A Compendium of Ligands Reported to Bind Alpha-Fetoprotein: A Comprehensive Review and Meta-Analysis
Gerald J Mizejewski*
New York State Department of Health, Wadsworth Center, New York
Submission: January 20, 2022; Published: January 28, 2022
*Corresponding Address: GJ Mizejewski, Division of Translational Medicine, Molecular Diagnostics Laboratory, Wadsworth Center, New York State Department of Health, PO Box 509, Empire State Plaza, Albany, NY 12201-0509, New York
How to cite this article: Gerald J Mizejewski. A Compendium of Ligands Reported to Bind Alpha-Fetoprotein: A Comprehensive Review and Meta- Analysis. Canc Therapy & Oncol Int J. 2022; 20(5): 556047. DOI:10.19080/CTOIJ.2022.20.556047
Abstract
The serum carrier/ transport function of mammalian alpha-fetoproteins (AFP) has long been known in the biomedical literature. Many published reports of AFP biological activities have noted in their introductory remarks that AFP is capable of binding, transporting, and delivering cargo ligands to many cells and tissues of the body. AFP, similar to albumin, can bind a multitude of ligands including fatty acids, steroids, drugs, and heavy metals. However, in lieu of the vast array of diverse ligands transported by AFP, the biomedical literature is devoid of a complete reference source detailing a listing, enumeration, classification, and description of all known ligands bound and transported by AFP. Unknowingly, there exists an umet need for a published review that has compiled all known ligands that bind AFP, together with their binding affinities and number of binding sites. The present review represents an attempt to provide a single reference source which could serve as a compendium of all known ligands that bind to mammalian AFP. Surprisingly, these ligands were found to number nearly 50 different biochemical compounds. The present comprehensive survey was designed to include ligands such as; non-esterified poly-unsaturated fatty acids, estrogens, dyes, toxins, flavonoids, phytoestrogens, commercial drugs, synthetic estrogens, heavy metals, retinoids, thyroid hormones, and plant/insect lectin agglutinins.
Keywords: Alpha-fetoprotein; Ligands; Fatty acids; Estrogens; Heavy metals; Lectins; Dyes; Flavonoids
Introduction
Alpha-fetoprotein (AFP) is a tumor-associated fetal glycoprotein (i.e., oncofetal protein) that serves as a serum ligand carrier transporter that also functions in the regulation of both ontogenic and oncogenic growth. The oncofetal protein is composed of 609 amino acids including a 19 amino acid NH2- terminal leader sequence segment [1]. AFP is a 69 kD single chain glycoprotein with 4.0% carbohydrate residues linked via N-glycan-associated amino acids. Mammalian molecular AFP has been reported to be a V-shaped three-domain cysteine-rich translated protein belonging to the albuminoid gene family consisting of 5 members: these include albumin (ALB), alpha-albumin, AFP, Vitamin-D binding protein, and the ARG-protein [2]. During fetal development, the single chain polypeptide is first synthesized in the fetal yolk sac and then in fetal liver and gastrointestinal tract during the course of mammalian pregnancy. In juvenile and adult life, AFP is synthesized and expressed in hepatoblastomas, hemangiomas, hepatomas, and germ cell tumors. It is the dual characteristic of its presence in the embryo/fetal compartment and in adult malignant tumors which has enabled AFP to serve as an oncofetal serum biomarker for pregnant, pediatric, and adult clinical patients [3,4]. AFP was the first protein to be employed as a clinical tumor marker and has remained the “gold standard marker for liver cancer since the late 1960s.
Objectives
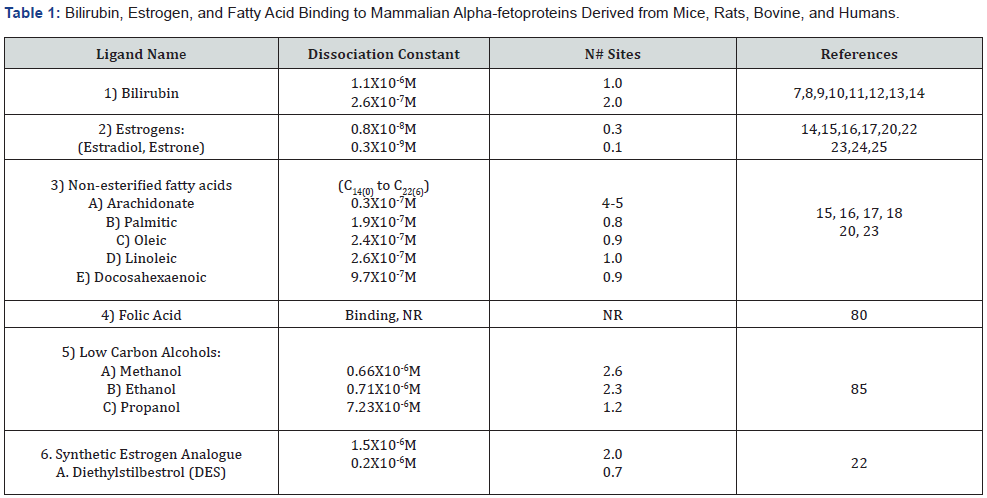
The purposes and objectives in the present review and meta-analysis are three-fold in purpose. First, this review was intended to provide an all-inclusive compendium of the multiple and varied ligands capable of binding to mammalian AFPs. However, present day biomedical literature is devoid of a single comprehensive survey reference source detailing all presently known ligands that bind AFP as a carrier transport protein. Such ligands include the following: 1) bilirubin; 2) fatty acids; 3) estrogenic compounds including flavonoids and synthetic steroids; 4) dyes; 5) toxins; 6) retinoids; 7) thyroid hormones; 8) commercial drugs; 9) heavy metals; and 10) plant and insect lectin agglutinins. Although not enumerated and described in the many literature reports, the AFPbound ligands total nearly 50 different bio-compounds. Secondly, the binding association/dissociation constants and binding site numbers on the AFP polypeptide, if reported, have presently been listed and enumerated in five text (Tables 1). Thirdly, many of the human methodologies and assays employed to identify, detect, and assay the ligands bound to AFP are discussed when such information was available. In summary, the surveyed information on ligand-binding to AFP should provide a valuable resource to the scientific community at large in attempting to more clearly elucidate the many biological activities of mammalian AFP.
Alpha-fetoprotein as a Carrier/Transport Protein
A major biological activity of AFP involves the functions of binding and carrier/transport of a multitude of ligands including both hydrophilic and hydrophobic compounds. In fact, during fetal development, AFP serves to function as a ligand shuttle protein being capable of entering a cell, delivering and dropping off cargo, with the AFP exiting back into the extracellular compartment [5]. In this fashion, AFP has been found to bind and transport a multitude of ligands including various drugs, dyes, steroids, hormones, fatty acids, and many others. An early study [6] determined that AFP bound ligands comparable to the Albumin’s site-I region (warfarin, phenyl-butazone) and albumin’s site II region (tryptophan), but not albumin’s site III region (cholic acid, digoxin). This prior study further reported that AFP was capable of binding low-carbon alcohols (methanol, ethanol, and propanol) depending on the ligands’ hydrophobicity. In addition to the ligands discussed above, AFP was also demonstrated to bind streptomycin, phenytoin, congo red dye, and 8-anilinonapthalenesulfonic acid (ANS). Conversely, AFP was not capable of binding to acetyazolamide, tetracycline, and amethopterin (see drug binding section below).
Both rodent (mouse/rat) and human AFP have been reported to bind a variety of ligands both hydrophilic and hydrophobic in nature. In the case of one organic compound, AFP has been demonstrated to bind bilirubin as does its albumin counterpart. The ALB binding site for bilirubin was found to be similar to that of AFP since both proteins are low affinity, high-capacity blood plasma transport/carrier proteins.
Bilirubin Binding to AFP
Bilirubin binding to rat AFP (RAFP) was first reported by Versee and Barel in 1978 [7] and later confirmed in a study by Berde et al the following year [8]. The latter study demonstrated that Bilirubin exhibited both a strong binding site and a weaker one on RAFP (See (Table 1) for binding dissociation constants). It was the team of Ruoslathti et al., [9] that further established the binding of bilirubin to human AFP (HAFP) at a single site in contrast to 2 sites on RAFP. However, a later study was able to elucidate that HAFP was indeed capable of binding bilirubin at one dominant site but also with a 2nd minor site of lower affinity [10]. In 1979, Aoyagi et al., [11] employed fetal-derived versus hepatoma-derived HAFP to demonstrate that both derived sources of AFP molecules bound bilirubin at a major site and a lesser minor site [12]. Subsequent studies on the competitive binding of bilirubin can be found in References [13] and [14]. A further study reported that neither the estrogen/fatty acid, nor the retinoid binding sites on RAFP were in competition with the major bilirubin binding site (15].
Fatty Acids in Competition with Estrogen Binding to AFP
It was discovered early in the 1970s that the binding of estrogens (estradiol, estrone) to rodent AFP showed the presence of a competitive inhibitor of estrogen which was not bilirubin [15,16]. The competitive inhibitor was further characterized as a non-polar, non-phenolic, dialysable, and thermostable substance. The serum competitor of the estrogen binding to RAFP was subsequently identified as a class of non-esterified long chain polyunsaturated fatty acids (PUFAs) from both fetal and pregnant rat sera and amniotic fluid [17] (Table 1). Such PUFAs included arachidonic, palmitic, oleic, linoleic, and docosahexaenoic acid, which were fatty acids ranging from carbon chains- C14 to C22 [17]. It is noteworthy that some of these fatty acid inhibitors bound to AFP even after intense purification and that the binding dissociation constants remained relatively high (10-7M). Finally, it is of interest that both human and bovine AFPs also displayed the similar PUFA inhibitory ligands as did the rodent AFPs [10].
The estrogen binding sites on rodent AFP were shown to display high binding dissociation constants equal or greater than those of the PUFAs [14]. While docosahexaenoic acid showed the highest affinity constant for AFP among the PUFAs (9.7 X 10-8 M ‘compared to 10-7M) for other fatty acids, the estrogens’ affinity constants were 1 X 10-8M or more (estradiol and Estrone) [18]. It is of interest that rodent ALB does not bind estrogens whatsoever [19]. Although Arachidonic (C20:4) and docosahexaenoic (C22:4- 6) acids are weaker binding ligands for mouse AFP (0.3 X 10-7M), they serve as efficient concentration-dependent inhibitors of estrogen binding to mouse AFP [20]. Interestingly, estradiol has been characterized as a high affinity, low-capacity ligand (0.8 X 10-8M) with 0.3 sites sites/mole of rodent AFP. None of the studied actions with rodent AFP were modified by bilirubin competitive binding even at high doses. It is important to note that full-length Human AFP does not bind estrogens (estradiol and estrone) in contrast to rodent AFP .(see concluding statements).
Diethylstilbesterol (DES) is a synthetic estrogen analogue previously used in human reproduction clinics [21]. DES was found to bind Human AFP, but with a weaker affinity than rodent AFP [22]. As discussed above, rodent and human AFP binding to estrogens are inhibited by endogenous hydrophobic compounds such as arachidonic and docosahexanoic acid and as such, it is also true for DES. The DES synthetic estrogen analogue binds to rodent AFP and display both low affinity and low capacity sites (0.2 X 10-7M and N = 0.7/mol of AFP), [20]. Arachidonic acid readily displaces both E2 and DES on rodent AFP due to the fact that these three components are able to compete with one other for binding to Rodent AFP. Estrogenic hormones , which normally compete with fatty acids for the high affinity binding site on rat AFP, were found unable to displace arachidonic acid bound to the human AFP at a single high affinity site and several low affinity sites [23]. Although human AFP does bind DES, rodent AFPs are also able to bind the synthetic estrogen with both low affinity and a high capacity (Ka = 1.5 X 10-6M at 2 sites/mol of RAFP) [19]. These data are noteworthy in that human AFP does indeed bind DES and had been used to treat women with a history of reduced fertility and miscarriages between 1940 to 1971 [21]. Furthermore, DES had also caused genital abnormalities and abnormal semen levels in males exposed to DES in clinical studies.
Binding of Alpha-fetoprotein to Bioflavonoids and Phytoestrogens
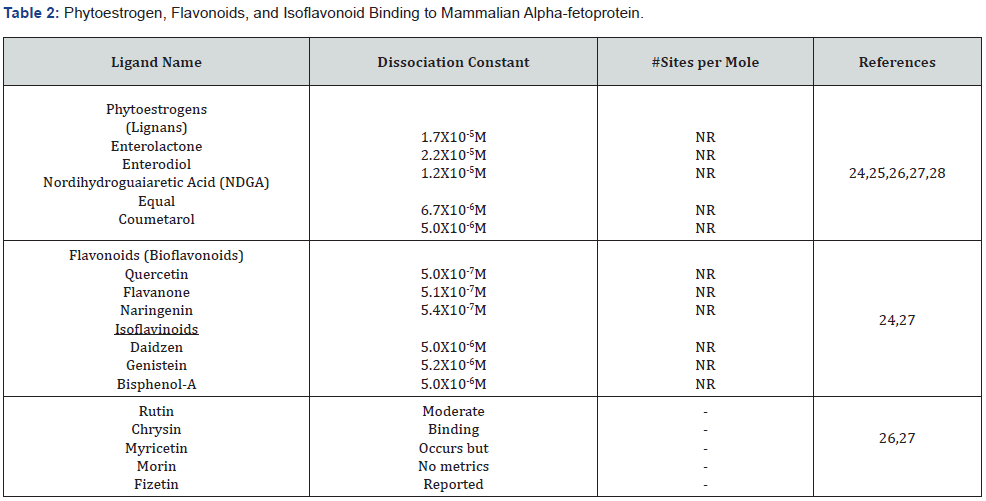
Bioflavonoids and phytoestrogen (environmental estrogens) are a group of natural plant, tea, and wine compounds with variable polyphenolic structures which are also found in fruits, vegetables, grains, and other plant substances. Flavonoids have a 15- carbon skeleton consisting of two phenyl rings and a heterocyclic ring (Table 2-4). Bioflavonoids are capable of influencing hormone action in mammals [24] and can affect growth of steroid-dependent breast and prostate tumors in addition to inducing abnormal development of steroid-sensitive tissues. Furthermore, bioflavonoids and isoflavinoids (at dietary levels) can inhibit estrogen binding to rodent AFP [25]. Phytoestrogenlike lignames, such as enterolactone, enterodiol, and equal also demonstrate inhibitory action on the binding of both estrogens and fatty acids to rodent and human AFP [26], (Table 4). As a further action, some phytoestrogen binding to AFP can induce a conformational change in the AFP molecule. Although RAFP binds estrogen at dissociation constants of 10-8M to 10-9M, flavonoids and phytoestrogens are known to display Kds of 10-6M to 10- 7M, as seen with both Daidzein and Genistein [24,27]. Finally, it is of interest that some flavonoids such as “Quercetin” has been reported to display anti-proliferative activities in cell culture cancer studies [28].
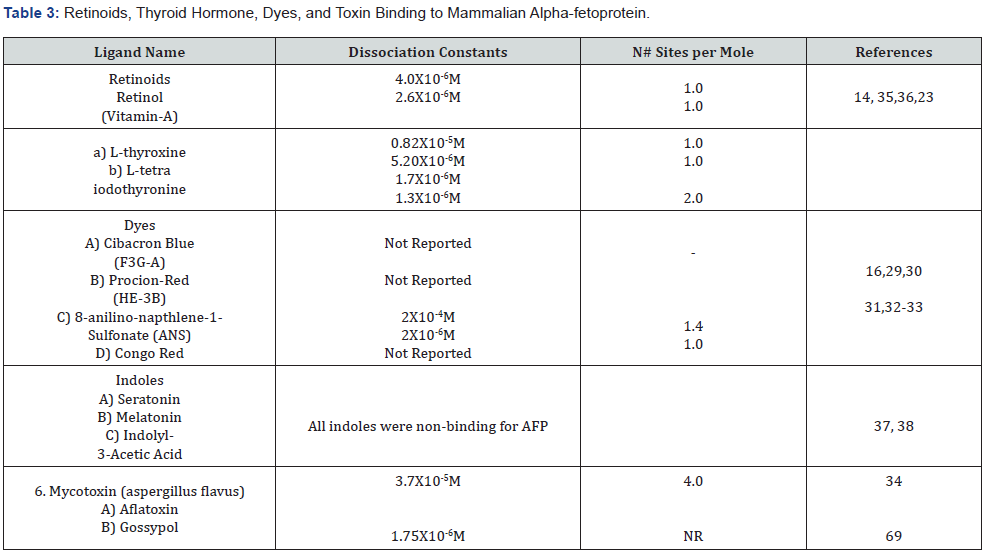
Dye and toxin Binding to Alpha-fetoprotein
It is of interest that various dye and toxin binding to AFP have been reported in the literature as early as 1978. Displacement experiments of bound radiolabeled estradiol with various dye compounds were used to investigate the specificity of the estrogen-binding site on rodent AFP. The fluorescent dye 8- anilinonaphthalene -1- sulfonate (ANS) was found capable of displacing estradiol binding to AFP at physiological concentrations [16]. In comparison, an early study by Cittanova et al., [29] reported that ANS displayed an association constant of 2 X 10-6 for dye binding to AFP. Further studies of AFP dye binding have utilized affinity chromatography columns utilizing triazine dyes such as Cibacron Blue F3G-A, congo red, and Procion-red HE-3B that bind human AFP [30-33]. Lastly, in a non-related separate study it was demonstrated that rat AFP was capable of binding aflatoxin, B1, a non-dye carcinogenic mycotoxin from the plant fungus Aspergillus flavus [34]. This fungal toxin is known to cause acute liver damage.
Binding of Retinoids, Indoles, and Thyroid Hormones to Alpha-fetoproteins.
Studies have demonstrated that rat AFP displays binding sites for three different and distinct classes of compounds that include: (1) retinoids including retinol, vitamin-A, and thyroid hormones; (2) fatty acids (Arachidonic acid) and estrogens (estradiol, estrone); and (3) indoles and tryptophan methyl esters [14]. The retinoids displayed a disassociation constant of 4X 10- 6M with one binding site on HAFP, while thyroid hormone binding affinity occurred at 0.8X10-5M for thyroxine and 1.7 X 10-4 M for triiodothyronine [35]. In comparison, estradiol displays one high affinity binding site on RAFP (1X10-8M), while Arachidonic acid displayed one high affinity site (2 X 10-7M) and multiple low affinity sites at 1.5 X 10-6M (14 [36]. It is noteworthy that the rodent AFP binding sites described above are quite distinct from the previously reported bilirubin binding site. It was also clearly shown that, in contrast to rat Albumin, neither rodent nor human AFP was capable of binding indoles such as serotonin and melatonin [36]. Finally, it was reported by Aussel & Myasseff [13,23] that the individual binding site locations of the three ligand, (i.e.), a) retinoids, b) fatty acids and estrogens, and 3) metals and tryptophan esters were located on and reflected the three domain molecular structure of full- length AFP.
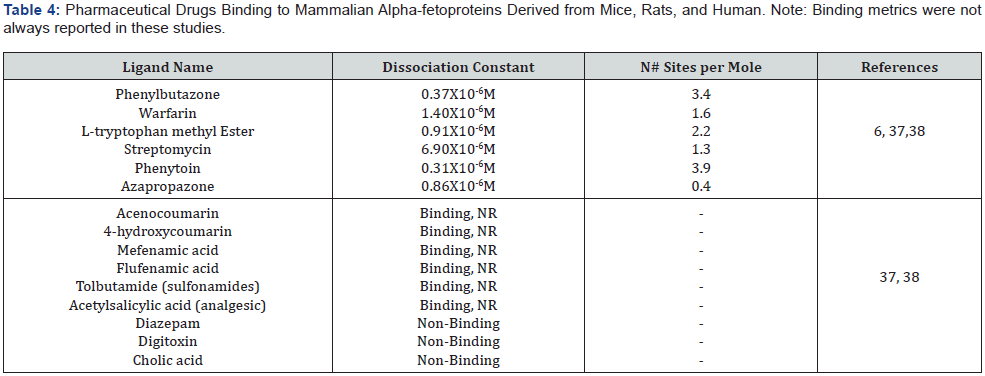
Drug Binding Properties of Alpha-fetoprotein
The drug binding properties of human and rodent AFP were investigated by standard photometric and fluorescence- spectral methods. As noted above, human AFP bound pharmacologic drugs at two of the three major sites comparable to those of human ALB. HAFP bound warfarin and phenlbutazone at ALB site-I and tryptophan at ALB site-II but did not bind cholic acid and digoxin at the comparable ALB site-III [6]. Moreover, cord serum-derived HAFP was further capable of binding Congo Red and ANS dyes, in addition to the low-carbon alcohols denoted above (Table 1). Rodent AFP is also capable of binding warfarin and phenylbutazone at two additional sites [37]. The warfarin site is also specific for the binding of coumarin and anthranilic drugs, whereas the phenylbutazone site binds more hydrophobic compounds such as estrogens and fatty acids; however, bilirubin is bound at a different distinct site [37,38]. In contrast to HAFP, rodent AFP lacks the ALB site-II as well as the ALB site III. Thus, RAFP was found to bind warfarin and phenylbutazone, in addition to azapropazone, at a site comparable to the ALB site-I exhibiting 0.5 sites per AFP molecule. Other drugs displacing the rodent AFP at the ALB site-I were sodium valproate, cloxacillin, Furosemide, and others [37,38] (Table 4).
Binding of Heavy Metals to Alpha-fetoprotein
The binding of heavy metals to AFP has been reported in the literature for the last 4 decades. An early study by Toyagi in 1978 [39] demonstrated that umbilical cord-derived HAFP could bind 1.0 mol of copper (III) ion per mol of protein at a pH range of 5.4 to 6.0. This range approximates the pK value of the imidazole group of histidine. Human AFP derived from amniotic fluid was further shown to bind zinc [40]. Subsequent purification of rat AFP was shown to be accomplished by means of a copper chelate column [41]. In a later study the total number of zinc binding regions on human AFP was demonstrated to occur at 16 different sites in which 4-5 of those sites were of moderately high affinity with Kds being 6.8 X 10-6M [42]. These latter authors showed that the zinc binding sites on HAFP were also capable of binding calcium, copper, and lead, while Anderson et al., [43] showed that HAFP could be purified on nickel as well as copper (but not cobalt) affinity columns. It is germane to this discussion that increased levels of zinc in the amniotic fluid of fetuses with neural tube defects approximated the high fluid levels found for AFP [40,44]. Finally, increased levels of serum AFP, correlating with copper and zinc levels have also been employed to serve as metallic barometers in hepatocellular carcinoma- bearing patients [45,46].
Lectin Binding to Alpha-fetoprotein
Both rodent and human AFP have been found to differ individually in their carbohydrate glycan forms and in their sialic acid content. The microheterogeneity of all three mammalian AFP (rat, mouse, human) molecules have been studied by a variety of separation methodologies using various lectins as carbohydrate (sugar chain) binding proteins [47]. The lectins are derived from plants and insects and bind specific sugar groups on certain protein molecules which cause agglutination of cells containing glycoconjugates and polysaccharides [48]. The various lectins that bind AFP and their source of origin (in parenthesis) are the following: Concanavalin-A (Jack Bean) [49]; Ricinus communis (Castor bean) [50]; Allomyrina dichotoma (beetle hemolymph) [51]; Phaseolus vulgaris (French bean) [51]; Lentil agglutinin (lentil plants) [52]; Lens culinaris (lentil bean) [53]; Pisum sativum (peas plants) [54]; Erthro-agglutinating phytohemagglutinin (legume plants) [55]; Erythrina cristagalli (legume stems) [56]; Cephlosporium curvulum (Pathogenic fungi) [57]; peanut agglutinin (Arachis hypogea) [58], and Datura stramonium (Jimsen weed) [59] (Table 3). The AFP separation methodologies employed to separate and identify lectin binding of AFP are included as follows: lectin affinity and immuneabsorbent chromatography, preparative PAGE electrophoresis, 2-dimensional affinity electrophoresis, and mixed lectin electrophoresis [60]. In addition to the agglutination and cell recognition functions, lectins have been employed to induce mitogenic blast transformation in cultured leucocytes. There are certain lectins that do not exhibit any interaction with AFP such as the following: Ulex, Lotus, and Wheat Germ lectins [61] (Table 5).
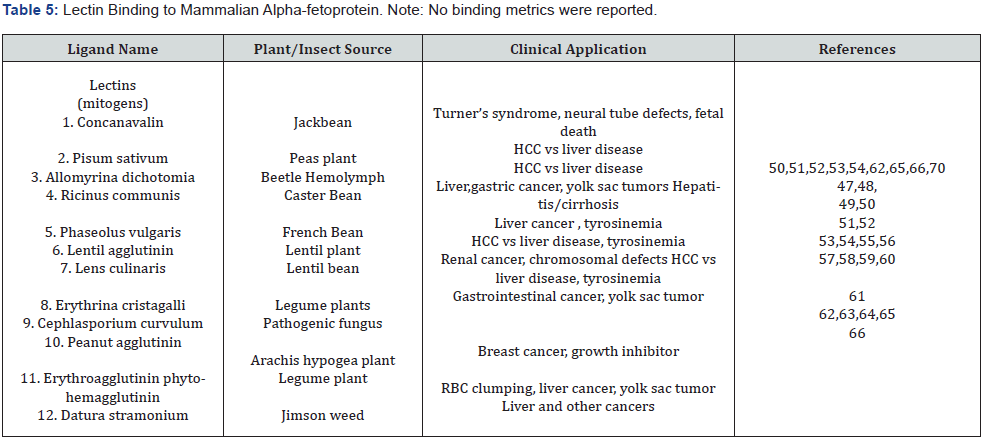
Hence, one source of the microheterogeneity of mammalian AFPs can be traced to its lectin affinity for various N-linked oligosaccharides containing beta-galactose, mannose, and N-acetylglucosamine [62]. The sugar chain (carbohydrate) structure of some tissue/tumor derived AFPs were found to contain a monosialyl-bicentennary sugar chain with additional fucosylation at the proximal N-acetylglucosamine [63]. The various altered gycoforms of AFP are due to their different carbohydrate structural sugar moieties found in rodent and human AFPs [61]. Several different AFP glycoforms have been detected, identified, and isolated from AFPs which are associated with various human disease states [64]. Some HAFP glycoform entities appeared to resolve into 5-6 forms numbered from L1 to L6 [65] with the L3 being the most utilized AFP glycoform for use in the clinic. For example, the L3 glycoform of HAFP has been employed in the clinical assays to differentiate between liver cancer versus benign liver disorders such as cirrhosis and hepatitis [53]. AFP subfractions which were non-reactive with certain lectins were found to contain the galactosialyl forms, while AFP- reactive subfractions were of monogalyectosidyl and diagalactosyl forms [66]. The lectin-reactive versus the lectin non-reactive AFP forms have been employed to detect and identify various human diseases and disorders such as hepatomas, yolk sac tumors, metastatic gastric tumors, germ cell tumors, pancreatic cancer, and various fetal malformations.
Concluding Statements and Summary
The many types of ligands bound by AFP have been studied and reported since 1970. However, no one report and/or team of researchers have attempted to chronicle the total number and different types of ligands that AFP is capable of binding to date. Although one of the first ligands reported to bind AFP was bilirubin, it was reported thereafter that estrogenic steroids, such as estradiol and estrone, were strongly bound by rat and mouse, but not human AFP. Subsequently it was reported that rodent AFP displayed natural competitive inhibitors of estrogen binding to rodent AFP, which were later identified as polyunsaturated fatty acids (PUFAs). Even though human AFP does not bind estrogens, HAFP does bind all the PUFAs that bind to rodent AFP. Moreover, the unfolded human AFP molecule does bind estrogens at an occult site concealed in a molecular crevice of the normally-folded HAFP molecule. This hidden site is exposed following a conformational change in the AFP molecule. The conformational change can be induced by intercellular environments of high concentrations of fatty acids, estrogens, and growth factors [67,68]. When many of the major ligand binding sites of AFP gradually become known, it was logical to compare these binding regions to those already known for the three major binding sites on rodent and human albumin. Many of the AFP and ALB binding sites were determined to be high capacity, low affinity binding sites which are essential for the on-off loading function of serum carrier/transport proteins. In comparison to ALB binding sites, AFP utilized the ALB-like binding sites I and II, but did not utilize the comparable ALB site-III for the binding to other ligands (see ref. 6 above).
It was later reported that mammalian AFPs could bind synthetic estrogen analogues, such as DES, as well as plant estrogenic compounds which included both the bioflavonoids and phytoestrogens [69-73]. These compounds were identified using AFP-ligand displacement experiments. The bound labeledestradiol displacements by phytoestrogens reinforced the concept that AFP could strongly bind ligand compounds that displayed the rigid structure of the oestratriene skeleton (i.e., estrone, estradiol). Binding to AFP has not been observed with compounds of a more flexible structure, as found with hexestrol and bisphenol [16]. As a final notation, chemical modification of the cysteine residues on AFP resulted in little effect on AFP binding to estrogenic compounds suggesting that the estrogen binding site on AFP did not contain cysteine amino acid resides [7].
Subsequent binding site displacement experiments on AFP binding to estrogens led to the use of competition studies involving dyes, toxins, retinoids, and thyroid hormone binding to the fetal protein. Such studies revealed three separate and distinct binding regions representing each of the three domains of AFP [14]. These binding regions encompassed three individual group sites for; 1) retinoids; 2) polyunsaturated fatty acids and estrogens; and 3) dyes, metals, and tryptophan methyl esters. In the following years, some investigations led researchers to uncover the ability of AFP, (similar to Albumin), to bind a multiplicity of synthetic drugs developed by the pharmaceutical industry [74-78]. Such drugs included warfarin and phenylbutazone and included the binding of coumarinic, anthranilic, and hydrophobic drugs [6]. These compounds were reported to bind AFP at the site comparable to the ALB site-I; such binding further included drugs such as sodium valproate, cloxacillin, furosemide, and others [37,38].
In the decades of the 1970-1990s, published reports revealed that human AFP could readily bind heavy metals such as copper, nickel, lead, and zinc [39,41-43]. Such studies further demonstrated that the high levels of metals in the amniotic fluids during development correlated with the increased levels of AFP, especially in fetuses with developmental abnormalities [40]. The metal-binding properties of AFP was further utilized in the construction of chelated metal affinity columns to purify mammalian AFPs [41,43]. Finally, the discovery of lectin agglutinin binding of AFP to a multitude of plant and insect-derived lectins opened a new vista to A) purify and separate glycoforms of AFP, and B) to harness such altered AFP glycoforms for use in clinical assays. For example, the separated AFP glycoforms used in clinical assays was utilized to differentiate primary liver cancer from other benign liver disorders, such as cirrhosis and hepatitis. The lectin-AFP interaction assays in one/study demonstrated that the quality control assay metrics showed 86% accuracy, 96% specificity, 82% sensitivity, 98% predictive values, and 82% positivity for hepatocellular carcinoma [79]. In closing, it is obvious from the above discussion, that mammalian AFPs bind a vast array of ligands for the purpose of carrier transport to target tissues and organs throughout the body during development and in oncogenic growth [80].
References
- Mizejewski GJ (2002) Biological role of alpha-fetoprotein in cancer: prospects for anticancer therapy, Expert. Rev. Anticancer Ther 2(6): 709-735.
- Mizejewski GJ (2009) Mapping the structure-function peptide sites on the alpha-fetoprotein amino acid sequence. Atlas of genetics and cyto-genetics in oncology and hematology. Deep Insights Section 1-65.
- Mizejewski GJ (2018) Head and neck germ cell tumors: Effectiveness of alpha-fetoprotein as a diagnostic biomarker. BAOJ Cancer Research and Therapy 4: 52-57.
- Lakhi NA, Mizejewski GJ (2017) Alpha-fetoprotein and Fanconi anemia: relevance to DNA repair and breast cancer susceptibility, Fetal Pediatr Pathol 36(1): 49-61.
- Mizejewski GJ (2004), Biological roles of alpha-fetoprotein during pregnancy and perinatal development. Exp. Biol. Med., 229(6): 439-463.
- Hirano K, Watanabe Y, Adachi T, Sugiura (1985) Carrier protein in human fetal serums: bilirubin binding abilities of albumin, alpha-fetoprotein, and ligandin. Chem Pharm Bull 32(2): 708-715.
- Versee V, Barel AO (1979) Interactions of rat alpha-fetoprotein with bilirubin. Biochem J 179: 705-707.
- Berde CB, Nagai M, Deutsch HF (1979) Human alpha-fetoprotein. Fluorescence studies on binding and proximity relationships for fatty acids and bilirubin. J Biol Chem 254(24): 12609-12614.
- Ruoslahti E, Estes T, Seppala M (1979) Binding of bilirubin by bovine and human alpha-fetoprotein. Biochim. Biophys. Acta, 578(2): 511-519.
- Hsia JC, Er SS, Tan CT, Estes, T, Ruoslahti E (1980) Alpha-fetoprotein binding specificity for arachidonate, bilirubin, docosahexaenate, and palmitate. J Biol Chem 255(9): 4224-4227.
- Aoyagi Y, Ikernaka, Ichida F (1979) Alpha-fetoprotein as a carrier protein and its bilirubin-binding ability. Cancer Res 39(9): 3571-3574.
- Bansal V, Kumari K, Dixit A, Sahib MK (1990) Interaction of human alpha-fetoprotein with bilirubin. Indian J Exp Biol 28(7): 697-698.
- Aussel C, Massayeff R (1984) Interaction of retinoids and bilirubin with the binding of Arachidonic acid to human alpha-fetoprotein. Biochem. & Biophys Res Com 119(3): 1122-1127.
- Aussel C (1985) Presence of three different binding sites for retinoids, bilirubin, estrogen, and arachidonic acid on rat alpha-fetoprotein. Tumor Biol 6(2):179-193.
- Benassayag C, Vallette G, Delorme J, Savu L, Nunez EA, et al. (1977) Rat and human embryo and post-natal sera contain a potent endogenous competitor of estrogen-rat alpha-fetoprotein interactions. Steroids 30(6): 771-785.
- Versee V, Barel AO (1978) Characterization of the binding properties of rat alpha-fetoprotein. Arch. Internatl. Physiol. Biochim 86: 971-972.
- Vallette G, Benassayag C, Savu L, Delorme J, Nunez E, et al. (1980) The serum competitor of estrogen-rat alpha-fetoprotein interactions. Biochem J 187(3): 851-856.
- Anel A, Calvo M, Naval J, Alava MA, Pineiro A (1989) Interaction of rat alpha-fetoprotein and albumin with polyunsaturated and other fatty acids: deterioration of apparent association constants. FEBBS Letters 250(1): 22-24.
- Savu L, Benassayag C, Vallette G, Nunez EA (1979) Ligand properties of diethylstilbesterol: Studies with native, fatty acid free rat alpha-fetoprotein & albumin. Steriods 34: 737-748.
- Savu L, Benassayag C, Vallette G, Nunez EA (1981) Mouse alpha-fetoprotein and albumin: A comparison of their binding properties with estrogens and fatty acid ligands. J Biol Chem 256: 9414-9418.
- Troisi R, Hyer M, Hatch EE (2013) Medical conditions among adult offspring prenatally exposed to diethylstilbesterol. Epidemiology 24(3): 430-438.
- Sheehan DM, Young M (1979) Diethylstilbesterol and estrogen binding to serum albumin and pregnancy plasma of rat and human. Endocrinol 104(5): 1442-1446.
- Aussel C, Masseyeff R (1983) Comparative binding properties of rat and human alpha-fetoproteins for arachidonic acid and estrogens. Res. Commun. Chem Pathol Pharmacol 42(2): 261-269.
- Baker ME, Medlock KL, Sheehan DM (1998) Flavonoids inhibit estrogen binding to rat alpha-fetoprotein. Proc Soc Exp Biol Med 217(3): 317-421.
- Milligan SR, Khan O, NASH M (1998) Competitive binding of xenobiotic estrogens to rat alpha-fetoprotein and to sex steroid binding proteins in rainbow trout. Gen Comp Endocrinol 112(1): 89-95.
- Garreau B, Vallette G, Aldercreutz H, Wahata K, Makela T, et al. (1991) Phytoestrogens:new ligands for rat and human alpha-fetoproteins. Biochim Biophys Acta 1094(3): 339-345.
- Teeguarden JG, Barton HA (2004) Computational modeling of serum-binding proteins and clearance in extrapolations across life stages and species for endocrine active compounds. Risk Analysis 24(3): 751-770.
- Ferry D R, Smith A, Malkhandi J, Baker J, Kerr DJ (1996) Phase-I clinical trial of the flavonoid quercetin pharmacokinetics and evidence for in vivo tyrosine kinase inhibition. Clin Cancer Res 2(4): 659-668.
- Cittanova N, Ulrich F, Nunez E, Jayle MF (1974) Estrogen displacement data for the association constant for ANS dye. 9th FEBS Meeting, Abstract #F8b40.
- Wong LT, Xu ZJ, Hsia CJ (1985) Simple purification procedure of rat alpha-fetoprotein by a combination of Cibacron blue gel affinity chromatography and anion-exchange high-performance liquid chromatography. J Chromatogr 338(2): 410-416.
- Huse K, Himmel M, Birkenmeier G, Bohla M, Kopperschager G (1983) A novel purification procedure for human alpha-fetoprotein by application of immobilized Cibacron blue F3G-A as affinity ligand. Clin Chim Acta 133: 335-340.
- Birkenmeir G, Usbeck E, Saro L, Kopperschlager G (1983) Triazine dye binding of human alpha-fetoprotein and albumin comparative study. J Chromatogr 265: 27-35.
- Kelleher PC, Smith CJ, Pannell R (1979) Chromatography of non-human albumins on Cibacron blue-agarose. Application to the separation of albumin from rat alpha-fetoprotein. J Chromatogr 173: 415-418.
- Evrain C, Cittaneva N, Jayle MF (1978) Fluorescence studies of the interactions of serum albumin and rat alpha-fetoprotein with aflatoxin B1. Specificity and binding parameters, Biochim. Biophys Acta 533(2): 408-414.
- Herve F, Grigorova AM, Rajkowski K, Cittanova N (1982) Differences in the binding of thyroid hormones and indoles by rat alpha-fetoprotein and serum albumin. Eur J Biochem 122: 609-612.
- Aussel C, Masseyeff R (1984) Interaction of retinoids and bilirubin with the binding of arachidonic acid to human alpha-fetoprotein. Biochim Biophys Res Commun 119(3): 1122-1127.
- Herve F, Rajkowski, Martin MT, Cittanva N (1986) Drug-binding properties of rat alpha-fetoprotein. Specificities of the phenylbutazone-binding and warfarin-binding sites. Biochem J 239(2): 451-458.
- Herve F, Rajkowski, Martin MT, Cittanova N (1984) Drug-binding properties of rat alpha-fetoprotein. Binding of warfarin, phenylbutazone, azapropane, diazepam, digitoxin, and cholic acid. Biochem J 221(2): 401-406.
- Aoyagi Y, Ikenaka T, Ichida F (1978) Copper (II)-binding ability of human alpha-fetoprotein. Cancer Res 38(10): 3483-3486.
- Laitinen R, Jalanko H, Kolho KL, Vuon E (1985) Zinc and alpha-fetoprotein in amniotic fluid from early pregnancies with fetal malformations. Amer J Obstet Gynecol 152(5): 561-562.
- Itani T, Ema M, Sakanoto J, Kowasaki H (1987) Microheterogeniety of rat alpha-fetoprotein on a chelate column. J Chromatogr 417(1): 168-172.
- Wu JT, Monir-Vagheti SM, Clayton F (1987) Human alpha-fetoprotein and albumin: differences in zinc binding. Clin Physiol Biochem 5(2): 85-94.
- Anderson L, Sulkowski E, Parath J (1987) Facile resolution of alpha-fetoprotein and albumin by immobilized metal affinity chromatography. Cancer Res. 47(14): 3624-3626.
- Mizejewski GJ, Antelman DE, Keenen JF, Preiss IL (1990) Effects of heavy metals on alpha-fetoprotein in maternal sera and amniotic fluid of pregnant mice. Toxicology 64(1): 19-32.
- Tami Y, Iwasa M, Eguchi A, Takei Y (2020) Serum copper, zinc, and metallothionein serve as potential biomarkers for hepatocellular carcinoma. Plos One 15(8): e 0237370.
- Naryak SB, Yashwanth S, Pinto M, Mayya S (2005) Serum copper, ceruloplasmin, protein thiols, and thio- barbituric acid reactive substances in liver cancer associated with elevated alpha-fetoprotein. Indian Physiol Pharmacol 49(3): 341-344.
- Kerchaert JP, Bayard B, Dubray H, Biserte G (1977) Rat alpha-fetoprotein heterogeneity. Comparative chemical study of the two variants and their Ricinus lectin-binding properties. Biochim Biophysica Acta 493(2): 293-303.
- Komath SS, Kavitha M, Swamy MJ (2006) Beyond carbohydrate binding: new directions in plant lectin research. Org Biomol Chem 4(6): 973-988.
- Noyes J, A Wood PJ (1981) Affinity chromatography of amniotic fluid alpha-fetoprotein using Concanavalin A-sepharose. Ann Clin Biochem 18 (Pt 6): 350-352.
- Ishiguro T, Takahashi Y (1989) Serum alpha-fetoprotein subfractions identified by Ricinus communis agglutinin in hepatic malignancies, Yolk sac tumors, beign liver diseases, and fetal stage. Disease Markers 7(4): 239-245.
- Taketa K, Ichikawa E, Umetsu K, Suzuki T (1986) Allomyrina dichotoma lectin-nonreactive alpha-fetoprotein carcinoma and other tumors: comparison with Ricinus communis agglutinin-I. Cancer Letters 31(3): 325-331.
- Wang SS, Lu RH, Lee FY, Chao Y, Lee SD (1996) Utility of lentil lectin affinity of alpha-fetoprotein in the diagnosis of hepatocellular carcinoma. J. Hepatol 25(2): 166-171.
- Chen S, Li J, Tan X, Xu Q, Wei Z (2020) Clinical role of combining alpha-fetoprotein and lens culinaris agglutinin-reactive fraction of alpha-protein for hepatocellular carcinoma. J. Clin Lag Anal 34(7): e23262.
- Yin ZF, Tu ZY, Cui ZF, Wu MC (1993) Alpha-fetoprotein in differentiation of benign liver diseases from hepatocellular carcinoma. Clin. Med. J 106: 615-618.
- Taketa K, Sekrya C, Namiki M, Kosaka K (1990) Lectin-reactive profiles of alpha-fetoprotein characterizing hepatocellular carcinoma and related conditions. Gastroeriterol 99(2): 508-518.
- Kanei M, Misawa A, Arai J, Kamakura K, Taketa K (1998) Erythrina cristagalli lectin-reactive alpha-fetoprotein-E2: a marker of hepatocellular carcinoma and other malignancies. Int. J. Biol. Markers 13(1): 24-29.
- Belur S, Jagadeesh N, Swammy B, Inander SR (2020) A core fucose specific lectin from Cephalosporium curvulum induces cellular apoptosis in hepatocellular and pancreatic cancer cells effective in detecting AFP. Glyconj. Journal 37(4): 435-444.
- Martin C, Daxenbichler G (1988) Peanut agglutinin inhibits proliferation of cultured breast cancer cells. Oncology 45(1): 47-50.
- Taketa K, Ichikawa E, Kato H, Taga H, Hirai H (1990) Datura stramonium agglutin-reactive alpha-fetoprotein isoforms in liver cancer. Tumor Biol 11: 220-228.
- Taketa K, Fuju Y, Toga H (1993) Characterization of E-PHA-reactive alpha-fetoprotein isoforms by 2-dimenional lectin affinity electrophoresis. Electrophoresis, 14(12): 1333-1337.
- Kerchaert JP, Bayard B, Biserte G, (1979), Microheterogeneity of rat, mouse, and human alpha-fetoprotein as revealed by poly-acrylamide gel electrophoresis and by crossed immuno-affino-electrophoresis with different lectins. Biochem. Biophys. Acta 576(1): 99-108.
- Yamashita K, Taketa K, Nishi S, Ohkura T (1993) Sugar chains of human cord serum alpha-fetoprotein: characteristic of N-linked sugar chains of glycoproteins produced in human liver and hepato-cellular carcinomas. Cancer Res 53(13): 2970-2975.
- Taketa K (1998) Characterization of sugar chains structures of human alpha-fetoprotein by lectin affinity electrophoresis. Electrophoresis J 19: 2595-25602.
- Taketa K, Izumi M, Ichikawa E (1983) Distinct molecular specifies of human alpha-fetoprotein due to differential affinities to lectins. Ann. NY Acad. Sci 417: 61-68.
- Nikolic JA, Stajic M, Cuperlovic M, Golubovic G (1990) Serum alpha-fetoprotein levels and microheterogeneity in patients with different liver diseases. J. Hepatol 11:252-256.
- Buaniah PK, Cornell C, Skillen AW (1987) Differential reactivity of alpha-fetoprotein with lectins and its usefulness in the diagnoses of various liver diseases. Clin. Chim. Acta 168: 69-73.
- Vakharia D, Mizejewski GJ (2000) Human alpha-fetoprotein peptides bind estrogen receptor and estradiol, and suppress breast cancer. Breast Cancer Res. & Treat 63: 41-52.
- Mizejewski GJ (2001) Alpha-fetoprotein structure and function: relevance to isoforms, epitopes, and conformational variants. Exp. Biol. Med. (Maywood) 226: 377-408.
- Baker ME (1984) Gossypol inhibits estrogen binding to rat alpha-fetoprotein. FEBS Lett 175: 41-44.
- Smith CJ, Kelleher PC, Belanger L (1979) Reactivity of amniotic alpha-fetoprotein with concanavalin- A in diagnosis of neural tube defects. Brit. Med J 1: 920-922.
- Yuen MF, Loi CL (2005) Serological markers of liver cancer. Brest. Proct. Res., Clin. Gastroenterol 19: 91-99.
- Sugiyama, H., Takeshita, H., Tachibana, K., Kawakami, S., (2019). Len culinaris agglutinin-reactive fraction of alpha-fetoprotein as a tumor marker in a patient with nonseminomatous germ cell tumor with normal AFP level. Clin. Genitourin. Cancer, 18:e309-e311.
- Yamamoto R, Azuma M, Wakui Fujimoto S (2001) Alpha-fetoprotein micro-heterogeneity: a potential biochemical marker for Down syndrome. Clin. Chim. Acta 304: 137-141.
- Minamoto T, Ritagawa M, Amano N, Mai M (1994) Renal cell carcinoma producing alpha-fetoprotein with a unique lectin affinity profile. J. Surg, Oncol 55: 215-221.
- Yamamoto R, Azuma M, Kishida T, Fujimoto S (2001) Total alpha-fetoprotein and Len culinaris agglutinin-reactive alpha-fetoprotein in fetal chromosomal abnormalities. B.J.O.B 108: 1154-1158.
- Baumann U, Duhme V, Auth MKH, Holme E (2006) Lectin-reactive alpha-fetoprotein in patients with tyrosinemia type-I, and Hepatocellular carcinoma. Pediatr. Gaestroenterol. Nuti 43: 77-82.
- Los FJ, Hagenaars AM, Gaillard JL, Brandelinbarg H (1992) Maternal serum alpha-fetoprotein levels and fetal outcome in early second trimester oligohydramnios. Prenatal diag 12: 285-292.
- Ishiguro T, Tsuchida Y (1994) Clinical significance of serum alpha-fetoprotein sub-fractionation in pediatric diseases. Acta. Paediatr 83: 709-713.
- Yin ZF, Tu ZX, Cui ZE, Wu MC (1993) Alpha-fetoprotein reaction to pisum sativum agglutinin in differentiation of beign liver diseases from hepatocellular carcinoma. Clin. Med. J 106: 615-618.
- Mizejewski GJ (1993) Alpha-fetoprotein and Folate deficiency. Amer. J. Ob. Gynecol 168: 279-280.






























