The Secretory Sanguine- Middle Ear Paraganglioma
Dr. Anubha Bajaj*
Consultant Histopathologist, A.B. Diagnostics Delhi, India
Submission: November 23, 2021; Published: December 01, 2021
*Corresponding Address: Dr. Anubha Bajaj, Consultant Histopathologist, A.B. Diagnostics Delhi, India
How to cite this article: Dr. Anubha Bajaj. The Secretory Sanguine- Middle Ear Paraganglioma. Canc Therapy & Oncol Int J. 2021; 20(2): 556034. DOI:10.19080/CTOIJ.2021.20.556034
Preface
Paraganglioma emerges as an exceptional neuroendocrine neoplasm which secretes catecholamine or norepinephrine. Tumefaction predominantly incriminates pre-aortic and paravertebral sympathetic plexus or base of skull. Paraganglioma arising in the head and neck, especially jugular foramen, ear or carotid body are minimally differentiated and preponderantly secrete norepinephrine.
Intra-abdominal tumours of the adrenal medulla such as neuroblastoma or pheochromocytoma or intra-adrenal paraganglioma predominantly secrete epinephrine are classify as differentiated neoplasms. Glomus jugulare and carotid body paraganglioma comprise of around 80% of paraganglioma emerging within the head and neck. Pheochromocytoma or paraganglioma arises as an extensively vascular neoplasm with parasympathetic or sympathetic differentiation. Parasympathetic paraganglioma situated within the skull base is generally non-secretory wherein below < 5% neoplasms are symptomatic and associated with catecholamine secretion.
Paraganglioma commonly emerges as a benign, singular, unilateral neoplasm although multiple tumefaction are discerned in sporadic or familial variants. Malignant neoplasms are prone to distant metastasis. Cogent tissue sampling is optimal for corroborating the diagnosis of paraganglioma. Therefore, preliminary tumour discernment and comprehensive surgical resection is recommended which ensures disease alleviation and favourable prognostic outcomes.
Disease Characteristics
The exceptional, catecholamine secreting, neuroendocrine paraganglioma originates during migration of neural crest cells. Nomenclature of paraganglioma is contingent to tumour location wherein glomus tympanicum paraganglioma arises within the middle ear although jugular foramen is spared and jugulotympanic paraganglioma or glomus jugulotympanicum or glomus jugulare emerges within the middle ear cavity or superolateral area of jugular foramen. Glomus tympanicum paraganglioma is confined to the middle ear cavity and is additionally designated as chemodectoma due to chemoreceptor function [1,2].
Majority of neoplasms arise within the jugular bulb and engender a tumefaction confined to the middle ear or external auditory canal. Glomus tympanicum paraganglioma generally arises from Jacobsen nerve or tympanic branch of glossopharyngeal nerve emerging at the cochlear promontory with consequent occurrence of a middle ear mass whereas occasional tumefaction arises from posterior auricular branch of vagus nerve or Arnold nerve and appears associated with a mass confined to the external auditory canal [1,2].
Glomus jugulotympanicum paraganglioma is frequently discerned, arises within the middle ear and manifests as a singular, sporadic neoplasm although familial tumefaction with autosomal dominant mode of inheritance is exceptionally discerned [1,2]. Typically, the neoplasm appears in middle age beyond > 40 years or within the sixth decade. Paraganglioma demonstrates a female predominance with a female to male proportion of 3:1. In contrast, hereditary paraganglioma is commonly observed in subjects within the third decade and demonstrates an equivalent female to male proportion of 1: 1[1,2]. Multiple neoplasms may depict germline mutations of succinate dehydrogenase gene subunits [1,2].
Clinical Elucidation
Parasympathetic paraganglioma is generally inactive and asymptomatic. Majority of neoplasms are situated within the skull base along the distribution of IX and X cranial nerves. As cranial nerves IX, X, XI and XII are usually incriminated, paralysis ensues within the distribution of implicated nerve. Cerebellar dysfunction may ensue.
Frequently, the neoplasm is discerned incidentally [3,4].
Symptomatic tumefaction engenders pulsatile tinnitus, otalgia or conductive hearing loss [3,4]. Glomus jugulotympanic paraganglioma can be discerned in an antecedent manner on account of characteristic clinical symptoms of pulsatile tinnitus which appears in association with or absence of conductive hearing loss [3,4]. Additionally, clinical symptoms may emerge due to sympathetic secretion of catecholamine or norepinephrine or as a direct mass effect. Classical clinical triad of headache, palpitations and profuse sweating is a common representation. Frequently, manifestations such as chronic constipation, dry mouth, dysphagia, chest pain, tachycardia, diaphoresis, episodic syncope, paroxysmal hypertension, hoarseness, cranial nerve palsies, pupillary dilatation, polyuria, polydipsia, panic attacks, fidgety movements, generalized fatigue or micturition syncope appear [3,4].
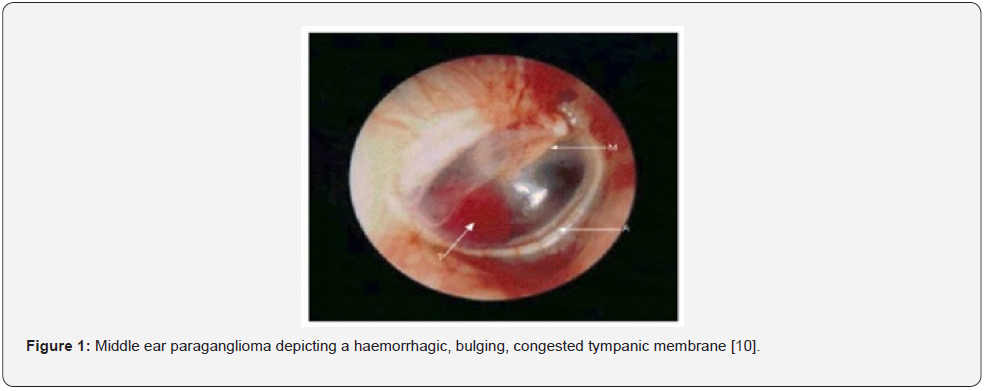
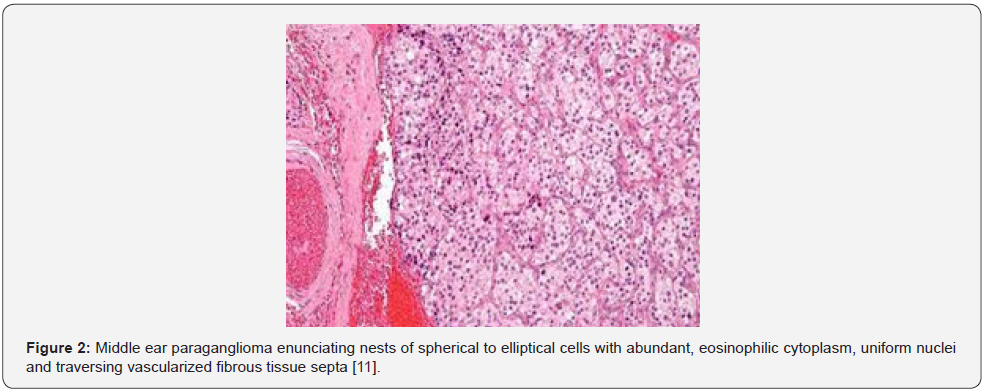
Paraganglioma is an extensively vascular, benign, symptomatic gradually progressive neoplasm which demonstrates fight-orflight sympathetic symptoms such as flushed facies, tachycardia, pupillary dilatation, headache, or tremors. Also, blurred vision, light-headed feeling, weight loss, excessive thirst, extreme hunger, mood disturbances, hyperglycaemia and weight loss ensue [3,4].
Histological Elucidation
Paraganglioma arises from chromaffin cells wherein sympathetic and parasympathetic paraganglioma appear morphologically identical [5,6]. Grossly, the neoplasm appears as a brown, firm, rubbery, polypoid, reddish, friable, pseudocapsulated or un-encapsulated tumefaction with a centric scar [5,6]. Upon microscopic examination, a classic organoid, nesting pattern or a “Zellballen” of tumour cells is observed. Tumour cell nests demonstrate centric spherical or elliptical chief cells imbued with abundant, eosinophilic, granular or vacuolated cytoplasm and uniform nuclei with dispersed chromatin. Spindleshaped, basophilic sustentacular cells circumscribe the periphery of tumour cell nests. Fibro-vascular stroma segregates tumour cell nests and appears prominent, as dense foci of vascularized fibrous tissue or exhibits foci of tumour infiltration. Mitotic activity or focal necrosis is exceptional. Foci of glandular or alveolar differentiation are absent [5,6]. Irregular clusters of tumour cells configure a distinct trabecular pattern or a “Zellballen” or tumour cell nests. Tumour tissue is extensively vascular and anastomosing bands of fibrous tissue may obscure the nesting pattern of tumour cells. Multinucleated tumour giant cells appear admixed with spherical or elliptical tumour cells imbued with abundant, granular, eosinophilic or basophilic cytoplasm and irregular nuclei. Tumour cells with sympathetic secretions depict hyaline globules.
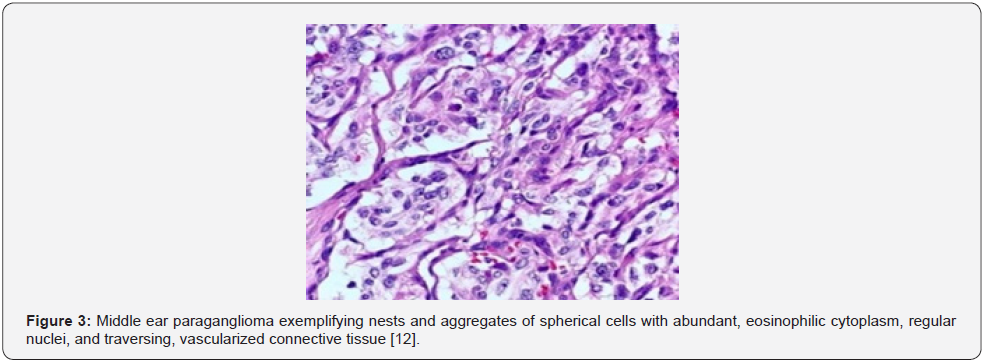
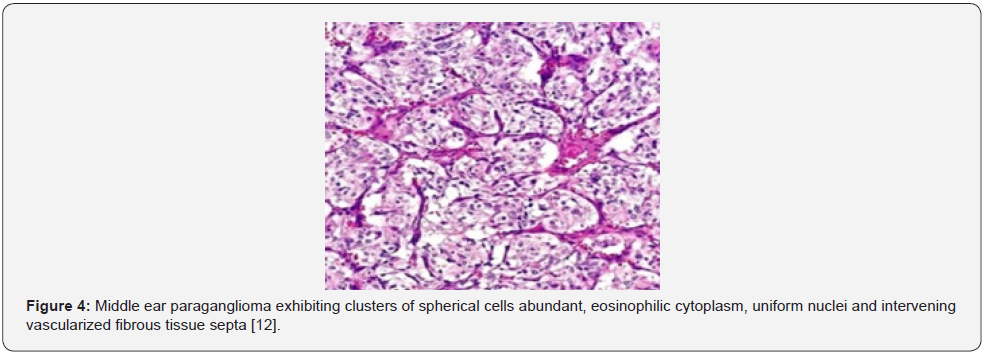
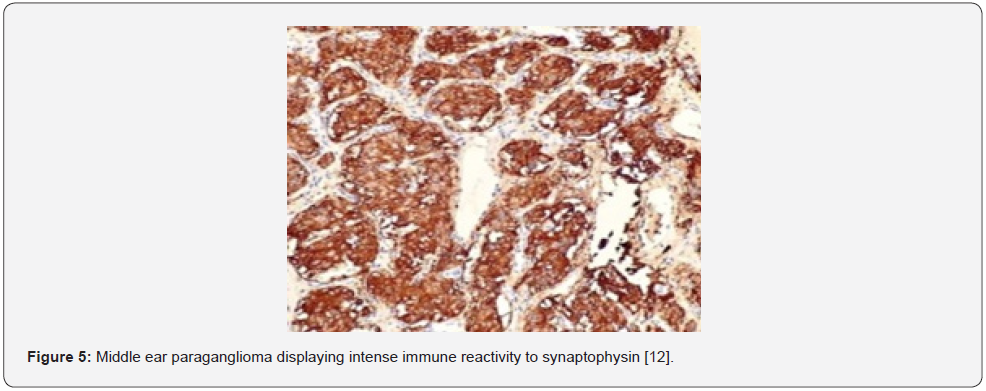
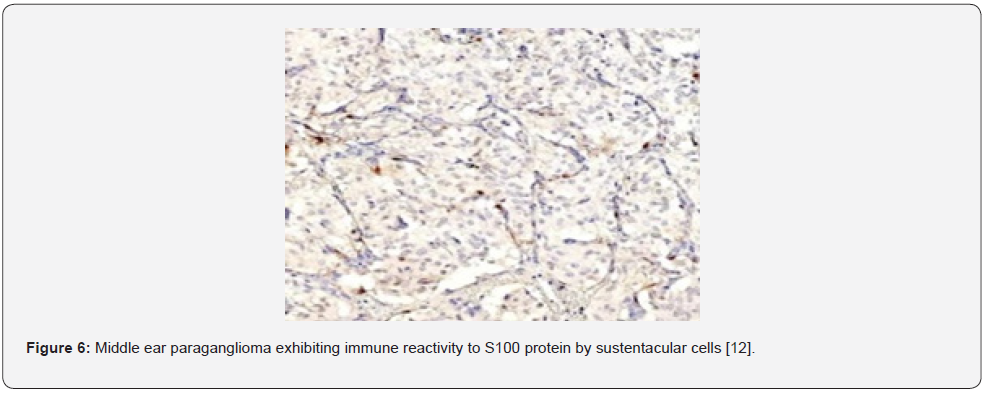
Features such as nuclear atypia, cellular or nuclear pleomorphism, necrosis, mitotic activity and localized or vascular invasion are commonly observed although are non-indicative of malignant metamorphosis [5,6]. Distant tumour metastasis is a singular feature which indicates malignant neoplastic transformation wherein metastasis occurs within cervical lymph node, pulmonary or hepatic parenchyma. Upon ultrastructural examination, giant mitochondria, neurosecretory granules, and para-crystalline intracellular inclusions are observed. Tumour cells or chief cells are immune reactive to chromogranin and synaptophysin whereas sustentacular cells are immune reactive to S100 protein. Chief cells and sustentacular cells are variably immune reactive to vimentin [5,6].
Reticulin stain is beneficial in highlighting fibrous tissue stroma and delicate nesting pattern of tumour cells [5,6]. Tumour cells are immune non-reactive to keratin, epithelial membrane antigen (EMA), human melanoma black 45 (HMB45), desmin and diverse myogenic markers. Stains such as mucicarmine or periodic acid Schiff’s (PAS) stain are non-reactive [5,6].
Differential Diagnosis
Upon imaging, glomus tympanicum paraganglioma requires a segregation from neoplasms such as •glomus jugulare paraganglioma which morphologically simulates glomus tympanicum paraganglioma. Nevertheless, the neoplasm incriminates jugular foramen, manifests a dehiscent jugular bulb, and demonstrates tumour permeation along with destruction of floor of the middle ear. The neoplasm enunciates a classic, organoid pattern with configuration of “Zellballen” or nesting architecture. Tumour cell aggregates appear as centric, spherical to elliptical chief cells imbued with abundant, eosinophilic, granular or vacuolated cytoplasm and uniform nuclei with dispersed chromatin. Spindle-shaped, basophilic, peripheral sustentacular cells circumscribe cellular nests which are segregated by prominent, fibro-vascular stroma. Occasionally, dense fibrous stroma may circumscribe tumour cell clusters. Foci of tumour cell infiltration are observed. Mitotic activity or focal tumour necrosis is exceptional. Glandular or alveolar differentiation is usually absent [7,8].
•Facial nerve schwannoma represents a pedunculated tumefaction which emerges from the tympanic segment of facial nerve. A biphasic, compact tumour is exemplified with distinct, hyper-cellular Antoni A areas commingled with myxoid, hypocellular Antoni B areas. Fibrillary processes with peripheral nuclear palisading (Verocay bodies) can be frequently discerned within cellular Antoni A areas. Enlarged, irregularly distributed blood vessels are prominent within Antoni B areas. Tumour cells are narrow, elongated, wavy with tapered edges, poorly defined cytoplasmic edges, blunt-ended nuclei with dense chromatin and are usually interspersed within collagen fibres. Degenerative manifestations with nuclear atypia can ensue in ancient tumours. Mitosis is infrequent. Foamy macrophages or lymphoid aggregates are delineated [7,8].
•Congenital cholesteatoma exhibits a lack of enhancement upon T1 weighted magnetic resonance imaging (MRI) following administration of gadolinium contrast. Essentially, cholesteatoma is layered by keratinized, non-dysplastic stratified squamous epithelium admixed with abundant granulation tissue and keratinous debris. The lesion is infiltrated with a chronic inflammatory infiltrate comprised of lymphocytes and histiocytes. Additionally, epithelioid cell granulomas, foreign body giant cell reaction, cholesterol clefts and foci of hemosiderin pigment deposition may be exemplified [7,8].
•Acoustic neuroma may be composed of foci of glandular metaplasia wherein glands appear haphazardly disseminated within zones of chronic otitis media. The neoplasm is configured of spindle-shaped cells with elongated nuclei and fibrillary cytoplasm. Specific zones of tumour configuration are described as Antoni A areas and Antoni B areas. Antoni A area is miniature and predominantly composed of organized, interwoven, elongated, bipolar cells. Antoni B area is represented by random accumulation of cells circumscribing foci of cystic change, necrosis, ancient haemorrhage, and vascular articulations. Lymphocytic infiltrate is variable and nuclear pleomorphism can be observed. Focal necrosis and mitotic figures are exceptional. Oedema, micro-cysts, macro-cysts, xanthomatous alterations and focal calcification are associated with degeneration of tumour tissue [7,8].
•Middle ear adenoma is a neoplasm demonstrating variable neoplastic configurations such as cellular sheets, solid areas, trabeculae, cystic articulations, cribriform pattern or glandular structures. Generally, papillary architecture is absent. Tumour glands or tubules are coated by uniform, singular layer of cuboidal or columnar epithelial cells incorporated with variable quantities of eosinophilic cytoplasm and spherical to elliptical, hyperchromatic nuclei with eccentric nucleoli. Neoplastic cells may exhibit significant pleomorphism. The circumscribing stroma is sparse, fibrotic or myxoid. Mitotic activity is minimal to absent. Tumour necrosis is absent. The neoplasm lacks infiltration or destruction of circumscribing bone [7,8].
•Middle ear carcinoma is constituted of tumour cells displaying significant cellular and nuclear pleomorphism, nuclear anaplasia, frequent mitotic activity, foci of tumour necrosis and infiltration of adjoining bone or soft tissue. Neoplastic glands depict an absence of dual cell layer, and a singular layer of luminal epithelial cells may be observed. Well differentiated neoplasms may simulate an adenoma although delineate an invasive pattern of tumour evolution. The neoplasm can represent as adenoid cystic carcinoma or muco-epidermoid carcinoma. Tumefaction expands within the meso-tympanum and is devoid of bony infiltration and destruction of circumscribing tissue [7,8].
•Malignant melanoma is composed of tumour cell nests comprised of spherical, elliptical, epithelioid or spindle shaped cells demonstrating significant nuclear pleomorphism, nuclear enlargement, hyperchromasia and a coarse, irregular pattern of chromatin dissemination with peripheral condensation and prominent eosinophilic nucleoli. Cellular cytoplasm is dusty and pigmented. Circumscribing stroma demonstrates variable quantities of inflammatory cell infiltrate along with focal fibrosis and a non-uniform distribution of melanin pigment [7,8].
•Meningioma exhibits hyper-ostotic foci of subjacent bone. Tumefaction is composed of spherical, elliptical or spindle-shaped cells configuring whorls, nests or a lobular pattern. Tumour cells enunciate indistinct cell boundaries and are imbued with pale cytoplasm, punched out nuclei and intra-nuclear cytoplasmic inclusions. Psammoma bodies may be discerned along with microscopic foci of bone infiltration. Tumour cells are immune reactive to epithelial membrane antigen (EMA) and vimentin [7,8]. Additionally, glomus tympanicum paraganglioma requires a segregation from lesions such as diverse neuroendocrine tumours, hyper-adrenergic spells, syncope, thyrotoxicosis, hypoglycaemia, hyperventilation, labile essential hypertension, sympathomimetic drug ingestion, diencephalic epilepsy (autonomic seizures), mast cell disease, somatization disorder, paroxysmal cardiac arrhythmia, carcinoid syndrome, recurrent idiopathic anaphylaxis, reno-vascular disease, anxiety, panic attacks, Valsalva manoeuver , autonomic neuropathy, migraine, stroke or unexplained spells of flushing [7,8].
Investigative Assay
Upon cogent physical examination, features of sympathetic overload or mass effect appear such as vocal cord paralysis with hoarseness, cranial nerve palsies, fidgety movements, tachycardia, diaphoresis, uncontrolled episodic hypertension, perspiration, cold or clammy skin, dilated pupils, dry mouth, or piloerection. Bradycardia, syncope, haemoptysis, dysphagia or chest pain may occur [8,9]. Plasma fractionated metanephrine levels are employed as a screening test. Assessment of plasma catecholamine is minimally sensitive although elevated values more than > twice the upper limit of normal is diagnostic of paraganglioma. Evaluation of 24-hour urine catecholamine and 24 hour urine metanephrines is beneficial [8,9].
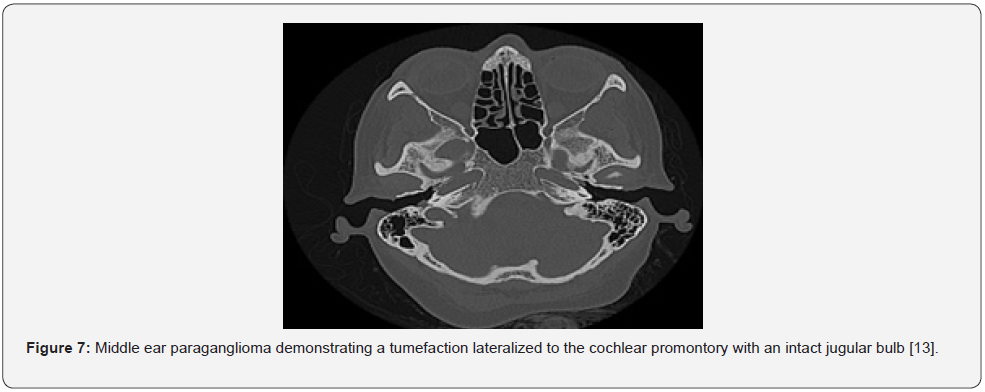
Non contrast computerized tomography (CT) or T2 weighted magnetic resonance imaging (MRI) can be adopted to visualize the neoplasm. Computerized tomography (CT) is an optimal modality for evaluating paraganglioma. Upon CT, a soft tissue mass appears lateralized to the cochlear promontory. Enlarged neoplasms may occupy the middle ear and infiltrate Eustachian tube or mastoid. Middle ear ossicles may be encompassed or disintegrated. Aggressive neoplasms depict devastation of circumscribing bone. The jugular bulb remains intact [8,9]. MRI depicts a typical salt and pepper appearance of the neoplasm wherein foci of haemorrhage and flow voids due to enhanced vascularity are observed [8,9]. Cogent tumour sampling is optimal to substantiate a vascular glomus tympanicum paraganglioma [8,9].
Therapeutic Options
The neoplasm can be subjected to embolization, surgical extermination, or radiotherapy. Paraganglioma of the head and neck may be challenging to treat wherein surgical resection, stereotactic radiosurgery or external beam radiation therapy are optimal therapeutic manoeuvers [8,9]. Comprehensive surgical eradication of the neoplasm is an optimal therapeutic strategy to treat middle ear paraganglioma [8,9]. Conservative surgical resection of the neoplasm in the absence of damage to delicate surrounding anatomical structures is necessitated. Instances where comprehensive surgical excision of the neoplasm is challenging can be treated with preoperative embolization or radiation therapy, strategies which decimate tumour vascularity and enhance tumour fibrosis. An estimated 50% of glomus tympanicum jugulare demonstrate localized tumour reoccurrence [8,9]. Appropriate control of hypertension and circumvention of intraoperative hypertensive crisis is necessitated [8,9]. Malignant metamorphosis is exceptional and is associated with an inferior prognostic outcome. Annual monitoring of the neoplasm with assessment of serum or urine fractional metanephrines is recommended. The neoplasm may be associated with minimal possibility of tumour reoccurrence [8,9].
References
- Ikram A, Rehman A (2021) Paraganglioma” Stat Pearls International 2021. Treasure Island, Florida, USA.
- Lumb R, Schwarz Q (2015) Sympathoadrenal neural crest cells: the known, unknown and forgotten? Dev Growth Differ 57(2): 146-157.
- Koopman K, Gaal J, Ronald R de Krijger (2019) Pheochromocytomas and Paragangliomas: New Developments with Regard to Classification, Genetics and Cell of Origin. Cancers (Basel) 11(8): 1070.
- Berends AMA, Buitenwerf E, Ronald R de Krijger, Anouk N A van der Horst Schrivers, Thera P Links, et al. (2018) Incidence of pheochromocytoma and sympathetic paraganglioma in the Netherlands: A nationwide study and systematic review. Eur J Intern Med 51: 68-73.
- Neumann HPH, Young WF, Charis Eng (2019) Pheochromocytoma and Paraganglioma. N Engl J Med 381(6): 552-565.
- Stenman A, Zedenius J, Carl Christofer Juhlin (2019) The Value of Histological Algorithms to Predict the Malignancy Potential of Pheochromocytomas and Abdominal Paragangliomas-A Meta-Analysis and Systematic Review of the Literature. Cancers (Basel) 15: 11(2).
- Canu L, Van Hemert JAW, Michiel N Kerstens, Robert P Hartman, Aakanksha Khanna, et al. (2019) CT Characteristics of Pheochromocytoma: Relevance for the Evaluation of Adrenal Incidentaloma” J Clin Endocrinol Metab 104(2): 312-318.
- Amodru V, Guerin C, Sarkis Delcourt, Pauline Romanet, et al. (2018) Quantitative 18F-DOPA PET/CT in pheochromocytoma: the relationship between tumour secretion and its biochemical phenotype” Eur J Nucl Med Mol Imaging 45(2): 278-282.
- Walz MK, Iova LD, Judith Deimel, Hartmut P H Neumann, Birke Bausch, et al. (2018) Minimally Invasive Surgery (MIS) in Children and Adolescents with Pheochromocytomas and Retroperitoneal Paragangliomas: Experiences in 42 Patients. World J Surg 42(4): 1024-1030.
- Image 1 Courtesy: NEJM.com
- Image 2 Courtesy: Wikipedia
- Image 3,4,5,6 Courtesy: Pathology outlines
- Image 7 Courtesy: Radiopedia






























