The Incited Helix- Chondrodermatitis Nodularis Chronica Helicis
Anubha Bajaj*
Histopathologist in A B Diagnostics, New Delhi, India
Submission: August 11, 2021; Published: August 26, 2021
*Corresponding Address: Anubha Bajaj, Histopathologist in A B Diagnostics, New Delhi, India
How to cite this article: Anubha Bajaj. The Incited Helix- Chondrodermatitis Nodularis Chronica Helicis. Canc Therapy & Oncol Int J. 2021; 19(4): 556017. DOI: 10.19080/CTOIJ.2021.19.556017
Preface
Chondrodermatitis nodularis chronica helicis is a benign, painful, inflammatory condition of the external ear. The idiopathic, non-neoplastic, ulcerative lesion of the auricle depicts incrimination of cutaneous surface and cartilage of the helix and antihelix of the pinna. Initially scripted by Winkler in 1915, the disorder is additionally denominated as Winkler disease, chondrodermatitis nodularis auricularis, ear pressure sore, nodular chondrodermatitis or chondrodermatitis nodularis anti helicis [1,2]. Chondrodermatitis nodularis chronica helicis is predominantly a clinical diagnosis, contingent to characteristic, tender, painful lesions situated upon the helix or antihelix of the pinna. Cogent histological tissue sampling may be required for disease confirmation. The benign condition is appropriately alleviated by localized surgical excision.
Disease Characteristics
Typically, helix of the pinna is affected although the condition may involve antihelix or segments of the auricle. Although a frequently discerned disease, exact incidence of chondrodermatitis nodularis chronica helicis remains obscure [1,2]. The condition is predominantly engendered in middle-aged or elderly males although younger adults may be implicated. An estimated 10% to 35% lesions may arise in women whereas the disorder is infrequent in children. Painful, tender lesions of chondrodermatitis nodularis chronica helicis commonly appear within the helix in males and the antihelix in females [1,2].
The disease lacks an ethnic predilection although lesions commonly emerge in fair-skinned individuals subjected to chronic sun exposure. Of obscure aetiology, chondrodermatitis nodularis chronica helicis is posited to preponderantly occur due to inadequate vascular perfusion or contiguous, excessive pressure upon the pinna as generated by dozing in a singular posture or prolonged utilization of hearing aids, headphones, associated headgear or mobile phones [1,2]. Anatomical malformations as a grossly protrusive helix or antihelix may engender the condition. Predisposing factors are repetitive trauma, localized ischaemia, actinic damage or exposure to cold weather. Additionally, the condition may be associated with autoimmune connective tissue disorders such as autoimmune thyroiditis, lupus erythematosus, dermatomyositis or scleroderma [1,2].
Disease Pathogenesis
Of obscure pathogenesis, diverse theories are proposed in the emergence of chondrodermatitis nodularis chronica helicis. It is surmised that perichondrial vasculitis enunciates the emergence of a secondary disease process contingent to arteriolar constriction within the perichondrium, especially in regions farthest from arterial perfusion as encountered in the helix. Consequently, ischemia, necrosis and extrusion of subjacent cartilage ensues. Eventually, foreign body giant cell reaction is induced along with severe inflammatory response and emergence of chondrodermatitis nodularis chronica helicis [3,4]. Besides, pinna is endowed with peculiar anatomical characteristics such as minimal subcutaneous tissue or restricted vascular perfusion and thereby appears prone to development of chondrodermatitis nodularis chronica helicis [3,4]. Decimated subcutaneous tissue reduces cushion and padding of subjacent cartilage initiating elevation of pressure and declining temperature with consequent emergence of cartilaginous injury. Decreased vascular perfusion delays wound healing and enhances possible emergence of ischemia which promotes development of the condition. Aforesaid predisposing factors generally expedite disease onset [3,4].
Clinical Elucidation
The lesion is predominantly located upon the border of right ear and arises due to compression by dozing. Apex of the helix of pinna is a frequent site of disease emergence. The lesion commonly arises upon the antihelix in incriminated females. Alternatively, the nodule can exceptionally be discerned upon external auditory canal or posterior segment of pinna [5,6]. Typically, individuals demonstrating chondrodermatitis nodularis chronica helicis manifest a spontaneously occurring unilateral, tender, painful nodule situated upon the helix or antihelix of the pinna of below <1 centimetre magnitude. Nocturnal pain is a frequently discerned symptom. Pain during the day or upon touching the nodule may occasionally ensue [5,6].
Generally, a solitary, firm, elliptical nodule with a centric, surface crust and circumscribing erythema is enunciated. Minimal haemorrhage or scaly material may exude from the lesion. Tumefaction expands briskly followed by stabilization with achievement of maximal magnitude. Nevertheless, bilateral lesions may appear [5,6]. Upon physical examination, a singular, elliptical or spherical nodule of magnitude 4 millimetres to 6 millimetres with an ulcerated, crusted centric region, a raised, rolled perimeter and a circumscribing erythematous zone is observed [5,6]. The nodule is firm, tender and adherent to subjacent auricular cartilage. The centric crust obscures a miniature channel wherein haemorrhage and exudate may appear upon eradication of the crust [5,6].
Histological Elucidation
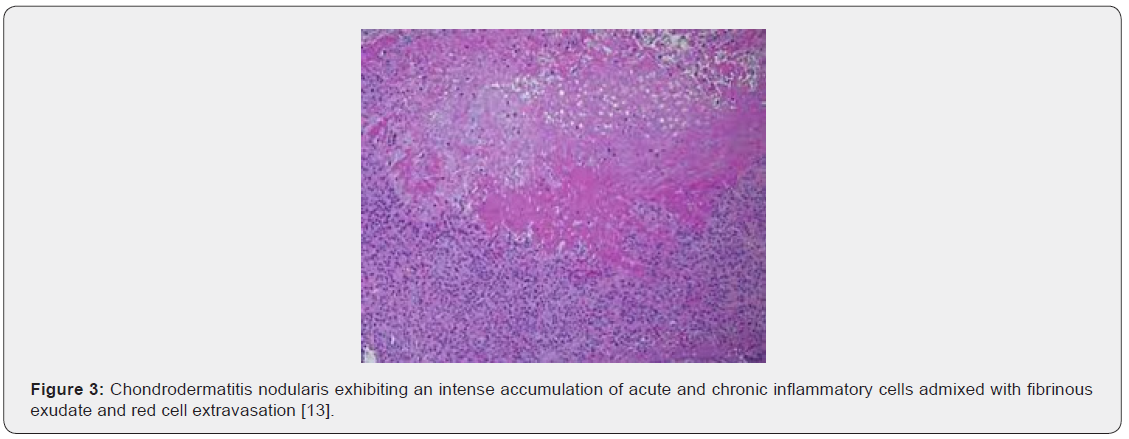
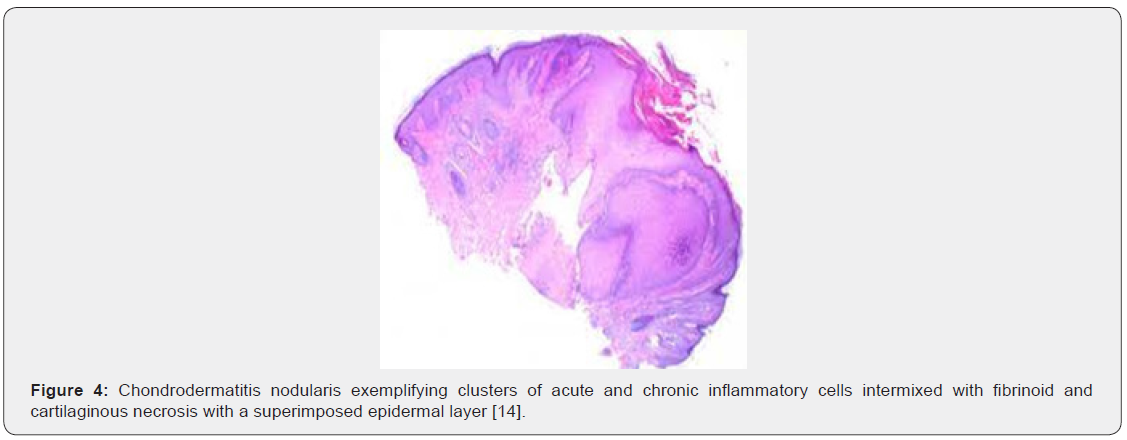
Upon gross examination, a dome shaped nodule of magnitude 0.3 centimetres to 1.8 centimetres with superimposed, crust-like scale enveloping a centric zone of ulceration is observed [4,6]. Centric foci of ulcerated stratified squamous epithelium appear adjacent to foci of acanthosis, hyperkeratosis, parakeratosis and pseudo-epitheliomatous hyperplasia. Site of lesion is usually devoid of dermal adnexa. Ulcer base demonstrates granulation tissue which extends to adjoining cartilage and perichondrium. Foci of fibrinoid necrosis may be observed. Associated peripheral nerves are hyperplastic and immune reactive to S100 protein [4,5]. Characteristically, the lesion depicts hyperkeratosis, parakeratosis, hyperplasia of adjacent stratified squamous epithelium along with significant destruction and replacement of dermal tissue with sclerosis. Proliferation of dermal vascular articulations and damage to subjacent cartilaginous tissue may ensue [5,6].
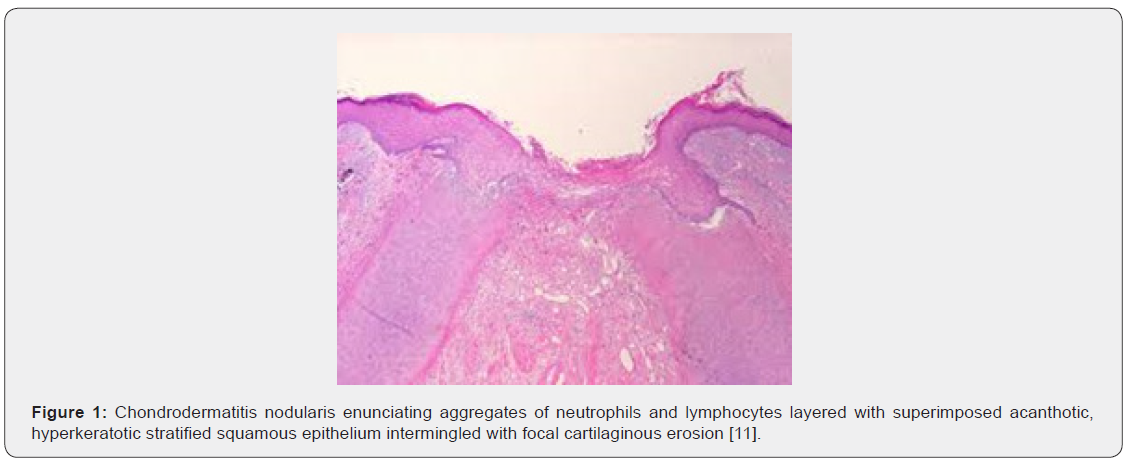
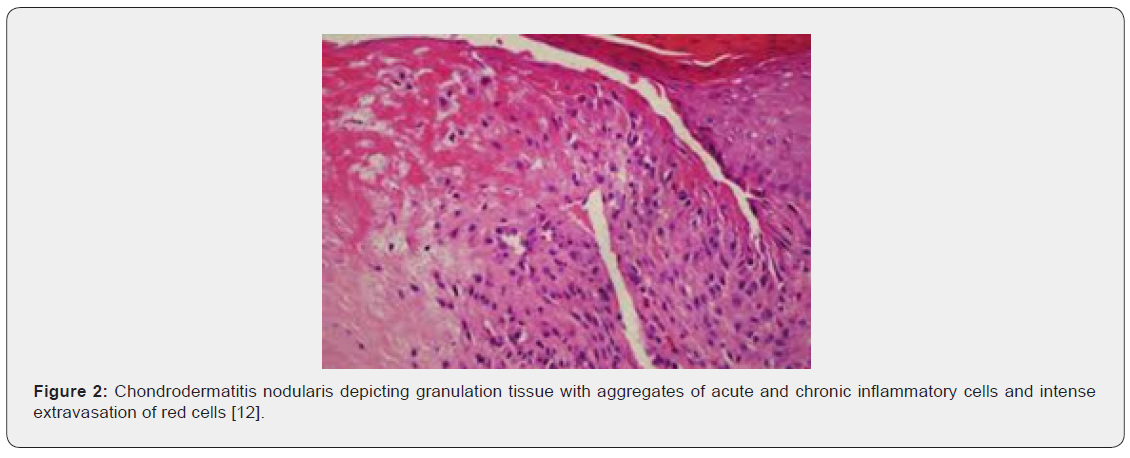
Upon microscopy, the centric, ulcerated, crusted region is circumscribed by acanthotic, parakeratotic stratified squamous epithelium. Fibrinous debris accumulated within base of the ulcer extends centrically towards the perichondrium and auricular cartilage. The debris appears encompassed by granulation tissue constituted of acute and chronic inflammatory cells with prominent vascular articulations. Cartilaginous erosion is induced and appears enveloped with necrosis and tissue basophilia. Reactive modifications as cellular atypia of the stroma and fibrosis can be exemplified [5,6] (Figures 1-8).
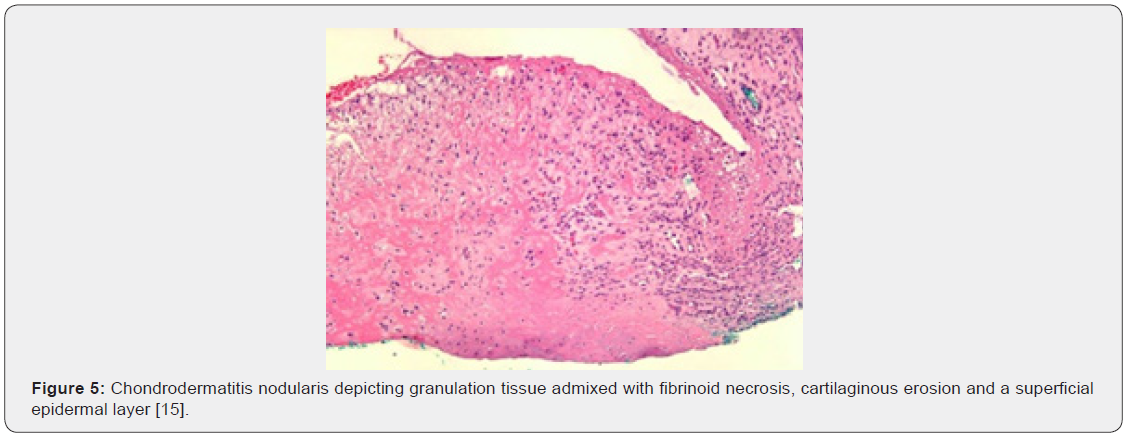

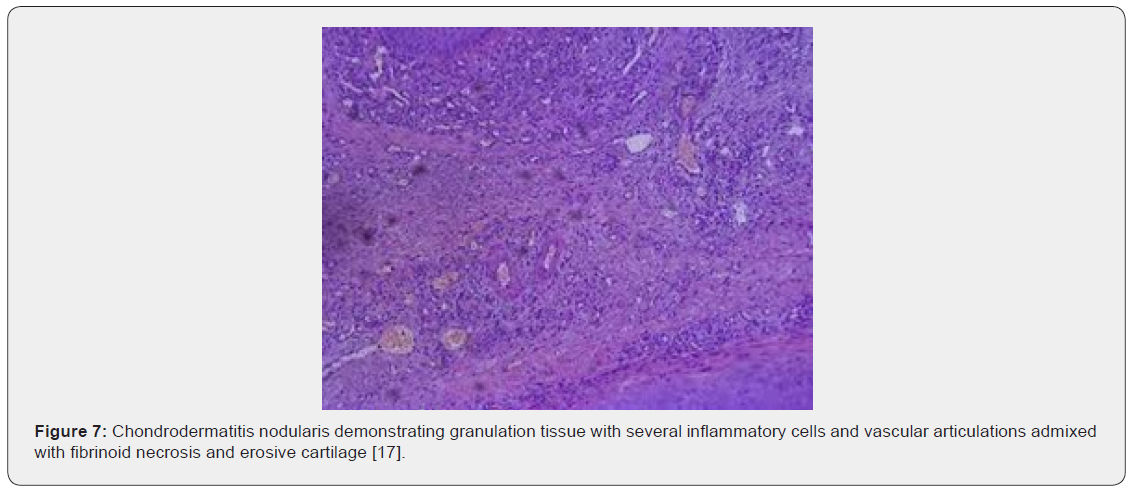
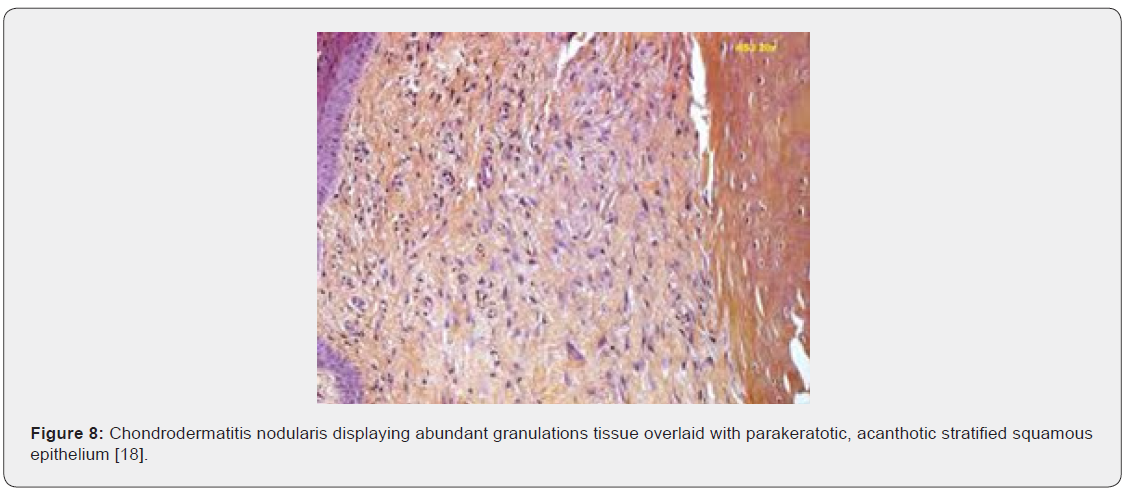
Differential Diagnosis
Chondrodermatitis nodularis chronica helicis requires a segregation from malignant cutaneous neoplasms such as •basal cell carcinoma which demonstrates a nodular appearance with centric crusting. Tumefaction is well circumscribed and displays variable adherence to superimposed epidermal layer or distinctive follicular attachment. Enlarged, basaloid tumour cell lobules depict peripheral nuclear palisading. Tumour cell aggregates can be solid or delineate centric cysts with the emergence of a definitive cleft between neoplastic lobules and circumscribing fibromyxoid stroma, configured due to excessive secretion of mucin. Cellular and nuclear pleomorphism is minimal, apoptotic cells, foci of tumour necrosis and mitotic activity is variable [7,8].
•Squamous cell carcinoma clinically simulates enlarged, inflamed lesions of chondrodermatitis nodularis chronica helicis. The carcinoma depicts cellular sheets or islands of enlarged, polygonal, malignant cells with pleomorphism, nuclear hyperplasia, hyperchromasia, anisocytosis, anisonucleosis and loss of intercellular bridges. Tumour cells are imbued with keratin discernible within individual cells or appearing as keratin pearls. Adjoining areas can display foci of carcinoma in situ. Neoplastic cell aggregates may be admixed with foreign body giant cells, palisading epithelioid cell granulomas, extensive neutrophilic infiltration, exceptional oncocytes or clear cell change due to accumulation of intracellular glycogen. The neoplasm is classified as well differentiated, moderately differentiated, or poorly differentiated contingent to proportionate or predominant intracellular or intercellular keratinization [7,8].
•Actinic keratosis is associated with atypia of basal keratinocytes demonstrating nuclear crowding, overlapping and loss of polarization. The lesion can extend up to mid-epidermal or upper epidermal layers, adnexal structures although is typically confined to infundibular region. Characteristically, mild acanthosis with occasional descending buds, focal parakeratosis, compact hyperkeratosis and attenuation of granular cell layer is observed. The dermis depicts solar elastosis and mild inflammatory infiltrate comprised of lymphocytes and plasma cells.
•Calcinosis cutis is composed of lobules of calcific substance circumscribed by histiocytic giant cells admixed with miniature, psammomatous aggregates of calcification. Irregular accumulation of intensely basophilic, acellular substance is confined to the dermis or subcutaneous tissue. Calcific deposits are characteristically well circumscribed and surrounded by an attenuated rim of eosinophilic hyalinisation and host-induced giant cell reaction [7,8].
•Viral wart generally protrudes above the epidermal surface and demonstrates focal hyperkeratosis, papillomatosis or hyper-granulosis. Columns of parakeratosis can overlay projections of dermal papillae. Superficial keratinocytes are vacuolated and imbued with pyknotic nuclei or koilocytes which are usually discerned in preliminary lesions. Rete ridges appear curvilinear. Dermal papillae exhibit dilated capillaries [7,8].
•Gouty tophus depicts multiple lesions which appear within peripheral sites such as fingers and toes. Preliminary lesions depict an oedematous synovium infiltrated with acute and chronic inflammatory cells. Late-stage lesions configure tophi which are enlarged aggregates of urate crystals intermingled with foci of granulomatous inflammation and hyperplastic, fibrotic synovial tissue. The crystals are elongated, slender, needle- shaped, water soluble and appropriately discerned with alcohol-based fixatives. Chronic disease delineates deposition of urate within soft tissue, ligaments or cutaneous surfaces. Crystal deposition is circumscribed by fibrous tissue, histiocytes and giant cells [7,8].
•keratoacanthoma is a rapidly progressive lesion which typically resolves spontaneously within a few months. Tumefaction is composed of enlarged, well differentiated, bland squamous cell aggregates with abundant, eosinophilic cytoplasm and enlarged, hyperchromatic or vesicular nuclei. Tumour cells configure a centric, keratin- filled crater. Circumscribing epidermis configures a “lip” around the tumour crater or invagination. Mitotic activity and cellular atypia is usually confined to the tumour periphery. Intra-epidermal, neutrophilic micro-abscesses and keratinous horn appear as distinctive components of the neoplasm [7,8].
•Seborrheic or keratotic dermatitis can simulate chondrodermatitis nodularis chronica helicis as the condition is associated with significant keratosis. Seborrheic dermatitis with variants of acute, subacute or chronic spongiotic dermatitis demonstrates focal, mild spongiosis with superimposed, folliculo-centric scales or crust imbued with few neutrophils or psoriasiform epidermal hyperplasia. Papillary dermis is minimally oedematous, superficial vascular articulations are dilated and a mild perivascular infiltrate of lymphocytes, histiocytes and occasional neutrophils can be discerned. Exocytosis of inflammatory cells is minimal [7,8].
•Glomus tumour simulates the vascular proliferation discerned in chondrodermatitis nodularis chronica helicis. Tumefaction is well circumscribed and constituted of distinctive components of spherical glomus cells, vascular articulations and smooth muscle cells along with admixed, branching, capillary-sized vasculature layered with endothelial cells. Aggregates of uniform glomus cells configure nests, sheets or trabeculae circumscribed with hyalinised or myxoid stroma. Spheroidal glomus cells depict an indistinct outline and are imbued with amphophilic or eosinophilic cytoplasm, spherical, sharp, punched- out nuclei with homogenous, bland chromatin and inconspicuous nucleoli. Mitosis is exceptional [7,8].
Investigative Assay
Chondrodermatitis nodularis chronica helicis can be adequately discerned with competent clinical examination. Also, confirmation with precise histopathological evaluation or examination of a cutaneous tissue sample is optimal for diagnosis. Besides, pertinent investigations to exclude systemic or autoimmune diseases such as scleroderma, thyroid disorders or collagen vascular diseases may be necessitated, especially in incriminated young adults below< 40 years [9,10].
Therapeutic Options
Chondrodermatitis nodularis chronica helicis can be appropriately managed with conservative treatment or surgical intervention [9,10]. Conservative therapy is an inexpensive methodology aimed at relieving pressure upon the pinna with the employment of a prosthesis or padding. Generally, posterior auricular, self-adhering foam sponges, foam bandages upon the head or doughnut pillows are adopted in order to relieve pressure upon pinna [9,10]. Chondrodermatitis nodularis ear protector can be employed during sleep with an elastic headband. Petroleum jelly or an antiseptic ointment can be applied with a light dressing, especially in infected or ulcerated lesions. Proportionate alleviation of clinical symptoms is contingent to compliance of incriminated subjects [9,10].
Topical or intra-lesional steroids decimate localized inflammation. However, glucocorticoid therapy may demonstrate unsatisfactory results. Collagen or hyaluronic acid fillers can be injected subcutaneously in order to cushion the cartilage. Localized injection of collagen can simulate subcutaneous tissue and function as a cushion in order to insulate the implicated site [9,10]. Photodynamic therapy generally necessitates multiple sessions. Carbon dioxide or argon laser can be utilized wherein the wound heals by secondary intention [9,10]. Topical application of 2% nitroglycerine or nitroglycerine patch is beneficial in alleviating clinical symptoms and ameliorating extraneous appearance of the lesion. Nitroglycerin ointment induces relaxation and vasodilation of arteriolar smooth muscle and is beneficial in reversing ischaemic alterations associated with chondrodermatitis nodularis chronica helicis [9,10].
Cryosurgery, electro-cauterization or curettage can also be employed. Curettage is adopted in order to eliminate diseased tissue and is associated with subsequent healing by secondary intention [9,10]. Chondrodermatitis nodularis chronica helicis can be subjected to surgical resection. Primary surgical excision with meticulous trimming of lesional cartilage is recommended [9,10]. Surgical options are comprised of eradication of cutaneous tissue and cartilage with minimised removal of tissue perimeter. Also, excision of the lesion can be obtained with wedge resection, or a punch biopsy followed by full thickness skin grafting [9,10]. Generally, extensive excision of enlarged lesions with reconstruction of the ear employing skin grafts or localized cutaneous flaps is advantageous. Excision of lesional cartilage, cutaneous and accompanying subcutaneous tissue can be adopted. Additionally, skin-sparing surgical techniques with eradication of cartilaginous tissue can be adopted [9,10]. Chondrodermatitis nodularis chronica helicis is accompanied by a favourable prognosis. Nevertheless, long-term disease associated morbidity is commonly discerned. Chondrodermatitis nodularis chronica helicis demonstrates around 10% to 30% proportionate lesion reoccurrence following surgery. Spontaneous resolution of the lesion may occur exceptionally within few months. Inadequately treated instances may undergo remission [9,10].
References
- Gupta G, Badri T, Eddie Kwan (2020) Chondrodermatitis Nodularis Helicis. Stat Pearls International 2020, Treasure Island, Florida, USA.
- Cohen PR, Erickson CP, Antoanella Calame (2019) Painful tumors of the skin: "CALM HOG FLED PEN AND GETS BACK" Clin Cosmet Investig Dermatol 12: 123-132.
- Morgado-Carrasco D, Fusta-Novell X, Sebastian Podlipnik, Lara Ferrandiz(2019)Dermoscopic Features of Chondrodermatitis Nodularis Chronica Helicis: A Case Series. Dermatol Pract Concept 9(1): 52-53.
- Darragh CT, Om A, Jeffrey P Zwerner (2018) Chondrodermatitis Nodularis Chronica Helicis of the Right Nasal Vestibule. Dermatol Surg 44(11): 1475-1476.
- Di Berardino F, Zanetti D (2018) The Direct Use of Mobile Phone and the Occurrence of Chondrodermatitis Nodularis in the Antihelix: An Exemplificative Case. Indian Dermatol Online J 9(6): 438-440.
- Garcia-Garcia B, Munguia-Calzada P, Javier Auban-Pariente, Susana Junceda-Antuna, Pedro Zaballos, et al. (2018) Dermoscopy of chondrodermatitis nodularis helicis. Arch Dermatol Res 310(7): 551-560.
- Salah H, Urso B, Amor Khachemoune (2018) Review of the Etiopathogenesis and Management Options of Chondrodermatitis Nodularis Chronica Helicis. Cureus 10(3): e2367.
- Elsensohn A, Getty S, Jessica Shiu, Sebastien de Feraudy (2018) Intradermal Proliferative Fasciitis Occurring with Chondrodermatitis Nodularis Helicis” Am J Dermatopathol 40(2): 139-141.
- Shah S, Fiala KH (2017) Chondrodermatitis nodularis helicis: A review of current therapies. Dermatol Ther 30(1).
- Juul Nielsen L, Holkmann Olsen C, Jorgen Lock-Andersen (2016) Therapeutic Options of Chondrodermatitis Nodularis Helicis. Plast Surg Int 2016: 4340168.
- Image 1 Courtesy: com
- Image 2 Courtesy: MD Edge
- Image 3 Courtesy: Wiley online library
- Image 4 Courtesy: Dermnet NZ
- Image 5 Courtesy: Dermatology advisor.
- Image 6 Courtesy: com
- Image 7 Courtesy: Docksci.com
- Image 8 Courtesy: Humpath.com






























