The Use of 18f-Fdg in Oncology and its Applications in Neuroblastomas
Maurino Coimbra de Azevedo Mendes¹ and Emerson Barbosa da Silva¹,²*
1 Department of Biomedical Sciences at Ibirapuera University, Brazil
2 Department of Pathology at Centro Universitário Saúde ABC, Brazil
Submission: July 29, 2020; Published: August 12, 2020
*Corresponding Address:Emerson Barbosa da Silva, Department of Biomedical Sciences, Ibirapuera University, São Paulo – SP, Brazil
How to cite this article:Maurino C d A M, Emerson B d S. The Use of 18f-Fdg in Oncology and its Applications in Neuroblastomas. Canc Therapy & Oncol Int J. 2020; 16(5): 555946.DOI: 10.19080/CTOIJ.2020.16.555946
Abstract
Neuroblastoma is the most common cancer in babies and the third most common cancer in children, behind only leukemia and brain tumor. Knowing this, oncology has been working hard with research and equipment to bring the best possible treatment and diagnosis as quickly as possible to combat not only neuroblastoma but also all cancers that the individual may contract. When we speak of neuroblastoma, cancer that originates from cells of the nervous system, but we do not necessarily find the same in the nervous system since the embryonic process, the majority of which develop in the adrenal gland, so most tumors are found in the region of the abdomen, it can manifest itself in cells near the spinal cord, it can also develop in toxin. It is important to highlight that we cannot think of it as a tumor that manifests in the brain, it only originates in cells of the nervous system. PET / CT is a hybrid technique that combines the nuclear medicine examination of PET (positron emission tomography) with conventional computed tomography, associating metabolic and anatomical information and, thus, allowing diagnoses that are more accurate. The radiopharmaceutical most frequently used for this technology is 18F-FD, a glucose analog, whose accumulation depends on the glycolytic metabolism in the various organs and tissues; it is currently the most used with most of its applications in Oncology and has been shown the most effective for diagnosing cancer beforehand.
Keywords: Neuroblastoma; 18F- FDG, PET / CT; Oncology
Abbreviations: 18F: Radioactive isotope of fluorine with 9 neutrons and mass number 18; CT: Computed Tomography; FDG: Fludesoxiglicose; PET: Positron Emission Tomography
Introduction
Neuroblastoma is a malignant tumor most common in childhood; it is diagnosed in the vast majority of times in the first years of life up to about 5 years of age, being in most cases children of the male. The main signs and symptoms that are usually late are specific and resemble other childhood diseases, making early diagnosis difficult for health professionals [1]. Neuroblastoma originates in primitive forms of nerve cells (usually found in the embryo or fetus) of the sympathetic nervous system, which, in turn, is part of the autonomic nervous system, which controls functions such as breathing, blood pressure, heartbeat and digestion . Most neuroblastomas develop in the adrenal glands, which are on top of the kidneys, in the abdomen or in the nerve cells near the spinal column, the so-called dorsal ganglia. They can also develop in the chest, neck or spine. Some are slow to develop; others progress rapidly [2].
This tumor considered an important challenge for health professionals because it accounts for 15% of the mortality rate when related to pediatric oncology. It is worth mentioning that, since neuroblastoma is a tumor of predominant childhood incidence, occurring in more than 90% of cases in children under 10 years of age, the clinical management of these patients and the development of therapeutic protocols are not part of the daily practice of adult oncologists [1]. The FDG-PET known as a very useful imaging modality in the field of clinical oncology. Numerous studies have reported the clinical significance of FDG-PET in the diagnosis, response to treatment and assessment of the prognosis of various types of tumors. Especially in pediatric neoplasms, the role of FDG-PET been reported in several studies. However, its clinical role for neuroblastoma is still less clear due to the scarcity of published studies that have shown the importance of FDG-PET [3]. 18F-FDG accumulates in primary and metastatic tumors of neuroblastoma, and the use of 18F-FDG PET or 18F-FDG PET/CT for the diagnosis of patients with neuroblastoma is increasing [4].
Discussion
Epidemiology
Neuroblastoma is the most common malignant extra cranial solid tumor in childhood, being the most common cancer diagnosed in the first year of life. The annual incidence is 10.5 per million children under the age of 15, with about 700 new cases per year in the United States. In Brazil, in a study based on the analysis of the population registry of cancer, which included 12 cities and the Federal District, the incidence of Neuroblastoma was 5.9 per million inhabitants under the age of 15 years, however studies that include registration nationwide for definitive conclusions [1]. Neuroblastoma occurs more commonly in children under 5 years of age, with a small predominance in males, and can be located anywhere along the chain of sympathetic ganglia, such as the paravertebral and posterior mediastina regions, but mainly in the medullary region of the adrenal gland. . This embryonic tumor, derived from precursor cells of the sympathetic nervous system, considered an important challenge for health professionals, as it is associated with 15% of mortality related to pediatric oncology [1].
Retrospective study, with analysis of clinical and epidemiological data of 263 patients with NB admitted to the service during the period from 1991 to 2012. Information was obtained through the analysis of medical records. Three patients with incomplete data and two admitted only for palliative care were excluded, being 258 cases in the study. The diagnosis of NB was performed by means of biopsy and anatomopathological examination, or by studying the bone marrow infiltrated by the tumor associated with the presence of catecholamine metabolites in the urine, according to international criteria.1,11 The patients were staged, according to the criteria of the International Staging System for Neuroblastoma (INSS)[5], in stages 1, 2, 3, 4 and 4S (Table 1). The World Health Organization (WHO) histological classification was used [1].
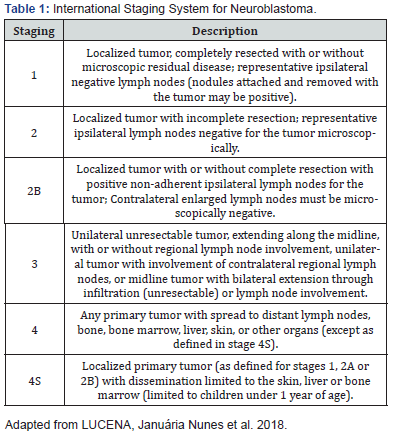
The age at diagnosis of the 258 patients with Neuroblastoma ranged from 4 days to 30 years, with a mean of 40.5 ± 46.4 months and a median of 28.9 months (interquartile range 42.2 months). Children from 1 to 4 years old represented the largest group (49%), followed by children under 1 year old (29%), those from 5 to 9 years old (17%) and, to a lesser extent, those older or older. equal to 10 years (5%). Of the patients, 148 (57%) were male, and 110 (43%), female, with a slight predominance of males and a ratio of 1.3: 1.1 [1]. Regarding the age of the patients, it is noteworthy that, since NB is a tumor of predominant childhood incidence, occurring in more than 90% of cases in children under 10 years of age, the clinical management of these patients and the development of therapeutic protocols they are not part of the daily practice of adult oncologists. For this reason, even older patients, outside the pediatric age group, can be referred to pediatric oncologists, with more experience in this pathology [1].
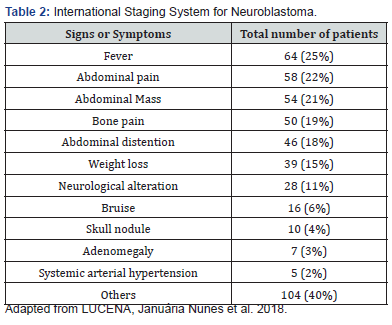
The most frequent signs and symptoms were fever, abdominal pain, abdominal mass and bone pain (Table 2). Of the patients admitted to the study, 3 (1.0%) had Horner’s syndrome, 5 (2.0%) Pepper’s syndrome, and 4 (1.0%) opsomioclonus / ataxia. The most common sites of metastasis were: bone marrow (37.0%), bones (33.0%), lymph nodes (13.0%), liver (10.0%), skin (0.4%) and other locations (5.0%). Of the 258 patients, 86 (33.0%) had metastasis in more than one location [1]. The recognition of signs and symptoms of NB is not always easy, due to the low incidence of the disease and the frequent mimication of the clinical picture with other common diseases in childhood. Knowledge of epidemiological characteristics, such as the predominant occurrence in childhood, anamnesis and physical examination, may suggest the suspicion of the tumor [1].
Physiopathology
Neuroblastoma can appear in the adrenal gland and/or in the cervical, thoracic, abdominal and/or pelvic regions, following the path of the entire chain of the sympathetic nervous system. The most frequent site of neuroblastoma is the abdomen, corresponding to 65% of cases, mainly in the adrenal gland (in 40% of children and in 25% of infants). Abdominal distension, associated with pain and a palpable mass on physical examination, indicates investigation with evaluation of the tumor size, its location and the involvement of other structures represented as findings of hepatomegaly, lymph enlargement and other signs of metastasis of the disease. The presence of massive hepatomegaly at diagnosis can lead to respiratory failure, especially in infants [2]. Thoracic neuroblastoma presents as mediastinal masses in the posterior region, some accidentally diagnosed by a radiological finding, or as Horner’s syndrome (miosis, unilateral prose and anhidrosis), or superior vena cava syndrome [2]. In cases of paravertebral tumors, it is important to observe and detect signs of spinal compression, such as acute and subacute paraplegia, intestinal or urinary dysfunction, or radicular pain. This condition considered a medical emergency, requiring immediate hospitalization and treatment [2].
Glioblastoma
Adult glioblastoma (GBM) is one of the most deadly and recalcitrant of all solid malignant tumors. In the United States alone, an estimated 12,120 patients were diagnosed with GBM in 2016, with a 5% 5-year survival rate. Despite considerable effort, little progress has been made towards prolonged survival in GBM, with much of the perceived improvement coming from the recognition of two prognostic biomarkers: mutations in the isocitrate dehydrogenase (HDI) and methylation of the O6- methylguanine-methyltransferase promoter [6]. For most GBM patients, there is no known cause of the disease. A small minority of patients (<5%) have a critical germline change, predisposing them to many types of tumors, including GBM, and less than 20% of patients with GBM have a strong family history of cancer. The only well-established causal exposure is ionizing radiation; however, only a small minority of cranial tumors caused by radiation exposure are GBMs. Other exposures (for example, cell phones), viral triggers (cytomegalovirus) and germline predispositions continue to explored but have not yet been explored. Clearly established as casual factors. No early detection of GBM is available. Currently, standard MRI provides the most sensitive tool for the initial detection of GBM; however, once a GBM-definable lesion identified with images, the tumor is in an advanced state. GBM clinical presentations are typically related to the functional aspect of the involved brain area [6].
Tumors in certain areas cause obvious symptoms, such as persistent weakness, numbness, loss of vision or language changes. With these symptoms, the size of the tumor in the image tends to be smaller. Tumors in other areas of the brain can result in more subtle symptoms, such as executive dysfunction, mood disorders, fatigue and mild memory disorders. Such tumors are often centered on the frontal lobe, temporal lobe or corpus callosum and tend to be larger after discovery. Seizures occur in only a minority of newly diagnosed GBM patients (approximately 25%) and are generally easy to control with anticonvulsants during the course of the disease; however, there is no clear role for anticonvulsants in patients without seizures. Headaches as an initial symptom are not uncommon and are usually associated with a significant mass effect, either directly from the tumor or through obstruction of the ventricular system [6].
Scintilography
Bone scintigraphy consists of the injection of a small amount of radioactive material into the patient’s vein, after a few hours this material attracted to bone tissue with disease. To record the areas where radioactive material captured, a special camera used, which detects radioactivity and creates an image of the skeleton [7]. The areas of bone damage appear as dark spots on the skeleton image. These points may suggest the presence of metastatic cancer. However, other diseases, such as arthritis, have the same image pattern. To differentiate the result from other diseases, additional imaging tests requested, such as simple radiographs, computed tomography or magnetic resonance imaging [7].
Fluor-18-Fluorodesoxyclycosis (18F-FDG)
The 18F-FDG radiopharmaceutical, fluorodeoxyglucose, is currently the most used with most of its applications in Oncology, Neurology and Cardiology. This radiopharmaceutical belongs to the group of medicines called radiopharmaceutical preparations for diagnosis [5]. 18F-FDG transported into the cell by glucose transporters (GLUT-1 to GLUT-7, GLUT-10 to GLUT-12). This molecule transported across the cell membrane in a manner analogous to glucose, however, only the first stage of glycolysis occurs, which results in 18F-FDG-6-phosphate that remains in tumor cells and not metabolized. In view of the slow dephosphorylation promoted by intracellular phosphatases, 18F-FDG-6-phosphate remains in the tissues for several hours. Its biodistribution has a particular incidence at the cerebral, cardiac, and more discreetly at the pulmonary and hepatic levels. Its elimination occurs via the kidney [5].
The first synthesis of deoxyglucose (DG) dates from the 1960s, having been applied as a chemotherapeutic agent that inhibits cancer cells. Later, in 1976, Dr. Wolf and colleagues were pioneers in the synthesis of the radiopharmaceutical Fluor- 18-fluorodeoxyglucose (18F-FDG), developed at Brookhaven National Laboratory in partnership with the National Institute of Health and the University of Pennsylvania, all located In the USA. Initially, the application was focused on the determination of glycolytic metabolism in the brain, but its usefulness in studies of myocardial and tumor glycolytic metabolism was quickly verified [8]. In biochemical terms, glucose is a monosaccharide-type carbohydrate, which represents an important source of primary energy for most organisms, from bacteria to humans. In addition, it is a precursor to several metabolic pathways and can be stored in the form of glycogen, mainly in the liver [9].
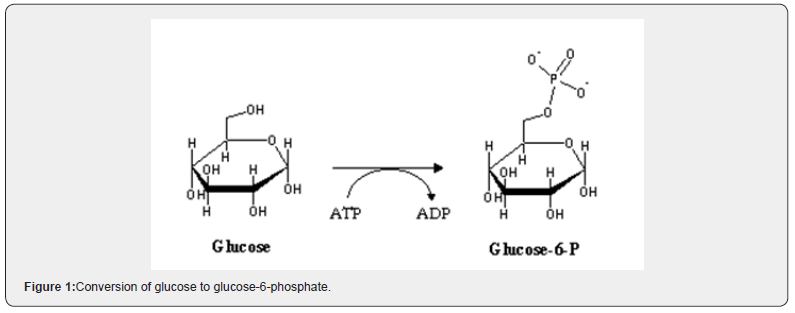
14 transporters, called GLUTs, facilitate the transmembrane transport of glucose into the cell. Once inside the cell, glucose metabolism starts in the cytoplasm in an anaerobic process, called glycolysis. In short, its objective is to break down the glucose molecule into two of pyruvate through the action of an enzymatic sequence that involves 11 reactions. At the end of the reactions, there is the generation of a stable product, lactate, and the energy generated in this process is converted into Adenosine Triphosphate (ATP) and Nicotinamide Adenine Dinucleotide (NADH). In the cytoplasm, the first stage of glycolysis is glucose phosphorylation, where the hexokinase enzyme is responsible for converting glucose into glucose-6-phosphate, by replacing the hydroxyl of carbon 6 with a phosphate group, derived from the hydrolysis of ATP in ADP (Figure 1). Several other steps follow, continuing the process of obtaining energy for the cell [9]. In 1954, Crane (1954) carried out research with the glucose analog, 2-deoxyglucose, concluding that the absence of hydroxyl in the position of carbon 2 does not influence the action of the enzyme hexokinase. On the contrary, the presence of this hydroxyl is indispensable for the performance of phosphoglycosis isomerase. Thus, 2-deoxy-D-glucose-6-phosphate cannot be metabolized and, consequently, accumulates intracellular, not serving as a substrate, such as glucose [10].
18F-FDG in Nuclear Medicine
Of all the radiolabeled molecules used in PET-CT, the FDG-18F is the pioneer and the most used worldwide. Fluorodeoxyglucose (FDG) is a glucose analog, eagerly consumed by malignant neoplastic cells, whose glycolytic metabolism extremely accelerated. Once captured and incorporated into the cell cytoplasm, a radioactive signal is emitted by its radioactive isotope, fluorine-18. This signal captured by the PET scanner, which transforms it into images. The good old PET- CT with FDG-18F represents approximately 99% of the studies carried out in the more than 180 PET- CT scanners existing in Brazil. Make a request for PET-CT that does not detail which radiopharmaceutical is to be used will likely using the glucose analog [6].
Diagnosis of Neuroblastoma by 18F-FDG Scintilography
Data from 40 pediatric patients (age: 5.5±5.6 years; male, 32; female, eight) with NB histopathologically submitted to PET-CT with 18F-FDG (staging, 21 patients; restaging/response monitoring (19 131I-MIBG scintigraphy data were available for 28/40 patients (mean interval of 15 days; staging, 20 patients; restaging/response monitoring, eight patients) [11]. 131I-MIBG scintigraphy and PET/CT images with 18F-FDG evaluated by two doctors in nuclear medicine in consensus and in separate sessions. Histopathology and/or clinical image monitoring taken as the reference standard [11]. The sensitivity, specificity, positive predictive value, negative predictive value and accuracy of PET-CT with 18F-FDG were 100, 50, 91, 89, 100 and 92.50%, respectively. 140 lesions were detected on the PET-CT. In 28 patients who underwent both imaging studies, sensitivity, specificity, positive predictive value, negative predictive value and PET-CT accuracy with 18 F-FDG were 100, 60, 92, 100 and 92.80%, respectively, and 131 I-MIBG were 95.65, 60, 91.67, 75 and 89.20%, respectively [11].
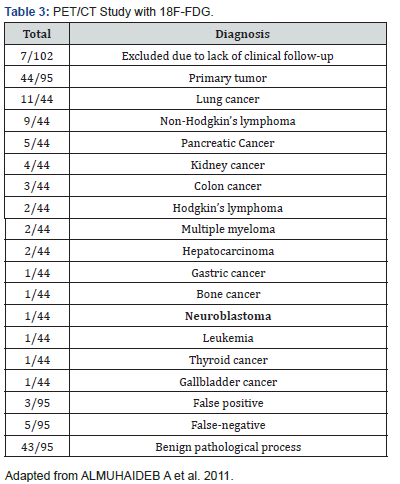
In these 28 patients, PET-CT detected 107 lesions and 131 I-MIBG scintigraphy detected 74 lesions. In a patient-based comparison, there was no significant difference between 18F-FDG PET-CT and 131 I-MIBG, but 18F-FDG PET-CT was greater than 131 I-MIBG in patients with injury compared. Although no difference was observed for primary lesions, PET-CT was greater than 131 Scintigraphy with I-MIBG for the detection of lymph node and bone lesions/bone marrow. PET-CT with 18F-FDG shows high precision in pediatric patients with newborns and demonstrates more injuries compared to scintigraphy with 131 I-MIBG [11]. The diagnosis made in the fusion study compared with the definitive pathological diagnosis. In addition, for the cases so required, the diagnosis issued in study 18F-FDG PET-CT fusion with clinical evolution of patients from the following 3 to 6 months. A cross analysis was performed between the results emitted by the two methods and the measurement was obtained according to the value. A study carried out on 102 patients, and then we can see an explanatory chart (Table 3) with the total number of cases and the diagnosis according to the PET-CT Study with 18F [12].
Main findings in the image
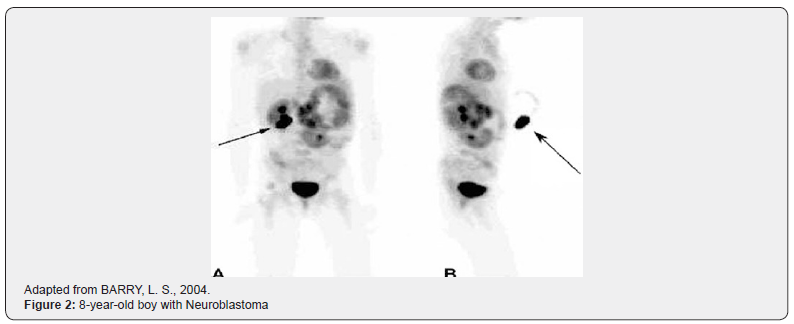
The high-quality PET image of pediatric patients is challenging and requires attention to the problems commonly encountered in the practice of pediatric nuclear medicine, but unusual in the image of adult patients. This includes intravenous access, fasting, sedation, consent and release of urinary tract activity. In figure 2 we have a PET image that is very characteristic of a pediatric neuroblastoma [13]. In a clinical case of an eight-year-old boy with Neuroblastoma. We can see in the presentation an image from the previous projection of the PET scan FDG (Figure 2). The activity superimposed on the right kidney (arrow) shown as representing a residual radiotracer in the injection tube (arrow) in the lateral projection image (B). The large uptake area is the upper left quadrant, at the same level as the right kidney, representing the primary tumor. The decreased uptake region within the mass is due to central necrosis [13].
In this other case, a 31-year-old man, a firefighter, apparently healthy until April 2013 when he noticed the appearance of a nodule in the cervical region to the lower left, so he went to the hospital in the area of residence. He also referred to swelling of the upper quadrants of the abdomen with about 2 months of evolution. He was in good general condition, physically robust (187 cm tall and weighed 78.5 kg, corresponding to a body mass index of 33) and was completely autonomous, so he was classified with Performance Status of 0. He had smoking habits of 10 units / packet / year, mild ethanolic habits and denied toxifilic habits. In the family history, a maternal grandfather who died with gastric neoplasia stood out [14].
The patient referred to an institution where he underwent a PET examination with 18F-FDG to stage the disease, as a hypothesis of diagnosis was lymphoma. This examination revealed a metabolically active disease, with abnormal radiopharmaceutical uptake in an extensive mass with an upper abdominal location, lateralized to the left, reaching a Standardized Uptake Value Maximum (SUVmax) of 6.1, with a heterogeneous aspect and with hypocaptizing areas suggestive of necrosis. Left supraclavicular adenopathies (SUVmax 3.8, retro-crural (SUVmax 4.6), celiac (SUVmax 5.6), leno-aortic and mesenteric adenopathic conglomerate was identified, extending to the aortic bifurcation (SUVmax 5 , 5 in joint evaluation) (Figure 3) [14].
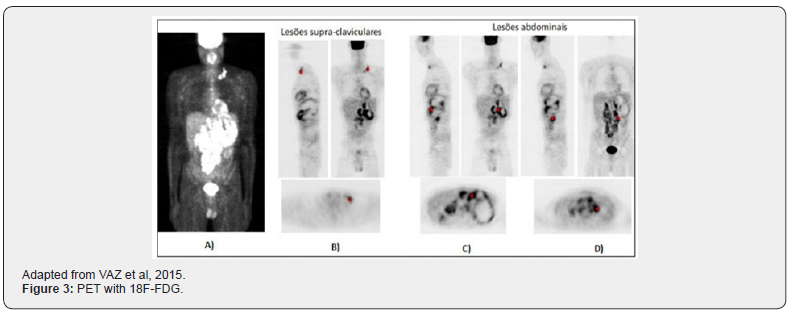
In this SPEC T with 18F-FDG we observe the image A that corresponds to the MIP (Intension Maximum Projection) the node identifies abnormal uptake in lesions above claviculares left and, more exuberant way, the abdominal level. The physiological distribution of the remaining radiopharmaceuticals visualized at the cerebral, cardiac, hepatic, intestinal, renal and bladder levels. The remaining images correspond to the triangulated sagittal, coronal and axial sections in the left supraclavicular lesions B), in the bulky abdominal lesion C) and in the lumbar-aortic ganglia D). The asterisk identifies the area that was triangulated [14].
In this image, 123I-mIBG was useful in the diagnosis because, despite clinical and laboratory suspicion and the cytological result of the cervical ganglion, the retroperitoneal biopsy was not conclusive. Considering the rarity of the case, the cintilograph it body spelling supported the diagnosis, as confirmed both injuries supraclavicular, such as abdominal injuries had neuroectodermal origin. On the other hand, it allowed defining the staging, excluding lesions of neuroendocrine differentiation in other locations and confirming the lesions known clinically and described in morphological exams. The heterogeneity of uptake in the primitive lesion was related to the presence of necrosis, cystic areas and calcification of the abdominal lesion [14].
Main benefits of the drug
Most radioactive isotopes for PET application are produced in cyclotrons. This equipment, in general, consists of an accelerator of charged particles, such as negative hydrogen ions (H-) or deuterons (d-). These particles emitted by an ion source, which is located in the central part of the equipment. Under the action of a strong perpendicular magnetic field, the charged particles describe a circular movement within two hollow metal cavities, the “Dees”, which consist of D-shaped electrodes, responsible for the generation of an alternating electric field, which in turn accelerates the particles [15].
The alternation of polarity occurs in a synchronized manner, causing the kinetic energy of the particles to increase each time they go from one electrode to another. They describe spiral paths, away from the center to measure its kinetic energy increases. When they reach the maximum point of their kinetic energy, an Extraction System, formed by an ultra-thin sheet of carbon guides the beam of particles [8]. On the outer part of the cyclotron, targets containing a stable element (which may be in gaseous, liquid or solid form) are positioned, which will be bombarded by the accelerated particle beam. The bombardment of the target by the particle beam leads to the production of radioisotopes, through a nuclear reaction. This process generates heat. Therefore, it is necessary to connect a cooling system, so that the target does not melt. This system based on a pump that circulates water or helium gas [8,15]. Specifically, for the production of 18F, the irradiated target is 18O, whose most common nuclear reaction is 18 O (p, n) 18F. The radioactive isotope 18F with fluoride can be used to mark deoxyglucose, giving rise to the radiopharmaceutical 18F-FDG [16].
The 18F Isotope
The element fluorine (F) inserted in the family of halogens in the periodic table. One of its most interesting characteristics is that this atom is the only one in its group that has a van der Waals radius very close to that of hydrogen (1.35 and 1.2 Angstrom, respectively). The fact implies that in any organic molecule the CF bond will behave similarly to the CH bond. In addition, fluorine is the most electronegative atom of halogens and promotes the formation of hydroxyl-like electrical dipoles in a molecule. Due to these characteristics, the 18F radioisotope is a positron emitter widely used in Nuclear Medicine, specifically ideal for PET radiopharmaceuticals [17].
The 18F is a radionuclide that emits positrons with low energy (0.64 MeV), therefore it has a short range in the tissues (maximum of 2.4 mm), factors that contribute to the provision of high-resolution images. Its decay occurs more than 97% due to positron emission, a fact that increases sensitivity in diagnosis. In addition, it produced in high specific activities, in addition to having appropriate labeling yields in the synthesis of radiopharmaceuticals for PET use. Its physical half-life of approximately 110 minutes is long enough to allow its transport to other locations, making it possible to carry out examinations in clinics further away from centers that have cyclotron [17].
Pharmacokinetics and Pharmacodynamics of 18F-FDG
According to Schmiedeberg, pharmacology defined as a science that studies “the effects of chemical substances under physiological conditions, which deals with the action of medicines regardless of their practical importance”. Among the subdivisions of this science are pharmacokinetics and pharmacodynamics [18]. Pharmacodynamics addresses the biological and physiological effects of drugs in the body, being concerned with aspects such as absorption, distribution, metabolism and excretion [19]. Specifically, the radiopharmaceutical 18F-FDG, which is a radioactive analog of glucose, crosses the cell membrane by facilitated diffusion through glucose membrane transporters, a process that occurs in competition with other sugars. Once inside the cell, the process of phosphorylation by the enzyme hexokinase occurs, giving rise to glucose-6-phosphate, as explained in the previous sub-item. The cell concentration of 18F-FDG, that is, the accumulation of radiopharmaceutical in a given region, is proportional to the cell’s glycolytic activity. Malignant cells are characterized by accelerated glycolysis and reduced capacity to produce energy aerobically, which implies high rates of glucose uptake, necessary to maintain the cells [20].
The areas of higher glucose uptake detected in the images are associated with an increase in the number of tumor cells; greater expression of glucose unit transporters in the GLUT family, specifically GLUT1, GLUT3 and GLUT5; and increased activity of some glycolytic enzymes, such as hexokinase, phosphofrutase and pyruvate dehydrogenase. Another parameter that differentiates tumor cells from healthy ones is the level of glucose-6-phosphate since glucose dephosphorylation occurs more slowly in cancer cells. Tumors frequently present hypoxia, a fact that stimulates the uptake of 18F-FDG, in order to accelerate glycolytic metabolism, especially anaerobic. In oncology, 18F-FDG is based on differential glucose metabolism in malignant and benign tissues [21].
The pharmacokinetic properties refer to the path taken by the drug in the body. After its intravenous administration, 18F-FDG in the vascular compartment obeys biexponential kinetics. This radiopharmaceutical has a distribution half-life of approximately 1 minute and 12 minutes for elimination. Cell uptake of 18F-FDG is done by partially insulin-dependent carriers, so they can be influenced by certain physiological conditions, such as diet, nutritional conditions and the existence of diabetes mellitus [22]. As 18F-FDG undergoes only the first phase of glycolysis, the fluordeoxyglucose18F-6-phosphate product accumulates in the cell and not metabolized. The 32 dephosphorylation processes carried out by intracellular enzymes occur slowly, so fluorooxyglucose18F-6-phosphate trapped in the tissue for several hours. In healthy individuals, 18F-FDG accumulates mainly in the brain (about 7% of the activity administered) within 80- 100 minutes after injection; heart (approximately 3%) after about 40 minutes; and a small incidence seen in the pulmonary and hepatic regions, corresponding to 0.3-2.4% of the total activity. This radiotracer also connects to the eye, pharynx and intestine muscle, but to a lesser extent [5].
In individuals with tumor, similarly, the radiopharmaceutical crosses the cell membrane, undergoes only the first phase of glycolysis and is retained intracellularly in the form of fluordoxyglucose18F-6-phosphate, thus marking the tumor region [20]. The 18F-FDG excretion pathway is predominantly renal, in which 20% of the activity eliminated by urine, approximately two hours after the injection [5].
Indications and limitations of 18F-FDG
18F-FDG is the most widely used PET radiopharmaceutical today, whose applications in oncology are aimed at differentiating between benign and malignant disease; identification of the tumor location for biopsy or surgery planning; detection of primary tumors; measurement of the volume of radiopharmaceutical captured to classify the degree of malignancy; tumor staging; distinction between residual and healed cancer in the posttreatment phase; detection of recurrence; determining the therapeutic response and using it to guide radiotherapy (KOSTAKOGLU, 2003; VIANA, 2012). This radiopharmaceutical is applicable in the detection of cancers of the respiratory system, head and neck, digestive system, breast, genitals, thyroid, central nervous system, lymphoma and hidden primary tumor [6,22], 18F-FDG, in the chemical concentration administered for diagnostic purposes, does not appear to have pharmacodynamic activity [2].
Despite being widely used, the 18F-FDG has some limitations. This radiopharmaceutical is not specific for the detection of neoplasms with high glycolytic metabolism, its absorption also occurs in inflammatory processes and 33 granulomatous lesions, which can result in false positive diagnoses. In addition, some cells have high glucose consumption even in the absence of pathologies, which leads to a high uptake of 18F-FDG in that region, such as brain cells, making it difficult to distinguish pathological from physiological marking. Physiological accumulation also seen in the myocardium, stomach and bladder [20]. The radiotracer distribution also altered in some behavioral states, such as in cases of stress or physical exercise, where there is an accumulation in skeletal muscle. Thus, it is interesting to develop methods to make the 18F-FDG more specific for the detection of tumors, in order to minimize false-positive diagnoses [5,8,20].
Synthesis of the 18F-FDG
In general, the 18F-FDG synthesized in automated equipment, called synthesis modules. Automated synthesis has several advantages over manual, such as, for example, greater reproducibility of the process, reduced synthesis time, and decreased operator exposure to radioactivity due to manipulation [23]. The greatest revolution in the synthesis of 18F-FDG occurred in 1986 with the implementation of the nucleophilic substitution technique, where the nucleophile is the 18F ion [22]. The 18F-FDG donor centers for carrying out this work - CRCNNE, IPEN-SP and CDTN-MG, all bodies linked to the National Nuclear Energy Commission (CNEN) - carry out the synthesis of this radiopharmaceutical in automated modules. Initially, 18F produced in the cyclotron and sent to the synthesis module in aqueous solution. To separate the 18F from the enriched water, the solution filtered through an anion exchange column, in which the fluoride ion retained in the resin while the cationic and watersoluble impurities go into the disposal bottle, together with the enriched water. Then, 18F eluted with a solution of Water: Acetonitrile (1:1) containing potassium carbonate and Kryptofix 222 (C18H36N2O6), in an equimolar ratio. The elution product is potassium fluoride. The next step is the evaporation of the residual solvents (water / acetonitrile), which occurs in a closed system with a vacuum of 1 mbar, under a flow of ultrapure nitrogen and a temperature of 95 ° C, obtaining the dry 18F [24,25].
Conclusion
The literature has shown that the Neuroblastoma most commonly occurs in children under 5 years of age, with a slight predominance in male. According to clinical cases, the most frequent signs and symptoms were fever, abdominal pain, abdominal mass and bone pain. The main advantage of PET-CT is that the exam provides detailed two-dimensional images, which show anatomy (through CT) and function (through PET). The two images (CT and PET images) can be viewed separately, or one can be overlaid on the other. Therefore, this technique provides useful information about anatomy and function and can help the doctor to identify abnormalities that affect anatomy and / or function, this technique is particularly useful for cancers, PET can show where a cancer is and where it is has spread and how it is responding to treatment. The main disadvantage is that when PET-CT performed during a single exam, the radiation dose is significantly increased and as the radionuclides used in PET release radiation for only a short time, PET can be performed only if the radionuclide is produced nearby and can be obtained quickly, PET is relatively expensive and is not widely available.
Fluorine-18 (18F) is a β + emitting radionuclide that has the most favorable physical and nuclear properties for the PET image: 97% β + decay, low maximum energy from the emitted positron and relatively short half-life. The drug is efficient in the screening of neuroblastoma because its biodistribution has a particular incidence at the cerebral, cardiac level, and in a more discreet way at the pulmonary and hepatic level, its elimination occurs through the kidney. There are some perspectives regarding the technique, for example, testing the efficiency of the nano radiopharmaceutical as a diagnostic probe of brain tumor, through ex vivo biodistribution assays and PET/CT images of mice with tumor.
Acknowledgement
We are grateful for the coordination of the undergraduate course in Biomedicine at Ibirapuera University.
Conflict of Interest
We declare to have no conflict of interest.
References
- Lucena J, Alves M, Abib S (2018) Aspectos clínicos, epidemiológicos e sobrevida de crianças com neuroblastoma: 21 anos de experiência do instituto de oncologia pediátrica, São Paulo, Brazil.
- Instituto Nacional Do Câncer (2020) Neuroblastoma - versão para Profissionais de Saú
- Seo k, Muhammad R, Yong-il k, Gi Jeong Cheon, Hyuong Jin Kang, et al. (2017) Significado clínico do pré-tratamento FDG PET/CT no neuroblastoma pediátrico MIBG-Avid. Nucl Med Mol Imaging 51(2): 154–160.
- Hiroaki I, Shingi I, Katsuhiko k (2018) Diagnostic performance of 18F-FDG PET/CT and whole-body diffusion-weighted imaging with background body suppression (DWIBS) in detection of lymph node and bone metastases from paediatric neuroblastoma. Annals of Nuclear Medicine 32: 348–362.
- Icnas: Instituto De Ciências Nucleares Aplicadas À Saúde: Radiofármaco: 18f-Fdg.
- Anjos D A (2018) PET/CT em oncologia. Diretrizes Oncoló
- GRAAC: Neuroblastoma – Diagnóstico Disponí
- Welch M J, Redvanly CS (2003) Handbook of Radiopharmaceuticals Radiochemistry and Applications.
- Nelson DL, Cox MM (2011) Princípios de Bioquímica de Lehninger, (5 edn)., Artmed.
- Crane RK, Sols A (1954) The noncompetitive inhibition of hexokinase by 6-glucose6-phosphatase and related compounds. Journal of Biological Chemistry, pp. 597-606.
- Dhull, Varun S, Sharma, Punit, Chetan, et al. (2020) Valor Diagnóstico De 18f-Fdg Pet/Ct No Neuroblastoma Pediátrico Em Comparação Com Cintilografia Com 131i-Mibg Disponí
- Almuhaideb A, Papathanasiou N, Bomanji J (2011) 18F-FDG PET/CT imaging in oncology. Ann Saudi Med 31(1): 3-13.
- Barry L, S Imagem PET em oncologia pediátrica Disponí
- Vaz S, Ferreira, C Silva A, R. Sousa, ID Patrocínio (2015) Caso clínico de Neuroblastoma do adulto: Utilidade diagnóstica e terapêutica da medicina nuclear.
- Jasinowodolinski D, Dimenstein R (2012) Bases Físicas e Tecnológicas PET e TC, 1 ed. São Paulo: Editora SENAC São Paulo, Brazil.
- Okuno E, Yoshimura E (2017) Física das Radiações, (2nd edn). Oficina de Textos.
- Vallabhajosula S (2011) A Broad Overview of Positron Emission Tomography Radiopharmaceuticals and Clinical Applications: What is new? Seminars in Nuclear Medicine. 41(4): 246-264.
- Bittencourt SC, Caponi S, Maluf S (2020) Farmacologia no século XX: a ciência dos medicamentos a partir da análise do livro de Goodman e Gilman. História, Ciências, Saúde – Manguinhos, Rio de Janeiro, 20(2): 499-520.
- Brunton, l L, knollman, Bjorn C (2011) chabner b.a. As Bases Farmacológicas da Terapêutica de Goodman & Gilman. (12 ed). Porto Alegre. McGraw-Hill, United States.
- G J Cook, I Fogelman, M N Maisey (1996) Normal physiological and benign pathological variants of 18-fluoro-2-deoxyglucose positron-emission tomography scanning: Potential for error in interpretation. Seminars in Nuclear Medicine 26(4): 308-314.
- Norbert A (2004) GLUT1 Expression in tissue and 18F-FDG uptake. The Journal of Nuclear Medicine 45(6): 930-932.
- Hamacher k, Coenen HH, Stocklin G (1986) Efficient stereospecific synthesis of no-carrier-added 2-[18F]-Fluoro-2-Deoxy-D-Glucose using aminopolyether supported nucleophilic substitution. J Nucl Med 27: 235-238.
- Nicolás Ricardo de Jesús Sánchez Casas, José Antonio Serna Macías, Óscar Quiroz Castro, Yancarla Conde Canaviri, Catalina Romo, et al. (2020) Papel del 18F-FDG PET/CT en la evaluación de tumores primarios de origen desconocido y síndrome paraneoplásico; experiencia del Hospital Ángeles del Pedregal.
- Welch MJ, Redvanly CS (2003) Handbook of Radiopharmaceuticals: Radiochemistry and Applications. John Wiley & Sons, Hoboken.
- Scott PJH, Hockley BG (2012) Radiochemical Synthesis-Radiopharmaceuticals for Positron Emission Tomography. New Jersey/EUA, John Wiley and Sons v 1: 3- 14.






























