Factors Influencing Long Term Survival in Metastatic Castration Resistant Prostate Cancer Patients: a Spanish Multicentre Experience
O Fernández Calvo1, T Alonso Gordoa2, N Láinez3, Á Rodríguez Sánchez4, G De Velasco5, U Anido6, R García Marrero7, E Martínez Ortega8, A Viana9, C Caballero Diaz10, J García Sánchez11, F Zambrana Tevar12, Á Pinto Marín13, R Lastra14, A Rodríguez15, C Perezagua16, Á Gaizka Montesa17, S Maciá18, R C Álvarez Llosa1 and S Vázquez Estévez19*
1Complejo Hospitalario Universitario Ourense, Ourense, Spain
2Hospital Universitario Ramón y Cajal, Madrid, Spainia
3Complejo Hospitalario de Navarra, Pamplona, Spain
4Complejo Asistencial Universitario de León, León, Spain
5Hospital Universitario 12 De Octubre, Madrid, Spain
6Hospital Clínico Universitario de Santiago de Compostela, Santiago De Compostela, Spain
7Hospital Universitario Tenerife, Tenerife, Spain
8Hospital Universitario de Jaén, Jaén, Spain
9Hospital Nuestra Señora del Prado, Talavera De La Reina, Spain
10Hospital General Universitario Valencia, Valencia, Spain
11Hospital Arnau de Vilanova, Valencia, Spain
12Hospital Infanta Sofía, San Sebastián De Los Reyes, Spain
13Hospital Universitario La Paz, Madrid, Spain
14Hospital San Jorge, Huesca, Spain
15Hospital General de La Palma, Spain
16Hospital de Henares, Coslada, Madrid, Spain
17Hospital Carlos Haya, Málags, Spain
18ICON Clinical Research, Alicante, Spain
19Hospital Universitario Lucus Augusti, Lugo, Spain
Submission: March 02, 2020 Published: March 10, 2020
How to cite this article: Fernández Calvo, Alonso Gordoa, Láinez, Rodríguez Sánchez, De Velasco et al. Factors influencing Long term Survival in Metastatic Castration Resistant Prostate Cancer Patients: a Spanish Multicentre Experience. Canc Therapy & Oncol Int J. 2020; 16(1): 555926. DOI:10.19080/CTOIJ.2020.16.555926
Abstract
Background: Cabazitaxel is approved as second line treatment for metastatic castration resistant prostate cancer (mCRPC) after docetaxel failure. However, it is difficult to establish which patients will reach benefit. In this study, we aimed to analyze retrospectively clinical characteristics for cabazitaxel long responding mCRPC patients.
Patients and methods: Observational, multicentric, retrospective study. All consecutive mCRPC patients receiving at least 10 cabazitaxel cycles were included from 19 centres in Spain. A descriptive analysis was performed. Overall survival (OS) was analyzed using a Kaplan-Meier model with 95% confidence intervals.
Results: 81 patients were included. The median age was 64.29 years, Gleason ≥8 in 63.9% patients, visceral involvement in 78.8%, 42.5% of patients had bone metastases, disease-related symptoms were present in 51.8% patients. The number of events at this cut-off point of the study is 66. Median time to progression to previous docetaxel was 5.3 months. Up to 38.09% of the patients had had a time to progression to prior docetaxel longer than 20 months. Cabazitaxel was well tolerated, being asthenia main adverse event. Median OS was 26.9 months (95% CI: 16.7; 37.2). For those patients with Gleason ≤ 7, median OS reached 29.3 months, versus 21.6 months for Gleason ≥ 8, (p=0.4). There were no differences based on presence of symptoms and visceral metastases. However, there was a trend to a longer survival in those patients who had received previous docetaxel for a more extended period (28.6 months for 3-6 docetaxel months compared to 21.6 in < 3 months).
Conclusion: This sample size is a weakness to assess prognostic factors, but there is a longer benefit in those patients with ECOG-PS 0-2 and longer treatment benefit from docetaxel. In addition, a trend to OS benefit for patients having previous treatment duration with ADT ≥20 months was observed; but longer follow-up is required. All patients regardless visceral metastases or symptoms achieved a meaningful benefit by receiving cabazitaxel for at least 10 cycles.
Introduction
Prostate cancer is the second most common tumor in men worldwide, counting 1,276,106 new cases and causing 358,989 deaths [1] in 2018. Mortality rates have been declining among the last years, mainly reflecting early diagnosis and treatments improvements [2]. There was a lack of treatments for advanced castration resistant prostate cancer until 2004, when the US Food and Drug Administration (FDA)-approved docetaxel as the first chemotherapeutic drug showing a benefit in overall survival (OS). After that moment, the number of approved therapeutic drugs has dramatically increased by incorporating sipuleucel-T, abiraterone, enzalutamide, radium-223, and cabazitaxel. In the absence of sequencing data for different alternatives, clinicians need to decide which therapies to use based on existing nontrial- based evidence. Differences in study designs make it difficult to draw cross-trial comparisons. Currently, there is little objective information other than the medical comorbidities and choice of the patient, in driving treatment sequence decisions; usually these agents are given sequentially, at the discretion of the treating physician. Cabazitaxel has recently proved its efficacy in the second and third line, as detailed below [3]. In addition, other therapies may be beneficial in some subsets of patients who harbor BRCA 1/2 and ATM mutations, as they can confer sensitivity to PARP inhibitor olaparib [4].
Cabazitaxel is a taxane used in the treatment of metastatic castration resistant prostate cancer (mCRPC) refractory to docetaxel [5]. Cabazitaxel was developed due to its ability to overcome tumor resistance to taxanes, as supported by the phase III TROPIC trial. Seven hundred fifty-five men with mCRPC who had developed progressive disease by Response Evaluation Criteria in Solid Tumor (RECIST) or prostate cancer working group 2 (PCWG2) criteria, during or after docetaxel were randomized to receive either mitoxantrone (12 mg/ m2) and prednisone 10 mg daily or cabazitaxel (25 mg/m2) plus prednisone. Median number of received cycles was 6, but patients could not receive over 10 cycles due to mitoxantrone cardiac toxicity. The primary endpoint of OS was meet and median OS in ITT population was 15.1 months for cabazitaxel versus 12.7 months for control arm (6. Progression-free survival (PFS) was also statistically better in the cabazitaxel group (2.8 vs 1.4 months, P<0.0001). A subgroup analysis showed that patients with measurable disease had significantly better tumor response rate (14.1% vs 4.4%, P=0.0005) and PSA response rate with cabazitaxel compared with mitoxantrone (39.2% vs 17.8%, P=0.0002). Given the overall benefit, the FDA-approved cabazitaxel for men with mCRPC with disease progression following docetaxel in 2010 [6].
Prophylaxis with supportive growth factor was strongly recommended after this study, as neutropenic sepsis was the most common cause of death (7/18 deaths in the cabazitazel arm). In other studies that were developed thereafter, supportive growth factor was administered. In the German Compassionate Use Program, a closer complete blood count monitoring was performed, with 17.1% of patients treated with granulocyte colony stimulating factor (G-CSF). There was 1.8% incidence of neutropenic fever among 111 patients, with four infectionor hematological-related deaths reported [7]. In the United Kingdom Early Access Programme (UK EAP), 112 docetaxel refractory mCRPC patients were treated included. A total of 84.8% of patients received G-CSF. The neutropenic sepsis rate was low (6.3%), occurring in patients who had not received prophylactic growth factor, and there were four infection-related deaths [8].
Additionally, cabazitaxel was compared versus abiraterone or enzalutamide recently in the CARD trial, showing a longer imaging-based progression free survival (PFS) for cabazitaxel (8 months, versus 3.7 for androgen signaling targeted inhibitor), better OS (13.6 versus 11 months) and other clinical outcomes, in patients with mCRPC who had been previously treated with docetaxel and the alternative androgen-signaling–targeted agent (abiraterone or enzalutamide). Hence, it can be said that cabazitaxel works in the third line, besides its proved efficacy in the second line [3].
Despite cabazitaxel got approval, some questions remained whether a lower dose could still be as efficacious with less toxicity. This led to the FDA-mandated PROSELICA study, a randomized Phase III noninferiority study of cabazitaxel 20 mg/ m2 (C20) vs 25 mg/m2 in 1200 patients with mCRPC previously treated with docetaxel. Of the 10 planned cycles, both groups completed a similar number of median cycles of therapy, with more dose reductions in the 25 mg/m2 arm. There were more grade 3-4 treatment-related toxicities and more treatmentrelated deaths in standard dose arm. However, patients treated on the 25 mg/m2 arm had significantly improved PSA response rates (42.9% vs 29.5%, P<0.001) and improved clinical response, but there was no difference in PFS. OS was not inferior in the lower dose arm (hazard ratio [HR] 20 vs 25, HR=1.024). This met the FDA-mandated prespecified noninferiority endpoint, which maintained 50% of the OS benefit of the dose in the originally reported TROPIC trial [9].
A majority of patients in these four studies completed 6 cycles of cabazitaxel treatment and almost 20% received up to 10 cycles of treatment. In addition, most patients received the full dose of study treatment (median relative dose intensity was almost 100% of the predicted dose intensity) [10]. Clinical data on cabazitaxel use beyond 10 cycles is limited. Data from docetaxel has suggested that more cycles may be associated with better outcomes, but there are no predictive factors to know which patients will get a long lasting PFS with cabazitaxel [11]. To assess these factors, we conducted a multi-institutional retrospective study and analyzed the efficacy and safety of realworld patients with mCRPC focusing on those patients that received, at least, 10 cycles of cabazitaxel.
Patients and Methods
A multicenter, epidemiological, retrospective, observational study was performed at 19 Spanish sites. All consecutive patients that received more than 10 cycles between 2007 and 2017 were included. Patients background, and treatment outcomes including PSA decline, progression free survival (PFS), OS, and adverse events (AEs) were investigated. Patients were scheduled to receive cabazitaxel (administered dose at physician’s discretion) as a 3-weekly regimen. Disease progression was defined according to PCWG2 [12]. Treatment failure was defined as discontinuation of cabazitaxel, which was determined by disease progression and/or AEs, or patient refusal. AEs were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0. Statistical analysis. All statistical analyses were performed using platform x86_64-apple-darwin15.6.0.
Results
Up to 81 patients were included. Baseline characteristics are detailed in table 1. The median age was 64.29 years, Gleason was ≥8 in 63.9% patients. Prior duration of androgen deprivation therapy (ADT) was at least 20 months in 60% patients. Visceral disease was present in 78.8% patients and most common metastatic location was bone; up to 48.2% patients had diseaserelated symptoms. Nearly all patients (97.6%) had received prior docetaxel, being 8 the median number of cycles. Only eleven patients had developed progressive disease while on docetaxel, and most patients (55) developed progressive disease at least three months after last docetaxel cycle. ECOG Performance Status (PS) was 0 in 25 patients (31.2%), 1 in 44 patients (55%) and 2 in 11 patients (13.8%). Patients’ backgrounds are shown in Table 1.
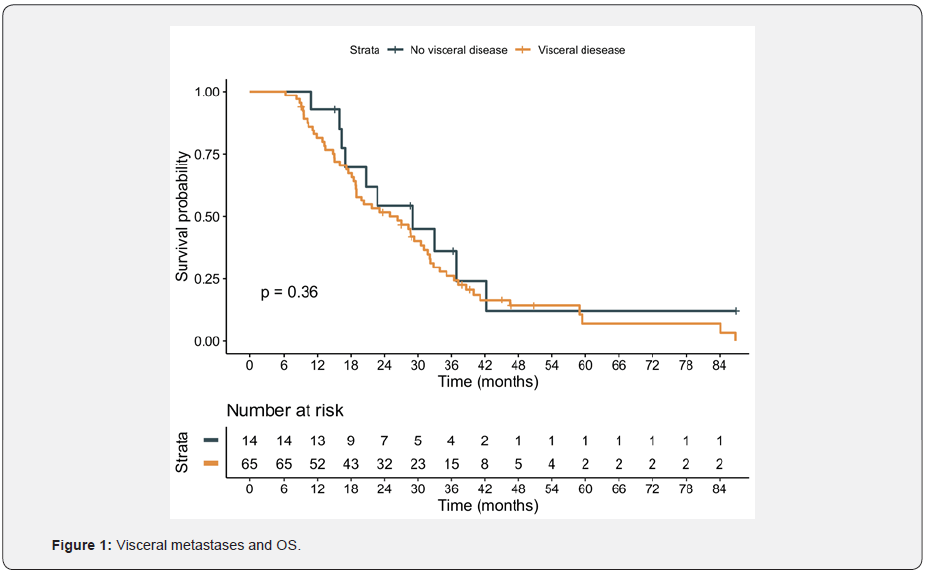
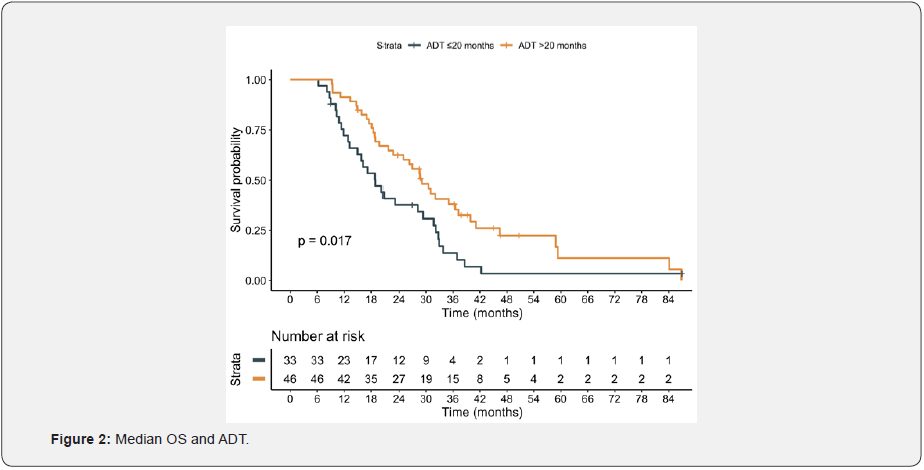
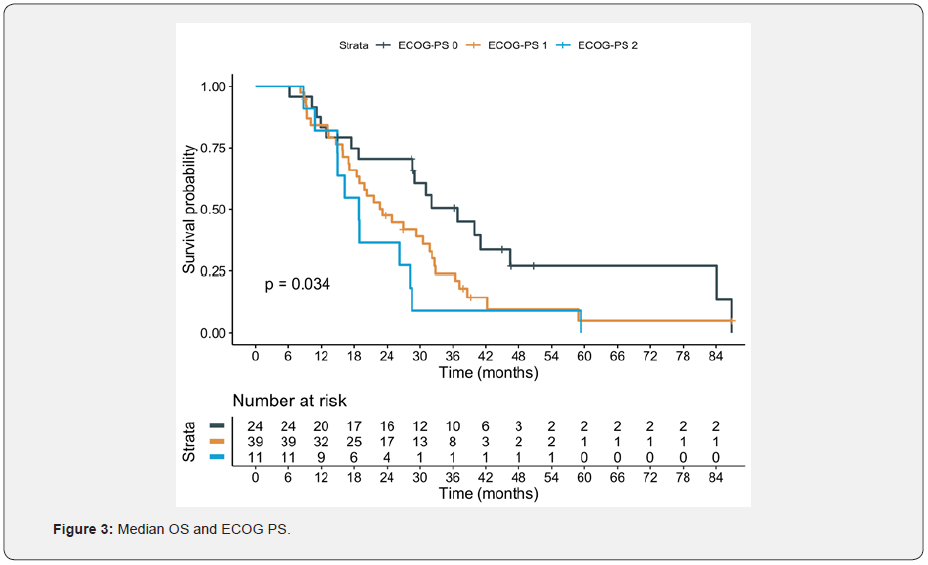
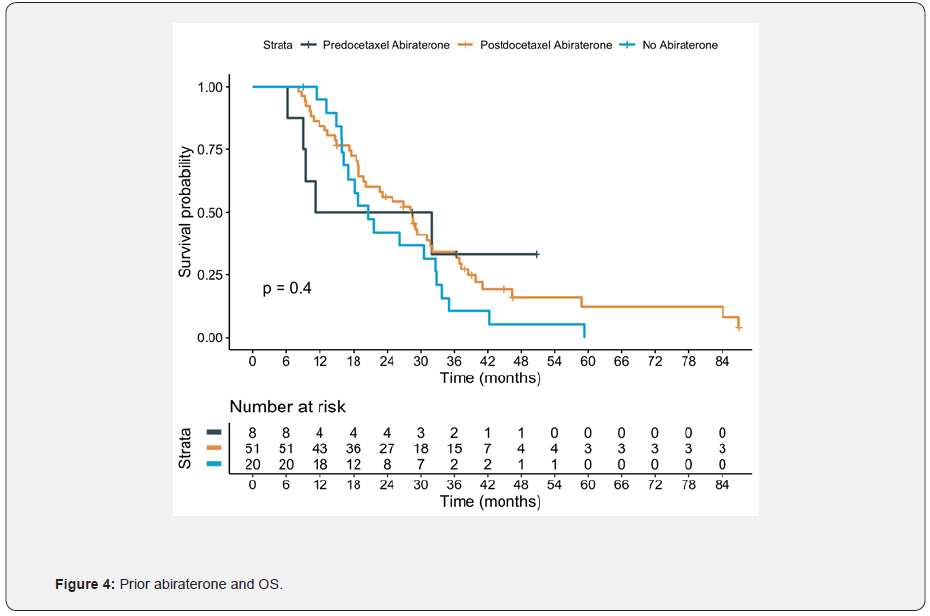
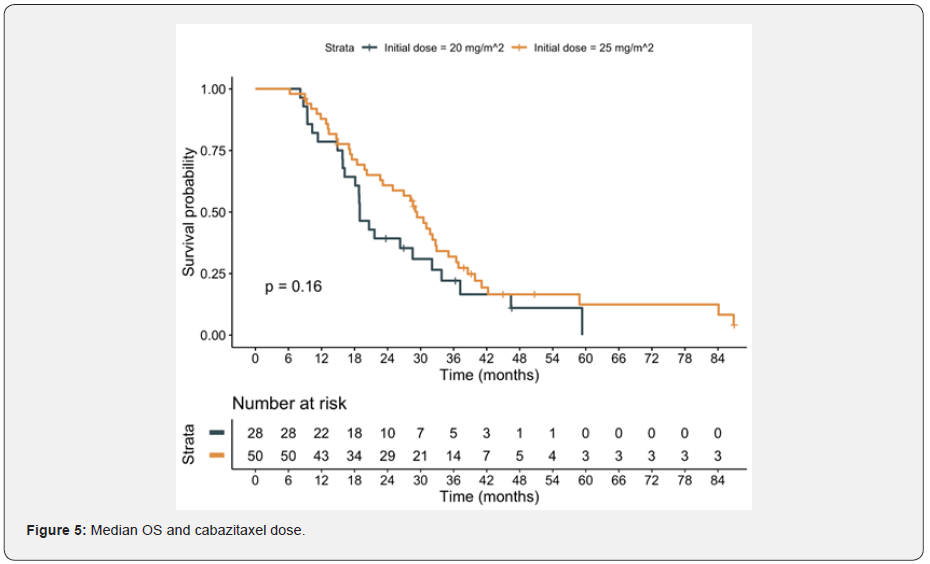
In this population, cabazitaxel was well tolerated. Forty-five (54.2%) patients received concomitantly G-CSF. Main adverse effect being asthenia (25.64%); other adverse effects were diarrhea, anemia, neutropenia, anorexia and nausea. Median OS was 26.3 months (95% CI: 19.8; 31.7). For those patients with Gleason ≤7, the median OS reached 29.0 months, compared with 22.7 months for patients with Gleason≥8, without statistically significant differences (p=0.16). There were no differences in survival regardless presence of disease related symptoms and visceral metastases (figure 1). However, patients with a duration of ADT ≥20 months had better OS (29 months; 95% CI: 22.7; 39.9, p = 0.017), compared with patients with duration of ADT < 20 months (18.9 months; 95% CI: 14.9; 31.7) and a hazard ratio (HR) of 0.55 (95% CI: 0.33; 0.90) figure 2. Additionally, despite there were not statistically significant differences, 32 patients (38.1%) had received previously more than 6 cycles of docetaxel. The most important prognostic factor was ECOG at the beginning of cabazitaxel, HR 1.64 (95% CI: 1.12; 2.39). Patients with ECOG 0 reached a median survival of 40 months, compared to 18 months for patient with ECOG 2 (p=0.034) figure 3. Focusing on prior treatments, sixty-five patients had also received prior abiraterone, mainly in the post-docetaxel setting (range 3-26 cycles). Seventeen patients had also received prior enzalutamide (range 1-11 cycles) and three patients prior Rad-223. There were no differences regardless the treatment line when prior abiraterone was administered. There were no differences in results based on the initial doses of cabazitaxel (20 versus 25 mg/m2) Figures 4 & 5.
Discussıon
The strength of this study was the broad eligibility criteria that encompassed all mCRPC patients treated with cabazitaxel for at least 10 cycles, whatever the treatment line. All included patients in this study received at least 10 cycles of cabazitaxel. As it has been shown, sample was quite heterogeneous, including patients with ECOG PS 0, 1 or 2, patients with visceral disease and patients who had received two or three prior lines. Regardless these factors, all included patients could be considered as good responders, not only to cabazitaxel, but also to prior therapies (ADT, docetaxel and abiraterone). This supports the idea of no cross resistance with prior treatments. Additionally, considering that patients had been pre-treated with other anticancer lines, it is especially important this clinical benefit in such a poor prognosis scenario. Our analysis has in addition a number of limitations, including its retrospective nature, the lack of sample size calculation, as well as the lack of assessment of peripheral neuropathy.
In our study, only eleven patients had developed progressive disease while on docetaxel, and most patients (55) developed progressive disease at least three months after last docetaxel cycle. This is quite consistent with TROPIC study, where 38% patients developed progressive disease at least 6 months after last docetaxel. Our study suggests that continuous chemotherapy with cabazitaxel beyond 10 cycles may be beneficial, keeping the benefit in survival without increasing adverse events significantly. These particular data cannot be compared with those extracted from TROPIC as, in the TROPIC study patients were not allowed to receive more than 10 cycles. Hence, our study supports the idea of further analyzing the benefit of extended cabazitaxel. Within this group, prognosis was especially good in those patients with ECOG 0 and patients who had received ADT for a period longer than 20 months. Despite survival differences did not reach statistical significance, there was also a trend of a higher benefit in patients who had received docetaxel for over 6 cycles and abiraterone for at least 12 months. The CARD study (3) showed that cabazitaxel is superior with regards to radiographic PFS in patients who relatively rapidly progress through first-line anti-androgen therapy. This supports the idea that patients who do not have extended responses to anti-androgen therapy need cytotoxic therapy rather than another anti-androgen agent.
Same results were seen by Shiota el al, who found, in a retrospective study including all patients treated with cabazitaxel at several institutions that, among men treated with >10 cycles of cabazitaxel, more cycles of prior docetaxel chemotherapy had been received. They also identified other characteristics in these patients, including absence of pain, and absence of bony and visceral metastases [13,14]. Interestingly, in our sample, visceral disease was present in 78.8 patients and most common metastatic location was bone; up to 48.2% patients had diseaserelated symptoms, which were completely different to Shiota’s sample characteristics. Therefore, we can say that close to 80% of our patients reached an important benefit (meaning at least 10 cabazitaxel cycles) despite having visceral metastases and disease-related symptoms, hence, being classically considered as poor prognosis patients.
There were no differences in efficacy regardless visceral metastases. In the TROPIC study, half of the patients enrolled had at least one site of soft tissue metastasis, with 25% having visceral disease. Additionally, Shiota et al also confirmed that PFS and OS were better in patients receiving at least 10 cycles of cabazitaxel, without an increase in the incidence of adverse events. This study included 63 Japanese patients with mCRPC who were treated with cabazitaxel from 2014 to 2017. The oncological outcomes and adverse events were documented, and prognostic factors for oncological outcomes and predictive factors for AEs were analysed. PSA decline was observed in 68.3% of patients, including 25.4% who achieved a ≥ 50% decline. The median PFS and OS were 4.3 and 9.0 months, respectively. More cycles of prior docetaxel therapy were identified as common favourable prognostic factors for PFS and OS. Severe neutropenia, febrile neutropenia, and severe nonhaematological AEs were commonly observed (73.0%, 33.3%, and 23.8% of patients, respectively). However, > 10 cycles of cabazitaxel was not associated with increased incidence of AEs. Notably, lethal adverse events did not occur in patients who received at least 10 cycles of cabazitaxel, while grade 4-5 adverse events were case of withdrawing treatment during the initial cycles. In conclusion, cabazitaxel chemotherapy can be considered as active in these patients treated with > 10 cycles of docetaxel chemotherapy, with an acceptable safety profile. Hence, treatment with cabazitaxel after > 10 cycles of docetaxel may be an appropriate option when it can be administered [14].
Presence of pain and short response to docetaxel had been previously reported as adverse predictive factors to cabazitaxel in other studies [13,14]. Performance status is a clearly prognostic factor, with a dramatic benefit for patients with ECOG 0, achieving a median of 40 months OS. There was no relation between ECOG and selected cabazitaxel dose, and there was no impact on clinical benefit for those patients receiving a dose of 25 mg/m2 as initial dose, however, there was a trend to a longer OS for the higher dose (30 versus 20 months, p=0.16).
Conclusion
Prostate cancer has had new approved treatments and landscape has been changing rapidly. Further studies are needed, to help optimizing the use of these agents by identifying those patients who most benefit and discovering the best way of giving as sequential single agents. Card trial showed that cabazitaxel improved clinical outcomes compared with the androgensignaling– targeted inhibitor after docetaxel. However, clinical trials have tested it being administered for up to ten cycles. Our results support that cabazitaxel may be well tolerated beyond ten cycles and may potentially provide survival benefit. Indeed, OS was dramatically longer in our study compared to TROPIC trial (23 versus 15.1 months), which is completely attributed to the sample selection, as only those patients who had at least 7.8 months PFS were included.
Comparative data between those patients who continue on treatment after ten cycles and patients who withdraw cabazitaxel are not available. However, as the incidence of AEs is aligned with those that appeared on TROPIC, it could be thought that number of AEs does not increase with a longer treatment duration, hence, no cumulative toxicities were seen. It should be noted that a high percentage of our patients had received two or more prior lines, oppositely to the TROPIC study, where all patients received cabazitaxel in second line after docetaxel.
Conflicts of Interest
Dr. Vázquez Estévez reports non-financial support and other from Pfizer, personal fees, non-financial support and other from Novartis, non-financial support and other from Bayer, personal fees, non-financial support and other from Ipsen, other from EUSA, non-financial support and other from Sanofi, grants and non-financial support from Pierre Fabre, personal fees and nonfinancial support from BMS, non-financial support from Roche, personal fees and non-financial support from Astellas, personal fees from Janssen.
Dr. Anido Herranz reports non-financial support and other from Pfizer, personal fees, non-financial support and other from Novartis, non-financial support and other from Bayer, personal fees, non-financial support and other from Ipsen, other from EUSA, non-financial support and other from Sanofi, non-financial support and other from Advanced Accelerator Applications, grants and non-financial support from Pierre Fabre, personal fees and non-financial support from BMS, non-financial support from Roche, personal fees and non-financial support from Astellas, personal fees from Janssen, personal fees from Kyowa Kirin, personal fees from Lilly, outside the submitted work.
Dr Fernández Calvo reports non-financial support and other from Astellas Pharma, Roche, Pfizer, Bristol-Myers-Squibb, Sanofi, EUSA Pharma, Sanofi, speaking honoraria from Pierre- Fabre, Novartis, Bristol-Myers-Squibb, Ipsen, Roche, Astellas Pharma, Bayer, Janssen; travel accommodations from Bristol- Myers-Squibb, Ipsen, Astellas. Rest of the authors have no relevant conflicts of interests.
References
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, et al. (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6): 394-424.
- Rawla P (2019) Epidemiology of Prostate Cancer. World J Oncol 10(2): 63–89.
- de Wit R, de Bono J, Sternberg CN, Fizazi K, Tombal B et al. (2019) for the CARD Investigators. Cabazitaxel versus Abiraterone or Enzalutamide in Metastatic Prostate Cancer. N Engl J Med 381(26): 2506-2518.
- Hussain M, Mateo J, Fizazi K (2019) PROfound: Phase 3 study of olaparib versus enzalutamide or abiraterone for metastatic castration-resistant prostate cancer with homologous recombination repair gene alterations. 2019 ESMO Congress 30: v851-v934.
- Zschäbitz S, Vallet S, Hadaschik B, Debatin D, Fuxius S, et al. (2017) Efficacy of Cabazitaxel Treatment in Metastatic Castration Resistant Prostate Cancer in Second and Later Lines. An Experience from Two German Centers. J Cancer 8(4): 507-512.
- de Bono JS, Oudard S, Ozguroglu M, Hansen S, Machiels JP, et al. (2010) TROPIC Investigators. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet 376(9747): 1147–1154.
- Heidenreich A, Scholz HJ, Rogenhofer S, Arsov C, Retz M, et al. (2013) Cabazitaxel plus prednisone for metastatic castration-resistant prostate cancer progressing after docetaxel: results from the German compassionate-use programme. Eur Urol 63(6): 977–982.
- Bahl A, Masson S, Malik Z, Birtle AJ, Sundar S, et al. (2015) Final quality of life and safety data for patients with metastatic castration-resistant prostate cancer treated with cabazitaxel in the UK Early Access Programme (EAP) (NCT01254279). BJU Int 116(6): 880-887.
- Eisenberger M, Hardy-Bessard AC, Kim CS, Géczi L, Ford D, Mourey L, et al. (2017) Phase III Study Comparing a Reduced Dose of Cabazitaxel (20 mg/m2) and the Currently Approved Dose (25 mg/m2) in Postdocetaxel Patients with Metastatic Castration-Resistant Prostate Cancer-PROSELICA. J Clin Oncol 35(28): 3198-3206.
- Sheel A Patel, Jean Hoffman-Censits (2017) Cabazitaxel in the treatment of metastatic castration-resistant prostate cancer: patient selection and special considerations. OncoTargets and Therapy 10: 4089-4098
- Shiota M, Nakamura M, Yokomizo A, Tomoda T, Sakamoto N, et al. (2019) Efficacy and safety of cabazitaxel for castration-resistant prostate cancer in patients with > 10 cycles of docetaxel chemotherapy: a multi-institutional study. Med Oncol 36(4): 32.
- Scher HI, Halabi S, Tannock I, Morris M, Sternberg CN, et al. (2008) Prostate Cancer Clinical Trials Working Group. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J Clin Oncol 26(7): 1148-1159.
- Halabi S, Lin CY, Small EJ, Armstrong AJ, Kaplan EB, et al. (2013) Prognostic model predicting metastatic castration-resistant prostate cancer survival in men treated with second-line chemotherapy. J Natl Cancer Inst 105(22): 1729-1737.
- Shiota M, Nakamura M, Yokomizo A, Tomoda T, Sakamoto N, et al. (2019) Therapeutic Outcome of >10 Cycles of Cabazitaxel for Castration-resistant Prostate Cancer: A Multi-institutional Study. Anticancer Res 39(8): 4411-4414.






























