The Suppression of DNA Repair Induced by PARP-1 Inhibitors Rucaparib and Olaparib in Combination with the Radiopharmaceutical 131I-MIBG in Noradrenaline Transporter-Expressing Xenograft Tumors
Donna L Nile1*, Colin Rae1, Colin Nixon2, Mark N Gaze3 and Robert J Mairs1
1Radiation Oncology, Institute of Cancer Sciences, University of Glasgow, United Kingdom
<2Beatson Institute for Cancer Research, University of Glasgow, United Kingdom
3University College London Hospitals, United Kingdom
Submission: April 26, 2018; Published: May 01, 2018
*Correspondence Address: Donna Nile, Radiation Oncology, Institute of Cancer Sciences, University of Glasgow, Glasgow, United Kingdom, Tel: 0141 330 4129; Email: Donna.Nile@glasgow.ac.uk
How to cite this article: Nile DN, Rae C, Nixon C, Gaze MN, Mairs RJ. PARP inhibitors enhance 131I-MIBG toxicity in vivo. Canc Therapy & Oncol Int J. 2018, 10(3): 555788. DOI: 005 : 10.19080/CTOIJ.2018.10.555788
Abstract
Radioiodinated meta-iodobenzylguanidine (131I-MIBG) is an effective treatment for tumors that express the noradrenaline transporter (NAT); including neuroblastoma, phaeochromocytoma, and gut neuroendocrine tumors. We have previously shown in vitro that the efficacy of 131I-MIBG was enhanced following its combination with PARP-1 inhibitors, rucaparib or olaparib. In order to assess in vivo the therapeutic benefit of combined radiotherapy and PARP-1 inhibition, we have first established a reliable protocol for the administration of olaparib and rucaparib, in NAT-expressing tumour xenografts. PARP-1 inhibition in combination with 131I-MIBG therapy was well tolerated with limited toxicity. Furthermore, administration of both PARP-1 inhibitors as single agents inhibited DNA repair in a time-dependent manner. These preliminary results highlight the therapeutic potential of PARP-1 inhibitors in vivo, and indicate the feasibility of combining rucaparib or olaparib with 131I-MIBG for the treatment of neuroendocrine tumors in preclinical models.
Keywords: Neuroblastoma; 131I-MIBG; PARP-1
Abbreviations: H&E: Haematoxylin and Eosin; HPBCD: 2-hydroxylpropyl-p-cyclodextrin; 131I-MIBG: Iodine-131 Metaiodobenzylguanidine; MBq: Megabecquerel; NAT: Noradrenaline Transporter; PARP-1: Poly(ADP-ribose) Polymerase-1; PBS: Phosphate Buffered Saline
Introduction
Radioiodinated meta-iodobenzylguanidine (131I-MIBG) is an effective treatment for the primordial neural crest-derived paediatric tumor, neuroblastoma [1-3]. A structural analogue of the catecholamine neurotransmitter, noradrenaline, 131I-MIBG is actively accumulated by tumors expressing the noradrenaline transporter (NAT) including neuroblastoma, the adrenal tumors phaeochromocytoma and paraganglioma [4], neuroendocrine tumors of the gut [5] and medullary thyroid carcinoma [6]. Whilst effective when administered as single agent therapy [2-4], maximal therapeutic benefit from 131I-MIBG will be obtained by its combination with chemotherapy [7]. Poly (ADP- ribose) polymerases (PARPs) mediate the post-translational modification of target proteins, thus signalling the site of DNA damage and initiating DNA repair. Indeed, PARP-1 inhibition was shown to exhibit synthetic lethality in BRCA-deficient cells [8,9], and has since proven an effective treatment in cancers that are deficient in BRCA-mediated DNA repair [10,11]. olaparib and rucaparib are FDA-approved PARP-1 inhibitors undergoing Phase II/III clinical evaluation [10-12].
We recently demonstrated that treatment with rucaparib or olaparib significantly reduced the IC50 dose of X-radiation or 131I-MIBG therapy required to kill neuroblastoma or NAT- expressing glioma cells [13]. We hypothesized that the PARP-1 inhibitors effectively lowered the threshold for radiation-induced cell death, by significantly delaying repair of radiation-induced DNA damage, and promoting cell cycle arrest. Our purpose is now to assess [1] whether PARP-1 inhibition aggravates 131I-MIBG toxicity [2] whether enhancement of the prolongation of DNA damage by PARP-1 inhibition also obtained in vivo.
Methods
Establishment and treatment of xenografts
In vivo experiments were performed in accordance with the Animals (Scientific Procedures) Act 1986. This research was reviewed and approved by the University of Glasgow's Ethical Review Board. Tumors derived from UVW glioma cells expressing NAT [14] were produced by subcutaneous injection of 3x106 cells into 6 week old female, athymic CD1 nude (nu/nu) mice (Charles Rivers plc, Kent, UK) as described previously [15]. Experimental therapy was initiated 3-5 weeks after injection, once the tumor volume had reached 60mm3. rucaparib (1 or 10mg/kg), olaparib (30mg/kg), or vehicle (10% v/v DMSO, 10% v/v HPBCD, 2-hydroxylpropyl-p-cyclodextrin, in PBS) were intraperitoneally injected. Tumors were excised 2, 6, or 24h after injection.
Assessment of Treatment Toxicity
131I-MIBG (10 MBq) was administered by intraperitoneal injection to 9 week old female, athymic CD1 nude [nu/nu] mice.
For combination therapy, mice were also intraperitoneally injected with rucaparib (10mg/kg), olaparib (30mg/kg), or vehicle (10% v/v DMSO, 10% v/v HPBCD in PBS) at the same time as 131I-MIBG administration, and for the next two consecutive days. Mice were weighed 3 times weekly and monitored daily to assess treatment toxicity.
Immunohistochemistry
Immunohistochemical analysis was performed on formalin- fixed paraffin-embedded 4|im sections of tumors according to standard protocols. Primary antibodies used were anti-yH2AX (1:50; Cell Signalling Technology, The Netherlands; 9718) and anti-Ki67 (1:100; Thermo Scientific, UK; RM-9106) after citrate buffer and antigen retrieval (25min, 98 °C) on a Pre-Treatment (PT) Module (Thermo Scientific, UK). All staining was performed on a Dako Autostainer Link 48 platform (Dako, UK). Slides were digitized using the SCN400F scanner (Leica Biosystems, UK) at x20 magnification and visualized using the Slide Path Digital Image Hub, version 4.0.1 (Leica Biosystems, UK).
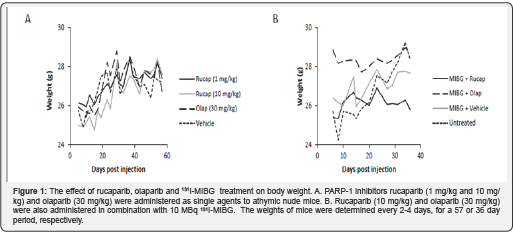
Results
No adverse toxic effect was observed following the administration of 1mg/kg or 10mg/kg rucaparib. Mice continued to gain weight for up to 57 days post injection, at a rate comparable to the tumors in the vehicle control group of mice. Negligible toxicity was also observed after treatment with 30mg/ kg olaparib (Figure 1A). Similarly, mice maintained weight for up to 36 days after treatment with 10mg/kg rucaparib or 30mg/ kg olaparib in combination with 10 MBq 131I-MIBG (Figure 1B). Therefore it was concluded that the administration of PARP-1 inhibitors did not aggravate 131I-MIBG toxicity.
We previously demonstrated that PARP-1 inhibitors, administered in combination with external beam radiation, promoted substantial DNA damage 2h after administration in vitro [13]. Moreover, this radiation-induced DNA damage persisted for 24h following treatment with PARP-1 inhibitors. We therefore resolved to determine the significance of delayed restitution of DNA in vivo. Tumor xenografts were excised 2, 6 and 24h after PARP-I inhibitor injection. Fixed, paraffin-embedded tumor sections were then stained with haematoxylin and eosin (H&E), anti-Ki67 or anti-yH2AX antibodies to observe gross tumor histology, cell proliferation and DNA damage, respectively. Exposure to rucaparib failed to affect the gross xenograft tumor histology, however, decreased Ki67-positive staining observed at 2h and 6h after treatment indicates decreased cell proliferation (Figure 2). Negligible DNA damage was apparent 2h and 6h after treatment, manifest by the predominance of counter staining. However, substantial DNA damage was observed 24h after treatment, according to yH2AX-positivity. Similarly, following exposure to olaparib, there was no change in tumor histology, whilst decreased cellular proliferation was observed 6h after treatment (Figure 3). Substantial DNA damage was also observed 24h following exposure to olaparib.
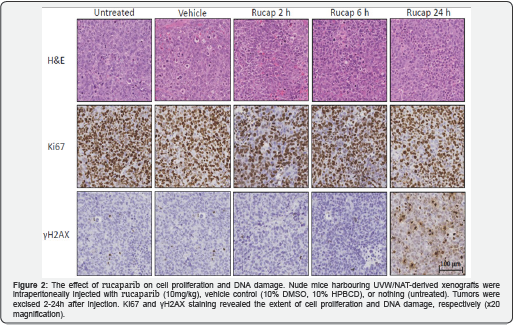
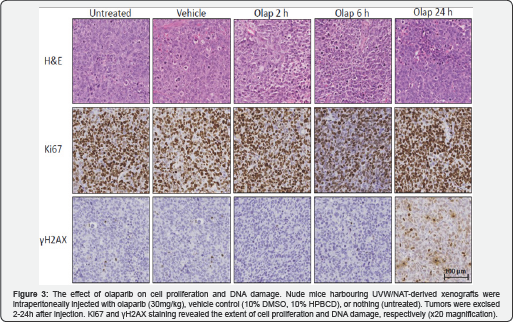
Conclusion
The noradrenaline analogue 131I-MIBG selectively accumulates within cells that express the noradrenaline transporter, NAT. This can be exploited therapeutically for the treatment of NAT-expressing tumors such as neuroblastoma, phaeochromocytoma, and gut neuroendocrine tumors. Whilst effective when administered as single agent therapy, maximal therapeutic benefit from 131I-MIBG is expected to be obtained by its combination with chemotherapy.
Activation of the PARP-1 DNA repair pathway follows genotoxic insult. We have previously shown that PARP-1 inhibition, following treatment with PJ34 [16] or rucaparib or olaparib [13], enhanced the antitumor efficacy of 131I-MIBG in NAT-expressing neuroblastoma and glioma cells. We hypothesized that PARP-1 inhibition effectively lowered the threshold for radiation-induced cell death, following the delayed repair of radiation-induced DNA damage, which prompted G2/M cell cycle arrest. Our current aim was to determine whether prolonged DNA damage by inhibition of PARP-1 also obtained in vivo. We observed that PARP-1 inhibitor single agent treatment prevented DNA repair in tumor xenografts in a time-dependent manner. yH2AX staining, which is indicative of DNA double strand breaks, was observed at 24h after administration of rucaparib or olaparib, but not at earlier time points. Jiang et al. [17] also demonstrated that olaparib single agent treatment significantly increased the number of yH2AX foci 24h after treatment in non¬small cell lung cancer xenografts. Interestingly, the DNA damage sustained was significantly enhanced following combination of 50mg/kg olaparib with 10Gy irradiation, particularly under hypoxic conditions [17].
The measurement of PARP-1 enzymatic activity in peripheral blood leukocytes has been used in clinical trials of rucaparib [12,18] and olaparib [19,20] as a pharmacodynamic biomarker. However, recent demonstration of poor correlation between leukocyte PARP-1 activity and the anti-tumor efficacy [18,19] suggest that this assay may be suboptimal. Therefore, alternative biomarkers, such as the yH2AX or RAD51 foci, may be more reliable indices of effectiveness [21].
Using a murine preclinical model [15,16,22], we have established an effective treatment regimen for the administration of olaparib or rucaparib in combination with 131I-MIBG. The treatment procedure, which mimics therapy schedules used clinically, did not aggravate the toxicity of either agent administered alone. This was manifest by the maintenance of murine body weight up to 57 or 36 days following administration of PARP-1 inhibitors as single agents or in combination with 131I-MIBG, respectively. Similarly, Min et al. [23] demonstrated no significant reduction in mouse weight compared to vehicle control mice, following administration of 50mg/kg olaparib. The drug concentrations used in this study are comparable to those used previously in experimental therapy of mice bearing xenografts derived from neuroblastoma, medulloblastoma and gastric cancer [23-25]. Furthermore, the administered doses are below the 400mg twice daily maximum tolerated dose of olaparib [10] and the 600mg twice daily recommended dose of rucaparib [18].
The results of this in vivo study have demonstrated that administration of PARP-1 inhibitors, rucaparib and olaparib, as single agents, inhibited DNA repair in mouse tumor xenografts in a time-dependent manner. This finding is in support of our published in vitro data [13]. We also established a reliable protocol for the administration of olaparib and rucaparib in NAT-expressing tumor xenografts established in athymic mice. The doses administered were similar to those used clinically, and were well tolerated. These preliminary results indicate the feasibility of combining rucaparib or olaparib with 131I-MIBG for the treatment of neuroendocrine tumors. It is expected that the combination of PARP-1 inhibition with 131I-MIBG will engender more effective therapy of NAT-expressing tumors.
Acknowledgements
The authors wish to thank Dr. Sally Pimlott and Dr. Sue Champion for radiopharmaceutical synthesis; Ms Nicola Munroe and Mr David McLaughlin for assistance with in vivo experimentation; Ms Gemma Thomson for assistance with image analysis. This work was supported by grant funding from Children with Cancer UK (W1057 and 15-191), Glasgow Children's Hospital Charity (YRSS/SPG/2015/01) and the Chief Scientist Office Scotland (TCS/16/38). Dr M N Gaze is supported by the National Institute for Health Research University College London Hospitals Biomedical Research Centre. Funding bodies played no role in the design of the study, collection, analysis, and interpretation of data, or in writing the manuscript.
References
- DuBois SG, Allen S, Bent M, Hilton JF, Hollinger F, et al. (2015) Phase I/II study of 131I-MIBG with vincristine and 5 days of irinotecan for advanced neuroblastoma. Br J Cancer 112(4): 644-649.
- Kang TI, Brophy P, Hickeson M, Heyman S, Evans AE, et al. (2003) Targeted radiotherapy with submyeloablative doses of 131I-MIBG is effective for disease palliation in highly refractory neuroblastoma. J Pediatr Hematol Oncol 25(10): 769-773.
- Matthay KK, Weiss B, Villablanca JG, Maris JM, Yanik GA, et al. (2012) Dose escalation study of no-carrier-added 131I-metaiodobenzylguanidine for relapsed or refractory neuroblastoma: new approaches to neuroblastoma therapy consortium trial. J Nucl Med 53(7): 1155-1163.
- Gedik GK, Hoefnagel CA, Bais E, Olmos RA (2008) 131I-MIBG therapy in metastatic phaeochromocytoma and paraganglioma. Eur J Nucl Med Mol Imaging 35(4): 725-733.
- Kölby L, Bernhardt P, Levin-Jakobsen AM, Johanson V, Wangberg B, et al. (2003) Uptake of meta-iodobenzylguanidine in neuroendocrine tumours is mediated by vesicular monoamine transporters. Br J Cancer 89(7): 1383-1388.
- Newton TD, Augustine T, Arumugam P, Malik RA (2008) Detection of medullary thyroid cancer with MIBG imaging for pheochromocytoma. Clin Nucl Med 33(5): 328-329.
- Mairs RJ, Boyd M (2011) Preclinical assessment of strategies for enhancement of metaiodobenzylguanidine therapy of neuroendocrine tumors. Semin Nucl Med 41(5): 334-344.
- Bryant HE, Schultz N, Thomas HD, Parker KM, Flower D, et al. (2005) Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 434(7035): 913-917.
- Farmer H, McCabe N, Lord CJ, Tutt AN, Johnson DA, et al. (2005) Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434(7035): 917-921.
- Audeh MW, Carmichael J, Penson RT, Friedlander M, Powell B, et al. (2010) Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer: a proof-of-concept trial. Lancet 376(9737): 245-251.
- Tutt A, Robson M, Garber JE, Domchek SM, Audeh MW, et al. (2010) Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and advanced breast cancer: a proof-of- concept trial. Lancet 376(9737): 235-244.
- Plummer R, Lorigan P, Steven N, Scott L, Middleton MR, et al. (2013) A phase II study of the potent PARP inhibitor, rucaparib (PF-01367338, AG014699), with temozolomide in patients with metastatic melanoma demonstrating evidence of chemopotentiation. Cancer Chemother Pharmacol 71(5): 1191-1199.
- Nile DL, Rae C, Hyndman IJ, Gaze MN, Mairs RJ (2016) An evaluation in vitro of PARP-1 inhibitors, Rucaparib and olaparib, as radiosensitisers for the treatment of neuroblastoma. BMC Cancer 16: 621.
- Boyd M, Cunningham SH, Brown MM, Mairs RJ, Wheldon TE (1999) Noradrenaline transporter gene transfer for radiation cell kill by 131I meta-iodobenzylguanidine. Gene Ther 6(6): 1147-1152.
- McCluskey AG, Boyd M, Ross SC, Cosimo E, Clark AM, et al. (2005) [131I] meta-iodobenzylguanidine and topotecan combination treatment of tumors expressing the noradrenaline transporter. Clin Cancer Res 11(21): 7929-7937.
- McCluskey AG, Mairs RJ, Tesson M, Pimlott SL, Babich JW, et al. (2012) Inhibition of poly (ADP-Ribose) polymerase enhances the toxicity of 131I-metaiodobenzylguanidine/topotecan combination therapy to cells and xenografts that express the noradrenaline transporter. J Nucl Med 53(7): 1146-1154.
- Jiang Y, Verbiest T, Devery A, Bokobza S, Weber A, et al. (2016) Hypoxia Potentiates the Radiation-Sensitizing Effect of olaparib in Human Non¬Small Cell Lung Cancer Xenografts by Contextual Synthetic Lethality. Int J Radiat Oncol Biol Phys 95(2): 772-781.
- Drew Y, Ledermann J, Hall G, Rea D, Glasspool R, et al. (2016) Phase 2 multicentre trial investigating intermittent and continuous dosing schedules of the poly(ADP-ribose) polymerase inhibitor rucaparib in germline BRCA mutation carriers with advanced ovarian and breast cancer. Br J Cancer 114(7): 723-730.
- Bundred N, Gardovskis J, Jaskiewicz J, Eglitis J, Paramonov V, et al. (2013) Evaluation of the pharmacodynamics and pharmacokinetics of the PARP inhibitor olaparib: a phase I multicentre trial in patients scheduled for elective breast cancer surgery. Invest New Drugs 31(4): 949-958.
- Fong PC, Boss DS, Yap TA, Tutt A, Wu P, et al. (2009) Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med 361(2): 123-134.
- Brown JS, Kaye SB, Yap TA (2016) PARP inhibitors: the race is on. Br J Cancer 114(7): 713-715.
- Rae C, Tesson M, Babich JW, Boyd M, Sorensen A, et al. (2013) The role of copper in disulfiram-induced toxicity and radiosensitization of cancer cells. J Nucl Med 54(6): 953-960.
- Min A, Im SA, Yoon YK, Song SH, Nam HJ, et al. (2013) RAD51C-deficient cancer cells are highly sensitive to the PARP inhibitor olaparib. Mol Cancer Ther 12(6): 865-877.
- Daniel RA, Rozanska AL, Mulligan EA, Drew Y, Thomas HD, et al. (2010) Central nervous system penetration and enhancement of temozolomide activity in childhood medulloblastoma models by poly (ADP-ribose) polymerase inhibitor AG-014699. Br J Cancer 103(10): 1588-1596.
- Daniel RA, Rozanska AL, Thomas HD, Mulligan EA, Drew Y, et al. (2009) Inhibition of poly (ADP-ribose) polymerase-1 enhances temozolomide and topotecan activity against childhood neuroblastoma. Clin Cancer Res 15(4): 1241-1249.






























