The Prediction of Survival by Standard Prognostic Scoring Systems and Comorbidity Indexes in Myelodysplastic Syndrome: A Hospital-Based Study
Selda Kahraman1*, Zeynep Guc2 and Mehmet Ali Ozcan3
Histopathologist in A B Diagnostics, New Delhi, India1 Department of Hematology, Medicana International Izmir Hospital, Izmir, Turkey
2 Department of Internal Medicine, Division of Medical Oncology, Dokuz Eylul University, Izmir, Turkey
3 Department of Internal Medicine, Division of Hematology, Dokuz Eylul University, Izmir, Turkey
Submission: September 16, 2021; Published: October 04, 2021
*Corresponding Address: Selda Kahraman, Medicana International Izmir Hospital Department of Hematology, Yenişehir, İşçiler street No:126, 35170 Konak/Izmir, Turkey
How to cite this article: Selda K, Zeynep G, Mehmet Ali O. The Prediction of Survival by Standard Prognostic Scoring Systems and Comorbidity Indexes in Myelodysplastic Syndrome: A Hospital-Based Study. Canc Therapy & Oncol Int J. 2021; 19(5): 556024. DOI: 10.19080/CTOIJ.2021.19.556024
Preface
Background: Many scoring methods developed as predictors of survival in Myelodysplastic Syndromes (MDS) and transformation to acute myeloid leukemia (AML). We aimed to examine the effects of disease-related and patient-related scorings systems on the survival of MDS patients.
Patients and Methods: 200 patients who were diagnosed with de novo MDS between the years 1990 and 2014 by our center were included in the study. The effects of International Prognostic Scoring System (IPSS, IPSS-R), WHO classification-based Prognostic Scoring System (WPSS/modified-WPSS), and comorbidity indexes as Charlson Comorbidity Index (CCI) and Hematopoietic Cell Transplantation Specific-Comorbidity Index (HCT-CI) that were calculated with available data on overall survival and leukemia-free survival were analyzed.
Results: The median age of patients was 70 (29-93) follow-up period was 18 months (5-168 months), and 132 (66%) patients died during the study. Median overall survival was 25 months (18.1-31.9 months), and median leukemia-free survival was 24 months (16-31.1 months). In 33 (16%) of patients, transformation to AML observed in median eight months (1-88 months). As IPSS, IPSS-R, WPSS and modified-WPSS risk scores increased, the occurring overall survival and leukemia-free survival decreased statistical significantly. Similarly, progress in CCI and HCT-CI risk groups was related to short survival times with statistical significance. In Cox regression modeling, significant prediction of survival by comorbidity indexes was independent from standard prognostic scoring systems (IPSS, IPSS-R, WPSS, modified-WPSS).
Conclusion: Comorbidity is an independent prognostic risk factor for survival in MDS patients and should be considered at the time of diagnosis for better review of clinical and treatment decisions.
Keywords: Myelodysplastic syndrome; Comorbidity index; Risk score; Overall survival; Leukemia-free survival
Introduction
Myelodysplastic Syndromes (MDS) are a heterogeneous group of neoplastic clonal stem cell diseases characterized by dysplastic morphologic features with a varying percentage of leukemic blasts and clinical bone marrow failure with increased risk of development of acute myeloid leukemia (AML). Many prognostic scoring systems which divided patients into different risk categories has been developed yielding estimates of overall and leukemia-free survival to guide clinical decision-making and also clinical study design [1].
Most commonly used prognostic scoring systems in MDS are the International Prognostic Scoring System (IPSS) [2] and WHO classification-based Prognostic Scoring System (WPSS) which has been developed by considering IPSS scoring with transfusion requirements. Subsequently the latter parameter was replaced by haemoglobin (Hgb) level by Malcovati et al. [3] (modified-WPSS model), changing the transfusion-dependency variable to Hgb <9 g/dL for males and <8 g/dL for females [3].
Disease-related factors as clinical, hematologic, morphologic, and cytogenetic parameters have been integrated into IPSS, IPSS-R, WPSS, modified-WPSS, however patient-related factors as comorbidity have not. The incidence of MDS increases with age, and in elderly patients, a high prevalence of comorbid conditions has been reported [4]. Accurate identification of comorbidity is essential in assessing the patient’s health status and quantifying the risk of mortality and morbidity. Comorbidities affect therapeutic plans and post-therapeutic outcomes of the primary disease. In many cases, comorbid conditions may be so severe as to impact directly on survival or prohibit the use of preferred antineoplastic therapies. The presence of comorbidity has been evaluated repeatedly as an important prognostic factor for survival in patients with MDS [5].
Mary Charlson has developed a classification index, which aims to predict one-year mortality in patients with solid tumors and includes the comorbid diseases in 1987 [6-8]. Comorbid diseases were scored in the extent of their severity according to this index, which is easily obtained retrospectively from patient files. The comorbidities were assigned a score of 1, 2, 3, six from mild to severe disease, and the comorbidity rating made according to the weighted score obtained by adding the scores of the comorbid diseases. According to this rating named as Charlson Comorbidity Index (CCI), the patients examined by dividing into four grades including 0, 1-2, 3-4, 5 and over.
Several papers evaluated comorbidities for stem cell transplantation performed secondary to malignant or non-malignant hematological diseases. CCI was modified for the evaluation of mortality after allogeneic stem cell transplantation, and as a result, The Hematopoietic Cell Transplantation-Specific Comorbidity Index (HCT-CI) was developed [9,10]. This scoring system is shown to be a better predictor of non-relapse mortality after allogeneic transplantation in patients with hematological malignancies. The HCT-CI has also been found to have prognostic relevance in MDS patients receiving best supportive care in which it can capture more comorbidities than the CCI [4]. As an elevated bone marrow blast percentage (>10%) or adverse karyotype are independently related to poor short-term outcome in MDS [11], these disease characteristics may limit the expected impact of comorbidities in assessing prognosis in older patients with MDS.
Prediction of the prognosis and the risk of leukemic transformation in MDS, which includes a wide spectrum of patients ranging from follow-up with supportive treatment to transformation to leukemia, is one of the main determining factors of the treatment. The study aimed to evaluate the prognostic value of comorbidity with the comorbidity indexes and compare with commonly used prognostic scoring systems in MDS patients.
Patients and Methods
Patients
A total of 200 patients (age between 29-93 years), who were diagnosed with MDS between February 1994 and September 2014 at DokuzEylul University, Faculty of Medicine, Department of Adult Hematology and were followed up in an outpatient or inpatient setting, were included in the study. The blood count, peripheral blood smear, bone marrow aspiration, the results of the biopsy, bone marrow cytogenetic examination findings and file records of the patients were reviewed. MDS patients were classified according to 2008 WHO classification as refractory anemia (RA), refractory anemia with ring sideroblasts (RARS), refractory cytopenia with multilineage dysplasia (RCMD), refractory cytopenia with multilineage dysplasia and ringed sideroblasts (RCMD-RS), refractory anemia with excess of blasts (RAEB) with subcategories RAEB-1 and RAEB-2, unclassifiable MDS (MDS-U), and myelodysplastic syndrome/myeloproliferative neoplasm (MPS/MPN).
All patients were given informed consent for study inclusion, and the study was approved by the Local Ethics Committee of DokuzEylül University.
Morphological Analysis
The peripheral blood smears and the bone marrow aspiration smears of the patients were evaluated by examining 100 and 500 cells, respectively. Dyserythropoiesis, dysgranulopoiesis, and dismegakaryopoiesis were assessed according to the FAB classification criteria [12-14].
Cytogenetic Analysis
Bone marrow aspiration material or peripheral blood was collected into 5 cc heparinized tubes for the study. Bone marrow aspiration samples were studied by applying 24 hour or overnight culture method. Peripheral blood samples were studied by modification of 72-hour culture method developed by Moorehead et al. [15] Prepared preparations were stained with GTL-banded method and evaluated according to ISCN (International System for Human Cytogenetic Nomenclature). Good prognosis includes patients with normal karyotype and -Y, 5q- or 20q- as single abnormalities, whereas poor prognosis includes patients with complex karyotype (more than three anomalies) or chromosome 7 anomalies. The other chromosomal abnormalities are considered as moderate prognostic factors.
Prognostic Scoring Systems and Comorbidity Indexes
All of the patients were assessed according to age, levels of hemoglobin, neutrophil, platelet count, MCV, LDH, erythropoietin, and ferritin, and the percentage of bone marrow blasts at the time of diagnosis. Cytogenetic risk scores, transfusion requirement (red blood cell suspension and platelets), previous treatments (chelation, supportive care, and chemotherapy), leukemia progression and time, and comorbid diseases (in line with comorbidity indexes) of the patients were reviewed.
Prognostic scoring systems, which stratified patients into risk groups for both overall survival and AML evolution, are based on different parameters as follows [1,2,14]:
IPSS: the percentage of blasts in the bone marrow, the type and number of chromosome abnormalities and cytopenias;
IPSS-R:the percentage of blasts in the bone marrow, the type and number of chromosome abnormalities , hemoglobin, platelet and neutrophil count.
WPSS: the type of WHO classification, the chromosome abnormalities, and whether or not the patient needs regular blood transfusions;
Modified-WPSS: the type of WHO classification, the chromosome abnormalities, and the severity of anemia.
Each factor is given a score, and MDS patients were classified into four risk groups for IPSS as low, intermediate-1, intermediate-2 and high, whereas patients were divided into 5 risk groups for IPSS-R, WPSS and modified-WPSS according to total scores as very low, low, intermediate, high, and very high risk group.
Comorbid characteristics of the patients were recorded retrospectively, considering the date of diagnosis. Nineteen different comorbid diseases were assessed in CCI, and comorbid diseases were scored according to CCI based on their severity. Comorbidities were given points from mild to severe disease as 1, 2, 3, and 6, respectively, and the comorbidity rating was made according to the weighted score achieved by adding scores of comorbid diseases. According to total scores, the patients were examined by dividing into four grades as 0 (very low), 1-2 (low), 3-4 (intermediate), 5 and over (high).
HCT-CI, an index developed to increase the sensitivity and specificity rates of CCI, includes criteria such as psychiatric condition associated with transplantation, obesity, and infection history. Seventeen different comorbid conditions were questioned and were given points graded from one to three in HCT-CI. The patients were examined by dividing into three groups based on their total scores as 0-2 (low), 2-5 (intermediate) and 6-11 (high).
Statistical Analysis
Data are demonstrated as mean±SD for normally distributed continuous variables, median for skew distributed continuous variables, and frequencies for categorical variables. Pearson chi-square test was performed for the comparison of categorical variables. Means of normally distributed continuous variables were compared by ANOVA. Skew distributed continuous variables were compared by Mann Whitney U test. Overall survival was calculated as the time relapsed from the date of diagnosis to the date of last contact or death. Leukemia-free survival was calculated from the diagnosis until last follow-up or leukemic progression. The patient groups formed according to IPSS, WPSS, and modified-WPSS were analyzed in terms of life expectancy. The comorbidity indexes of patients were evaluated with the survival curves in accordance with the groups. Survival curves were performed with Kaplan-Meier method and were compared with the long-rank test. Cox regression analysis was used for the multivariate analysis. Statistical Package for Social Sciences (SPSS) for Windows version 15.0 (SPSS Inc., Chicago) was used for the analysis and two-sided p value of <0.05 was considered as significant.
Results
Characteristics of patients
Of 200 patients included in the study, 120 (60%) were male and 80 (40%) female (Male/female ratio: 1.5). The median age at the time of diagnosis was 70 years(range: 29-93 years).According to 2008 WHO classification, RA were diagnosed in 49 of the patients (24.5%), RCMD in 33 patients (16.5%), RARS in 15 patients (7.5%), RAEB-1 in 23 patients (11.5%), RAEB-2 in 35 patients (17.5%), RCMD-RS in 5 patients (2.5%), MDS-U in 5 patients (2.5%), and MDS/MPN in 35 patients (17.5%). Clinical characteristics of patients, median peripheral-blood counts, LDH and ferritin values, and bone marrow cellularity rates at the time of diagnosis are listed in Table 1.
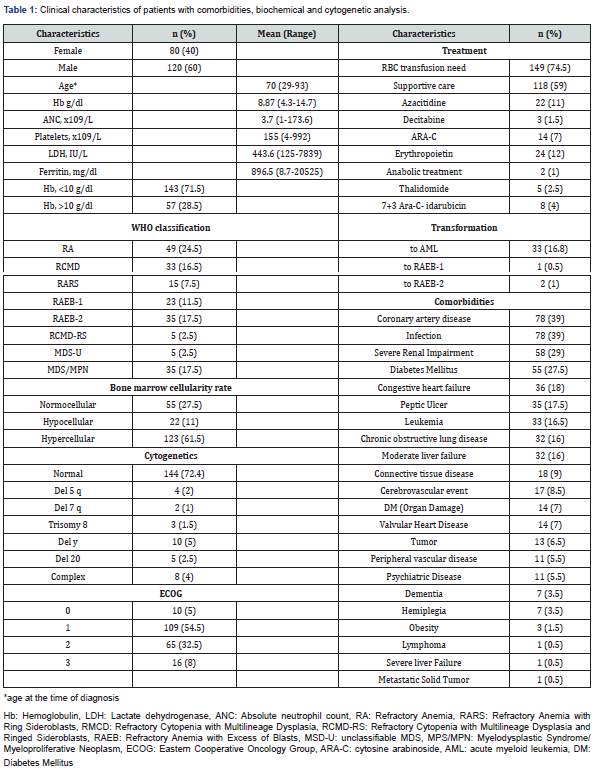
119 (59.5%) of all patients had grade 0 and 1 ECOG performance status, and the most common chromosomal abnormality was found as del Y (5%), followed by complex karyotype (4%), del 20 (2.5%), del 5q (2%), trisomy 8 (1.5%), and del 7q (1%). Supportive treatments were administered to the majority of patients, and the transfusion need of 149 patients requiring erythrocyte transfusion was determined as 13.13 units (1-90 U) on average. Azacitidine was administered to 11% of patients, followed by ARA-C (7%). 33 patients (16.8%) were observed to be transformed to AML, two patients (1%) to RAEB-2, and one patient (0.5%) to RAEB-1.
The most common comorbid diseases among patients were coronary artery disease and infectious disease, followed by severe renal impairment. While a total of 58 patients (29%) with severe renal impairment, being of the serum creatinine level over 2 mg/dl, being of the patient in dialysis program or having a history of renal transplantation were considered as the evaluation criteria. The third most common comorbid disease was type II Diabetes Mellitus (DM), which was found in 55 patients (27.5%). The number of patients with type II DM associated with organ damage was 14 (7%). ECOG performance status, cytogenetics, treatment modalities, and comorbidities are also summarized in Table 2.
Prognostic Scoring Systems
A total of 195 patients were grouped according to IPSS, it was seen that there were 74 patients (37.2%) in the low, 77 patients (38.7%) in the intermediate-1, 37 patients (18.6%) in the intermediate-2, and 7 patients (7%) in the high-risk group. In the assessment made on a group basis, a significant difference was found between IPSS diagnostic groups in terms of overall survival time (OAS) and leukemia-free survivial time (LFS) and (p=0.002, 0.001) (Table 3).
A total of 188 patients were grouped according to IPSS-R, it was seen that there were 80 patients (42.6%) in the very low, 36 patients (19.1%) in the low, 38 patients (20.2%) in the intermediate, 26 patients (13.8%) in the high, and 8 patients (4.3%) in the very high-risk group. In the assessment made on a group basis, a significant difference was found between IPSS-Ra diagnostic groups in terms of OAS and LFS(p=0.001) (Table 3).
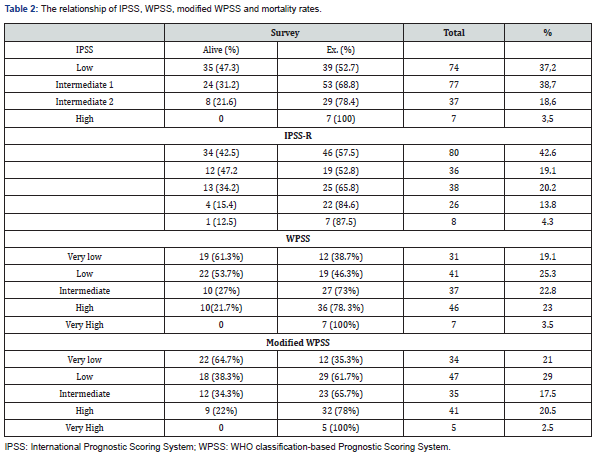
A total of 162 patients were grouped according to WPSS and modified-WPSS. According to WPSS, 31 patients (19.1%) were observed to be in the very low, 41 patients (25.3%) in the low, 37 patients (22.8%) in the intermediate, 46 patients (23%) in the high, and 7 patients (3.5%) in the very high risk group. According to modified-WPSS, 34 patients (21%) were observed to be in the very low, 47 patients (29%) in the low, 35 patients (17.5%) in the intermediate, 41 patients (20.5%) in the high, and 5 patients (2.5%) in the very high-risk group. A significant difference was found among risk groups of WPSS and modified-WPSS in terms of OAS and LFS and (p=0.003- 0.001, 0.002-0.001) (Table 3).
Comorbidity Scoring Systems
All of the patients were grouped using two different comorbidity indexes according to comorbid diseases. Sixteen patients (8%) with a score of 0 according to CCI were in the very low, 66 patients (33%) with a score of 1-2 in the low, 67 patients (33.5%) with a score of 3-4 in the intermediate, and 51 patients (25.5%) with a score of ≥5 in the high-risk comorbidity group. The patients received scores ranging from 0 to 11 after the classification done according to comorbid diseases in accordance with HCT-CI. Seventy patients (35%) with a score of 0-2 were classified as the low, 89 patients (44.5%) with a score of 3-5 as the intermediate, and 41 patients (20.5%) with a score of 6-11 as the high comorbidity index. The relationship between the comorbidity indexes and the ferritin levels was statistically evaluated, no correlation was found between the increase in comorbidity index and the elevation in ferritin levels (for CCI p: 0.272, for HCT-CI p: 0.991). Similarly, no correlation was found between the comorbidity index and erythrocyte transfusion frequency (for CCI p: 0.499, for HCT-CI p: 0.411).
The relationship between prognostic scoring systems and comorbidity indexes was statistically compared. No correlation was found between IPSS, IPSS-R and comorbidity indexes (for CCI p:0.127, p:0,457; for HCT-CI p:0.555, p:0.406), whereas comorbidity indexes was found to correlate with WPSS and modified-WPSS (for CCI p: 0.018, 0.035; for HCT-CI p: 0.036, 0.048, respectively).
Survival Analysis
Median overall survival of the patients was 25 (18.1-31.9) months according to Kaplan-Meier. Of 200 patients, 132 (66%) died and 66 (34%) survived during course of the study. When evaluated in terms of mortality rates, the mortality rate was detected to be higher in the male gender. The death rate was observed to be 73.3% for males and 50% for females. In terms of ECOG evaluation, the mortality rate was seen to be 60% for ECOG-0 and 93.8% for ECOG-3. It was observed that the mortality rate increased with increasing of performance scale as well. Independently of the age and gender, hemoglobin level of less than 10 g/dl, neutrophil count of less than 0.5x109/L, and platelet count of less than 50x109/L were seen to have a statistically significant effect on the survival (p: 0.039, p: 0.005, and p: 0.001, respectively).
When the presence of cardiac disease, type II DM, severe renal impairment and moderate pulmonary failure were evaluated in terms of mortality rates, no statistically difference was observed in the analysis between the median survival time and the presence of DM, cardiac disease, moderate pulmonary disease, and severe renal impairment (p: 0.190, p: 0.901, p: 0.103, p.0.205 respectively).
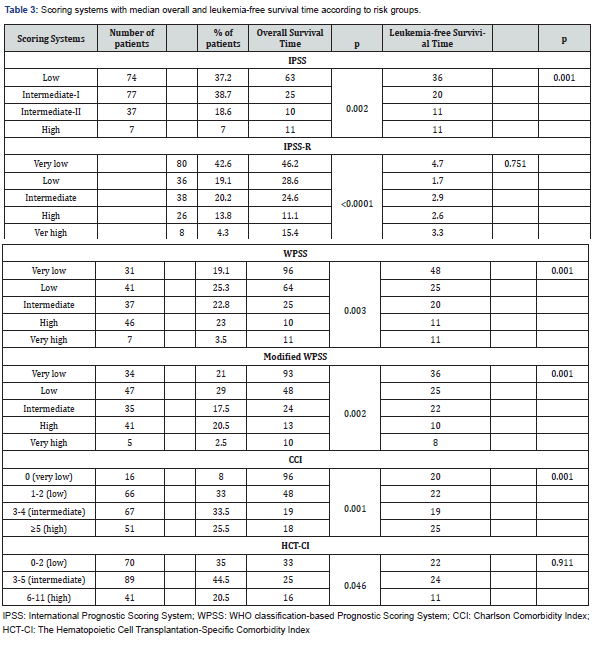
Overall Survival: When the patients were evaluated according to IPSS, median overall survival in the low-risk group was 63 months, 25 months in the intermediate-1, 10 months in the intermediate-2, and 11 months in the high-risk group. According to IPSS-R, median overall survival in the very low risk group was 46.2 months, 28.6 months in the low, 24.6 months in the intermediate, 11.1 months in the high, 15.4 months in the very high-risk group. According to WPSS and modified-WPSS, the median survival time in the very low, low, intermediate, high, and very high-risk groups was 96, 64, 25, 10, and 11 months in WPSS; whereas 93, 48, 24, 13, and 10 months in modified-WPSS, respectively. Survival time was seen to be significantly decreased inversely proportional to the risk between the groups classified according to IPSS, WPSS, and modified-WPSS (p<0.01 for all prognostic scores) (Figure 1 A-D).
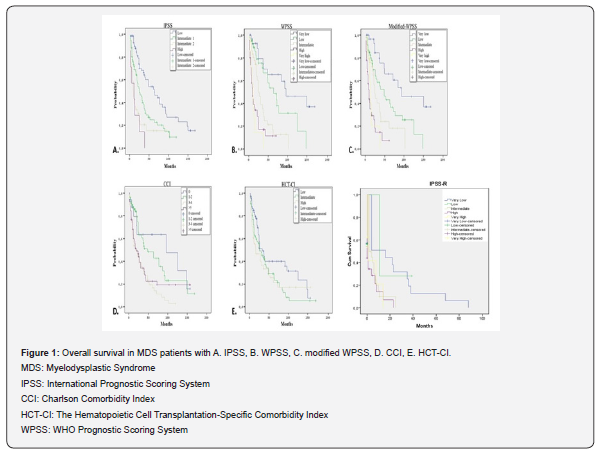
According to CCI, it was observed that median overall survival of the very low (CCI 0) risk group was 96 months, 48 months in the low (CCI 1-2), 19 months in the intermediate (CCI 3-4), and 18 months in the high (CCI ≥5) risk group. A total of 200 patients were divided into three groups according to the HCT-CI with 33, 25, and 16 months in the low, intermediate and high-risk groups, respectively. When the groups were compared regarding survival time, a statistically significant difference was found between the risk groups for both CCI and HCT-CI (p: 0.001, p: 0.046, respectively) (Figure 1 E-F).
Leukemia-free survival: The transformation to acute myeloid leukemia was observed in 33 (16.5%) of 200 patients within 18 (5-168) months mean follow-up period. Of the patients, 11 (33.3%) were female and 22 (66.7%) were male (male / female ratio was 2). The median age was 64.4 (35-85) years. When assessed in terms of distribution of diagnostic groups, 9 (27.3%) patients with the diagnosis of RAEB-1, 9 (27.3%) with RAEB-2, 12 (36.4%) with MDS/MPN, 1 (3%) with RCMD, and 2 (6.1%) with RARS were observed. It was seen that 31 (93.9%) of the patients transformed to AML were dead, however only two (6.1%) patients were alive.
The patients were assessed according to IPSS, IPSS-R, WPSS, modified-WPSS, and comorbidity indexes in terms of leukemia-free survival. The median leukemia-free survival of the low risk group was 36 months, 20 months in the intermediate-1, 11 months in the intermediate-2, and 11 months in the high risk group according to IPSS. The median leukemia-free survival in the very low, low, intermediate, high, and very high-risk groups was 46, 23, 18, 12 and 10 months in IPSS-R, 48,25,20,11 and 11 months in WPSS; whereas 36, 25, 22, 10, and 8 months in modified-WPSS, respectively. The leukemia-free survival time was also seen to be significantly decreased inversely proportional to the risk between the groups classified according to IPSS, IPSS-R WPSS, and modified-WPSS risk groups (IPSS, IPSS-R, WPSS, modified-WPSS, p: 0.001) (Figure 2 A-D).
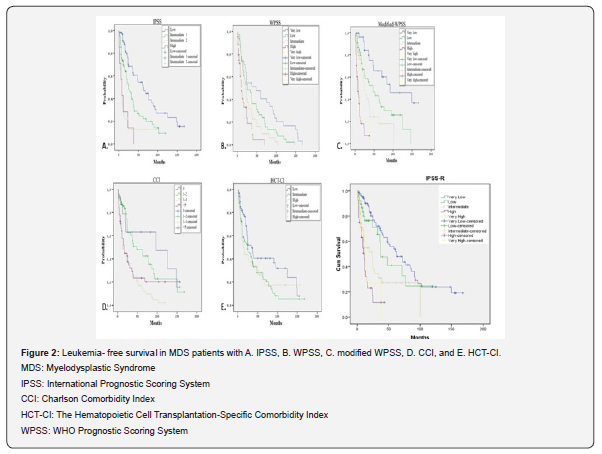
Leukemia-free survival for CCI was found to be 20, 22, 19, and 25 months in the very low, low, intermediate, and high-risk groups, respectively. According to HCT-CI, median leukemia-free survival of the low-risk group was 22 months, 24 months in the intermediate, and 11 months in the high risk group. Similar to IPSS, IPSS-R, WPSS and modified-WPSS, a statistically significant difference was found between risk groups of CCI and leukemia-free survival (p.0.001), whereas there was no significant difference in the analysis of the groups of HCT-CI (p:0.911) (Figure 2 D-E).
Prognostic scoring systems with median overall and leukemia-free survival time according to risk groups are summarized in Table 3.
Discussion
In the present study in which six different prognostic scoring systems including disease-and patient-related scorings systems were compared, our important findings are as follows; all of the scoring systems showed that overall survival time was seen to be significantly decreased inversely proportional to the risk between the groups. The survival of comorbidity indexes continues to be significant predictors independent of standard scoring systems.
The CCI was found to be the predictor for overall and leukemia-free survival, while the HCT-CI was for only overall survival. MDS is usually a disease of older age groups with the median age of onset is usually reported to be the sixth-seventh decades with a little more prevalent in males. In this study involving 200 patients, the median age and male/female ratio appear to be compatible with the literature [14-19].
When our patient group is divided into subgroups according to WHO, showed a higher rate of patients with RA, RAEB-2 and MDS/MPN and a lower rate of ones with RCMD-RS and MDS-U. Although MDS/MPN formed a rare subgroup in most series [20-22], the highest incidence reported in the literature belongs to Nöstlinger et al. with 23% [22]. The high incidence of MDS/MPN in our study may be either a regional feature or may be observed due to referring of the patients previously diagnosed to our hospital as a referral center. Transformation to AML was observed in 33 (16.5%) of our patients during the follow-up, and median leukemia-free survival time was found to be 24 (16-31) months. The diagnostic distribution of the patients who had a transformation to AML during the follow-up in the study of found similar to present study [23].
Prognosis in MDS shows a great deal of variation. The median survival time, which was determined as 25 months (18-31.9) in our patient group, was longer than the study of Sperr et al., that may be caused by variations of the follow-up periods of patients and patient subgroups [24]. The median survival time was found to be 22 months in the series of Germing et al., [21] 28 months in the study of Morel et al., and 21 months in the study involving 227 patients of Lee et al. [19,25,26,27].
There are many parameters with prognostic significance in MDS. Bone marrow blast percentage, serum LDH levels, erythrocyte count, hemoglobin concentration, age, platelet count, degree of cytopenias, karyotype, MCV, and degree of neutropenia were revealed as the variables that influence prognosis [15,27,28]. In a study of Aul et al. involving 235 patients, bone marrow blast percentage, LDH, hemoglobin concentration, age, and platelet count were identified as independent variables affecting the prognosis. FAB classification was observed not to have any impact [27-29]. In a study performed by Rigolin et al., [30] degree of cytopenias, bone marrow blast percentage, age, gender, and karyotype were found to be a predictor of survival series [28]. Independent of age and gender, hemoglobin level of less than 10 g/dl, neutrophil count of less than 0,5x109/L, and platelet count of less than 50x109/L were shown to be negative prognostic factors for survival in univariate and multivariate analyses in the present study. It was emphasized that the degree of cytopenias may also have a prognostic effect in MDS as in the study of Breccia et al. [31].
The prognostic value of cytogenetic analysis in MDS patients was highlighted many years ago [30]. Karyotyping has taken its place in prognosis and treatment with the introduction of IPSS in 1997. In our study, of 200 patients, 192 were assessed by cytogenetic analysis. The analysis results revealed a presence of normal karyotype in 144 (72.5%) of these cases. Since most patients who did not have cytogenetic results were excluded from the study, current data were found to be insufficient to reflect the overall patient population. Evaluation for survival analysis with cytogenetic data was not carried out.
In most studies, although IPSS classification is taken as basis, the need of additional prognostic classification comes to the fore because different clinical patterns may be observed even in any IPSS subgroup. IPSS, IPSS-R, WPSS and modified-WPSS, which are the most commonly used criteria for determining the prognosis in MDS, were applied in our patient group. When IPSS groups were assessed in terms of mortality rate and survival analysis, mortality rates were observed to be increased in parallel with risk groups. Similarly, overall and leukemia-free survival were seen to be reduced significantly as the WPSS scores increased. In a way confirming the previous studies [24,25,27]; IPSS, IPSS-R, WPSS and modified-WPSS are independent prognostic parameters on overall and leukemia-free survival and showed a significant effect in predicting survival.
Comorbidity is the presence of one or more diseases in addition to an index disease. In the United States, 45% of the general population and 88% of the over 65 years population have at least one chronic disease [31]. The average number of comorbidities is stated as three in patients over the age of 70 with cancer [32]. MDS patients are a group of elderly with an average age of 70 years and patient-related factors such as comorbid diseases have also a determinant influence on the survival [21,33]. Coronary artery disease, chronic obstructive pulmonary disease (COPD), and dementia were demonstrated to shorten the life span significantly in the study of Wang et al. [35]. Although heart failure, liver, and lung diseases were shown to increase non-leukemia mortality risk in the MDS group in previous studies [33,35,36], no correlation was found between the presence of cardiac pathology, COPD, renal failure, and diabetes and mortality rates in our MDS patients. Increased risk of infection and anemia developed in patients in whom coronary artery disease, COPD, and dementia are observed, may also affect the reduction of survival. As it may determine the performance of the patient, it may also cause limitations on the treatment choice.
As the prevalence of chronic diseases also increases with age, different scoring systems have been developed in various studies carried out in this direction. CCI is a scoring system widely used for prognostic risk assessment of comorbid conditions, and is applied in different disorders as AML, breast cancer, colorectal cancer, esophageal cancer, and non-Hodgkin's lymphoma [32,34,37]. However, HCT-CI is a different scoring system, which was adapted for patients undergoing hematopoietic stem cell transplantation. A multicenter, prospective national registry from Canada (MDS-CAN prospective study) reported frailty and comorbidity assessed by CCI to be independently related to survival. Bammer et al. [38] demonstrated that HCT-CI risk grouping remained an independent prognostic parameter for survival in multivariate analysis in the study of 616 well-defined patients from the Austrian MDS platform Sperret al. [39]. also analyzed 419 MDS patients with HCT-CI and CCI and concluded that HCT-CI and CCI was found to be a significant prognostic factor for overall survival estimation, whereas neither CCI nor HCT-CI were of prognostic significance when calculation AML-free survival.
Unlike other studies, our study was conducted in order to investigate the survival effects of patient-specific scoring systems (CCI and HCT-CI) and disease-associated scoring systems (IPSS, IPSS-R, WPSS and modified-WPSS) in our own pool of de novo MDS patients. Increasing of risk scores in the CCI was associated with both shorter overall and leukemia-free survival, whereas HCT-CI was observed to be a determinant for overall survival, but not for leukemia-free survival. Therefore, the usage of HCT-CI for the purpose of survival evaluation may give better results in patients who underwent stem cell transplantation following the diagnosis of MDS. When CCI and HCT-CI were evaluated for the content, transformation to leukemia is seen to be included among the parameters of the CCI. Since the development of leukemia in patients with MDS affects treatment and survival significantly, CCI may be found more effective on survival.
Conclusion
The comorbidity indexes are associated with short survival times similar as standard prognostic scoring systems, and the significant predictiveness of comorbidity indexes for survival persists independently of the standard risk scores. The comorbidity at the time of diagnosis may predict the survival in patients with MDS, and the determination of the comorbidity index may provide a review of treatment decisions and options more properly.
Conflict of Interest
The authors declare that they have no conflict of interest.
References
- Komrokji RS, Bennett JM (2006) Who is WHO in myelodysplastic syndromes? Clinical implications of the WHO classification. Current Hematologic Malignancy Reports 1(1): 9-15.
- Greenberg PL, Tuechler H, Schanz J, Guillermo Sanz, Guillermo Garcia-Manero, et al. (2012) Revised international prognostic scoring system for myelodysplastic syndromes. Blood 120(12): 2454-2465.
- Greenberg P, Cox C, LeBeau MM, Fenaux P, Morel P, et al. (1997) International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood 89(6): 2079-2088.
- Malcovati L, Della Porta MG, Strupp C, Ilaria Ambaglio, Andrea Kuendgen, et al. (2011) Impact of the degree of anemia on the outcome of patients with myelodysplastic syndrome and its integration into the WHO classification-based Prognostic Scoring System (WPSS). Haematologica 96(10): 433-1440.
- Yates JW (2001) Comorbidity considerations in geriatric oncology research. CA Cancer J Clin 51(6): 329-336.
- Zipperer E, Pelz D, Nachtkamp K, Andrea Kuendgen, Corinna Strupp, et al. (2009) The hematopoietic stem cell transplantation comorbidity index is of prognostic relevance for patients with myelodysplastic syndrome. Haematologica 94(5): 729-732.
- Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5): 373-383.
- Charlson M, Szatrowski TP, Peterson J, Gold J (1994) Validation of a combined comorbidity index. J Clin Epi demiol 47(11): 1245-1251.
- Sorror ML, Sandmaier BM, Storer BE, Michael B Maris, Frédéric Baron, et al. (2007) Comorbidity and disease status based risk stratification of outcomes among patients with acute myeloid leukemia or myelodysplasia receiving allogeneic hematopoietic cell transplantation. J Clin Oncol 25(27): 4246-4254.
- Sorror ML, Maris MB, Storb R, Frederic Baron, Brenda M Sandmaier, et al. (2005) Hematopoietic cell transplantation (HCT)–specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 106(8): 2912-2919.
- Sorror ML, Giralt S, Sandmaier BM, Marcos De Lima, Munir Shahjahan et al. (2007) Hematopoietic cell transplantation specific comorbidity index as an outcome predictor for patients with acute myeloid leukemia in first remission: combined FHCRC and MDACC experiences. Blood 110(13): 4606-4613.
- Schanz J, Tüchler H, Sole F, Mar Mallo, Elisa Luño, et al. (2012) New comprehensive cytogenetic scoring system for primary myelodysplastic syndromes (MDS) and acute myeloid leukemia after MDS derived from and international database merge. J ClinOncol 30(8): 820-829.
- Bennett JM, Catovsky D, Daniel MT, G Flandrin, D A Galton et al. (1982) Proposals for the classifıcation of the myelodysplastic syndromes. Br J Haematol. 51(2): 189-199.
- Chatterjee T, Dixit A, Mohapatra M, Gupta PK, Mishra P, et al. (2004) Clinical, haematological and histomorpological profile of adult myelodysplastic syndrome. Study of 96 cases in a single institute. Eur J Haematol 73(2): 93-97.
- Moorhead PS, Nowell PC, Mellman WJ, Battips DM, Hungerford DA, et al. (1960) Chromosome preparations of leukocytes cultured from human peripheral blood. Exp Cell Res 20: 613-616.
- Malcovati L, Germing U, Kuendgen A, Matteo G Della Porta, Cristiana Pascutto, et al. (2007) Time dependent prognostic scoring system for predicting survival and leukemic evolution in myelodysplastic syndromes. J Clin Oncol 25(23): 3503-3510.
- Blank J, Lange B (1981) Preleukemia in children. J Pediatr 98(4): 565-568.
- Bader-Meunier B, Mielot F, Tchernia G, Buisine J, Delsol G, et al. (1996) Myelodysplastic syndromes in childhood: report of 49 patients from multicentre study. Br J Haematol 92(2): 344-350.
- Miescher PA, Farguet JJ (1974) Chronic myelomonocytic leukemia in adults. Semin Hematol 11(2): 129-139.
- List AF, Doll DC, The myelodysplastic syndromes, Wintrobe's Clinical Hematology 10th edition page 2320-2341
- Germing U, Gattermann N, Strupp C, Aivado M, C Aul et al. (2000) Validation of the WHO proposals for a new classification of primary myelodysplastic syndromes: a retrospective analyis of 1600 patients. Leuk Res 24(12): 983-992.
- Malcovati L, Porta MG, Pascutto C, Rosangela Invernizzi, Marina Boni, et al. (2005) Prognostic factors and life expectancy in myelodysplastic syndromes classified according to WHO criteria: a basis for clinical decision making. J ClinOncol 23(30): 7594-7603.
- Lee JH, Shin YR, Lee JS, Kim WK, Chi HS, et al. (2003) Application of different prognostic scoring systems and comparison of the FAB and WHO classifications in Korean patients with myelodysplastic syndrome. Leukemia 17(2): 305-313.
- Nösslinger T, Reisner R, Koller E, Grüner H, Tüchler H, et al. (2001) Myelodysplastic syndromes, from French-American-British to World Health Organization: comparison of classifications on 431 unselected patients from a single institution Blood 98(10): 2935-2940.
- Demirkan F, Alacacioglu I, Piskin O, Hayri G Ozsan, Baris Akinci, et al. (2007) The clinical, haematological and morphological profile of patients with myelodysplastic syndromes: A single institution experience from Turkey. Leuk Lymphoma 48(7): 1372-1378.
- Sperr WR, Wimazal F, Kundi M, C Baumgartner, T Nösslinger, et al. (2010) Comorbidity as prognostic variable in MDS: comparative evaluation of the HCT-CI and CCI in a core dataset of 419 patients of the Austrian MDS Study Group. Ann Oncol 21(1): 114-119.
- Lee JJ, Kim HJ, Chung IJ, Kim JS, Sohn SK, et al. (1999) Comparisons of prognostic scoring systems for myelodysplastic syndromes: a Korean multicenter study. Leuk Res 23(5): 425-432.
- Morel P, Hebbar M, Lai JL, Duhamel A, Preudhomme C, et al. (1993) Cytogenetic Analysis Has Strong Independent Prognostic Value in De Novo Myelodysplastic Syndromes and Can be Incorporated in A New Scoring System: A Report on 408 Cases, Leukemia 7(9): 1315-1323.
- Aul C, Gattermann N, Heyll A, Germing U, Derigs G, et al. (1992) Primary Myelodysplastic Syndromes: Analysis of Prognostic Factors in 235 Patients and Proposals for An Improved Scoring System. Leukemia 6(1): 52-59.
- Rigolin GM, Cuneo A, Roberti MG, Bardi A, Castoldi G et al. (1997) Myelodysplastic syndromes with monocytic component: hematologic and cytogenetic characterization. Haematologica 82(1): 25-30.
- Breccia M, Carmosino I, Biondo F, Marco Mancini, Eleonora Russo, et al. (2006) Usefulness and prognostic impact on survival of WHO reclassification in FAB low risk myelodysplastic syndromes. Leuk Res 30(2): 178-182.
- Fenaux P, De Botton S (2002) Prognostic Factors in Myelodysplasia Syndromes. Pathol Biol 50(4): 256-260.
- Extermann M, Overcash J, Lyman GH, J Parr, L Balducci, et al. Comorbidity and functional status are independent in older cancer patients. J ClinOncol 16(4): 1582-1587.
- Stauder R, Wimazal F, Nösslinger T, et al. (2008) Die individualisierte Riskoeinschatzung und Therapie planung bei myelodysplastischen Syndromen. Wien KlinWochenschr 120: 523-537.
- Wang R, Gross CP, Halene S, Xiaomei Ma (2009) Comorbidities and survival in a large cohort of patients with newly diagnosed myelodysplastic syndromes. Leuk Res 33(12): 1594-1598.
- Fritz (2000) International classification of diseases for oncology. 3rd World Health Organization.
- Hoffman D, Rice, Sung HY (1996) Persons with chronic conditions. Their prevalence and costs. JAMA 276(18): 1473-1479.
- Jacot W, Colinet B, Bertrand D, S Lacombe, M-C Bozonnat, et al. (2008) Quality of life and comorbidity score as prognostic determinants in non-small-cell lung cancer patients, Ann Oncol 19(8): 1458-1464.
- Buckstein R, Wells RA, Zhu N, Heather A Leitch, Thomas J Nevill, et al. (2016) Patient-related factors independently impact survival in patients with myelodysplastic syndromes: an MDS-CAN prospective study. Br J Haematol 174(1): 88- 101.
- Bammer C, Sperr WR, Kemmler G, Friedrich Wimazal, Thomas Nösslinger, et al. (2014) Clustering of comorbidities is related to age and sex and impacts clinical outcome in myelodysplastic syndromes. J Geriatr Oncol 5(3): 299-306.
- Sperr WR, Wimazal F, Kundi M, Baumgartner C, Nösslinger T, et al. (2009) Comorbidity as prognostic variable in MDS: comparative evaluation of the HCT-CI and CCI in a core dataset of 419 patients of the Austrian MDS Study Group. Ann Oncol 21(1): 114-119.






























