Nivolumab Induced Thrombocytopenia; A Case Report
Asmaa El Hussainy* and Jasem Al Barrak
Department of medical oncology, KCCC, Kuwait
Submission: September 02, 2021; Published: September 09, 2021
*Corresponding Address: Asmaa El Hussainy, Department of medical oncology, KCCC, Kuwait
How to cite this article: Asmaa El H, Jasem Al B. Nivolumab Induced Thrombocytopenia; A Case Report. Canc Therapy & Oncol Int J. 2021; 19(4): 556020. DOI: 10.19080/CTOIJ.2021.19.556020
Abstract
Background: Immune checkpoint inhibitors (ICIs) are a class of drug that are increasingly being used in different cancers. The extent of response to treatment with ICIs differs among individuals. Immune checkpoint inhibitors (ICIs) induce various immune-related adverse events. Life-threatening hematotoxicity can be provoked by ICI therapy. Although ICI-related endocrinopathy and interstitial lung disease have been well documented, hematotoxicity is relatively rare. We describe a case of nivolumab induced thrombocytopenia after two courses of therapy for hepatocellular carcinoma.
Case presentation: is a 70 year old male patient with a hepatocellular carcinoma since November 2017 was treated by nivolumab as a second line systemic therapy at our institute. His laboratory data indicated thrombocytopenia suspected to be an immune-related adverse event following two courses of nivolumab.
Conclusion: hematologic toxicities induced by ICIs, including thrombocytopenia, are viewed as rare irAEs, and increased application of ICIs in advanced malignancies contributes to increased reports of immune thrombocytopenia.
Keywords: Nivolumab; Immunotherapy; Gastric cancer; Thrombocytopenia
Abbreviation: ICI: Immune Checkpoint Inhibitors; Iraes: Immune Related Adverse Effects
Introduction
Immune checkpoint inhibitors such as anti-programmed cell death protein 1 (PD-1) antibodies or anti-cytotoxic T-lymphocyte–associated-4 (CTLA-4) antibodies are monoclonal antibodies that combine immune checkpoint molecules and inhibit their function. The PD-L1 molecules on the cell surface of cancer cells or stromal cells inhibit T lymphocytes from attacking oneself and evade immune-system self-monitoring. Therefore, T-lymphocytes could attack cancer cells if anti-PD-1 antibodies inhibited the mechanism [1].
Nivolumab is a programmed death receptor-1 (PD-1)-blocking antibody indicated for the treatment of patients with hepatocellular carcinoma who have been previously treated with sorafenib, as a single agent or in combination with ipilimumab according to CHECKMATE-040, a multicenter, multiple cohort, open-label trial. The most frequent serious adverse reactions (reported in ≥4% of patients) were pyrexia, diarrhea, anemia, increased AST, adrenal insufficiency, ascites, esophageal varices hemorrhage, hyponatremia, increased blood bilirubin, and pneumonitis [2]. Events such as immune-related adverse events (irAEs) have been reported. However, the occurrence of hematological irAEs is rare. Some studies have reported on nivolumab-induced thrombocytopenia in lung cancer and melanoma, renal cell carcinoma. But our case today has documented thrombocytopenia as an effect of nivolumab for hepatocellular carcinoma treatment [3,4].
Case Presentation
November 2017: Mr. A was investigated for uncontrolled Rt hypochondrial pain, where CT scan had shown multicentric hepatocellular carcinoma at seg II, III, VIII. Figure 1. He was in good general condition, child Pugh score = A, AFP= 35 he was fit to start treatment but unfortunately, he declined treatment. May 2018: again, he was presented to us to start treatment, at beginning he received Y90 (TARE) to seg II. that was followed by TACE to lesions at seg III, and VIII. With marked decrease in AFP from 35 to 19, CT scan post ablation showed no active disease, so he was kept under follow up (Figure 2) during his course of follow up his CT scan has shown stable disease.
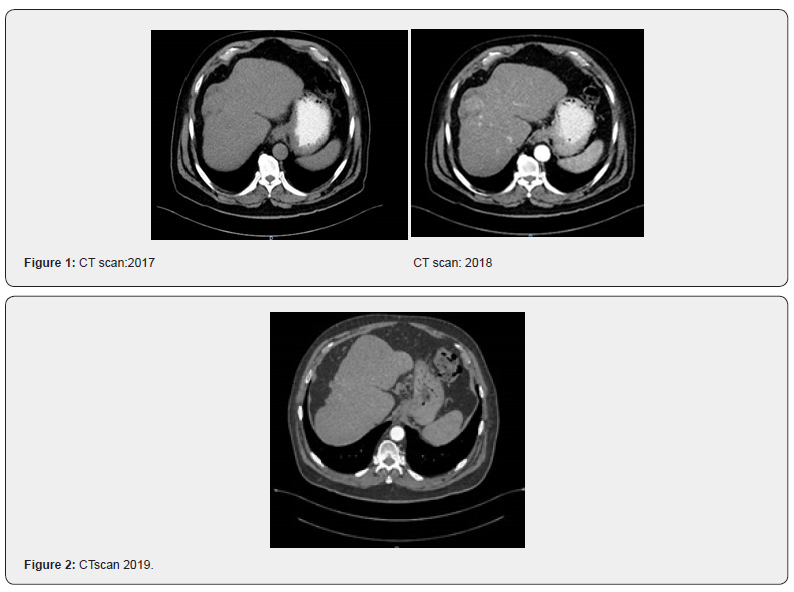
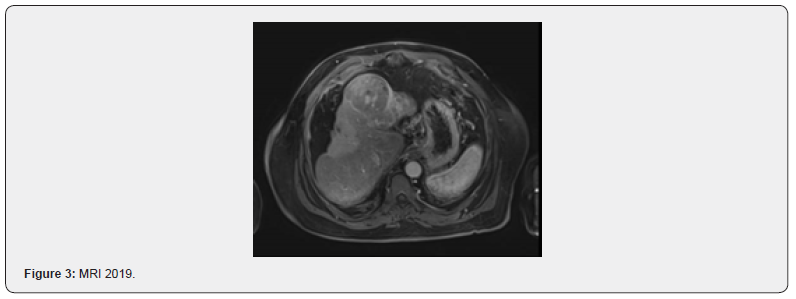
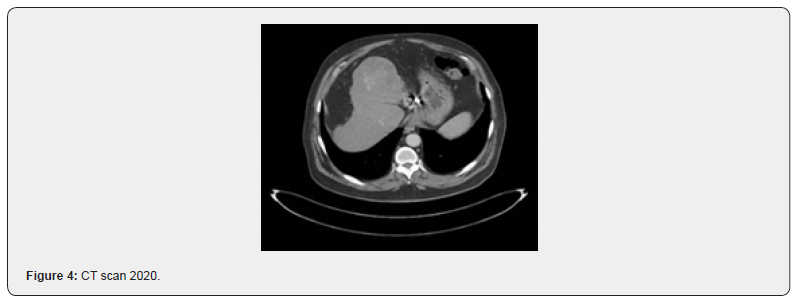
October 2019: CT scan has shown variable tumour response, MRI has confirmed disease recurrence, at seg III, and VIII (1.6 cm and 1 cm respectively) (Figure 3). He was started on LENVATANIB with good response tolerating treatment well till September 2020 where CT scan has shown progression in his disease; multiple new hepatic focal lesions, AFP 8.3, patient was in good condition ECOG PS= 1, child Pugh score = A, the decision was made for 2nd line of treatment (immunotherapy -nivolumab) (Figure 4). he received the 2 doses of that treatment without complication.
Nov 2020: he presented for his due 3rd cycle, but unfortunately his platelet count was low = 45, even not recovered after one week holiday of treatment. Immunotherapy was stopped at that time, and He was seen by haematology team where blood film has shown no abnormal cells; true thrombocytopenia. Bone marrow aspirate: no dysplastic changes, megakaryocytes adequate without dysplasia CG; diploid karyotype. So as his liver disease is stable, with no stigmata of chronic liver disease, no hepatosplenomegaly this isolated thrombocytopenia mostly immunotherapy induced he was received a boluses dose of steroid with gradual increase of platelet count, his platelet count was recovered to 56.
CT scan for disease evaluation has shown further disease progression, so the decision was to start him on 3rd line of treatment (regorafenib) (according to REAL trail). he tolerates treatment well, platelet count has increased to 90, his liver disease is under control. He was kept under the same line of treatment till April 2021 where CT scan has shown further disease progression with marked rise in AFP 1200. he is still maintained his vitals with preserved liver function. he is still fit for further active treatment of his cancer, there was a plan to start him on 4th line of treatment Ramucirumab according to (REACH-2 trail) but he refuses to continue treatment and he preferred conservative and symptomatic treatment only.
Discussion
Cancer immunotherapy modulates the host’s tumour-directed immune response and has been an effective strategy in treating various tumours. Specifically, PD-1 and its associated ligand (PD-L1) have been two of the most targeted checkpoints, effectively overcoming tumour’s immune escape mechanisms in a large subset of patients [5]. In fact, the number of clinical trials using PD1/PDL-1 inhibitors have increased over 600% from 2015 to 2017 alone [6]. Moreover, the estimated number of cancer patients eligible for immune checkpoint inhibitors (ICIs) increased from 1.5% in 2011 to 43.6% in 2018 [2,6]. Thrombocytopenia has been reported in approximately 15–37% of patients receiving ICI therapy [7,8]. The precise pathogenesis of this immune-mediated thrombocytopenia is not entirely understood [9]. The toxicity profile of nivolumab observed in patients with advanced HCC was generally like that observed in patients with other cancers with the exception of higher incidence of elevation in transaminases and bilirubin levels. For patients with NSCLC, PD-1/PD-L1 inhibitors are generally safer and better tolerated than cytotoxic chemotherapy, though a 0.7% incidence of thrombocytopenia has been reported [3]. In a retrospective chart review of 2360 patients with melanoma treated with an ICI, < 1% experienced thrombocytopenia and, of those, most showed spontaneous resolution and did not require treatment [4]. In some cases, however, the thrombocytopenia reportedly persisted for an extended period and was not resolved by standard treatment protocols; intravenous administration of immunoglobulin and a thrombopoietin-receptor agonist was required [10-12].
In our patient the clinical course and laboratory results suggests the thrombocytopenia was caused by nivolumab induced antiplatelet autoantibodies autoimmune activation. the thrombocytopenia developed shortly after initiating systemic therapy with nivolumab, and a bone marrow examination confirmed that the thrombocytopenia was peripheral. Moreover, the lack of effectiveness of platelet infusion and the effectiveness of steroids suggests a diagnosis of ITP. Nivolumab may induce or increase production of platelet-specific IgG autoantibodies. Interestingly, it was reported that patients with irAEs experienced significantly greater antitumor effect than patients without an irAE [7]. The flare up of an immunoreaction may reflect a marked antitumor effect.
Conclusion
Autoimmune hematologic toxicities induced by ICIs, including thrombocytopenia, are viewed as rare irAEs, and increased application of ICIs in advanced malignancies contributes to increased reports of immune thrombocytopenia, but it should not be neglected in treating patients with ICIs because it is potentially life threatening in some cases. Oncologists should bear in mind that decreasing platelet counts represent an early sign of immune-related thrombocytopenia.
References
- Petersen SL (2007) Alloreactivity as therapeutic principle in the treatment of hematologic malignancies Studies of clinical and immunologic aspects of allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning. Dan Med Bull 54(2): 112–139.
- Sanchez-Guijo FM, Orfao A, Canizo MC (2012) Bone Marrow Transplantation Extends Its Scope. Adv Exp Med Biol 741: 121–134.
- Hoyle C, Goldman JM (1994) Life-threatening infections occurring more than 3 months after BMT 18 UK Bone Marrow Transplant Teams. Bone Marrow Transplantation 14(2): 247–252.
- Majhail NS, Rizzo JD, Lee SJ, Aljurf M, Atsuta Y, et al. (2012) Recommended Screening and Preventive Practices for Long-Term Survivors after Hematopoietic Cell,Hematol Oncol Stem Cell Ther 5: 1–30.
- George B, Mathews V, Srivastava A, Chandy M (2004) Infections among allogeneic bone marrow transplant recipients in India. Bone Marrow Transplantation 33(3): 311–315.
- Hughes WT, Armstrong D, Bodey GP, Brown AE, Edwards JE, et al. (1997) 1997 guidelines for he use of antimicrobial agents in neutropenic patients with unexplained fever. Infectious Diseases Society of America. Clin Infect Dis 25(3): 551–573.
- Kameda K, Kimura SI, Misaki Y, Yoshimura K, Gomyo A, et al. (2019) Associations between febrile neutropenia-related parameters and the risk of acute GVHD or non-relapse mortality after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 54(5): 707-716.
- Lipari FG, Zarate AH, Garcia JJ, Basquiera AL, Caeiro JP (2017) Bloodstream infection in patients receiving hematopoietic stem cell transplant. Seven years of experience with adults and children. Rev Chilena Infectol 34(6): 535-538.
- Vavilov VN, Averyanova MY, Bondarenko SN, Stancheva NV, Zubarovskaya LS, et al. (2015) Bacterial infections in the early period after allogeneic bone marrow transplantation. Ter Arkh 87(7): 88-93.
- R Safayi, F Shahi, M Ghalamkari, M Mirzania, M Khatuni, et al. (2018) A Survey of Infection in Allogenic Hematopoietic Stem Cell Transplantation in Patients with Acute Myeloid Leukemia Int J Organ Transplant Med 9(3): 112–116.
- Ninin E, Milpied N, Moreau P, Andre-Richet B, Morineau N, et al. (2001) Study of bacterial, viral and fungal infections in adult recipients of bone marrow transplants. Clin Infect Dis 33(1): 41–47.
- Hovi L, Saarinen-Pihkala UM, Vettenranta K, Saxen H (2000) Invasive fungal infections in pediatric bone marrow transplant recipients: single center experience of 10 years. Bone Marrow Transplant 26(9): 999–1004.
- Gil L, Styczynski J, Komarnicki M (2007) Infectious Complication in 314 Patients after High-Dose Therapy and Autologous Hematopoietic Stem Cell Transplantation: Risk Factors Analysis and Outcome, Infection 35(6): 421–427.
- Winston DJ, Gale RP, Meyer DV, Young LS (1979) Infectious complications of human bone marrow transplantation. Medicine(Baltimore) 58(1): 1–31.






























