Effects of Acute Submaxımal Exercıse On Blood Coagulatıon And Fıbrınolytıc Systems In Male Patıents Wıth Type 2 Dıabetes Mellitus
Selda Kahraman1* and Fatih Demirkan2
1Medicana International İzmir Hospital , Department of Hematology, Turkey
2Dokuz Eylül University Department of Hematology, Turkey
Submission: February 28, 2022; Published: March 09, 2022
*Corresponding Address: Mirza Qaiser Baig, Associate professor, J K Cancer Institute Utter Pradesh Govt, India
How to cite this article: Selda K, Fatih D. Effects of Acute Submaxımal Exercıse On Blood Coagulatıon And Fıbrınolytıc Systems In Male Patıents Wıth Type 2 Dıabetes Mellitus. Canc Therapy & Oncol Int J. 2022; 21(1): 556053. DOI:10.19080/CTOIJ.2022.21.556053
Abstract
Objectıve: Alterations in coagulation and fibrinolytic system are considered among the causes of increasing cardiovascular morbidity and mortality in diabetic patients. We assessed how acute submaximal exercise affects fibrinolytic parameters in type 2 diabetic patients by also discussing limited number of studies in the literature on effects of exercise in diabetic patients.
Methods: Fifteen Type 2 diabetic and 12 healthy volunteer non-smoking male individuals between 30-60 years old and not regularly exercising were included in this study. Treadmill VO2Max test was done in all participants at baseline in order to determine personal anaerobic threshold. The exercise intensity at which anaerobic threshold value occurred was determined. 3-7 days after this test, submaximal exercise on treadmill for 30 minutes at this threshold value was performed. Hemogram, PT, aPTT, INR, d-dimer, fibrinogen, PAI-1 antigen (ag) level and activity, TAFI ag and activity levels were measured before the exercise, immediately after and 1 hour after the exercise.
Results: PAI-1 ag level after submaximal exercise has declined significantly in the control group during exercise (p:0,041), on the other hand increased during rest (p:0,010). In the patient group PAI-1 ag response was blunt and there was statistically insignificant minimal increase (p:0,125) There was no significant change in PAI-activity during exercise in both groups. TAFI antigen level has increased in both groups and was rather prominent in patient group but it was still statistically insignificant. TAFI activity has decreased significantly in the control group (p.0,005) but didn’t change in diabetic group during exercise, increased during the rest period after the exercise (p.0,031). In the control group TAFI activity values have significantly decreased (p.0,005); but increased after the exercise (p.0,388).In the patient group TAFI activity didn’t change by exercise (p.0,910); on the other hand, it increased significantly at rest (p. 0,031) however, compared to baseline the change wasn’t statistically significant (p.0,125).
Discussion: During acute submaximal exercise, alterations in fibrinolytic parameters show a different pattern in diabetic patients living a sedentary life style than healthy control group. In diabetic patients, non-decreasing PAI-1 and TAFI ag levels and activities and inability to recover to normal levels during and after exercise but submaximal exercise was well tolerated in diabetic patients.
Keywords: Coagulation system; Fibrinolytic system; Diabetes mellitus; Submaximal exercise
Introduction
There can be some changes in coagulation and fibrinolytic system varying with the type, duration and intensity of exercise [1,2]. Coagulation time and Activated Partial Thromboplastin Time (aPTT) shortens after exercise and prothrombin time (PT) and thrombin time shortens or stay constant. Mild exercise activates only fibrinolytic system but heavy exercise activates both fibrinolysis and coagulation [3]. Usually it’s assumed that during exercise fibrinolysis is activated as a result of t-PA release from endothelium. 45-60 minutes after heavy exercise t-PA return to normal levels [4]. Earlier research have shown that PAI-1 activity decreases following aerobic or anaerobic exercise [5-7] but in other studies after tiring aerobic exercise protocols there was no significant change in PAI-1 levels; thus it was considered that PAI-1 response to exercise may vary from person to person [7]. Plasma factor 8, fibrinogen levels vary according to exercise intensity and duration and exercise capacity of an individual [8]. D-dimer increased more after a brief maximal exercise following submaximal exercise or after heavy exercise [9-11].
IAT (Individual aneorobic threshold) is a sound indicator of aerobic exercise used in order to check intensity of exercise. It’s the point at which lactate production and elimination are in equilibrium. Over this threshold value lactate production dominates and anaerobic exercise begins. In longer periods of exercise (50-120 min) standardized by IAT (90% IAT) it has been shown that coagulation wasn’t activated but fibrinolysis was activated.
Fibrinolytic system begins with binding of t-PA to fibrin surface. This binding occurs via specific C-terminal lysine residuals causing partial fibrin degradation. TAFI (Thrombinactivatable fibrinolysis inhibitor) is released to plasma by liver [12]. TAFI is activated by thrombin and this is the evidence that coagulation system has control over fibrinolytic system [13,14]. In theory, when TAFI release increases intrinsic coagulation system is activated and thus incidence of thrombotic disease increases [15,16]. Increasing myocardial infarction incidence following uncontrolled heavy exercise directed attention of researchers to this issue. The subsequent studies have shown that after maximal exercise at aerobic conditions coagulation system and platelets were activated but fibrinolytic system didn’t change or minimally increased, however after performing exercise under aerobic conditions at standardized exercise intensity fibrinolytic system increased markedly and this increase persisted after the exercise [2].
Diabetes Mellitus is an important risk factor for coronary artery disease. We have assessed how acute submaximal exercise affects fibrinolysis parameters in type 2 diabetic patients by also discussing limited number of studies in the literature about effects of exercise on diabetic patients.
Materials and Method
Volunteers
Fifteen Type 2 diabetic and 12 healthy volunteer non-smoking male individuals between 30-60 years old and not regularly exercising were included in this study. BMI were <30 kg/m2, HbA1C levels were <%8, thyroid function tests, liver and renal function tests were in normal range. All of the patients were taking same type oral anti-diabetic medications. Medical history of patients revealed no diabetic complication requiring treatment. All of the patients were taking acetyl salicylic acid 100 mg and atorvastatin 20 mg/day. Hypertensive patients were taking ACE inhibitory medication. All participants didn’t drink alcoholic beverages 24 hours prior to exercise and patients taking beta-blocker weren’t included in the study. The control group wasn’t permitted to take any drugs for the past 6 weeks. In all participants; body mass index and waist-to-hip ratio were calculated, physical examination were done, ECG, exercise ECG and echocardiography were taken. After the cardiac examinations participants without a condition precluding them from exercising were included in the study. Blood samples were obtained immediately and 1 hour after exercise.
Experimental design
The study was done in Dokuz Eylül University Physiology exercise laboratory on January-March 2018. All participants came to laboratory three separate days. In the first day serum biochemistry profile was determined, physical and cardiologic exams were completed. Second and third visits, each visit was 3-7 days apart, all participants were asked to be ready at the laboratory at 10:00 a.m. In the second visit, maximal exercise test was done on a treadmill. In the third visit, 30 minutes of submaximal exercise test with work load based on IAT values determined in maximal exercise test was done. Venous blood samples were obtained from participants before, immediately and 1 hour after submaximal exercise test. The samples were stored at -80 ͦ C for further evaluation of PT-aPTT-INR-D-dimer-fibrinogen- PAI-1 ag and activity, TAFI ag and activity tests.
Metabolic measurements and determination of VO2max
Expiratory air of the participant produced during maximal exercise test collected via one-way valve mask and transferred to a metabolic analyser (Fitmate, Cosmos, Italy). Gas measurements were done by open-ended mixing chamber method and VO2 values were calculated. The measurements at exercise intensity at which maximum load is achieved by the volunteer was recorded as VO2max.
Determination of lactate threshold
During maximal test before each load increase capillary blood sample was taken from fingertip of participants by automated lancets and immediate lactate analysis was performed. Lactate levels were also measured at 0, 1, 3, 5, 10 min after exercise was terminated and measurements have continued until post-exercise lactate levels decreased under a certain value. Lactate analyses were done by ECG Biosen Sport (Germany) device using enzymatic method for measurements. Lactate values were put into a grap via computer and IAT values were calculated by the method reported by Stegmann [17]. Statistical evaluation: All data were recorded to SPSS 15. The evaluations between different times (basal versus postexercise 0th, basal versus postexercise 60th, postexercise 0th versus 60th) between each other have been done non-parametric Mann-Whitney U test, whereas the correlation analysis has been done Pearson correlation analysis. P values that were less than .05 have been considered as statistically significant.
Measurement of coagulation and fibrinolytic parameters
In the day of submaximal exercise when the participants were at rest before the exercise, immediately and 60 min after the exercise 8 cc blood was taken to citrate tubes. Centrifuged blood was separated into 0.5 cc aliquots and were stored at -80 ͦ C until analysis time. PAI-1 ag and activity, TAFI ag and activity levels were measured using available ELISA kits (PAI-1 ag and activity: American Diagnostica, Stamford, Conn., USA, and TAFI-Ag and activity : Affinity Biologicals, Ancaster, Ont., Canada). For PAI-1 ag the intra-assay coefficient of variation is 4% and the interassay coefficient of variation is 4.3%. The intra-assay and interassay coefficients of variation for TAFI-Ag are 6%.
Results
Age, height, weight, body fat percentage, body mass index (BMI), waist-to-hip ratio, exercise total time on Bruce test, VO2 max., lactate threshold value, maximal lactate, maximal heart rate and heart rate at submaximal exercise of the patient and control group were shown in Table 1. As expected, total time on Bruce test, (p.0,001), VO2 max.(p.0,006), maximal lactate (p.0,019) and maximal heart rate (p.0,032) were observed to be statistically higher in the control group (Table 1). Hemogram, PT, aPTT, INRD- dimer, fibrinogen, PAI-1 ag and activity, TAFI ag and activity levels were measured by using blood samples obtained from the patients and controls before , immediately after and 1 hour after the exercise (Tables 2-3). Baseline PAI-1 ag was found to be lower in patient group (p.0,003) and PA1-1 activity was significantly lower in controls after maximal exercise(p.002). There was no difference regarding other markers between both groups.
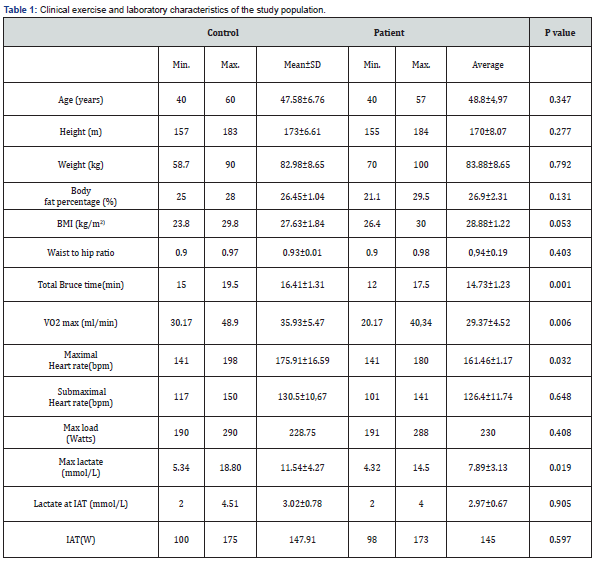
BMI: Body mass index, IAT: Individual anaerobic threshold
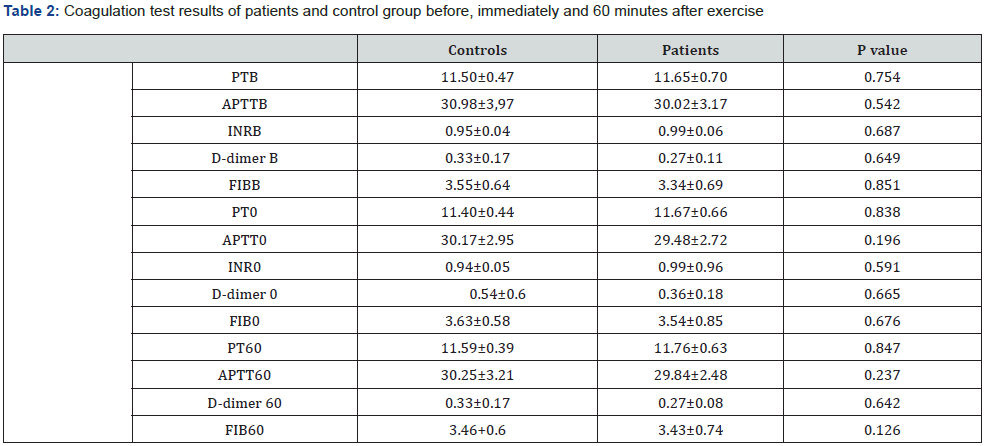
PT-Prothrombin time
APTT-Activated partial thromboplastin time
INR-International normalization ratio
FIB-Fibrinogen
B-basal (Before exercise)
0-immediately after the exercise,
60-60 min after the exercise
Normal values range: PT: 11.23-14.44 seconds; APTT: 25.9-36.6 seconds; D-dimer: 0-0.5 mg/L, FIB: 1.75-4 g/L
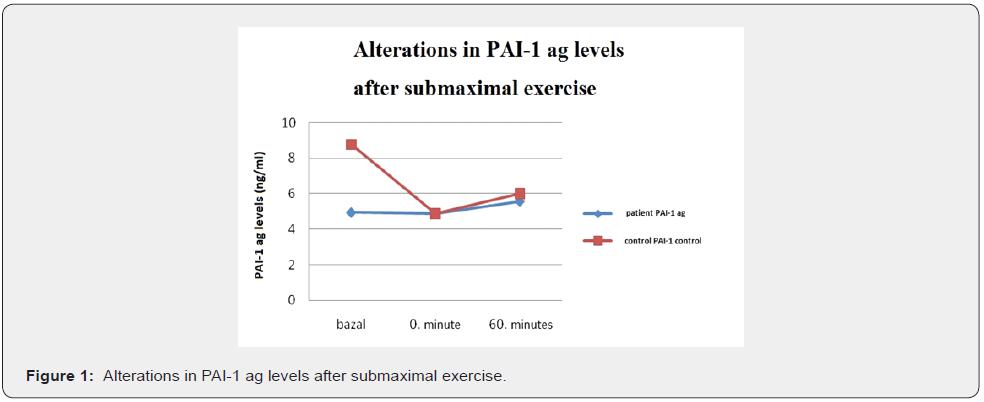
When alterations observed in coagulation and fibrinolytic systems in both groups during and after the exercise test are shown in a graphics; It was observed that in the control group PAI- 1 ag markedly decreased during exercise (p.0,041) but increased at rest (p.0,003); however the reduction persisted compared to baseline. In the patient group PAI 1 ag response was blunt and during and after the exercise test minimal increase which was statistically insignificant was observed (Figure 1). In the control group, during the exercise PAI-1 activity minimally increased (p.0,583), but decreased after the exercise (p.0,388) and returned to baseline level. (p.0,638) In the patient group, during the exercise PAI-1 activity minimally decreased (p.0,776) but increased after the exercise (p. 0,112), however the increase wasn’t statistically significant.(p.0.233) (Figure 2) After submaximal exercise in the control group TAFI ag levels minimally increased during the exercise test (p.0,875) and the increase persisted up to 60 minutes (p.0,583); however the increase wasn’t statistically significant compared to baseline (p.0,814). In the patient group TAFI ag has increased by exercise (p.0,256) and decreased minimally at rest (p. 0,865); however, the increase wasn’t statistically significant compared to baseline. (p.0,363) (Figure 3).
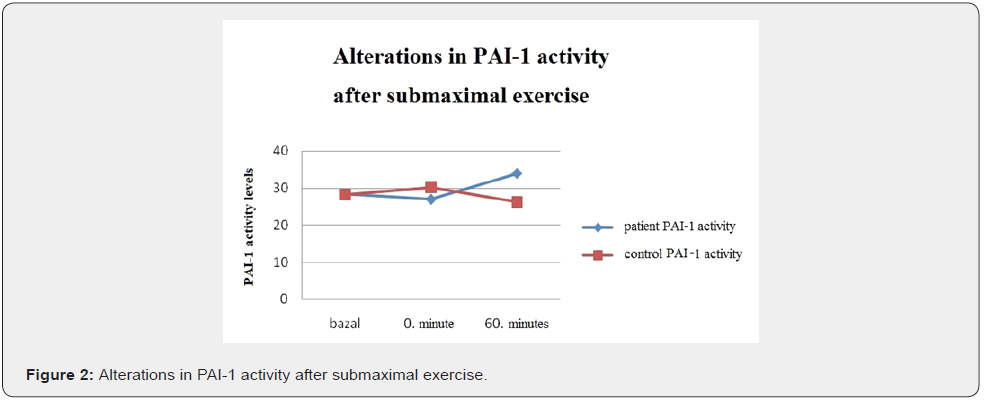
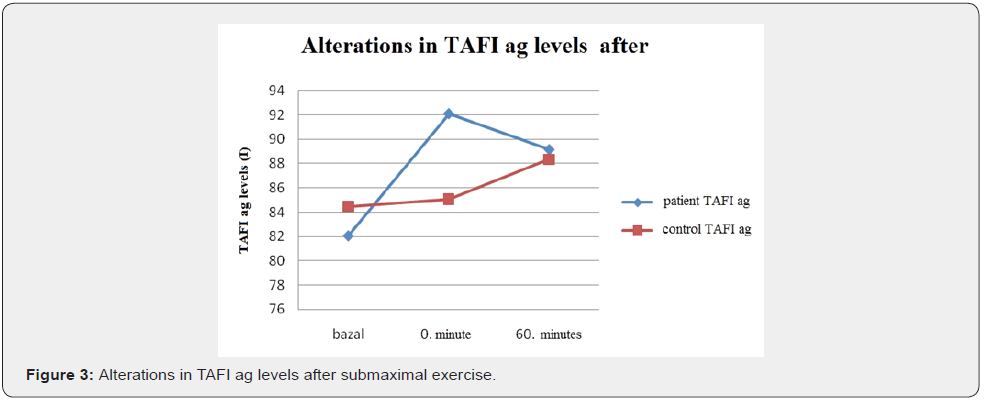
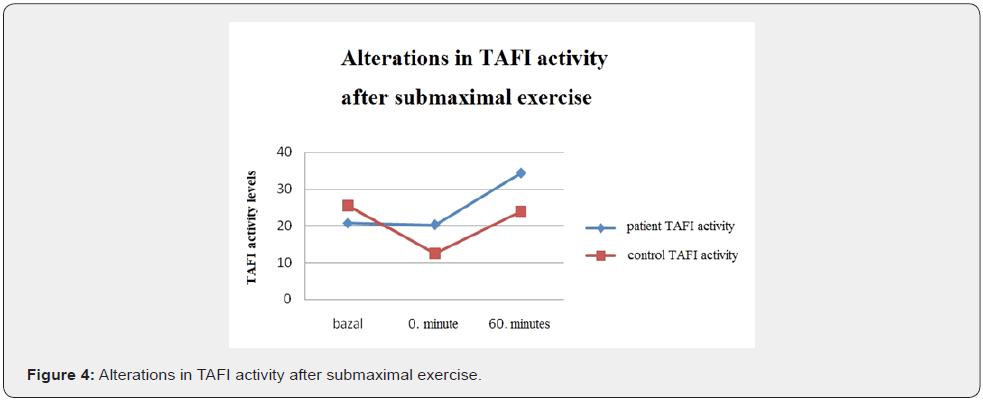
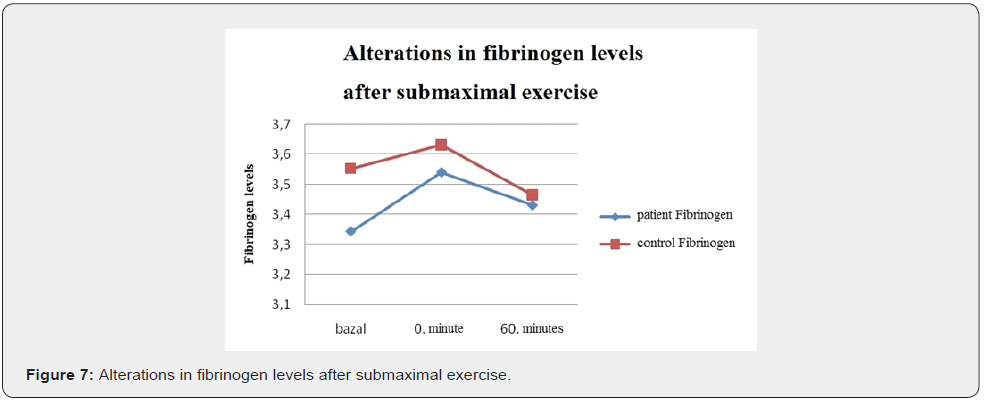
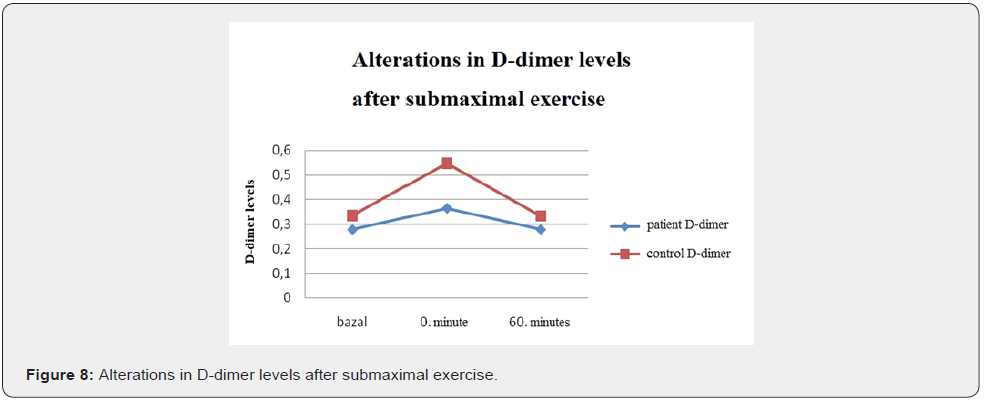
In the control group during submaximal exercise test TAFI activity values have significantly decreased (p.0,005) ; but increased after the exercise (p.0,388). Compared to baseline the alteration in TAFI activity values wasn’t statistically significant. (p. 0,937) In the patient group TAFI activity didn’t change by exercise (p.0,910); on the other hand, it increased significantly at rest (p. 0,031) however, compared to baseline the change wasn’t statistically significant (p.0,125) (Figure 4). During submaximal exercise fibrinogen levels have increased in the control group (p.0,05). It decreased at rest significantly (p.0,010); compared to baseline this decrease was statistically significant (p.0,037). In the patient group fibrinogen levels have increased significantly during exercise (p.0,022) but decreased at rest (p. 0,012); however, compared to baseline this alteration wasn’t statistically significant(p.0,572) (Figure 5). During submaximal exercise D-dimer levels have increased in the control group (p.0,239) and at rest returned to baseline levels within an hour (p.0,410); compared to baseline this alteration wasn’t statistically significant (p.0,480). In diabetics D-dimer levels have increased during exercise (p.0,007)) but returned to baseline levels at rest(p.004). (Figure 6).
Discussion
Hypofibrinolysis and hypercoagulation are important findings causing macro and micro-vascular complications in diabetic patients [18-20]. PAI-1 ag is one of the most important parameters inhibiting fibrinolytic system. TAFI can be activated by thrombin and has been isolated from the plasma recently [21]. Higher plasma TAFI Ag levels and increased TAFI activity are considered as a marker of hypofibrinolysis. [22].
Data obtained from large cohorts have shown that higher levels of haemostatic factors are associated with presence and progression of atherosclerosis [23]. Association of decreased fibrinolytic activity with atherosclerosis has been shown in various studies [24]. Hamsten et al. have determined high levels of plasma PAI-1 and low t-PA activity in young survivors of myocardial infarction [25]. The same group has shown in another study that high plasma PAI-1 ag level is an independent risk factor for reinfarction [26]. Similarly, Thögersen et al. have shown that high plasma PAI-1ag level is associated with increase in myocardial infarction risk [27].
Previous studies have suggested that plasma TAFI level may be associated with atherosclerosis. Genetic polymorphism is also seen as a probable determinant of plasma TAFI levels [28]. Studies investigating association between TAFI levels and glucose intolerance are limited in the literature. Some researchers have suggested robust association between TAFI ag level and glucose intolerance; on the other hand some others have concluded that there was no significant association between TAFI ag level and glucose intolerance [29]. However, in these studies only TAFI ag levels were investigated. Because of TAFI gene polymorphism, TAFI activity levels in individuals may vary. Thus, along with TAFI ag level TAFI activity should also be determined.
In our study we studied how acute submaximal exercise pattern and intensity affect fibrinolysis in type 2 diabetic patients. Fibrinogen level increased in both groups by exercise but decreased to baseline level during rest. Fibrinogen is an important protein increasing aggregation and having a role in the final step of coagulation factor [30]. There are conflicting results on fibrinogen levels following acute exercise [31]. Bartsch et al. have found that fibrinogen levels of 19 athletes who ran decreased and hyperfibrinogenolysis was suggested as a reason for this. However, considering the fact that fibrinogen concentration is affected from plasma dilution, fluid intake of participants should be standardized [32].
D-dimer levels increased in the control group after the exercise and decreased to baseline level in one hour with rest; however, there was no significant change. In diabetic patients d-dimer levels increased significantly during the exercise (p.0,007), but returned to baseline after rest. (p.004) Hilberg et al, have studied acute maximal exercise in diabetic patients and have found that D-dimer levels increased significantly with exercise [33]. This increase may be a marker of fibrinolytic activity during exercise in diabetic patients.
PAI-1 ag level as a fibrinolytic system parameter was found to be lower at rest in diabetic group (table 3-p.0,003); however, in other parameters including PA1-1 activity, TAFI ag and activity no significant difference was observed. Search on studies in the literature on diabetic patient cohorts have revealed that PAI-1 ag and TAFI ag is higher in patients with uncontrolled blood glucose levels, high BMI and truncal obesity [28,29]; however PAI-1 ag was lower than control group in studies done in non-obese diabetic patient group with well controlled metabolic status and without macro- or micro-vascular complication. [33].
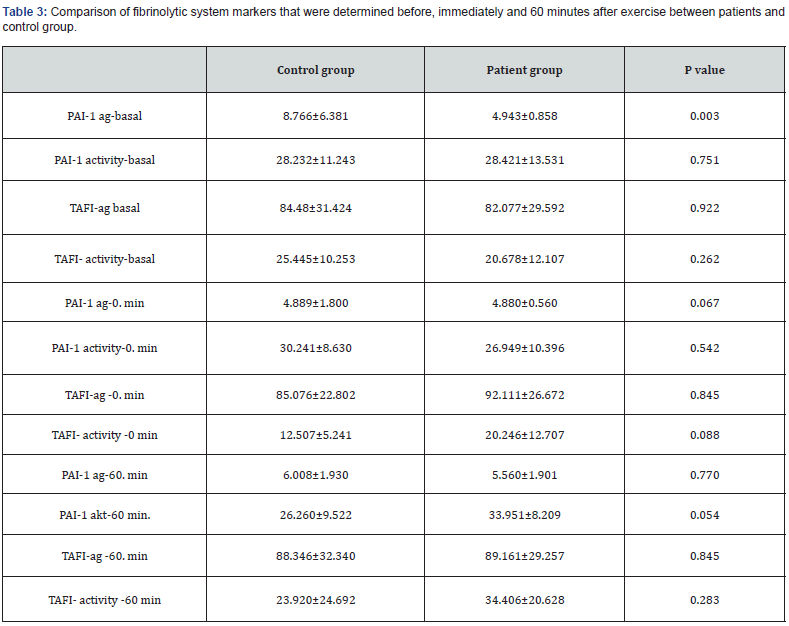
B-basal,
0-immediately after the exercise
60-60 min after the exercise
PAI-1-ag-Plasminogen activator inhibitor-1 antigen
PAI-1-activity- Plasminogen activator inhibitor-1 -activity
TAFI-ag-Thrombin activatable fibrinolysis inhibitor-antigen
TAFI-activity-Thrombin activatable fibrinolysis inhibitor-activity
Our patient group with well-controlled metabolic status, without diabetic complication and with a BMI <30 kg/m2 may explain the lower baseline PAI -1 ag levels observed in the diabetic group. Our diabetic group had a better lipid profile (cholesterol total and LDL) than the control group due to atorvastatin treatment. In the study of Konduracka et al. [34] it was showed that atorvastatin treatment reduced PAI-I ag levels in diabetic patients.
In our study; the control group PAI-1 ag decreased during exercise significantly (p.0,041) but increased at rest (p.0,003); but, compared to the baseline value the reduction wasn’t statistically significant. In the patient group baseline PAI-1 ag was lower and response to exercise was blunt, but during and after the exercise statistically insignificant increase was seen. This results show that fibrinolytic system becomes active and this activity continues at rest. There were no significant alterations in fibrinolytic system in the diabetic group. PAI-1 activity levels didn’t significantly change during and after acute submaximal exercise in both groups that has a sedentary life style.
We have observed significant decreases in TAFI activity scores during submaximal exercise in control group (p:0.005). An increase has been detected following the exercise (p.0.388) but we have found no statistically significant correlation compared to the baseline. In the patient group TAFI activity didn’t change with exercise (p.0,910) but significantly increased at rest (p. 0,031); however, change wasn’t significant compared to baseline values. (p.0,125) We may speculate TAFI activity response occurs after acute submaximal exercise at a later time and coagulation becomes dominant by development of hypofibrinolysis after the exercise in patients living a sedentary lifestyle. TAFI ag has a regulatory role between coagulation and fibrinolytic system and is a reliable fibrinolytic system marker with its minimal biological variability.
In conclusion, during acute submaximal exercise, alterations in fibrinolytic parameters show a different pattern in diabetic patients living a sedentary life style than healthy control group .Submaximal exercise was well tolerated in diabetic patients and further studies, with increased number of patients may yield more significant results to evaluate the role of fibrinolysis in this type of exercise. A defect in fibrinolysis may be multifactorial and it may not be explained by a single parameter. Changes in fibrinolytic parameters during various exercise intensities and duration should also be investigated.
References
- Van den Burg P J, J E Hospers, M van Vliet, W L Mosterd, B N Bouma, et al. (1995) Changes in haemostatic factors and activation products after exercise in healthy subjects with different ages. Thromb Haemost 74(6): 1457-1464.
- Weiss C, G Seitel, P Bartsch (1998) Coagulation and fibrinolysis after moderate and very heavy exercise in healthy male subjects. Med Sci Sports Exerc 30(2): 246-251.
- Weiss C, Seitel G, Bartsch P (1998) Cogulation and fibrinolysis after moderate and very heavy exercise in healthy male subjects. Med Sci Sports Exercise 30(2): 246-251.
- Molz AB, Heyduck B, Lill H, Spanuth E, Rocker L (1993) The effect of different exercise intensities on the fibrinolytic system. Eur J Appl Physiology 67(4): 298-302.
- Jootar S, Chaisiripoomkere W, Thaikla O, Kaewborworn M (1992) Effect of running exercise on haematological changes, hematopoietic cells (CFU-GM) and fibrinolytic system in humans. J Med Association Thai 75: 94-98.
- El-Sayed MS, Jones P, Sale C (1999) Exercise induces a change in plasma fibrinogen concentration: fact or fiction? Thromb Res 96(6): 467-472.
- Szymanski LM, Pate RR, Durstine JL (1994) Effects of maximal exercise and venous occlusion on fibrinolytic activity in physically active and inactive men. J Appl Physiology 77(5): 2305-2310.
- De Paz JA, Lasierra J, Villa JG, Vilades E, Martin-Nuno MA, et al. (1992) Changes in the fibrinolytic system associated with physical conditioning. Eur J Appl Physiology 65(5): 388-393.
- Arai M, Yorifuji H, Ikematsu S, Nagasawa H, Fujimaki M, et al. (1990) Influences of strenuous exercise on blood coagulation and fibrinolytic system. Thromb Research 57(3): 465-471.
- Rocker L, Taenzer M, Drygas WK, Lill H, Heyduck B, et al. (1990) Effect of prolonged physical exercise on the fibrinolytic system. Eur J Appl Physiology 60: 478-481.
- Prisco D, Paniccia R, Bandinelli B, Fedi S, Cellai AP, et al. (1998) Evaluation of clotting and fibrinolytic activation after protracted exercise. Thromb Research 89(2): 73-78.
- Eaton DL, Malloy BE, Tsai SP, Henzel W, Drayna D (1991) Isolation, molecular cloning, and partial characterization of a novel carboxypeptidase BJ Biol Chem 266(32): 21833-21838.
- Wang W, Boffa MB, Bajzar L, Walker JB, Nesheim ME (1998) A study of the mechanism of inhibition of fibrinolysis by activated thrombin-activable fibrinolysis inhibitor. J Biol Chem 273(42): 27176-27181.
- Redlitz A, Tan AK, Eaton DL, Plow EF (1995) Plasma carboxypeptidases as regulators of the plasminogen system. J Clin Investigation 96(5): 2534-2538.
- Boffa MB, Wang W, Bajzar L, Nesheim ME (1998) Plasma and recombinant thrombin-activable fibrinolysis inhibitor (TAFI) and activated TAFI compared with respect to glycosylation, thrombin/thrombomodulin-dependent activation, thermal stability, and enzymatic properties. J Biol Chem 273(4): 2127-2135.
- Meijers JC, Tekelenburg WL, Bouma BN, Bertina RM, Rosendaal FR (2000) High levels of coagulation factor XI as a risk factor for venous thrombosis. N Engl J Medicine 342(10): 696-701.
- Stegmann H, Kindermann W, Schnabel A (1981) Lactate kinetics and individual anaerobic threshold. Int J Sports Medicine 2(3): 160-165.
- Fuller JH, Keen H, Jarrett RJ, Omer T, Meade TW, et al. (1979) Haemostatic variables associated with diabetes and its complications. Br Med J 2(6196): 964–966.
- Christe M, Fritschi J, Lammle B, Tran TH, Marbet GA, et al. (1984) Fifteen diabetes mellitus and in patients with vasculopathy. Thromb Haemost 52: 138–143.
- Kannel WB, D’Agostino RB, Wilson PWF, Belanger AJ, Gagnon DR (1990) Diabetes, fibrinogen and risk of cardiovascular disease: the Framingham experience. Am Heart J 120(3): 672–676.
- Bajzar L, Manuel R, Nesheim ME (1995) Purification and characterization of TAFI, a thrombin-activable fibrinolysis inhibitor. J Biol Chem 270(24): 14477-14484.
- Eichinger S, Schonauer V, Weltermann A, Minar E, Bialonczyk C, et al. (2004) Thrombin-activatable fibrinolysis inhibitor and the risk for recurrent venous thromboembolism. Blood 103(10): 3773-3776.
- Haverkate F (2002) Levels of haemostatic factors, arteriosclerosis and cardiovascular disease. Vascul Pharmacol 39(3): 109-112.
- Meade TW, Ruddock V, Stirling Y, Chakrabarti R, Miller G (1993) Fibrinolytic activity, clotting factors, and long-term incidence of ischaemic heart disease in the Northwick Park Heart Study. Lancet 342(8879): 1076-1079.
- Hamsten A, Wiman B, de Faire U, Blomback M (1985) Increased plasma levels of rapid inhibitor of tissue plasminogen activator in young survivors of myocardial infarction. N Engl J Medicine 313(25): 1557-1563.
- Hamsten A, de Faire U, Walldius G, Dahlen G, Szamosi A, et al. (1987) Plasminogen activator inhibitor in plasma: risk factor for recurrent myocardial infarction. Lancet 2: 3-9.
- Thogersen AM, Jansson JH, Boman K, Nilsson TK, Weinehall L, et al. (1998) High plasminogen activator inhibitor and tissue plasminogen activator levels in plasma precede a first acute myocardial infarction in both men and women: evidence for the fibrinolytic system as an independent primary risk factor. Circulation 98(21): 2241-2247.
- Morange PE, Juhan-Vague I, Scarabin PY, Alessi MC, Luc G, et al. (2003) Association between TAFI antigen and Ala147Thr polymorphism of the TAFI gene and the angina pectoris incidence. The PRIME Study (Prospective Epidemiological Study of MI). Thromb Haemost 89(3): 554-560.
- Akinci B, Celtik A, Yener S, Genc S, Tunali S, et al. (2011) Plasma TAFI levels are not associated with glucose intolerance and subclinical atherosclerosis in women with previous gestational diabetes. Clin Appl Thromb Hemost 17(6): E224-E230.
- Ernst E, Koenig W (1997) Fibrinogen and cardiovascular risk. Vasc Medicine 2(3): 115-125.
- El Sayed M (1996) Fibrinogen levels and exercise: is there a relationship. Sports Med 21(6): 402-408.
- Bartsch P, Haeberli A, Straub PW (1990) Blood coagulation after long distance running. Antithrombin III prevents fibrin formation. Thromb. Haemostasis 63(3): 430-434.
- Hilberg T, Eichler E, Glaser D, Prasa D, Sturzebecher J, et al. (2003) Blood coagulation and fibrinolysis before and after exhaustive exercise in patients with IDDM. Thromb Haemostasis 90(6): 1065-1073.
- Konduracka E, Galicka-Latala D, Cieslik G, Rostoff P, Fedak D, et al. (2008) Effect of atorvastatin on endothelial function and inflammation in long-duration type 1 diabetic patients without coronary heart disease and arterial hypertension. Diabetes Obes Metab 10(9): 719-725.






























