When planning IMRT Technique for Head and Neck Cancer by Radiation Oncologist what needs to know?
Mirza Baig*
Associate Professor department of Radiation oncology, MLB Medical College Jhansi, UP, India
Submission:November 19, 2020; Published:December 09, 2020
*Corresponding Address:Mirza Baig, Associate Professor department of Radiation oncology, MLB Medical College Jhansi, UP, India
How to cite this article:Mirza B. When planning IMRT Technique for Head and Neck Cancer by Radiation Oncologist what needs to know?. Canc Therapy & Oncol Int J. 2020; 17(3): 555965.DOI:10.19080/CTOIJ.2020.17.555965
Abstract
For practicing highly conformal therapy like Intensity Modulated Radiation Therapy, defining the target (GTV, CTV and PTV) accurately is of overwhelming importance due to a sharp dose gradient between the tumor and the surrounding critical structures. Unrecognized gross or microscopic disease can lead to higher marginal” recurrences and taking undue margins can result in high dose to surrounding critical organs and normal tissues leading to higher toxicity. Therefore, accurate definition of GTV and CTV is a prerequisite for a successful IMRT planning and delivery.
Keywords: IMRT; GTV; CTV; PTV
Definition of Gross Tumor Volume (CTV)
Recognition of gross tumor volume is the first step in defining target for IMRT planning. A lot of time and effort should go in this very important first step Inputs required for GTV delineation?
a.Physical examination findings
b.Endoscopic findings
c.CT / MRI details
d.Per operative details
e.Histopathology
Clinical Examination
A through clinical examination and its documentation is Mandatory since often we contour the GTV few to several days after first clinical Examination. A lot of clinical findings may not be picked on radiological investigations which are critical to target definition. Involvement of skin around the tumor or neck nodes or superficial spreading mucosal carcinoma in the oral cavity may not be picked on CT scan or MRI. Clinical examination findings should be carefully integrated with radiological studies at the time of contouring. Post-op Target Delineation: It requires full details of the operative findings specially with regards to the involvement of the underlying bone (Mandible) and significant neck nodes with the levels of involvement and evidence of extra capsular extension (ECE) should be documented. A detailed histopathology report should be available for documenting neck nodes involvement, detection of ECE and adequacy of the margins. Any evidence of residual disease or high-risk area should be discussed with the surgeon and the histopathologist. This area can be boosted to a higher dose for better loco-regional control.
Endoscopic Examination: Direct laryngoscopic and Naso-pharyngoscopic examination should be carefully studied and documented. Often the blind biopsy from Nasopharynx can be positive although CT scan or MRI are reported normal. On the other hand, submucosal disease may be better appreciated on CT/MRI but may be unrecognized on endoscopic study. Thus, both clinical and endoscopic examinations should be available at the time of CT/ MRI evaluation and at the time of contouring GTV and CTV since they usually act complementary to each other.
CT Scan
Today, Radiation Oncologists must possess thorough knowledge of Radiological anatomy because he is the one who can best correlate the natural history of the disease with findings on the CT scan or MRI. A high-quality contrast enhanced CT scan with 3-5mm slice thickness is mandatory for both GTV and CTV definition and inverse planning (Figure 1).

While contouring appropriate “level and window” setting is essential to visualize maximum dimension of gross disease. One should seek the help of Radiologist to delineate the tumor correctly. MRI scan in Target Delineation in Head and Neck Cancers: In order to improve the accuracy of GTV, MRI has been integrated in head and neck target definition. It results in significant reduction in uncertainties in defining GTV and CTV and reduces inter-observer and intra-observer variation in target volume definition (Figure 2).

Ultimately it would result in further reduction of marginal recurrences and normal tissue toxicity by reducing the volume of normal tissue encompassed in the radiation field.
MRI gives better information in following circumstances:
a. Minimum artifacts from Dental amalgam, densely calcified or ossified cartilage
b. Ideally suited for evaluation of masticator, Parapharyngeal, Parotid spaces, Skull Base and Floor of mouth.
c. Bone marrow invasion
d. Soft tissue Contrast
e. Mucous Vs Tumor
f. Boundaries related to GTV in cranio-caudal direction.
g. Multiplanar display – Coronal and sagittal reformation capability is better with MRI.
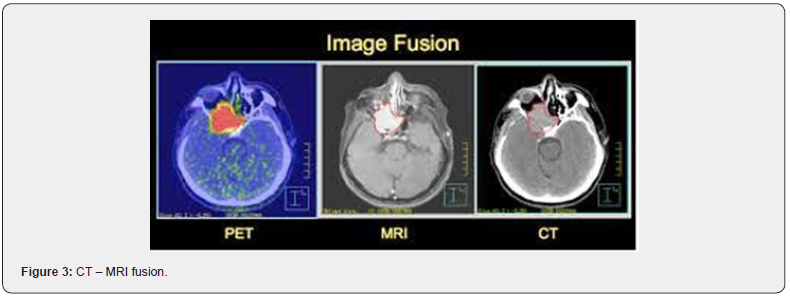
GTVs outlined on MRI are in general more consistent than GTVs derived from CT. Inclusion of MRI in radiotherapy treatment planning reduces the uncertainty in delineating the GTV and consequently less margin needs to be included in the CTV. On the other hand, CT scan is better for Cortical Bone Invasion and Tumor fat boundaries e.g Localization of tumor in the orbital fat and paranasal sinus tumors are better recognized on CT scan as compared to MRI. CT scan also has short scanning time and has less motion artifacts. gives better information than either modality alone since it depicts relationship between bone and tumor better than either modality alone [1]. MRI has added advantage of multiplaner display. Extension through the base of skull is clearly visible on the coronal MRI but very poorly on the axial MRI. This difference can be explained by the partial volume effect, slice gap and poor visibility of orientation structures in the axial MRI (Figure 3).
Target Delineation for Neck Nodes
For delineation of the neck nodes several recommendations are available, for each site, different levels of neck nodes are to be included in the target volume depending upon the lymphatic drainage and likelihood of microscopic involvement, for nodes with ECE, the CTV needs to be extended up to the skin surface while for ECE negative neck, a gap of 2-3mm can be left between the nodes and skin surface for skin sparing. On the side of N2, N3 disease, cranial margin of the neck node volume is taken high up into the base of the skull in the post - styloid region [2]. In the low-risk neck cranial extent can be taken up to upper border of C2 vertebra. Clinical Target Volumes (CTV): CTV1 is composed of GTV-T (tumor) and the surrounding volume of Subclinical disease (CTV-T), GTV – N+ (nodal) and surrounding extra nodal disease. In postoperative cases, CTV1 is composed of original gross tumor, adjacent soft tissue of the surgical bed, grossly enlarged lymph nodes or regions with extracapsular extension. Usually a margin of 5-15mm is taken around gross tumor and neck nodes. CTV2 is composed of prophylactically treated regions usually ipsilateral / contralateral lymph nodes. Radiation oncologist considers the microscopic extensions of the disease near GTV and the natural history of local spread, perivascular and perineural extension and lymph node involvement while deciding CTV.
ICRU 62 has further refined the definition of target with the addition of margin due to setup uncertainties
i. Internal margin (IM) – To consider size, shape and position of the CTV in reference to patient’s co-ordinate system, e.g., respiratory movement, filling of rectum.
ii. Set up margin (SM) – To consider all uncertainties in patient – beam positing in reference to treatment machine co-ordinate system. e.g., set up uncertainties, Immobilization, Mechanical stability of the machine.
PTV
It is the margin created around CTV to account for geometric uncertainties. Its size and shape depend upon GTV and CTV, Internal motions of tumor & normal organs, inaccuracies in beam and patient set up, Patient fixation and Beam Orientation. PTV is 3D – envelope fixed in space and GTV & CTV move within this envelope. Margins around CTV to create PTV are also not an exact science. The Solid data for internal organ motion & set up error for most sites are still lacking [3] and the internal organ motion and set up uncertainties tend to be asymmetrical (anisotropic). After PTV is defined, appropriate beam sizes to account for Penumbra and beam arrangements must be selected. Risk of the missing part of CTV needs to be balanced against the risk of complications due to PTV which is too large. According to [4,5] to ensure CTV is within PTV 95% of the time – CTV should be expanded 1.65 times the SD in each direction. CRAIG et al have proposed that coverage of 95% of PTV is a reasonable goal for PTV design. At times, the margin for PTV is reduced near the critical organ and is more liberal in rest of the volume.
Delineation of Organs at Risk
These are the organs whose radiation sensitivity may significantly influence treatment planning and / or prescribed dose e.g Spinal cord, Brain Stem, Optic Nerves / Chiasma, Lung, Liver, Kidneys etc. PRV: ICRU - 62 has introduced planning organ at Risk Volume. - It is the margin taken around ‘organ at risk’ due to internal organ movements & set up uncertainties (Analogous to PTV). PTV and PRV may often overlap and a search for compromise is resorted to in planning process e.g., Spinal cord, Parotid gland.
Imrt - Dose Prescription
Two general approaches are undertaken while prescribing the tumor dose to various targets, the greatest advantage of IMRT is its ability to deliver variable doses to different targets in one plan compared to several plans required in conventional radiation therapy to achieve that goal. When a single plan is prescribed the gross tumor receives both a higher total dose and a higher dose per fraction compared with sub clinical disease. Due this difference a correction of the total dose to yield the biologically equivalent doses (BEDs) is required [6]. Dose – volume specification is made to impose constraints on the DVH of the targets, critical organs and noninvolved tissue of interest. Prescription dose: RTOG –H- 0022 protocol specifies it to be dose that encompasses at least 95% of the PTV. In plan evaluation not more than 20% PTV can receive more than 110% of the dose and not more than 1% PTV can receive less than 93% of the dose [7]. Hot Spot is defined as volume outside PTV that receives more than 100% prescribed dose, and it should be more than 15mm diameter.
References
- Hill et al, (1994) Radiology91: 447-54
- Eisbruch A, Marsh LH, Dawson LA, Carol R Bradford, Theodoros N Teknos, (2004)Recurrence near base of skull after IMRT for Head and neck cancer: Implications for target delineation in high neck and for parotid gland sparing. J Radiation Oncology BiolPhys59(1): 28-42.
- Martinez (1999) Radiology 211:815-828.
- Antolak (1999) IJROBP 44:1165.
- Rosenzweig (2000) IJROBP 48:81-87.
- Mohan (2000) IJROBP 46:619-630.
- Chao KSC, Wippold FS, Ozygit G (2002) Determination of oral delineation on nodal target volumes for head and neck cancers based on pattern of failure in patients receiving definitive and postoperative IMRT. Inj J Radiat OncolPhys53(5): 1174 -1184.






























