Traumatic Ulcerative Granuloma with Stromal Eosinophilia
Anubha Bajaj*
Histopathologist in A B Diagnostics, New Delhi, India
Submission: September 18, 2021; Published: September 29, 2021
*Corresponding Address: Anubha Bajaj, Histopathologist in A B Diagnostics, New Delhi, India
How to cite this article: Anubha B. Traumatic Ulcerative Granuloma with Stromal Eosinophilia. Canc Therapy & Oncol Int J. 2021; 19(5): 556023. DOI: 10.19080/CTOIJ.2021.19.556023
Preface
Traumatic ulcerative granuloma with stromal eosinophilia (TUGSE) is an exceptional, reactive, benign, chronic, self-limiting ulcer arising from the oral mucosa. It may be misinterpreted as a microbial infection or malignant disorder on account of rapid progression. Traumatic ulcerative granuloma with stromal eosinophilia (TUGSE) was initially scripted by Riga in 1881 and infantile lesions were histologically described by Fede in 1890. Thus, infantile, or neonatal traumatic ulcerative granuloma with stromal eosinophilia is denominated as Riga-Fede disease [1,2]. Traumatic ulcerative granuloma with stromal eosinophilia is additionally designated as traumatic granuloma of the tongue, sublingual granuloma, traumatic granuloma, eosinophilic granuloma, eosinophilic ulcer, traumatic eosinophilic granuloma (TEG), eosinophilic ulcer of the oral mucosa, eosinophilic ulcer of the tongue or ulcerative eosinophilic granuloma. Definitive disease discernment necessitates combined evaluation of cogent clinical history, T cell phenotype, specific cellular characteristics as immune non reactivity to CD30, lack of Epstein-Barr virus infection, clinical symptoms, and histology.
Keywords: Granuloma; Ulcerative; Eosinophilia; Traumatic
Disease Characteristics
Of obscure aetiology and pathogenesis, localized trauma is posited to engender the condition. Nevertheless, majority (>50%) of instances are devoid of a traumatic factor contributing to disease occurrence. A locotypic immune response may be conducive to disease emergence [3,4]. Degranulation of eosinophils and toxic elements secreted by cytotoxic T cells engenders typical, mucosal degeneration of arising in traumatic ulcerative granuloma with stromal eosinophilia [3,4]. Besides, expression of transforming growth factor alpha (TGF-α) or transforming growth factor beta 1 (TGF-β1) within the eosinophils is significantly decimated. It is hypothesized that delayed healing within the lesion is associated with lack of synthesis of transforming growth factor (TGF) by infiltrating eosinophils [3,4].
Decimated production of eosinophil generated transforming growth factors (TGFs) in chronic oral ulcers is associated with reduced proportion and consequently delayed healing in traumatic ulcerative granuloma with stromal eosinophilia [3,4]. It is theorized that traumatic ulceration permits influx of microorganisms, toxins, and foreign debris into circumscribing soft tissue. Additionally, a severe inflammatory response triggered on account of mast cell‐eosinophil interaction may exacerbate localized inflammation with recruitment of eosinophils [3,4]. Viral infection, toxic agents, accidental bites, sharp tooth margins, ill‐fitted dentures, incisional biopsy, or repetitive trauma to the tongue are posited to engender the condition. Reoccurring injury alters tissue antigens or induces toxins, microorganisms, endogenous degradation products or foreign proteins into the soft tissue, factors which evoke a localized immune reaction wherein cell mediated immunity is a significant contributor to the inflammatory process [3,4]. Possible interaction with mast cells, secretion of eosinophil chemotactic factors and tissue eosinophilia are hypothesized to configure the granulomatous ulcer [3,4].
Generally, traumatic ulcerative granuloma with stroma eosinophilia emerges within 2 years accompanied by teething or within the fifth decade to seventh decade. Lesions commonly arise due to trauma from natal or neonatal teeth or newly erupted primary teeth. Minimal female predominance is observed [3,4]. Tongue is frequently incriminated although solitary ulcer or lesions arise within buccal, vestibular, or palatal mucosa, retromolar area, gingiva, and floor of the mouth [3,4]. Generally, lesions are sublingual or situated upon ventral side of anterior tongue. Dorsal or lateral surface of the tongue is frequently incriminated although lesions can be discerned upon the lip, palate, gingiva, vestibular mucosa, or floor of the mouth. Indurated lesions represent an antecedent phase followed by emergence of the typical ulcer. Regional lymphadenopathy is exceptional [3,4].
Clinical Elucidation
Traumatic ulcerative granuloma with stromal eosinophilia may be asymptomatic or demonstrate mild to intense pain. Clinically, an isolated, solitary, asymptomatic or painful ulcer or an indurated, submucosal tumefaction is observed. The flattened, inadequately healing, or self-healing ulcer with a benign clinical course is associated with moderate, continuous pain which may radiate to the cheek and neck region [5,6]. Majority of instances manifest a rapidly progressive, solitary ulcer with elevated or indurated periphery. Additionally, a solitary white plaque submucosal mass or multifocal lesions may appear. Reoccurrence may ensue [3,4]. Symptomatic ulcers depict mild to severe pain, indicative of malignant lesions or infective conditions such as deep-seated fungal infection, tuberculosis, or primary syphilis. The ulcer persists for weeks or months and resolves spontaneously [3,4]. An ulcer with elevated, indurated perimeter and a yellowish, fibrinous base is delineated. Draining cervical lymph nodes may be palpable. Swelling of the cheek is observed. Intraoral lesion demonstrates centric, superficial ulcer with an indurated base. Rapid progression of lesion may indicate a malignant ulcer. Lesion may undergo spontaneous retrogression [3,4].
Histological Elucidation
Generally, the smooth, tender, firm, erythematous, indurated, well circumscribed ulcer of ~ one centimeter magnitude appears circumscribed by a whitish perimeter. Fixation to deep seated soft tissues or regional lymphadenopathy is absent. Typically, the mosaic-like, ulcerative lesion depicts a diffuse, polymorphic, inflammatory infiltrate composed of histiocytes, small lymphocytes, predominantly activated T lymphocytes, streaked T lymphoblasts and eosinophils admixed with a mild granulomatous response [7,8]. Frequently, inflammation associated ulceration extends into the submucosa, deep seated skeletal muscle or salivary glands. Upon microscopy, a diffuse, polymorphic inflammatory cell infiltrate predominantly constituted of eosinophils, B lymphocytes, T lymphocytes, macrophages and enlarged, atypical cells appears to infiltrate the superficial mucosa, subjacent submucosa and engenders degeneration of circumscribing skeletal muscle [7,8].
The deep-seated ulceration is superimposed with stratified squamous epithelium and a fibrino-purulent membrane. Ulcerated area may invade skeletal muscle fibres with inflammatory cells as eosinophils, neutrophils, and fibroblasts. Infiltrating polymorphic lymphocytes or activated T cell blasts appear immune reactive to CD3+, CD2+, CD4+, CD5+ and immune nonreactive to CD8‐, CD7‐ and CD30-. However, infiltrating T lymphocytes immune reactive to CD30+ are indicative of nonspecific T cell or B cell activation [7,8]. The ulcer displays an expansive, infiltrative, mildly granulomatous lesion confined to superficial mucosa or adjoining adipose tissue and may extend between deep seated striated skeletal muscle fibres [7,8].
The ulcer is superimposed with hyperplastic, hyperkeratotic stratified squamous epithelium. Superficial squamous epithelial cells of the centric ulcer exhibit accumulated mucopolysaccharides with keratin dystrophy. The diffuse, polymorphic, inflammatory infiltrate is predominantly composed of eosinophils, histiocytes, numerous enlarged mononuclear T cells, B cells and macrophages imbued with abundant cytoplasm, irregular nuclear contour, miniature nucleoli and fine chromatin. Along with inflammatory invasion of superficial mucosa, submucosa, skeletal muscle and salivary glands, a pseudo-lymphomatous countenance may be observed [7,8]. Ulcerated area is densely infiltrated with a mixed inflammatory infiltrate of eosinophils and macrophages.
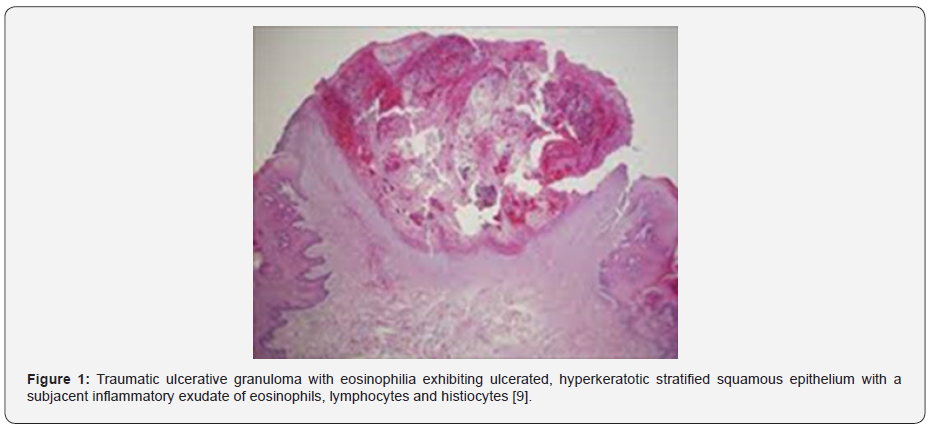
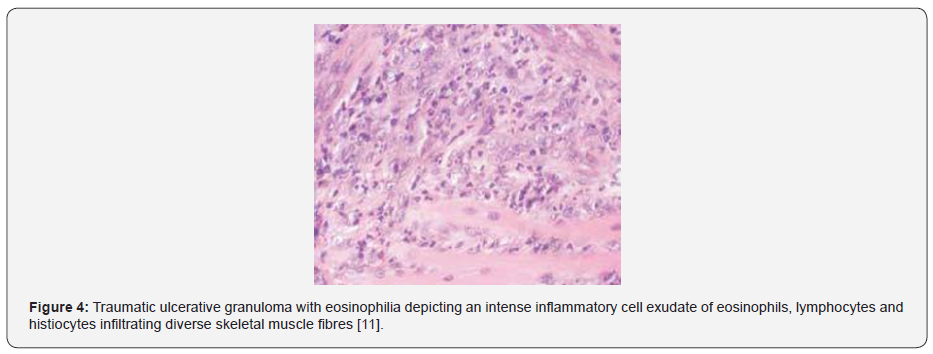
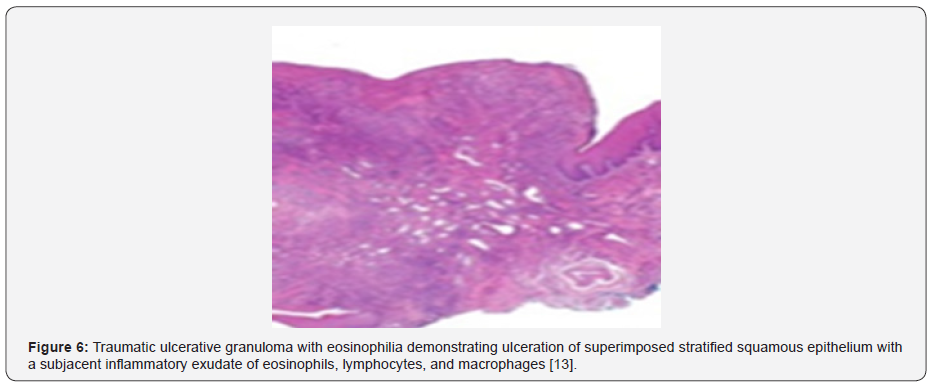
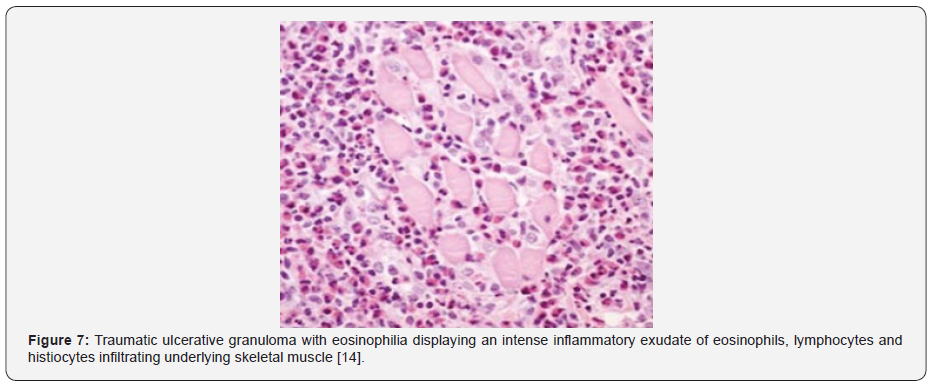
The inflammatory cell exudate extends and infiltrates into vascularized, deep seated, skeletal muscle fibres of the tongue. Atypical cells are usually absent [7,8]. Enlarged, atypical T lymphocytes appear disseminated or clustered within the ulcerated lesion. Lymphocytes infiltrating traumatic ulcerative granuloma with stromal eosinophilia frequently express markers of T cell, cytotoxic T cell and display clonal T cell receptor gene rearrangements [7,8]. The lesion may undergo spontaneous retrogression or may resolve following elimination of possible triggers such as micro-trauma as encountered with artificial denture within weeks or months [7,8-14] (Figures 1-7).



Differential Diagnosis
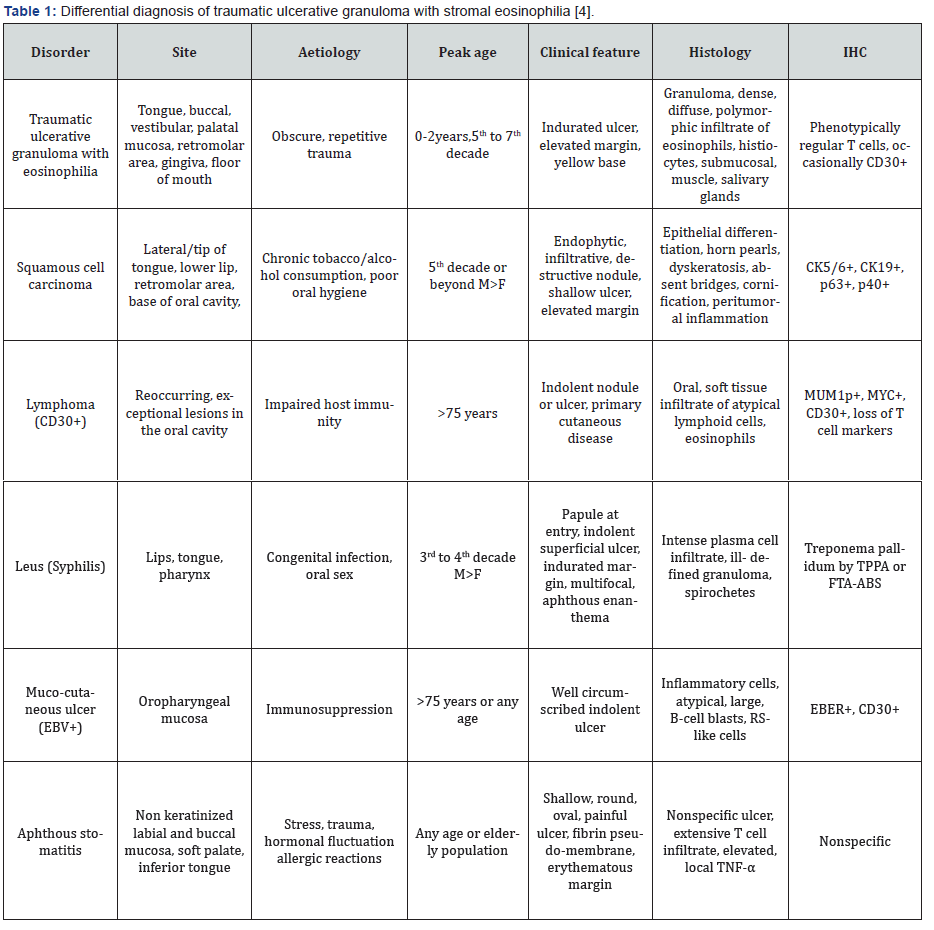
Traumatic ulcerative granuloma with stromal eosinophilia requires segregation on clinical grounds from conditions such as traumatic neuroma, lymphoma, lymphangioma, salivary gland neoplasms, primary syphilis, metastatic malignancies, atypical histiocytic granuloma or proliferative myositis [3,4]. Histological demarcation is necessitated from reactionary, proliferative processes such as atypical histiocytic granuloma, Langerhans cell histiocytosis, proliferative myositis, angiolymphoid hyperplasia with eosinophilia (ALHE), Kimura’s disease, certain lymphomas, allergic reactions, or parasitic infestation [3,4].
Tlymphocytes immune reactive to CD30+ may be discerned in lymphoproliferative disease, Hodgkin’s lymphoma with Reed Sternberg cells, non-neoplastic cutaneous disorders as atopic dermatitis, adverse drug reactions, molluscum contagiosum and insect bites. Therefore, aforementioned disorders mandate a demarcation [3,4]. Segregation is required from squamous cell carcinoma (SCC), lymphoproliferative disorder (LPD) immune reactive to CD30+, infectious disease as primary syphilis, tuberculosis, aphthous stomatitis, necrotizing sialometaplasia or Epstein-Barr virus (EBV) associated mucocutaneous ulcer [3,4]. As traumatic ulcerative granuloma with stromal eosinophilia demonstrates a rapid onset, gradual alleviation and rolled ulcer perimeter, segregation from an oral squamous cell carcinoma may be challenging [3,4]. Appropriate monitoring for aforesaid infectious diseases is recommended [3,4].
Investigative Assay
In order to differentiate traumatic ulcerative granuloma with stromal eosinophilia from diverse infectious or malignant disorders, cogent tissue sampling is mandatory. Concurrence with an extensive clinical examination and screening for infectious diseases as syphilis, Epstein-Barr virus, or human immune deficiency virus (HIV) infection is beneficial and necessitated [15,16].
Therapeutic Options
Alleviation of the lesion may occur in up to a year. Also, majority of instances resolve spontaneously. Application of topical corticosteroids remains ineffectual [15,16]. Delayed healing of oral traumatic ulcerative granuloma with stromal eosinophilia may be due to lack of synthesis of transforming growth factor alpha (TGF-α) and transforming growth factor beta 1(TGF-β1) by infiltrating eosinophils [15,16].
Traumatic ulcerative granuloma with stromal eosinophilia can be treated conservatively with topical steroids, topical antibiotics, mouthwash, curettage, and cryotherapy [15,16]. A frequently adopted therapeutic strategy is simple surgical extermination of the lesion. Brisk amelioration of the ulcer is observed, and localized reoccurrence is usually absent [16,17]. Cogent surgical extermination of the lesion with prophylactic extraction of respective tooth is recommended. Exclusion of diverse, malignant disorders is necessitated following comprehensive eradication of the lesion [16,17]. Regular monitoring or incisional biopsy can be adopted for definitive disease discernment as resolution of the lesion may occur within weeks, months, or a year [16,17]. Frequency of monitoring is recommended at quarterly within first six months, subsequently twice a year and annually over next two years. Micro-trauma or punctate haemorrhage may persist. Although documented, lesion reoccurrence is usually absent (Table 1) [16,17].
References
- Fede F (1891) Della produzione sottolinguale, o malattia del Riga; Atto Congresso italiano di pediatria. Atto Congresso italiano di pediatria Napoli pp. 251-260.
- Popoff S (1956) Documents Iconographiques, fig. 450. Annales Dermatologie et Syphilographie pp. 83.
- Tufayl Ahmed Hannan, Muhammed Umer, Labib Syed, Mohamed Areeb Anis-Alavi, Umer M (2020) A case report of traumatic ulcerative granuloma with stromal eosinophilia (TUGSE) in a 21-year-old” Clin Case Rep 8(11): 2214-2216.
- Benitez B, Mülli J, Alexandar Tzankov (2019) Traumatic ulcerative granuloma with stromal eosinophilia-clinical case report, literature review and differential diagnosis. World J Surg Onc 17: 184.
- Dhanrajani P, Cropley PW (2015) Oral eosinophilic or traumatic ulcer: a case report and brief review. Natl J Maxillofac Surg 6(2): 237-240.
- Chandra S, Raju S et al. (2014) Traumatic ulcerative granuloma with stromal eosinophilia. Arch Iran Med 17(1): 91-94.
- Feller LL, Khammissa RR, et al. (2013) Oral squamous cell carcinoma in relation to field precancerisation: pathobiology. Cancer Cell Int 13(1): 31.
- Marszalek A, Neska-Dlugosz I (2011) Traumatic ulcerative granuloma with stromal eosinophilia. A case report and short literature review. Pol J Pathol 62(3): 172-175.
- Image 1 and 2 Courtesy: MDEdge.com
- Image 3 Courtesy: Twitter
- Image 4 Courtesy: Academic oup.com
- Image 5 Courtesy: Wikimedia commons
- Image 6 Courtesy: Wikipedia
- Image 7 Courtesy: Research gate
- Dojcinov SD, Venkataraman G, et al. (2010) EBV positive mucocutaneous ulcer--a study of 26 cases associated with various sources of immunosuppression. Am J Surg Pathol 34(3): 405-417.
- Sharma B, Koshy G, et al. (2016) Traumatic Ulcerative Granuloma with Stromal Eosinophila: A Case Report and Review of Pathogenesis. J Clin Diagn Res 10(10): ZD07-ZD09.
- Segura S, Pujol RM (2008) Eosinophilic ulcer of the oral mucosa: a distinct entity or a non-specific reactive pattern? Oral Dis14(4): 287-295.






























