Radiation Therapy Decision and Field Design after Neoadjuvant Chemotherapy in Breast Cancer: A Review of Neoadjuvant Approach
E Elif Ozkan*
Suleyman Demirel University, Department of Radiation Oncology, Turkey
Submission: September 14, 2021; Published: September 23, 2021
*Corresponding Address: E Elif Ozkan, Suleyman Demirel University, Department of Radiation Oncology, Isparta, Turkey
How to cite this article: E Elif O. Radiation Therapy Decision and Field Design after Neoadjuvant Chemotherapy in Breast Cancer: A Review of Neoadjuvant Approach. Canc Therapy & Oncol Int J. 2021; 19(5): 556022. DOI: 10.19080/CTOIJ.2021.19.556022
Abstract
Locoregional control and overall survival benefit from postmastectomy radiotherapy (PMRT) particularly in women with node-positive breast cancer is demonstrated by plenty of randomized trials and meta-analysis. However, these data are insufficient to date to individualized treatment decisions regarding PMRT after neoadjuvant chemotherapy (NAC) although a fair amount of data indicated some clinical and pathological features to determine the patients who might benefit. Interpretation of published data including non-randomized studies suggest that extent of initial disease and response to NAC are the mostly affirmed factors predicting locoregional recurrence risk in these patients. Initial stage, pathologic response and additional features such as grade, biomarker status should be meticulously taken into consideration to recommend PMRT.
In this review, plenty of previous prospective and retrospective data is reviewed to provide a guidance for rationalization of suggesting or omitting PMRT after NAC in different clinical scenarios. To ease the patient evaluation, imaging for pretreatment clinical staging, radiologic and pathologic axillary investigation are briefly summarized as separate topics. Additionally, the discordance on the definition of term pathologic complete response and radiation portal design is briefly discussed.
Keywords: Breast cancer;Neoadjuvant chemotherapy; Pathologic complete response; Post-mastectomy radiotherapy; Radiation portal design
Introduction
Neoadjuvant chemotherapy (NAC) was incorporated into breast cancer treatment strategy mainly to allow breast-conserving surgery (BCS) for operable patients and downstage tumor to become operable for patients who were deemed inoperable due to skin or chest wall involvement consequentially improving surgical outcome [1-3]. Eradicating micrometastatic disease earlier than adjuvant setting, mitigating the hypothetical stimulatory effect of surgery on occult disease [4] and providing predictive information about the chemosensitivity that guides subsequent drug selection are some of the proven evidence that rationalizes the ever-increasing wide utilization of neoadjuvant approach particularly for large tumours [1-3,5,6]. Taking the uncontrovertible role of genetic profiling on individualizing treatment strategy into consideration, providing extra time for genetic profile evaluation is another advantage of NAC. Providing a chance for planning reconstruction is another advantage of NAC in terms of surgical aspect. Pathologically confirmed achievement of clear axilla after NAC via sentinel lymph node biopsy (SLNB) enables adjuvant radiation with smaller fields. To sum up; advantages of NAC are, higher chance of undergoing breast conserving therapy [7], gaining time for convenient type of surgery (breast conserving surgery or mastectomy with or without reconstruction) and genetic analysis if indicated, achieving information about resistance to systemic treatment, chance of less extensive axillary intervention while tumour-positive (axillary) lymph nodes can be converted into ypN0 in 30–45% of the patients [8-11].
NAC was compared with the same chemotherapy given postoperatively in plenty of randomized trials [12-16]. Accurate interpretation of these data is hardly available because: firstly, frequency of breast-conserving surgery differed between groups and in some trials [17,18] some of the good responders even did not receive surgery. Second drawback is substantial downstaging bias caused by analysis of postsurgical characteristics data in the studies investigating influence of tumour characteristics on outcome [19]. However, NACT is shown to provide equivalent survival to adjuvant chemotherapy in National Surgical Adjuvant Breast and Bowel Project Protocols B-18 and B-27 trials [20].
NACT enables successful BCS with negative margins in patients having too large primary tumor relative to the breast size which is a well-accepted indication for mastectomy [21]. Despite all above mentioned advantages, NACT should not be considered as a substitute for radiotherapy or other adjuvant treatments.
Guidelines for PMRT particularly for patients who have pathological complete response (pCR) after neoadjuvant therapy is less clear. Because of increasing utilization of NAC and more targeted therapies, the pCR rates have also increased [20,22,23]. Current evidence is insufficient to omit PMRT in case of pCR and pretreatment clinical stage is referred to guide this decision rather than post chemo pathological stage. Several retrospective studies comparing PMRT versus observation in patients who have had nodal pCR in tems of locoregional recurrence rates, disease free and overall survival failed to show any superior outcome with PMRT [16-20]. But these studies are underpowered while they include smaller high-risk subgroups of patients [24].
NAC strategy is preferred for inoperable cases such as T4, inflammatory breast carcinoma or with bulky N2,3 lymph nodes and in case of Her 2 overexpression or triple negative breast cancer patients (TNBC) if T>T2/N (+). Patients with relatively large tumor in small breast willing breast conserving surgery may also be appropriate candidates for NAC. The purpose in this review is to analyze the published literature investigating indications and portal design of adjuvant radiotherapy after NAC considering data on adequate pretreatment imaging, postsurgical complete response evaluation and risk factors for locoregional recurrence.
Imaging and Nodal Evaluation
Pretreatment Imaging
Accurate staging before NAC, is a prerequisite while clinical T and N stage are the most important factors determining locoregional recurrence (LRR) risk which consequentially guides locoregional therapy decision after NAC.
About 30% of patients who are considered as cN0 found to have occult nodal disease by SLNB and that warrants supportive imaging tools for sufficient pretreatment clinical staging [25]. Axillary ultrasound is one of these imaging modalities used for nodal staging. Longitudinal greatest dimension/transverse greatest dimension ratio of < 2, absence of regular hilum, eccentrically widening of cortex, and cortical thickening are well known reported features of pathological lymph node [26]. Alterations in morphology apart from dimentional changes, may help to detect malignancy with a greater specificity [27]. Magnetic resonance imaging (MRI) and 18-fluorodeoxyglucose positron emission tomography (18FDG-PET) are additional imaging modalities that can be particularly helpful to assess nodal disease burden accurately [28-33]. MRI can also determine the disease extent in breast with high accuracy even in the absence of axillary nodes [34]. The sensitivity and specificity of MRI for nodal disease were reported as 83% and 90% respectively [28]. The sensitivity and specificity of 18FDG-PET reported to be 61% and 80% respectively in prospective study [31]. In their review on postmastectomy radiotherapy, Kishan et al. [34] conclusively recommended pretreatment axillary ultrasound and biopsy for suspicious lymph nodes and clipping it for all patients with pathologically confirmed breast carcinoma. In our previous study with 18FDG-PET, the sensitivity and specificity for ALN metastases were determined as 78.8% and 92.6%, respectively. With the cut-off value of 1.79 for the maximum standardized uptake value (SUVmax), PPV, NPV and the accuracy was calculated as 0.933 (93.3%), 0.75 (75%) and 0.837 (83.7%) respectively [35]. In case of clinically positive nodes, 18FDG-PET is also referred as an advanced imaging modality to guide adjuvant radiation therapy in terms of providing more targeted approach to axilla [36].

ESMO Guidelines recommends all three of mamography, breast/axillary ultrasound and MRI to evaluate the pretreatment locoregional disease extent accurately. And iteration of all three modalities is also recommended after NAC [37].
Axillary Evaluation After NAC
Radiologic evaluation of axilla after NAC is investigated by American College of Surgeons Oncology Group (ACOSOG) Z1071 (ALLIANCE) trial reporting the utility of axillary ultrasound after NAC and its impact on postNAC sentinel node biopsy [38]. The authors demostrated a reduction in FNR from 12.6% to 9.8% when postNAC SLNB is limited to patients with normal AUS and with at least 2 SLNs removed. And a suggestion of adoption of SLN surgery for node-positive patients treated with neoadjuvant chemotherapy was given conclusively. Standard imaging strategy after NAC is still a matter of debate [39]. However, MRI (or ultrasound if there is no initial MRI) might be used to evaluate which patients have not responded well and should receive locoregional therapy.
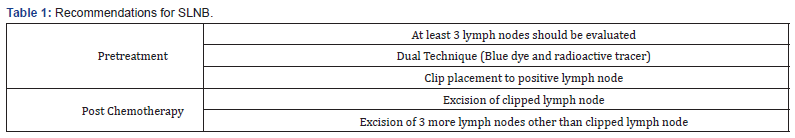
Axillary lymph node dissection (ALND) is currently replaced by SLNB for both patients with initial cN0 and cT1–2 disease and involvement of up to two nodes on pretreatment SLNB [40-42]. Supportively, European Organisation for Research and Treatment of Cancer (EORTC) 10981–22023 AMAROS randomized trial reported that axillary radiotherapy can replace ALND in patients with a positive SLNB and a 0.5–3 cm primary [43]. As 20–40% axillary downstaging can be seen after NAC, the use of SLNB in this context is concern of controversies. Lower sentinel node identification rate after NAC compared to prereatment procedure is reported in plenty of single institution reports and multicenter studies [44-47]. In a metaanalysis by Fu et al., overall sentinel node identification rate and overall false negative rate (FNR) was found as 89% and 14% respectively [48]. In the ALLIANCE trial, which is phase II trial including 649 women with cT0–4N1–2 disease who received NAC, the overall FNR was 12.6%. After reevaluation according to number of removed nodes; FNR was 21.1% in patients with <3 lymph nodes removed, where it was 9.1% in patients with at least three sentinel nodes removed. Also, dual identification method (blue dye and radiolabeled colloid), reduced the FNR to 10.8% [49].
In the SENTINA study, SLNB was performed before NAC (for initially cN0 and ycN0 after NAC), before and after NAC (for cN0 that were pathologically N0 by pretreatment SLNB) or after NAC (for cN+ which were ycN0 after NAC) [50]. It was concluded that FNRs were 24% and 18%, respectively if patients only had one or two sentinel nodes removed, the, but if at least three nodes were removed, FNR was less than 5%. Additionally, dual identification methodology reduced FNR to 8.6% [50]. They attributed this FNR, in part, after NAC to the impaired lymphatic drainage due to systemic treatment. Results of SENTINA trial [50] eventuated in several inference;
i. Information about initial histologic status provided by SLNB before NAC guides adjuvant locoregional treatment,
ii. Changes in lymphatic drainage caused by fibrosis and nonuniform tumor regression may result in false negative results with SLNB after neoadjuvant chemo,
iii. Dual identification technique (both dye and colloid) should be used to reduce FNR,
iv. Initially biopsied and positive lymph node should be clipped and dissected after NAC
v. In patients with small volume metastasis such as isolated tumor cells or micrometastasis in SLNB, axillary dissection may not be obligatory.
The accuracy of SLNB after NAC was investigated prospectively with 153 biopsy-proven T0–3N1–2 breast cancer patients in SN-FNAC study [51]. Immunohistochemistry was mandated in this study and patients with ypN0(i+) with SLNB were considered as positive. The sentinel node identification rate and FNR were 87.6% and 8.4% respectively. FNR would increase to 13.3% if ypN0(i+) patients were taken as negative. The FNR was 18.2% for patients with only one node identified and it was 14.2% for patients with T3 tumors.
Kishan et al. [34] suggested completion ALND if only 1 or 2 sentinel nodes were removed and reported to be negative. The interpretation of the published data allows certain suggestions to takeaway such as a FNR of less than 10% can be achieved in cN+ patients after NAC with SLNB if three or more sentinel nodes are removed, dual identification method should be used, and pretreatment pathologically positive nodes should be clipped and excised. If pathologically positive lymph node is not clipped before NAC, patient should be reviewed by the surgeon and pathologist in terms of axillary sampling. In cases particularly with cN (+), axillary dissection should be considered if < 2 sentinel nodes were examined and reported to be negative.
Recommendations directed by the inference from published data is summarized in Table 1.
Surgery after neoadjuvant chemotherapy
As this is out of the main topic of this review, only some surgical issues related with postsurgical radiotherapy will be discussed under this subheading. If breast conserving surgery is planned; tumor bed, radiologically suspicious residual microcalcifications should be clipped to guide radiotherapy boost volume. Mastectomy should be preferred in case of large tumor relative to breast volume, multicentric tumor, unavailibility of (-) margin in presurgical evaluation and willingness of mastectomy by the patient [52].
One of the controversial issues is optimal time of surgery after completion of chemotherapy. Usually, operation in 2-4 weeks after chemotherapy is recommended where 6 - 8 weeks are also suggested by some other authors. Lai and colleagues reported no statistically significant difference among breast cancer patients receiving surgery < 4 weeks, 4-8 weeks, or > 8 weeks after the last dose of NAC in terms of pCR, DFS, OS, surgical complications, and rates of conversion from mastectomy to BCS [53]. On the contrary Sanford et al. found that patients who received surgery>8 weeks after NAC have worse overall survival [53]. Variable time intervals ranging between 1 and 5 weeks from the last chemotherapy dose are suggested in trials of neoadjuvant setting. Wildiers et al. recommends surgery as early as 1–3 weeks after NAC particularly in patients with locally advanced disease [54]. Sparano et al. [55] suggests time interval of about 4 weeks for stage IIB-IIIC patients. Some other studies determine the optimal time interval as 28 days [56,57].
To accurately decide on this issue, recovery of the patient to suitably recover from possible myelosuppressive effects, extent and time of surgery must be taken into consideration. In a single institution retrospective research with 324 patients who had residual N1 nodal disease following NAC underwent axillary evaluation with either sentinel lymph node biopsy (SLNB) only or ALND were investigated. ALND was preferred for patients with clinically positive nodes at presentation, multiple positive nodes (micrometastasis or macrometastasis in 1 node or 2 nodes) during SLNB. SLNB was performed for good responders with limited nodal burden after NAC. After 71 months of follow up, no statistically significant differences between the two groups were found in terms of axillary relapse free survival (ARFS), distant metastasis free survival (DMFS), overall survival (OS), and breast cancer specific survival (BCSS) in both univariate and multivariate analyses. Conclusively, SLNB was suggested as a possible option for ALND in patients with breast cancer who have limited axillary residual disease after NAC without compromising outcomes [58].
According to American Society of Clinical Oncology (ASCO) guidelines SLNB was not recommended after NAC in the previous decade, however it is currently offered before or after NAC and a full ALND is considered as standard of care in case of confirmed nodal disease after SLNB [59,60]. Axillary dissection is also recommended as a standard approach for patients with residual tumor in nonsentinal lymph nodes even is sentinel node is found to be negative in ACOSOG Z1011 trial [61]. In patients with SLNB (+) in axillary evaluation after NAC, nonSLNB (+) rate is reported to be 40-70%; therefore, ALND is suggested in such patients. St Gallen and ESMO recommendations also in line suggesting ALND in case of macro or micro nodal residual disease [37].
Definition and Importance of pCR
Definition of pCR
Association between pCR and improved survival outcome after NAC has been reported plenty of trials [5,20] which necessitates a proper defining of the degree of response to chemotherapy. There is yet no accepted validated standard for clinical response assessment. Mukherjee et al. investigated the correlation between the clinical, radiological, and pathological parameters to assess tumor response after NAC. Clinical evaluation had a sensitivity and specificity of 73.5% and 88.5% respectively while sensitivity and specificity of radiological evaluation were 14.2% and 100% respectively. Authors suggested that clinical assessment shows a higher sensitivity compared to radiological assessment. However, the overall low sensitivity and specificity rates warrants a better method of evaluation [62]. Despite its extensive use in literature and decision making by physicians, definition of pCR is still a matter of debate. Typically, it is accepted as having no residual invasive disease in the breast or nodes on postsurgical specimen.
Three common definitions used by different investigators are:
i. ypT0 ypN0: absence of invasive cancer and in situ cancer in the breast and axillary nodes
ii. ypT0/is ypN0: absence of invasive cancer in the breast and axillary nodes, irrespective of carcinoma in situ
iii. ypT0/is absence of invasive cancer in the breast, irrespective of ductal carcinoma in situ or nodal involvement [63].
EMA reported 3 main definitions in their change proposal report about pCR:
Total pCR which means the eradication of invasive cancer from the breast and lymph nodes (ypT0/is ypN0, aka tpCR), breast pCR defines eradication of invasive cancer from the breast (ypT0/is, aka bpCR) and the specific definition of German Breast Group that necessitates eradication of invasive and in situ disease from breast and lymph nodes (ypT0 N0, aka GBGpCR). There are controversial suggestions on involving in situ disease in the definition of pCR. In the EMA recommendations, depending on the study by Cortazar et al. in situ disease is not included in pCR definition. Currently EMA recommends defining pCR as the absence of any residual invasive cancer on hematoxylin and eosin evaluation of the resected breast specimen and all sampled ipsilateral lymph nodes after NAC aka tpCR for registration of neoadjuvant trials [63-66].
FDA [67] recognizes one of the following pCR definitions for trial registration U.S. marketing approval:
i. The absence of residual invasive cancer on hematoxylin and eosin evaluation of the complete resected breast specimen and all sampled regional lymph nodes after NAC (i.e. ypT0/Tis ypN0 in the current AJCC staging system)
ii. The absence of residual invasive and in situ cancer on hematoxylin and eosin evaluation of the complete resected breast specimen and all sampled regional lymph nodes following NAC (i.e. ypT0 ypN0 in the current AJCC staging system) [67].
Impact of pCR on Treatment Outcome
Response to NAC is considered as an endpoint for survival in breast carcinoma, and association between attaining pCR and favorable long-term survival rates is reported previously [20,68,69]. Consequentially FDA recommended pCR as a primary end point in neoadjuvant trials. Mamounas et al. [70] reported tumour response as an independent predictor for LRR at 10 years. Chest wall recurrences were seen in 1/94 patients after mastectomy who achieved breast pCR with pathologic negative nodes irrespective of initial tumour size and clinical nodal status. Interpretation of the results from two prospective NSABP trials revealed lowest LRR rates of 0 and 6% for tumours of 5 cm or less, and larger than 5 cm, respectively in patients achieving pCR, pN0 after NAC [70]. Authors concluded that LRR rates significantly increases with tumor size irregardless of initial clinical nodal status and pathologic response to chemotherapy (cN0: 11.2vs 14.6%; cN+: 17.0 vs 22.5%). Two subgroups of patients with low LRR risk, denoted as <10% even without PMRT defined by Fowble et al. [71] are: (a) Clinical Stage II (T1-2 N0-1), pN0or 1–3, ER+, no ECE, LVI; >40 years; (b) T3N0 (Stage IIB). Patients presenting with T3N1 with pCR and >40 years.
In a meta-analysis of 1955 patients, it is confirmed that, patients who achieved ypT0-TisN0 status after NAC had improved event-free survival and OS [5]. Deferring PMRT can be suggested for patients with pCR, however there is considerable amount of data reporting high rates of LRR despite pCR in select group of patients. Most prominent long-term benefit of pCR achievement was shown in patients with aggressive subtypes such as triple negative, Her 2 overexpressed, high-grade ER (+) and Her 2 negatives. This finding makes sense taking the published evidence about variable pCR response in different molecular subtypes. ER-positive tumors (Luminal A) are found to be less responsive to chemotherapy [72] and only disease-free survival improvement is reported after pCR in triple-negative/basal-like, HER2-positive (nonluminal), or luminal B (HER2-negative) tumors [22]. Basal-like tumors is the only one among six identified subtypes of triple negative breast cancer, revealing the existence of association between genomic signature, pCR, and survival [73]. Therefore, it’s noteworthy to mention the necessity of stratification according to biological subtype for accurate individualization of whole treatment strategy [62].
Indications for PMRT in Neoadjuvant Approach
After NAC, an indisputable consensus has been formed in the direction of strongly suggesting postoperative radiation therapy after breast-conserving surgery, regardless of the pathological response. However, PMRT following NAC is a matter of debate while prospective data to guide our decision is scarce and not supported with high-level evidence. Therefore, mostly extrapolated data from retrospective trials and received information on locoregional recurrence predictors from previous adjuvant chemotherapy studies are used to help for critical decisions.
PMRT was recommended according to initial stage in the early times of neoadjuvant treatment era. This approach has been largely abandoned due to published reports on high rates of pCR following NAC [74]. The retrospective studies and several prospective investigations which are consulted to develop the guidelines have many limitations such as heterogeneity of sample sizes, and initial stage at presentation, application of outdated chemotherapy schedules, and PMRT decisions directed by nonuniform indications [75].
Retrospective data is obtained from the analyses on women who were not given PMRT after NAC to assess the effect of specific patient and tumor features on LRR. Some additional studies reported the retrospective comparison of groups with or without PMRT. Interpretation of a recent study investigating the relaps pattern after NAC leads us to number of conclusions as: higher recurrence rate in triple negative breast cancer (TNBC) patients, earlier (within 3 years) recurrence TNBC and HR (hormone receptor)-/HER2+ subtypes, initial stage III disease, grade 3 tumours, non-complete pathologic response and no adjuvant radiotherapy were independent predictors of inferior recurrence-free survival [76]. A data from 416 patients with stage II/III also confirmed the association of triple-negative subtype, stage III disease, and non-pCR with inferior DFS and OS [77]. Buccholz et al. [78] examined 150 patients which of 55% were clinical stage IIIA disease or higher, in terms of risk factors for postmastectomy LRR after NAC (78). Conclusively, presentation with clinical stage IIIB or higher disease, >4 pathologically positive nodes, and no use of tamoxifen were stated as predictors of predictors of LRR. Huang et al. stated skin/nipple involvement, supraclavicular nodal disease, no tamoxifen use, extracapsular extension, and estrogen-receptor negative disease as predictor of 10-year LRR in this cohort in their study [79]. 10-year LRR rate was less than 8%, for patients with < 2 of these factors, where it was found 28% in patients with > 2 of the abovementioned 5 risk factors.
NSABP-18 and NSABP-27 are the two large prospective randomized controlled trials which provides evaluation of LRR pattern after NAC and mastectomy [8,10,20]. Kishan et al. [34] investigated the predictors of LRR in a multivariate analysis. They stated clinical tumor size >5 cm (HR 1.58), clinically positive nodal status (HR 1.53), pathologic incomplete nodal response (HR 4.48) and pathologic incomplete breast tumor response (HR 2.2) as significant predictors. However, clinical tumor size was not a significant predictor for patients treated with breast-conserving therapy. Only 7.5% of initial cN+ disease achieved pCR in the breast and nodes although 10-year LRR was 0.0% in that subgroup. Ten years LRR rates were greater than 10% for patients with ypN + disease regardless of initial nodal status, initial tumor size or pathologic N stage. Conclusive interpretation determined that: Age<50y, tm>5 cm, cN +, noncomplete response in primary and/or nodal disease are independent predictors for locoregional recurrence [20]. Considering all data, it can be stated that the risk of LRR depends on both initial clinical stage and pathologic factors emerged in postmastectomy specimen. And pathologically confirmed response does not rebut locally advanced initial clinical status.
The benefit of PMRT in patients with T1-2N1M0 who were ypN0 after NAC was assessed in a prospective randomized study on 142 eligible patients. After median follow up of 72 months. Univariate analyses showed that 5-year RFS was 88.7 % in PMRT group where it was 72.4% in patients without PMRT (p = 0.028), however, no difference was detected in terms of LRFS, and OS rates between the two groups. PMRT was also confirmed as a significant prognostic factor for RFS in multivariate analyses (HR, 0.411;95% CI, 0.175–0.968; p = 0.042). After a Propensity score matched analysis PMRT remained significant. In the subgroup of patients with pathologic complete responses (ypT0, and ypN0) after NAC, PMRT was not effective on RFS. Conclusive statement of this study was for initially pT1-2N1M0 patients who achieved were ypT0, and ypN0 after NAC; omitting PMRT might be considered [80]. In the analysis of 8,321 patients with cN1 and cN2 disease adjusted survival analysis revealed improved overall survival with PMRT for both cN1 (P < 0.01) and cN2 (P < 0.01) disease. In the patients who achieved ypN0 after NAC, no survival difference was detected except in case of hormone receptor negative status who had survival benefit with PMRT (P < 0.01) (24). Cao et al. [81] also reported significant improvement in locoregional recurrence-free survival (a 5-yearrate of 94.7% vs. 72.9%), distant metastasis-free survival (a 5-year rate of 92.8% vs. 75%) and disease-free survival (a 5-year rate of 92.9% vs. 62.5%) with PMRT who were T1-2N1 at presentation in case of achieving ypN0. Conclusively postmastectomy radiotherapy was indicated as the only significant prognostic factor affecting locoregional recurrence-free survival via univariate analysis in T1-2N1 patient subgroup [81]. Wang et al. [82] suggested a nomogram to determine the patients who will benefit from PMRT after NAC among stage cT1-2N0-1. According to multivariate analysis pathological N stage, lymph-vascular invasion, and histological grade were reported as independent prognostic factors for LRR. Based on this data, a nomogram was generated for cT1-2N0-1 patients, and they were classified as low- and high-risk. No improvement was detected in LRR for low-risk group with PMRT, where adjuvant radiotherapy significantly decreased LRR in high-risk patients.
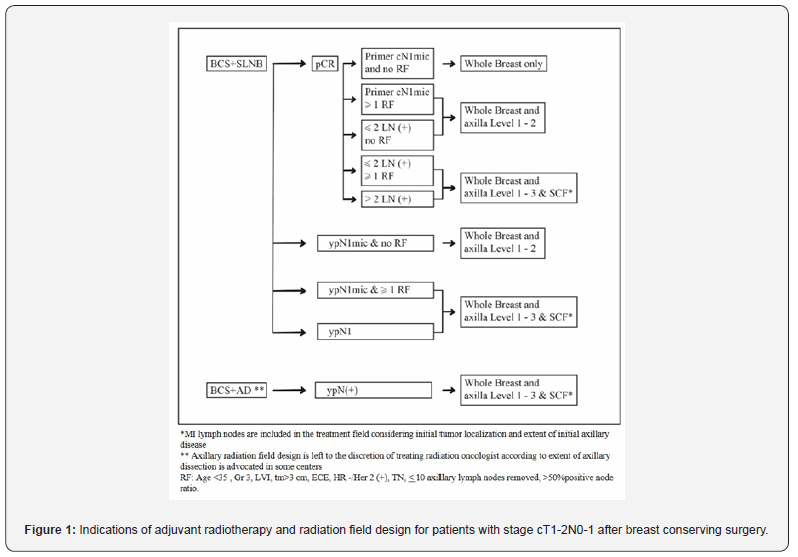
Interpretation of the above-mentioned data regarding indications of adjuvant radiotherapy and radiation field design for patients with stage cT1-2N0-1 is summarized as diagrams after breast conserving surgery and mastectomy in Figures 1 & 2 respectively. Acquired conflicting data on LRR benefit to date is not sufficient to end the debate for patients with stage II disease who achieve a pCR in terms of suggesting PMRT or not. However, in stage II or stage III disease who achieve a nodal pCR, specific features possibly portending higher LRR rates such as residual breast disease, young patient age, may support the necessity for PMRT [70]. Locoregional control and overall survival benefit with PMRT after NAC was reported in one of the earliest studies on this issue by Abdel-Wahab et al. [83]which investigated 55 breast cancer patients with cT3–4 or cN2–3 [83]. After 47 months of median follow up LRR rates were 7% and 31% for patients with and without PMRT respectively which turned out a 3-year OS benefit (88% versus 46%).
A recent retrospective analysis on 554 clinical stage II-III breast cancer patients who underwent NAC and modified radical mastectomy investigated the relationship between LRR, DFS, and clinical pathological characters. After 65 months of median follow-up time 5-year cumulative incidence of LRR and DFS was 9.2% and 74.2%, respectively. 5-year LRR of the patients with PMRT (7.3% vs. 14.1%, P = 0.01) decreased significantly. Benefit of PMRT was more significant particularly in patients with ypN1 and ypN2-3. However, no significant benefit was found with PMRT in patients who were ypN0 after NAC. The authors concluded that PMRT after NAC significantly reduced the LRR of patients with clinical stage II-III breast cancer particularly those with ypN1 and ypN2-3 [84].
Huang et al. found that 10-year LRR rate was 22% without PMRT while it was 11% with PMRT for patients with stage II - Stage IV (ipsilateral supraclavicular lymph node) disease [79]. Although multivariate analysis revealed a significant benefit in favor of PMRT; 10-year cause-specific survival (CSS) was identical between these groups. Additionally, in the subgroup of patients with stage IIIB or higher disease, cT4 tumors, and four or more involved nodes significant improvement in CSS was demonstrated with PMRT. Adjuvant radiotherapy indications after surgery in patients who underwent NAC is mostly investigated according to specific subsets of interest. In a report by MDACC 10-year LRR rate was found 10% in 132 patients with clinical stage I - II disease without PMRT after NAC [85]. Five years LRR was 5%; for patients with cT1–2 disease and one to three positive nodes in the same study. The authors reported cT3N0 disease, four or more nodes at surgery, and age 40 years or younger as significant predictors of LRR.
In the ACOSOG Z1071 trial which enrolled cT0-4N1-2 breast cancer patients RT was associated with a trend toward improved locoregional control however, no association was found between RT and overall survival, BC-specific survival, or Disease Specific Survival. PMRT is suggested for triple-negative BC patients due to higher locoregional relapse rates [61]. Nagar and colleagues investigated the benefit of adjuvant radiotherapy after NAC in cT3N0by comparing 119 patients who underwent PMRT with 43 who did not [86]. Despite higher rates of <40 years of age and more ypN+ disease in the PMRT group, 5-year LRR rate was 24% and 4% for patients without and with PMRT respectively. The 5-year LRR was 14% for patients who were ypN0, and the subset analysis led to the conclusion of significant locoregional control benefit for patients with ypN+ or high-grade disease with PMRT [86].
The overall 5-year LRR rate was reported as 16.1% for cT1 to T3/N0 to N3 in the series by Nagar and colleagues. PMRT provided a significant reduction in LRR (HR 0.25), which translated into a 5-year disease-free survival improvement (91.3% versus 64.8%) [87]. Pathologically evaluated primary tumor and nodal status after NAC were significantly associated with an increased LRR. The authors attributed their results revealing no significant association between initial clinical factors and LRR to inadequate upfront staging.
Retrospective analysis from MDACC [79,88] investigating Stage III–IV patients who had pCR after NAC found 10-year LRR rates of 3 and 33% in the subgroups with or without PMRT respectively (p = 0.006). A recent study by Zhang et al. [89] investigated the outcome with PMRT after NAC. The authors concluded that PMRT significantly improved overall survival in initial T3N0-T4N3 disease and revealed a significant benefit in terms of LRR-free survival and DFS for patients presented with stage T2N0-T4N3.
Ohri et al. [90] investigated impact of PMRT for patients without pathologic complete nodal response. In patients with ypN1 and ypN2 5-year OS rates were similar with or without PMRT but in ypN3 patients PMRT provided significant survivaş benefit (66% vs. 63%, p=0.042). After adjusting for patient and tumor variables in multivariate analysis, PMRT was assessed as an improving factor for overall survival only among ypN3 patients [90]. Another analysis from National Cancer Database on clinical stage II/III breast cancer patients with ypN0 found no benefit of PMRT for the entire cohort [91]. However, in the subgroup of patients with cT3/4, ypT1/2, or clinical stage IIIB/C, OS was significantly better with PMRT. In the study of Japanese Breast Cancer Registry 3226 cT1–4 cN0–2 patients who underwent NAC and mastectomy were investigated in terms of effect of PMRT on outcomes. LRR, DDFS, or OS were not improved with PMRT in ypN0-1 patients. However, in patients with ypN2–3 radiotherapy improved LRR-free survival (p\0.001), DDFS (p = 0.01), and OS (p\0.001). An authors concluded that PMRT may be beneficial only for patients with ypN2–3 after NAC [92].
A significantly lower 10-year LRR rate with PMRT (33% versus 3%) was reported by Huang et al., in 46 patients with clinical stage III disease or higher who experienced a pCR [87]. On the contrary patients with clinical stage, I and II disease and a pCR, did not seem to have significant benefit from PMRT in terms of LRR. In another study investigating LRR rates in 106 patients with noninflammatory breast cancers treated with NAC and mastectomy who had pCR, confirmative results were reported [93]. The authors found 10-year LRR rate of 0% for clinical stage I–II disease, without considering PMRT. However, PMRT significantly improved 10-year LRR rate (33.3% versus 7.3%) in 74 patients with stage III disease. Additionally, this improvement eventuated in significant improvements in CSS (40% versus 87%) and OS (33.3% versus 77.3%). Meattini et al. [94] reported their results of 170 patients with clinical stage II–III breast cancer after NAC. They found no significant survival benefit of PMRT for patients with pCR or downstaging. Authors concluded that PMRT improved overall survival only in patients with initial T3 disease. Xin et al investigated the prognostic significance of number of negative lymph nodes (NLN) removed after NACT in a retrospective study [95]. The authors found that, PMRT improved OS (p < 0.05) and DFS (p < 0.05) in the subgroup of patients with ypN1 stage, and ≤13 NLN, however no benefit of PMRT was detected in patients with >13 NLN even they were ypN1 after NAC.
These abovementioned data conclusively suggest PMRT for patients with clinical stage III disease without considering their pathologic response. Some other studies reported refutatory results making PMRT decision more complicated. In a study from Institut-Curie, 134 patients with a nodal pCR after NAC were evaluated. These patients underwent PMRT following mastectomy or mastectomy alone [96]. Ten years LRR was 3.8% and 13.2% for patients with and without PMRT respectively. OS was reported 77.2% and 87.7% for the same groups and none of them were statistically significant. It is worth to note that in the subset analysis of 50 patients with initial clinical stage III disease, also no significant difference was found. Similarly, Shim et al. from Korea investigated 151 patients with clinical stage II–III disease who had a nodal pCR after NAC and could not report any significant difference in 5-year LRR rates with or without PMRT [97]. In this study, age (⩽40 years old versus >40 years old) and pathologic T stage were associated with higher LRR and CSS, while no impact of PMRT was found on either.
Despite the lack of molecular subtype information and concurrent administration of tamoxifen with chemotherapy, results from NSABP-27 determined that 10-year LRR rate following NAC and mastectomy is relatively low without PMRT. To interpret this study accurately, it must be kept in mind that NSABP-18 and 27 enrolled higher rate of lower risk patients compared to other retrospective reports mentioned above. The cT1–2N0 disease conformed 55% of the cohort, 20% were cT1–2N1, 16% had cT3N0 and only 9% of patients were cT3N1. Nevertheless, these data encouraged some studies to investigate omission of PMRT in patients with clinical stage II disease who achieve a pCR [98-100]. Fowble et al. used ACR Appropriateness Criteria modified Delphi methodology to determine PMRT indication in their review [71]. The authors stated that patients with stage T1–2N0–1, older than 40 years, ER (+), < 4 positive axillary nodes, without lymphovascular space invasion or extracapsular extension may not benefit from PMRT while the expected 5-year LRR of these patients were less than 10% [71].
Evidence about other specific subsets are scarce. Clinical multifocality and multicentricity or Her2 positivity do not appear to be associated with increased risk of LRR [86,100,101] where the risk of LRR was increased in triple negative status [79,103]. Wright et al. [103] reported that seven of nine regional recurrences were seen with triple negative histology [103]. And they found that LRR and DFS curves differed significantly amongst the three molecular subtypes (HR+ and HER-2−, HER-2+, and TNBC). Five-year LRR of TNBC patients was found to be significantly higher compared to HR+ and HER-2− patients (15.3% vs. 5.6%, P = 0.006). LRR and DFS was not significantly different in any of three subtypes with PMRT. However, the benefit of PMRT was indicated in the survival curve of TNBC patients (P = 0.065). Pathologic complete response rates were higher in TNBC and HER + patients than HR+ patients after NAC (P = 0.001; and P = 0.02) [84]. In their study with 227 cT1-T3N0 and pN0 patients, Crown et al. [104] also found 3-year actuarial LRR rates 10.1% where it was 3.2% for hormone receptor-positive and human epidermal growth factor receptor 2-negative (HER2-) patients. The authors indicated the necessity of PMRT/RNI for node-negative TN patients. The behavioral pattern of different molecular subtypes in terms of response to NAC and benefit from PMRT is also investigated. In Korean study of 189 patients PMRT was associated with greater LRC in the luminal subtype (p = 0.046), but not in other subtypes. As an additional comment, the authors indicated that PMRT provides no additional survival benefit in patients with ypN0 regardless of molecular subtype [105].
Effect of age is investigated in a focused study on patient younger than 35 years. The authors reported improved 5-year LRR (12% versus 37%) and OS (67% versus 48%) for patients who received PMRT despite a greater ratio of adverse features in this group [106]. Although higher survival results achieved in favor of PMRT in all stages, it reached statistical significance for patients with clinical stage IIB disease. Age younger than 40 years is determined as a significant predictor of LRR also in another study from Korea [97]. Impact of delay in PMRT after NAC is investigated by Desai et al. [107]. Clinical stage II, and III 248 patients were retrospectively reviewed. Patients were evaluated in three groups as PMRT within 8 weeks, 12 weeks, and 16 weeks. No significant difference was found in locoregional outcome between patients who underwent PMRT within 8 weeks versus >8 weeks (p = 0.634), < 12 versus >12 weeks (p = 0.332), or < 16 versus >16 weeks (p = 0.549) after surgery. Conclusively initiation of PMRT up to at least 16 weeks was suggested, however PMRT should be offered even after 16 weeks in those without early locoregional recurrence [107].
Post-NAC PMRT portal design: Individualization of Radiation Treatment Fields
PMRT fields after NAC is not fully defined to date especially in patients with <4 lymph nodes and data regarding selection and designation of radiation portals for PMRT after NAC is scarce. The pioneer of publications investigating the role of regional nodal irradiation (RNI) after NAC was the one by Daveau et al. [108]. This retrospective study enrolled 249 patients presented with initial cN0-2 disease who were ypN0 after NAC. RNI added to whole-breast irradiation in 63.7% of these patients. Results of this cohort was not able to detect any subgroup who has a benefit in terms of LRRFS, DFS, or OS in univariate and multivariate analyses. However, as expected there was a selection bias as higher rate of clinical lymph node involvement, younger median age and more medial/central tumor location in patients treated with RNI. In their recent study, Fayanju et al. [109] found no significant benefit with nodal radiation in ypN0 patients, but it was almost significant for ypN1 patients (hazard ratio (HR) 0.83, 95% CI 0.69–0.99, p=0.04, overall p-value=0.11). After lumpectomy, nodal radiation improved OS both in patients with ypN0 (HR 0.38, 95% CI 0.22–0.66) and ypN1 (HR 0.44, 95% CI 0.30–0.66, both p<0.001), but the benefit provided by nodal irradiation was not significantly distinct compared to the benefit of breast-only radiation. In another retrospective study investigating the same issue by analyzing 98 patients with cT3/4 or cN+ breast cancer with ypN0/1-status after NAC, supraclavicular lymph nodal region was treated in 57% of patients in addition to whole-breast or chest wall irradiation [110]. Confirming the results of the above-mentioned study no benefit of RNI was detected for ypN0-patients in terms of LRRFS or DFS. However, DFS was significantly worse for ypN1 patients without RNI. In the Korean experience of RNI in 260 stage II/III patients who were ypN0 after NAC [111] all patients had BCS. Regional nodes were treated in 52% of the cohort, 5% including IMN. Regional radiotherapy had no impact on LRRFS or DFS.
In the study including 464 patients who underwent PMRT after NAC, reported by Wright et al. [103] 17.5% of the cohort underwent only chest wall radiation. Overall, 5-year LRR rate and LRR rate for ypN0 patients was 5.8%, and 1.9% respectively. Chest wall only irradiation, ypN+ status and triple negative histology was found significantly associated with LRR in multivariate analysis (HRs of 3.39, 10.23 and 8.5, respectively). Conclusively, no subgroup of patients without any locoregional control benefit by irradiating supraclaviculary region was indicated. However, the authors stated likely limited benefit for patients who achieved nodal pCR due to low rate of LRR.
National Cancer Database (NCDB) analysis is the largest study on this concern with 15,315 cT1-3 cN1 breast cancer patients included [112]. Detail of regional radiotherapy field extent (axilla, supra-/infraclavicular region, IMN) was not given in the full text. Propensity score matching was used to obviate the imbalances in patient characteristics. No benefit was provided with adding regional fields to whole breast or chest wall portals in terms of OS regardless of pathologic nodal response to NAC or type of surgery. Due to lack of information, locoregional control or DFS were not reported.
NSABP B18 and B27 trials reported nodal failures below 5% without axillary irradiation [70]. This result is compatible with general acquired data determining those regional nodal recurrences are uncommon after NAC [79,83]. Wright et al. [103] also reported 1.9% recurrence rate after NAC confirming this background. Besides, none of the previous studies has provided evidence conferring a lower locoregional control or survival rates for cN0 and pN0 patients by deferring axillary radiotherapy when an optimal dissection is performed [108,113]. Some high-risk clinical features are defined in the literature as indication of regional nodal irradiation. Truong et al. showed that age <45 years, Stage T2, histologic Grade 3, ER-negative disease and positive node ratio >25% are associated with a 10-year LRR risk of >20% in patients with 1-3 positive nodes [114]. In IBCSG 23-01 (42) study 934 patients with T2 or less invasive breast cancer and at least one sentinel lymph node containing micrometastatic disease were randomized to ALND versus no ALND. Omission of ALND was noninferior in terms of 5-year DFS or OS.
The Canadian study MA 20 [115] randomized 1832 node positive or high-risk (>T3 or greater, > 2 cm or greater with fewer than 10 nodes removed, G3 or greater, LVI positive, or ER) node negative breast cancer patients to receive WBI alone or WBI plus RNI, including axillary, supraclaviculary and IMN regions. For patients with less than 10 nodes removed or more than 3 positive axillary nodes, the lateral aspect of tangential field was extended to include axillary levels 1 and 2. At a median of 9.5 years of follow-up, no significant difference in OS between the two groups was detected (82.8% and 81.8% in the experimental and control arms respectively). However, DFS was statistically higher in nodal radiotherapy arm (82% vs 77%. HR 0.69, p = 0.05) which reveals a relative improvement of 24% regarding DFS with the addition of RNI. These results ensure persuasive evidence of a significant reduction in both locoregional and distant disease recurrence with nodal irradiation in node positive or high-risk node negative patients although no survival advantage has been indicated. Gilliot et al. compared PMRT including RNI and tangent-only [116]. The authors reported similar 10-year LRR rates in both groups (95% without and 91% with RNI). However, both 10-year OS rate (96% versus 75% p < 0.05) and distant-metastasis-free survival (97% versus 78%) were significantly higher without RNI. This unexpected result is attributed to imparity between groups such as significantly higher rates of cT3–4 tumor, larger residual tumors among patients who underwent RNI.
Kishan et al. [34] suggests RNI after NAC for patients with ypN+ disease, and for patients initially with cN2–N3 disease. Unless adverse factors such as <10 nodes removed, >50% rate of positive nodes and vascular adherence, whole axilla would not be covered if full axillary dissection is performed. In patients initially cN0–N1 who achieved nodal pCR after adequate axillary staging, to decide supraclavicular or IMN treatment factors such as lymphovascular space invasion, age, initial clinical nodal status, size and biologic features of breast residual disease. In case undissected axilla or inadequate number of evaluated lymph nodes, RNI must be considered.
Shahar et al. [117] suggested to limit the axillary radiation fields with levels I-II for stage II disease with 1 positive sentinel node in case of no lymphovascular invasion, however; they recommended extending the treatment portal to encompass both the upper level of the axilla and the supraclavicular areas in the presence of >1 axillary nodes. Another study reported a significant association between extending irradiation fields to supraclaviculary region and LRR on multivariate analysis (HR: 3.39; p = 0.024) [103]. As their date revealed higher isolated regional nodal failure rate in triple negative patients, the authors also recommended systematical irradiation of supraclavicular area in this subgroup. Wright et al investigated the same issue in a larger study [103] and stated the necessity of including a supraclavicular field but no comment was mentioned in terms of IMNs while only 5% of patients were estimated to have IMN irradiated.
Two trials investigated the effect of nodal irradiation in outcome: The EORTC 22922/10925 trial, randomized patients who either had medial tumors (regardless of nodal status) or pN+ disease to receive RNI or no RNI [118]. The National Cancer Institute of Canada Clinical Trials Group (NCIC CTG) MA.20 trial randomized women with node-positive disease, T3 disease, T2 with <10 nodes removed in addition to at least one of the following: grade 3 histologic categorization, estrogen receptor negativity or lymphovascular invasion to RNI or no RNI [115]. Nodal field includes supraclavicular fossa/level III axilla, the IMNs and level I–II of the axilla in selected circumstances in MA.20 trial, medial supraclavicular nodes, IMNs, and axilla in a small subgroup (7.4–8.3%) in the EORTC trial. Both trials reported a significant improvement in disease-free survival, locoregional control and distant metastasis free survival however no difference was found in 10-year OS.
Investigations addressing the challenge of selecting patients who will benefit from IMN irradiation after NAC are lacking in the literature. MDACC reported that IMN adenopathy was clinically detectable by imaging in up to 10% of patients with locally advanced disease and including IMN in those patients results in excellent 5-year IMN and locoregional control [119]. Importance of involving internal mammary node region is reported in the study by European Institute of Oncology in Milan [120]. In this cohort of patients IMC positive rate was 10% in tumours located in medial quadrants which is 27% of all cases with regional node metastases. Adding IMC to radiotherapy port in those patients resulted in excellent survival rates (95% at 5 years), indicating the importance of including this region in treatment volume in cases judged necessary (120). Kim et al. consequently addressed the issue of targeting of the internal mammary nodes after NAC in stage II-III patients [121]. The 55% of 521 patients enrolled underwent radiotherapy including the IMN. After propensity score adjustment to eliminate the imbalances between groups, treating IMN was found to improve 5-year DFS from 64.1 to 80.5%, particularly in patients with cN1-2, ypN1-2, inner/central tumors, and triple-negative tumors. Additionally, patients with ypN1 also had a significantly improved OS (hazard ratio (HR)) 0.28, 95% confidence interval (CI) 0.11–0.70) with IMN radiotherapy.
Considering the published data patients with;
A- Initially cT3–4 disease regardless of pathologic response at the time of surgery
B- Initially cN2–3 disease regardless of pathologic response at the time of surgery
C- Residual nodal disease after NAC
D- Initial clinical stage II disease who achieved pCR but ⩽40 years old
E- Residual tumor > 2 cm in the breast
F- with high grade histology
G- with triple negative histology
are suggested to have PMRT [34,71,122].
When these reviews, retrospective studies are totally interpreted; detailed evaluation of pre - NAC clinical stage with circumstantial physical examination and appropriate imaging tools, post-NAC pathologic stage and individual patient and tumor factors are warranted for proper individualization of PMRT decision and radiation field design after NAC. The randomized NSABP51/RTOG 1308 trial and nonrandomized RAPCHEM study are 2 ongoing trials addressing the issue of PMRT indications and risk adapted radiotherapy after NAC stratified according to clinical and biologic risk factors.
Limitations
Due to mutual effect between tumor biology, treatment response, clinical stage, and postoperative pathologic tumor stages, the interpreted studies investigate heterogeneous patient subgroups when reporting patients received PMRT/RNI and who did not. Second limitation is the selection bias resulting in more favorable patient population in terms of clinical and pathologic stages, age and treatment response for the observation group [123]. Another limiting issue is outdated chemotherapy regimens and lack of HER2-targeted therapy [124,125], both of which could result in misinterpretation of the adjuvant radiotherapy effect such as over- or underestimation of its potential benefit. As the beneficial role of PMRT irregardless of treatment response in inflammatory breast cancer is out of dispute [126,127], studies with inflammatory breast cancer were not evaluated in this review.
Conclusion
Decision of administrating PMRT after NAC is a controversial issue and warrants evaluation by a multidisciplinary team particularly with a radiation oncologist. Careful and detailed consideration of initial clinical stage, post NAC pathologic stage, and individual patient and tumor factors is recommended for individualization of treatment in terms of PMRT after NAC. Accurate initial staging is a sine qua non for designation of adequate treatment strategy customized for every individual patient. Axillary ultrasound enabling FNA of suspicious lymph nodes where available, MRI for all patients regarding detailed breast evaluation and PET-CT for all node-positive patients are recommended to achieve satisfactory initial clinical staging. Assessing axillary status after NAC with SLNB with dual-tracer method and removing at least three nodes is deemed sensible. Decision regarding radiation portal design is extrapolated from the data on standard PMRT situation. Interpretation of previous retrospective and prospective data in the literature indicates high LRR after mastectomy in patients with cT3–4 disease, cN2–3 disease and ypN+ disease, therefore PMRT is justified for these patients. Initially clinical stage II disease (excluding cT3N0) with pCR, some suggests omitting PMRT. However, for patients with young age, greater than 2 cm breast tumor residue, high-grade or triple negative histology, PMRT should be offered. Patients with stage III disease are suggested to receive adjuvant RT regardless of pathological response. However, there are still controversies regarding the role of PMRT in patients with stage II disease with clinical nodal involvement or cT3N0 who achieved pCR or ypN0. With the available evidence to date, PMRT should be considered in patients with additional risk factors such as young age, ER/PR-negative, lymphovascular invasion, residual tumor in the breast for stage II disease and clinically involved lymph nodes in case pCR/ypN0. RNI is recommended for patients presented with clinically positive lymph nodes regardless of neoadjuvant treatment response particularly in cases with adverse features as young age, ER/PR-negative, lymphovascular invasion, residual tumor in the breast. For patients with ypN1-3 after NAC, PMRT should be performed because of known high regional recurrence rate of this subgroup [70].
References
116. Gilliot O, Durando X, Abrial C, Bellière A, Gimbergues P, et al. (2010) Does regional lymph node irradiation improve the outcome of N0 and pN0 breast cancer? Cancer Invest. 28(2):195-200.
118. Poortmans P, Struikmans H, Collette S, Kirkove C, Budach V, et al. (2014) Lymph node radiotherapy improves survival in breast cancer: 10 year results of the EORTC ROG and BCG phase III trial 22922/10925. Radiother Oncol 111: S206.






























