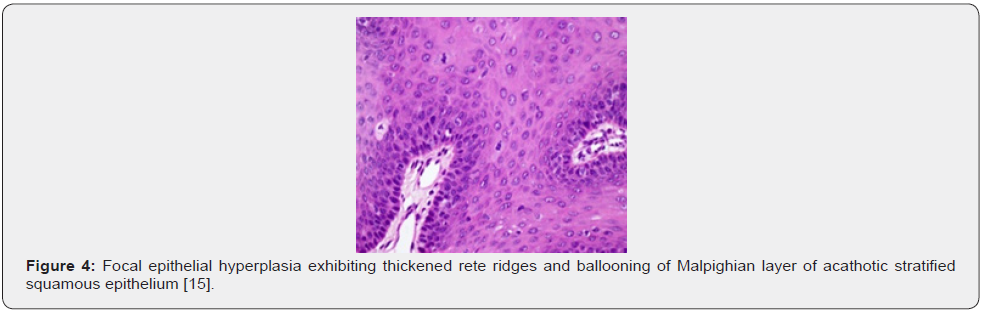
Upon macroscopic examination, well circumscribed, sessile plaques and pale elevation of buccal mucosa may be observed [7,8]. Pathognomonic features emerge as koilocytosis, ballooning degeneration, epithelial cells with clear cytoplasm, parakeratosis, acanthosis, epithelial hyperplasia, broad, thick, rete ridges, ballooning degeneration and mitosoid bodies with fragmented nuclear deoxy ribo-nucleic acid (DNA) [7,8]. Upon microscopy, acanthosis, focal parakeratosis, broad rete ridges and foci of typical koilocytic change are observed within the superimposed stratified squamous epithelium. Also, nuclear features simulating “mitosoid” bodies are delineated. Malpighian layer exhibits aggregates of balloon cells. Zones of mucosal epithelial hyperplasia demonstrating significant acanthosis and parakeratosis can be exemplified [7,8]. Segregation is necessitated from HPV-induced conditions such as verruca vulgaris or squamous cell papilloma [7,8] (Figures 1-8).
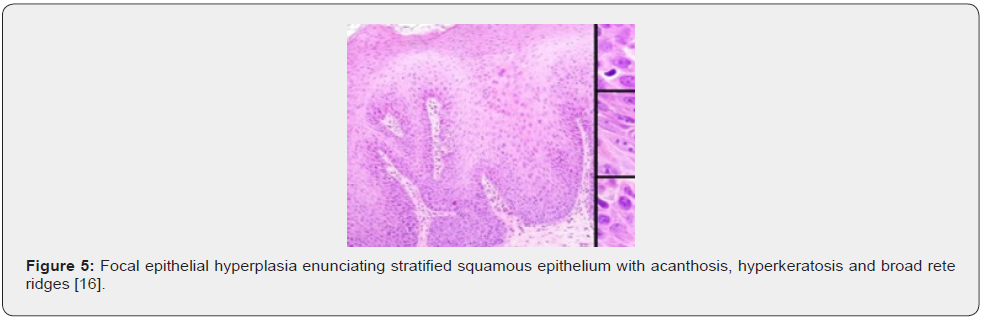
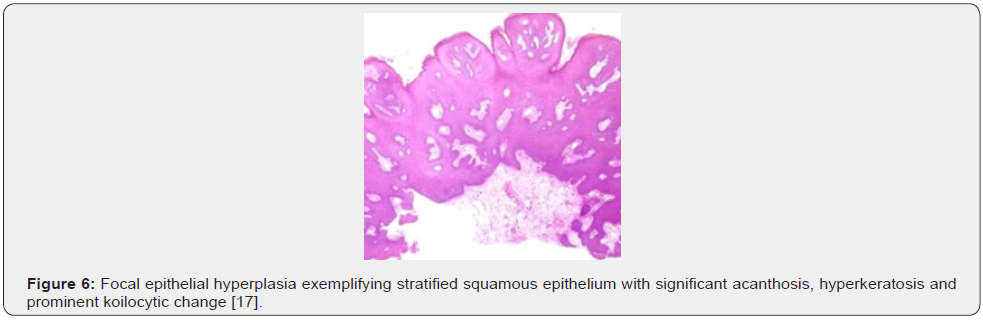
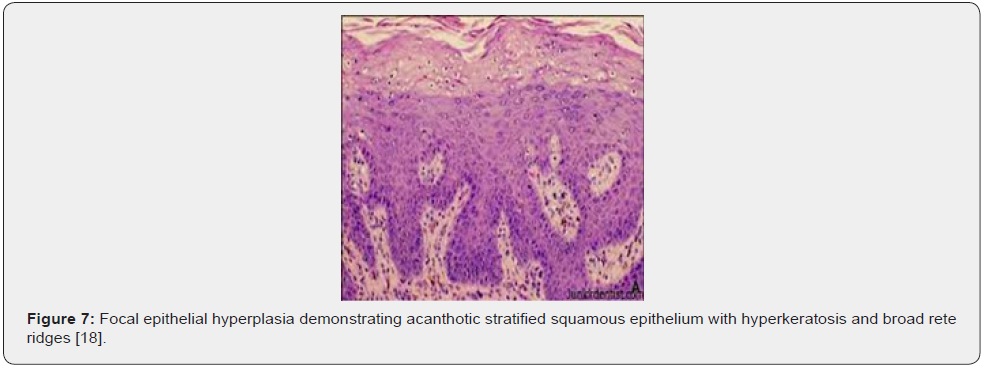
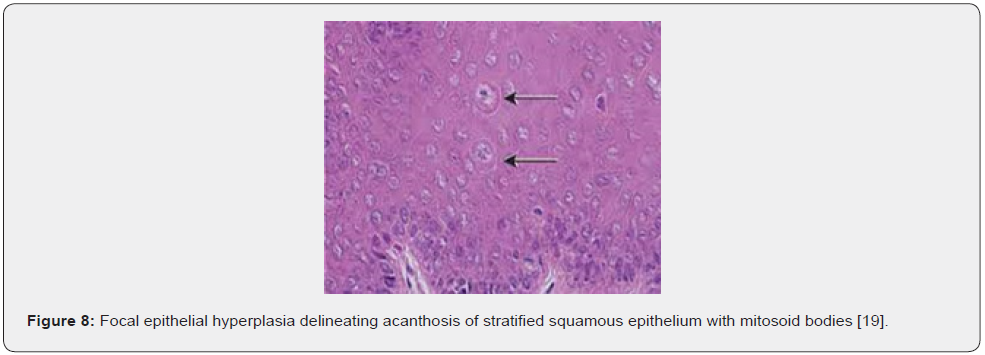
Investigative Assay
Focal epithelial hyperplasia can be adequately determined upon cogent clinical representation and histopathological assessment. Nevertheless, appropriate evaluation of viral (HPV) contribution towards lesion emergence may be challenging. Pertinent morphological assessment of tissue samples for HPV infection may be unwarranted. A cogent human papilloma virus (HPV) panel assessment may be unwarranted in lesions determined upon clinical or histological examination [8,9]. A pananti- human papilloma virus (HPV) antibody can be employed for immunohistochemistry upon specific tissue samples [8,9]. Furthermore, low-risk and minimally asymptomatic viral variants as HPV13 and HPV32 are usually non-assessable with commonly employed genetic probes [8,9]. Additionally, infection with HPV13 and HPV32 may be determined with polymerase chain reaction (PCR) with specific primers. Polymerase chain reaction (PCR) is an optimal, recommended investigative modality to demonstrate HPV13 and HPV32. However, findings in certain instances may be non-confirmatory [8,9]. Pertinent instances may be subjected to DNA sequencing.
The non-invasive cyto-brush technique can be adopted to collect exfoliative oral squamous epithelial cells. Children and field workers are especially amenable to the technique [9,10]. A specific allele of human leukocyte antigen (HLA) family, the HLA-DR4 may exhibit an enhanced frequency of focal epithelial hyperplasia. Commonly, HLA-DR4 is exemplified among indigenous South American population [9,10]. Upon dermoscopy, well defined lobules of squamous epithelium appear demarcated by whitish septa. The lobules delineate prominent, polymorphic vascular configurations designated as linear, branching vessels, dendritic vessels, and hairpin vessels. The vascular articulations are disseminated upon an orange-yellow background [9,10]. Apart from characteristic, prominent, polymorphic vascular pattern, lobules and septa arising in focal epithelial hyperplasia probably represent acanthosis and parakeratosis of constituent squamous epithelial layer. Multiple vascular configurations and vascular distension as described upon morphological assessment is indicated by vascular articulations discernible with dermoscopy [9,10].
Therapeutic Options
Circumvention of co-infection with high-risk genotypes of human papilloma virus (HPV) may be managed with prophylactic HPV vaccination. Aforesaid manoeuver may provide crossprotection for related HPV genotypes [10,11]. Contemporary, prophylactic vaccines against low-risk and high-risk HPV genotypes are constituted of a bivalent vaccine against HPV16 and HPV18, a quadri-valent vaccine comprising of HPV6, 11, 16 and 18 and a nine-valent vaccine protective for infection with HPV6, 11, 16, 18, 31, 33, 45, 52 and 59 [10,11]. Antecedent vaccination procedures are recommended as focal epithelial hyperplasia is significantly prevalent in children and young adults and coinfection with high-risk HPV genotypes require circumvention. Following HPV vaccination, prevalence of oral HPV infection is significantly decimated, especially infections induced by pertinent HPV genotypes. However, the contemporary, approved vaccines against HPV infection are protective against prevalent genotypes of focal epithelial hyperplasia such as HPV13 and HPV32. Certain degree of vaccine cross-protection between HPV strains of genomic similarity may be discerned [10,11]. Enlarged lesions or nodules encountered in locations warranting therapy can be subjected to diverse treatment strategies such as 5% topical imiquimoid, topical retinoid, localized cryotherapy with application of liquid nitrogen, cauterization, surgical extermination of the lesion, ablative laser therapy, laser excision or utilization of 80% trichloro-acetic acid and intralesional interferons. Cryotherapy with liquid nitrogen can be suitably adopted to treat focal epithelial hyperplasia which may be subsequently monitored [10,11]. Lesion reoccurrence may be treated with imiquimod which induces disappearance of lesions [10,11].
References
- Waldman GH, Shelton DW (1968) Focal epithelial hyperplasia (Heck’s disease) in an adult Caucasian. Oral Surg Oral Med Oral Pathol26: 124–127.&
- Bendtsen SK, Jakobsen KK, Amanda-Louise Fenger Carlander, Christian Gronhoj, Christian von Buchwald (2021) Focal Epithelial Hyperplasia. Viruses 13(8): 1529.
- Al Ameer A, Kaliyadan F, Yusef M Almarzooq, Maryam Imran, Hassan F Alkhars, et al. (2020) Dermoscopy of Focal Epithelial Hyperplasia. Indian J Dermatol 65(5): 429-430.
- Sethi S, Ali A, Xiangqun Ju, Annika Antonsson, Richard Logan, et al. (2021) An update on Heck’s disease-A systematic review. J Public Health2021: fdaa256.
- Bozca B, Ozbudak I, Erkan Alpsoy (2020) A case of Heck’s disease with primary intestinal lymphangiectasia treated with imiquimod. Indian J Dermatol Venereol Leprol 86(6): 724–725.
- Brehm MA, Gordon K, Miahil Firan, Peter Rady , Nnenna Agim, et al. (2016) Case Report of Focal Epithelial Hyperplasia (Heck’s Disease) with Polymerase Chain Reaction Detection of Human Papillomavirus 13. Pediatr Dermatol 33(3): e224–e225.
- Arbelo-Ramos N, Anderson K, Avni Joshi, Carilyn Wieland, Stephen Tyring, et al. (2021) Heck’s disease occurring after Epstein-Barr virus-associated smooth muscle tumors in an immunosuppressed child. Pediatr Dermatol 38(1): 257–259.
- Puriene A, Rimkevicius A, Mindaugas Gaigalas (2011) Focal Epithelial Hyperplasia: Case Report. Stomatologija13(3): 102–106.
- Said AK, Leao JC Stefano Fedele, Stephen R Porter (2013) Focal epithelial hyperplasia—An update. J Oral Pathol Med42(6): 435–442.
- Nguyen JT, Allen CT, Joshua T Dodge, Koenraad Van Doorslaer, Alison A McBride, et al. (2021) HPV32-related Heck’s disease in a chronic graft-versus-host disease patient with long-term successful KTP laser treatment: A rare case report. Clin Case Rep9(5): e04253.
- Jimenez Aguilar SM, Rodriguez DL, Víctor Fernando Muñoz Estrada, Silvestre Guadalupe Cazarez Salazar, Jesus Salvador Velarde Felix, et al. (2019) Great diversity of oncogenic human papillomaviruses is revealed in an outbreak of multifocal epithelial hyperplasia. J Am Acad Dermatol S0190-9622(19): 33318-323.
- Image 1 Courtesy: Dentowsome.com
- Image 2 Courtesy: Indiamart.com
- Image 3 Courtesy: Turkish Journal of Pathology
- Image 4 Courtesy: Wiley online library
- Image 5 Courtesy: Grepmed.com
- Image 6 Courtesy: Twitter.com
- Image 8 Courtesy: Dr Lucian Schiazza






























