Nerve Encrusted Sinew - Benign Triton Tumour
Anubha Bajaj*
Histopathologist in A B Diagnostics, New Delhi, India
Submission:September 30, 2020; Published:November 25, 2020
*Corresponding Address:Anubha Bajaj, Histopathologist in A B Diagnostics, New Delhi, India
How to cite this article:Anubha B. Nerve Encrusted Sinew - Benign Triton Tumour. Canc Therapy & Oncol Int J. 2020; 17(3): 555963.DOI:10.19080/CTOIJ.2020.17.555963
Preface
Tumours categorized as generic admixture of neural tissue and skeletal tissue are benign triton tumour, neurofibroma and commonly discerned rhabdomyosarcoma. Benign triton tumour is an exceptional, developmental lesion comprised of commingled mature skeletal muscle tissue and mature neural tissue. The neoplasm is additionally nomenclated as “neuromuscular hamartoma” or “neuromuscular choristoma” “Triton” terminology is a derivative of amphibian triton salamander wherein normal nerve can induce regeneration of skeletal muscle. Initially scripted by Louhimo and Rapola in 1972, benign triton tumour was documented as a hamartoma. Neuromuscular choristoma was a terminology adopted by Bonneau and Brochu as skeletal muscle fibres are not contemplated to be normal anatomic articulations of peripheral nerves [1,2]. Simple observation and cogent tissue sampling are appropriate for diagnosis of the biologically benign neoplasm.
Disease Characteristics
The neoplasm is frequently discerned within cranial nerve trunks or as miniature nodules within peripheral nerve trunks, especially brachial plexus, sciatic nerve, or proximal median nerve or may emerge as an intracranial mass. The neoplasm can arise within spinal intra-dural space or as cutaneous nodules. Exceptionally, tumefaction is detected within head and neck, incriminating enlarged, central cranial nerves or miniature, peripheral nerve twigs. The tumour can demonstrate aggressive biological behaviour [3]. Benign triton tumour is commonly detected within first decade of life, infants, or children although the condition is infrequent in adults. The neoplasm usually appears in children below < 2 years. Factors contributing towards mesenchymal differentiation with engenderment of skeletal muscle are incompletely defined. Preliminary differentiation of skeletal muscle is independent of neural influence [4]. Environmental and neural factors are crucial in deciding extent of evolution, organization, and maintenance of skeletal muscle. Therapy can engender residual nerve dysfunction upon attempted surgical extermination in certain tumours, whereas specific neoplasms can retrogress spontaneously. Thus, appropriate tumour discernment is crucial for satisfactory treatment planning [3,4].
Disease Pathogenesis
Benign triton tumour commonly emerges within enlarged nerves. Precise mechanism of emergence of benign triton tumour is inconclusive. Tumour cell lines appear to be derivatives of primitive, neural crest cells or are posited to represent a skeletal muscle spindle. Alternatively, an epigenetic or sporadic alteration of motor end- plate engenders a reactive proliferation of neural and skeletal muscle tissue, configuring an organoid pattern [3,4]. Despite a controversial genesis, it is argued that skeletal muscle can originate from limb mesenchymal cells which are entrapped within nerve sheaths during embryogenesis [3]. As per an alternative theory, neuro-ectoderm or ectomesenchyme can engender mature skeletal muscle, akin to generation of iris muscle emerging from neuro-ectodermal eye cup [4]. With cessation of trophic stimuli initiating pertinent muscle differentiation, the muscular component degenerate, and atrophies. Degenerated neural and muscular component are reinstated with fibrotic tissue. Thus, spontaneous retrogression or postoperative fibromatosis of the hamartoma can arise, particularly in concurrence with common inciting factors such as previous surgical intervention or irradiation. Dense fibrous tissue is intermingled and can be restored by skeletal muscle and neural tissue [3,4]. Endoneurial cells of neuroma, impacted with organising influence of motor nerve fibres, can differentiate into skeletal muscle tissue, a hypothesis which is cogent in emergence of malignant triton tumour. Triton tumour denominates a peripheral nerve sheath tumour demonstrating skeletal muscle differentiation engendered by neoplastic nerve sheath cells or neoplastic Schwann cells [4].
Clinical Elucidation
The neoplasm appears as a painless, asymptomatic nodule extending into subjacent soft tissue with consequent architectural distortion or occlusion of abutting viscera. An absence of localized depigmentation, hypertrichosis, freckles, cafe au lait macules or an accompanying bruit is observed. Neurological dysfunction is usually absent [5]. Clinically, asymptomatic tumefaction or a palpable, solitary or multinodular mass is enunciated. However, pain or neurologic dysfunction represented by muscular weakness, paraesthesia or headache along with oculomotor ophthalmoplegic migraine can be exemplified, pertaining to site of tumour incrimination [5,6]. Contingent to tumour magnitude, location and proportionate impingement upon circumscribing anatomic structures, the neoplasm is accompanied by minimal to striking functional disability. Foot deformities can ensue, especially unilateral deformity, thus mandating stringent evaluation of cogent organ functions [5]. Incriminated sciatic nerve can delineate fibromatosis or retrogression of the hamartoma or an accompanying persistent neurologic deficit. Benign triton tumour emerging within head and neck is distinctly categorized as •an aggressive, central subtype incriminating enlarged cranial nerve trunks, predominantly V, VII and XII. The category usually implicates infants and children. •a non aggressive, peripheral subtype manifesting asymptomatic, gradually evolving, cutaneous nodules. Generally, adults are incriminated [5,6].
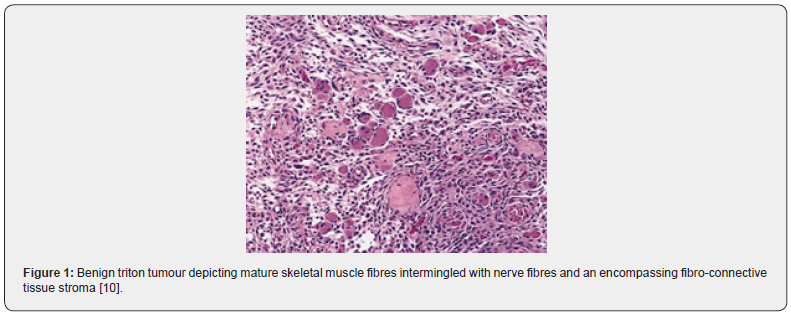
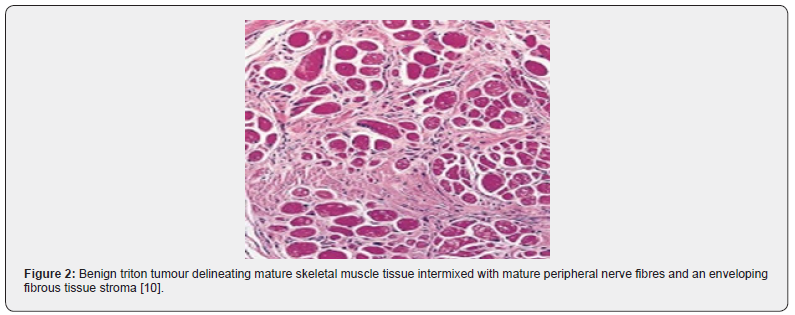
Histological Elucidation
Grossly, a tan coloured, non tender, firm, ill-defined nodule is discerned. Also, an irregular, smooth, protruding tumefaction with normal skin tone can appear upon a mucosal surface. The tumefaction enunciates as a circumscribed, grey/ brown, or grey/white, multinodular mass adherent to an adjacent nerve. Cut surface is solid and grey white [6,7]. On microscopy, a sub epithelial tumefaction with cutaneous superimposition is denominated. The ill-defined neoplasm is composed of fascicles of mature neural tissue disseminated within a multinodular tumour architecture subdivided by fibro-collagenous septa. Tumour cells can circumscribe a pilo-sebaceous unit. Nerve fascicles are intricately admixed with mature, striated, skeletal muscle fibres which appear as bundles of magnitude varying from 15 micrometres to 20 micrometres. Certain muscle fibres are incorporated within perineurial sheath. Foci of myxoid change are discerned. Several capillary- sized blood vessels are intermingled with minimal exudation of chronic inflammatory cells.
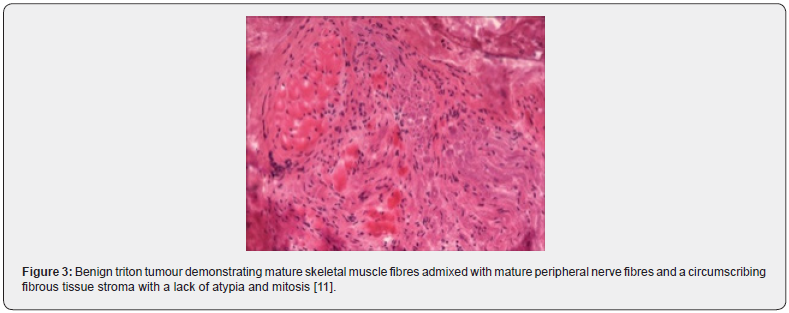
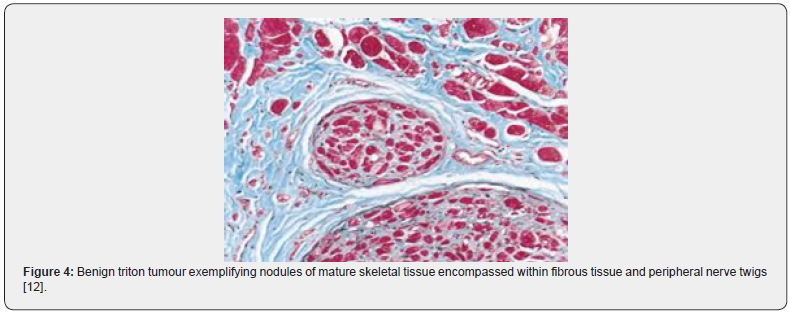
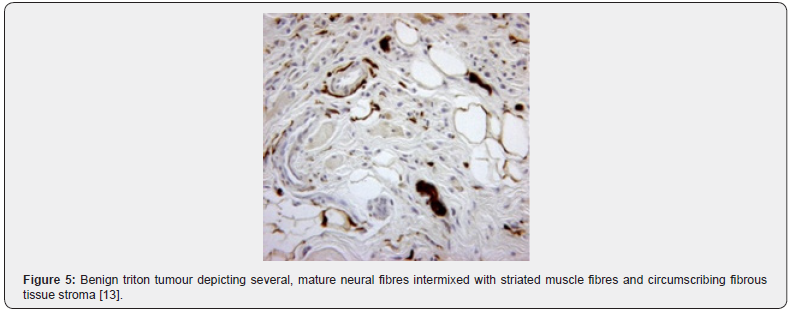

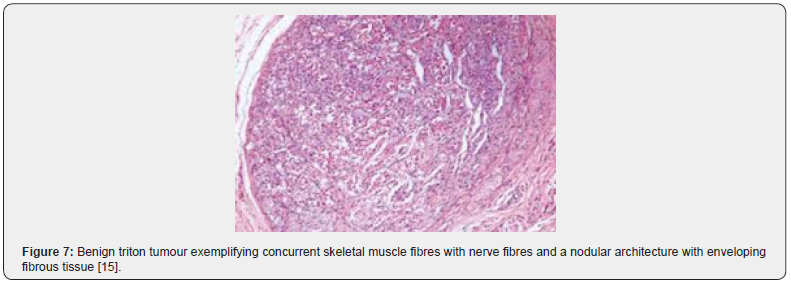

The multinodular neoplasm is subdivided into miniature lobules and fascicles by collagenous, fibro-connective tissue septa. Tumour fascicles are comprised of mature, striated muscle fibres intimately intermingled with neural fibres circumscribed by a singular perimysial- like fibrous sheath. Tumours with a preponderance of fibrous tissue can be challenging to discern and classify [5,7]. Multiple nodules of 3 centimetre to 5-centimetre magnitude are segregated by narrow bands of fibro-connective tissue. Nodules are constituted by striated muscle bundles of variable dimension entangled with myelinated or non myelinated nerve fibres enveloped within a solitary perimysial fibrous sheath. Encompassing stroma can be hyper-cellular with bland, spindle-shaped cells and recapitulate foci of fibromatosis [6,7]. As the neoplasm is composed of miniature nodules and fascicles, is traversed by dense, fibrous tissue septa and is incorporated with well developed, striated muscle fibres along with variable proportion of myelinated and non myelinated nerve fibres, tumour circumscription ensues with flattened, projected layering of perineurial cells, immune reactive to epithelial membrane antigen(EMA) [6,7]. Baseline cellular components of nerve fibres and skeletal muscle fibres are intimately commingled and appear immune reactive to S100 protein and desmin, respectively. Foci of transition of nerve fibres into striated muscle fibres are discerned [6,7].
Collagenous stroma can display individually dispersed nerve fibres and skeletal muscle fibres. Random distribution of cells originating from cellular fascicles or disintegration of tumour nodules, with encompassing, protrusive, epithelial membrane antigen (EMA) reactive perineurial cells is denominated. Degenerative manifestations within the muscular compartment and deteriorating miniature nerve bundles or neural component can induce initial fibrosis [6,7]. Muscle fibres delineate a spherical, elliptical, or spindle-shaped cellular configuration wherein intracytoplasmic cross striations are observed. Muscular differentiation or maturation is predominant within periphery of tumour nodules. As individual dissemination of nerve fibres and skeletal muscle fibres within collagenous stroma is discerned, the randomly dispersed cellular component is engendered from fascicles or nodular degeneration [5,7]. Dense fibrous tissue is variably admixed and appears to replace muscular and neural component. Skeletal muscle cells can undergo atrophy, apoptosis, or degenerative change in the absence of regeneration of muscle fibres. Miniature nerve bundles enunciate focal, myxoid degeneration [5,6]. Dense, cellular fibrous tissue can circumscribe the lesion and is indicative of fibromatosis replacing mature skeletal muscle and nerve tissue. Intra-lesional fibrosis concurs with degeneration of neural component [7]. Diverse quantities of Schwann cells merge with striated muscle cells and demonstrate variable immune reactivity to S100 protein within the muscular component. Striated muscle emerges on account of divergent metaplastic manifestation of Schwann cells or progenitor neural crest cells [6]. Benign triton tumour exemplifies tumour nodules encompassed within an expansive perineurium with concomitant muscular proliferation or replacement. Subsequently, a fascicled architecture is denominated with fragmentation of perineurium. Neural component can also degenerate or disappear [5].
Immune Histochemical Elucidation
The neoplasm can be highlighted with special stains such as Masson’s trichrome or phospho-tungstic acid haematoxylin (PTAH). Striated muscle fibres of benign triton tumour are immune reactive to desmin and muscle specific actin (MSA). Nerve fibres are immune reactive to S100 protein, neuron specific enolase (NSE) and neurofilament protein [3,4]. Perineurial cells are immune reactive to epithelial membrane antigen (EMA). Skeletal muscle component confined to tumour nodules is variably immune reactive to S100 protein. On cytogenetic analysis, a normal karyotype is denominated which is indicative of a malformation rather than a true neoplasm [3,4].
Differential Diagnosis
Benign triton tumour necessitates differentiation from diverse head and neck neoplasms. Several benign, soft tissue neoplasms of head and neck emerge from adipocytes, fibro-histiocytes, vascular tissue or neural tissue. Thus, benign triton tumour mandates a segregation from neurofibroma, Schwannoma, neuroma, or uncommon neoplasia such as meningioma, granular cell tumour or ganglioneuroma. Benign triton tumour requires a demarcation from embryonal rhabdomyosarcoma which is composed of poorly and moderately differentiated skeletal muscle cells [7,8]. Foetal rhabdomyoma is a neoplasm which lacks an intimate association with nerve fibres. The neoplasm is immune non-reactive to S100 protein [8].
Investigative Assay
Computerized tomography is a superior guide which discerns and defines entire spectrum of lesions, especially central subtype of benign triton tumour [8]. Magnetic resonance imaging (MRI) depicts a well circumscribed tumefaction. Axial T1 and T2 weighted imaging delineates minimal signal intensity within the tumour, in contrast to skeletal muscle. Contrast- induced imaging demonstrates mild heterogeneous enhancement [8].
Therapeutic Options
Comprehensive surgical extermination of the tumefaction is recommended. Surgical excision is an optimal treatment option wherein an aggressive procedure is adopted for alleviating central subtype of benign triton tumour and conservative management is appropriate for peripheral tumours [9]. Surgical eradication of the neoplasm demonstrates neural elements entangled within the tumefaction [9]. Following surgical resection, neural palsy of incriminated nerve can occur, thus nerve sparing surgery is recommended. Peripheral category of benign triton tumour can be alleviated with simple surgical extermination of the lesion. Occasional, localized reoccurrence is observed [9]. Although comprehensive surgical resection is curative, tissue sampling or inadequate surgical excision can induce an amelioration of clinical symptoms and progressive decline in tumour magnitude. Adequate treatment is accompanied with superior prognosis. Fibromatosis can ensue following tissue sampling or comprehensive surgical excision. As the neoplasm can exceptionally reappear, regular monitoring following surgical extermination is mandated [8,9]. Therefore, with appropriate discernment of the lesion, suitable conservative treatment is adopted to maintain integrity of incriminated nerve [9].
References
- Louhimo I, Rapola J (1972) Intra-neural muscular hamartoma- report of two cases in small children. J Pediatr Surg 7(6): 696-699.
- Bonneau R, Brochu P (1983) Neuromuscular choristoma- a clinicopathologic study of two cases. Am J Surg Pathol 7(6): 521-528.
- Markel SF, Enzinger FM (1982) Neuromuscular hamartoma – a benign triton tumour composed of mature neural and striated muscle element. Cancer 49(1): 140-144.
- Kim SY, Kwon HP, Kyoung Duck Kwak, Kee BaekAhn(2005) NeuromuscularChoristoma of the Sciatic Nerve- a Case Report” The Korean Journal of Pathology 39: 192-196.
- Daley TD, Darling MR, Bret Wehrli(2008) Benign triton tumour of the tongue. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 105(6): 763-766.
- Akimoto J, Fukami S, Ryo Hashimoto, Jo Haraoka(2012) Neuromuscular hamartoma is a possible primary pathology of oculomotor ophthalmoplegic migraine. Cephalalgia 32(2): 171-174.
- Thakrar R, Robson CD, Sara O Vargas, John G Meara, Reza Rahbar, et al. (2014) Benign triton tumour – multidisciplinary approach to diagnosis and treatment. Pediatr Dev Pathol 17(5): 400-405.
- Barh A, Mukherjee B, KirthiKoka, Subramanian Krishnakumar (2019) Triton tumour of orbit. Orbit 39(6):418-421.
- Krishnappa A, Vijay Shankar S, Kuchangi C Nischal, Haleuoor B Basavaraj(2013) Benign Triton Tumour –a Rare Entity in Head and Neck Region. The Korean Journal of Pathology 47(1): 74-76.
- Image 1 and 2 Courtesy: Pathology Outlines
- Image 3 Courtesy:Research Gate
- Image 4 Courtesy: JAMA network
- Image 5 Courtesy:Science direct
- Image 6 Courtesy: Journal of Neurosurgery
- Image 7 Courtesy: Jpatholtm.com
- Image 8 Courtesy: Oral Surg Oral Med Oral Pathol.com






























