Post-Mastectomy Lymphocele after Breast Cancer Surgery: Risk Factors Evaluation
Sidy KA*, Mohamed Ezzet Charfi, Jaafar Thiam, Adja Coumba Diallo, Papa Souleymane Dieng and Ahmadou Dem
Department of Oncology, Cheikh Anta Diop University, Senegal
Submission: October 02, 2020; Published:October 29, 2020
*Corresponding Address: Sidy KA, Department of Oncology, Cheikh Anta Diop University, Senegal
How to cite this article: Sidy K, Mohamed E C, Jaafar T, Adja C D, Papa S D, et al. Post-Mastectomy Lymphocele after Breast Cancer Surgery: Risk Factors Evaluation. Canc Therapy & Oncol Int J. 2020; 17(2): 555957. DOI:10.19080/CTOIJ.2020.17.555957
Abstract
Aim of study: The main postoperative complication of mastectomies with axillary lymph node dissection is the lymphocele that can last many months after the operation. The purpose of our study was to analyze the factors that can contribute to its production.
Methods: Sixty-one patients have been included in our study. The follow-up was 6 months. All patients had a mastectomy with axillary node dissection following the Madden technique. All quantities of lymphoceles during postoperative hospitalization and ambulatory care have been noted. Factors linked to patient, disease and treatment have been collected.
Results: Sixty-one patients (61) operated at Joliot Curie Institute of Dakar between January and June of 2018 for breast cancer have been included in our study. Amongst the factors linked to patients, the lymphocele production is more significant for obese patients and those with prolonged aPTT (partial thromboplastin time). The weight of surgical specimens from 1001 to 1250 g was associated with greater production. It was the same for a more extensive dissection. Neither surgical factors nor chemotherapy influenced the lymphocele production. However, the extended use of a suction drain causes a more significant production.
Conclusion: The production of postoperative lymphocele is heterogenous, varying from a patient to another. Nevertheless, several factors may influence the production of this lymphocele. These factors are bound to patient, sickness and treatment.
Introduction
Breast cancer is the most widespread women cancer worldwide [1]. In many cases, mastectomy with axillary node dissection is required [2-4]. The main complication of this surgery is the lymphocele that can last several months after the operating procedure. It deeply affects the patient’s quality of life. The lymphocele weighs down patient care and can delay the administering of adjuvant treatment [5]. Preventing a lymphocele currently relies on the use of a suction drain. However, numerous risk factors may influence the production of postoperative lymphocele. These factors are linked to patient, disease and the surgery itself. Several factors have been reported in literature [6]. The aim of our study was to identify the main lymphocele risk factors in our practice.
Methods
This was a prospective study which evaluate the production of lymphocele post-mastectomy and axillary dissection and its relative risk factors. Inclusion criteria were the following: unilateral mastectomy, documented biological and clinical history. All patients have been hospitalized postoperatively until the quantity of lymphocele produced did not exceed 50 mL. Then, each patient had been monitored for 6 months after surgery. We had indexed many parameters linked to patient, sickness, and treatment.
Results
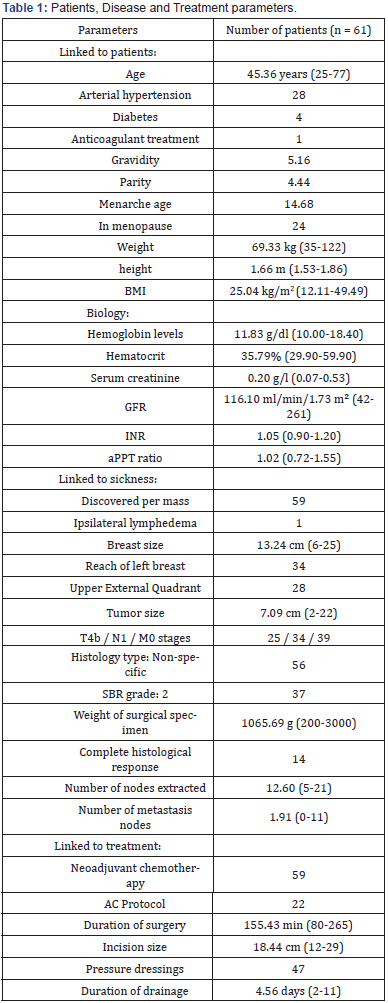
The study occurred at Joliot Curie Institute of Dakar. The period of inclusion was from the 1st January to 30th June 2018. In total, 61 patients have been included in our study. The characteristics linked to patient, sickness and treatment are summed up in Table 1.
Factors linked to the patient
We had noticed that the total production of lymphocele grew with BMI. The mean quantity moved from 725.00 mL to 1992.25 mL for obese patients. Furthermore, this same quantity was affected by aPPT ratio. The more prolonged the aPPT was, the more significant was the quantity, varying from 633.33 mL if the ratio was less than 0.80 to 2213 mL if the ratio was more than 1.21.
Factors linked to the disease
We had noted that surgical specimens weighing between 1001 and 1250 g corresponded with the most important productions (2409.67 mL). This quantity lowered as the weight decreased. On the other hand, the more nodes the dissection brings, the more significant the quantity was, independently of nodes metastasis.
Factors linked to the treatment
No factors linked to surgery or chemotherapy had an important effect on production. However, the more prolonged the use of the suction drain was, the more the total production after 6 months was significant, going from 176.37 mL for drainage of fewer than 2 days to 1839.18 for drainage surpassing 6 days.
Discussion
The early complications of mastectomies mainly include the lymphocele which is a lymphatic effusion from the armpit and mastectomy compartment [7]. Extremely frequent, it can vary in volume, being very abundant and even rebellious in some cases [6-10]. It can be responsible for prolonging the duration of hospitalization, as well as an infection, sutures disunion and a delay in administering adjuvant treatment. It can also originate plastic skin lesions and chronic pain [11,12].
Currently, the prevention of lymphocele hinges on setting up a postoperative suction drain, but this procedure elongates the duration of hospitalization until its ablation [13]. However, this production is extremely variable between individuals and is largely influenced by several factors.
Amongst the factors linked to the patient, we have not noticed a major influence of age, gravidity and parity, menarche age, menopausal status and age of menopause, weight, height, breast size, arterial hypertension, diabetes, diagnosis method (biopsy or cytology) and of the following biological parameters: hemoglobin, hematocrit, number of white blood cells, number of platelets, serum creatinine, GRF and INR.
Hypertension may play a role in the production of lymphocele [14,15,16]. This phenomenon is probably due to the excessive formation of exudate by detached surfaces. This result has not been confirmed by all studies. Likewise, they show that diabetes and the use of anticoagulants do not modify the quantity of lymphocele either in the short or the long run [17]. On the other hand, factors such as age, diabetes, tobacco, and breast size, with varying grades of evidence do not influence the quantity of lymphocele [18,19]. For BMI and aPPT ratio our notice shows a correlation with lymphocele production with strong evidence. it was not well studied parameters by other authors excepted the patient weigh. It was shown in prospective studies as well in metaanalysis, a strong link between weigh and lymphocele production [20-23], and between obesity and lymphocele [24].
In our study, analysis shows that aPPT ratio is linked to the production of lymphocele. This production goes from the simple to the double, 633.33 mL, 1030.16 mL and 2231.00 mL respectively for the groups inferior to 0.80, 0.81-1.20 and superior to 1.21. The more the ratio rises, and thus the aPPT elongates, the more the risk of lymphocele increased. However, the role of the biological factors on the production of lymphocele had not been studied.
For the factors linked to the disease, prognostic factors such as tumor size, TNM stage, specimen size, node involvement, immunohistochemistry and lymphatic embols status seem not link to lymphocele occurrence [18,21]. But the specimen weight and the number of involved nodes had been found determinant to predict positively the quantity of lymphocele [22,25]. Concerning the factors linked to the treatment, the fact that the patient receives neoadjuvant chemotherapy or not has no effect in the production of lymphocele [19,20]. However, the duration of surgery does affect the quantity of lymphocele. However, the use of the dressing compression, contrary to what is commonly thought, do not affect the lymphocele production [26,27].
Conclusion
Some factors can influence the total production of lymphocele after mastectomy for breast cancer. These factors are relative to the patient such as BMI and aPPT ratio, to the disease such as weight of surgical specimen and number of extracted nodes, and to the treatment such surgical drainage. It is the same for other factors like clinical tumor size and the number of metastasis nodes. It is unclear whether other factors like patient weight, hypertension, duration of surgery and the use of a pressure dressing have a link with lymphocele volume and duration.
References
- Coughlin SS (2019) Epidemiologyof Breast Cancer in Women. Adv Exp Med Biol1152:9-29.
- Abass MO, Gismalla MDA, Alsheikh AA, Elhassan MMA (2018)AxillaryLymph Node Dissection for BreastCancer:Efficacy and Complication in Developing Countries.J GlobOncol4:1-8.
- Hennigs A, Riedel F, Feißt M, Köpke M, Rezai M, et al. (2019) Evolution of the Use of Completion AxillaryLymph Node Dissection in Patients with T1/2N0M0 Breast Cancer and Tumour-Involved Sentinel LymphNodesUndergoing Mastectomy: A CohortStudy. Ann SurgOncol 26(8):2435-2443.
- Arisio R, Borella F, Porpiglia M, Durando A, Roberto Bellino, et al. (2019) AxillaryDissection no Axillary Dissection in Breast Cancer Patients With Positive Sentinel Lymph Node: A Single Institution Experience.In Vivo33(6):1941-1947.
- Marszalek D, Noël JC, Simon P (2016) Post operative complications afterneoadjuvantsystemictherapy in primarybreast cancer treatment - Case-control study.Rev Med Brux 37(6):469-475.
- Van Bemmel AJM, van de Velde CJH, Schmitz RF, Liefers GJ (2011) Prevention of seroma formation after axillary dissection in breast cancer : A systematic review. European Journal of Surgical Oncology 37(10): 829-835.
- Ouldamer L, Bonastre J, Brunet-Houdard S, Body G, Giraudeau B, et al. (2016) Dead space closure with quilting suture versus conventional closure with drainage for the prevention of seroma after mastectomy for breast cancer (QUISERMAS): protocol for a multicentrerandomised controlled trial. BMJ Open 6(4): e009903.
- Douay N, Akerman G, Clément D, CMalartic, O Morel, E Barranger et al. (2008) Priseen charge des lymphocèlesaxillaire dans le cancer du sein. GynécologieObstétrique&Fertilité 36(2): 130-135.
- Katsumasa K, Kojiro S, Tetsuya T, Hirohisa Imai, HiroyasuYamashiro, et al. (2005) Pathophysiology of seroma in breast cancer. Breast Cancer 12(4): 288-293.
- Zaidi S, Hinton C (2017) Breast cancer surgery without suction drainage and impact of mastectomy flap fixation in reducing seroma formation. European Journal of Surgical Oncology 43(5): s32.
- Classe JM, Dupre PF, François T, Serge Robard, Jean Loup Theard, et al. (2002) Axillary padding as an alternative to closed suction drain for ambulatory axillary lymphadenectomy: a prospective cohort of 207 patients with early breast cancer. The Archives of Surgery 137(2): 169-173.
- Delmar R, Aitken MD, John P, Minton MD (1983) Complications associated with mastectomy. Surgical Clinics of North America 63(6): 1331-1352.
- Trefoux-Bourdet A, Body G, Jacquet A, T Hebert, I kellal,et al. (2015)Intérêt du capitonnage de la loge de mastectomie dans la prévention des séromes post-opératoires : étude prospective. GynécologieObstétrique&Fertilité 43(3): 205-212.
- Salmon RJ, Cody HS, Vedrenne JB, B Asselain, J C Durand,et al. (1985) Prevention of postoperative lymphocele after breast amputation. La Presse Médicale 14(1): 27-29.
- Kumar S, Lal B, Misra MC (1995) Post-mastectomy seroma: a new look into the aetiology of an old problem. Journal of the Royal College of Surgeons of Edinburgh 40(5): 292-294.
- McCaul JA, Aslaam A, Spooner RJ, I Louden, T Cavanagh,et al. (2000) Aetiology of seroma formation in patients undergoing surgery for breast cancer. Breast 9(3): 144-148.
- Garbay JR, Picone O, Baron-Merle G, SYacoub,S Lasry, et al. (2004) Le curage de l’aisselle avec capitonnagemusculaire sans drainage. GynécologieObstétrique&Fertilité 32(12): 1039-1046.
- van Bemmel AJM, van de Velde CJH, Schmitz RF, Liefers GJ (2011) Prevention of seroma formation after axillary dissection in breast cancer: A systematic review. European Eur J Surg Oncol37(10): 829-835.
- Hashemi E, Kaviani A, Najafi M, MandanaEbrahimi, HomeiraHooshmand, et al. (2004) Seroma formation after surgery for breast cancer. World J SurgOncol2:44.
- Unalp HR, Onal MA (2007) Analysis of risk factors affecting the development of seromas following breast cancer surgeries. The Breast Journal 13(6): 588-592.
- Hidar S, Mabrouk H, Harrabi I, M Jerbi, H Gahannem, et al. (2005) Volume et durée du drainage post-opératoire après mastectomieradicalemodifiée. Imagerie de la femme 15(1-2): 72-75.
- Theunissen D, Cant PJ, Dent DM (2001) factors that incluence volume andduration of wound drainage after mastectomy and level III axillary node clearance. Breast 10(6): 538-539.
- Forouhi P, Dixon JM, Leonard RC, Chetty U (1995) Prospective randomized study of surgical morbidity fllowing primary systemic therapy for breast cancer. British Journal of Surgery 82(1): 79-82.
- Banerjee D, Williams EV, Ilott J, IJ Monypenny,D J Webster,et al. (2001) Obesity predisposes to increased drainage following axillary node clearance: a prospective audit. Annals of the Royal College of Surgeons of England 83(4): 268-271.
- Barwell J, Watkins RM, Campbell L, Teasdale C (1997) How long should suction drains stay in after breast surgery with axillary dissection? Annals of the Royal College of Surgeons of England 79(6): 435-437.
- Gonzalez EA, Saltzstein EC, Riedner CS, Nelson BK (2003) Seroma formation following breast cancer surgery. The Breast Journal 9(5): 385-388.
- O'Hea BJ, Ho MN, Petrek JA (1999) External compression dressing versus standard dressing after axillary lymphadenectomy. Am J Surg177(6): 450-453.






























