Olaparib: A Tale of Two Dosage Forms
Trevor N Christ*
Department of Pharmacy, University of Chicago Medical Center, USA
Submission: February 20, 2018; Published: February 23, 2018
*Correspondence Address: Trevor N Christ, Department of Pharmacy, University of Chicago Medical Center, UChicago Medicine, 5841 S. Maryland Ave, Room TE026 / MC0010, Chicago, IL 60637, Tel: [773) 834-7870; Email: trevor.christ@uchospitals.edu
How to cite this article: Trevor N Christ. Olaparib: A Tale of Two Dosage Forms. Canc Therapy & Oncol Int J. 2018; 9(4): 555766. DOI: 10.19080/CTOIJ.2018.09.555766
Abstract
Olaparib is the first poly (ADP-ribose) polymerase (PARP) inhibitor approved by the FDA, with three approvals in two different cancer types. The original dosage form of olaparib was a 50 mg capsule, requiring 16 capsules per day for patients at full dose therapy which prompted development of a tablet dosage form with improved bioavailability. Herein, major trials for olaparib are reviewed and the two dosage forms are compared from a pharmacokinetic and clinical perspective.
Keywords: PARP inhibitor; BRCA; Ovarian cancer; Breast cancer; Pharmacokinetics
Abbreviations: PARP: Poly(ADP-ribose) Polymerase; BRCA: Breast Cancer Susceptibility Gene
Introduction
Olaparib is a poly (ADP-ribose) polymerase (PARP) inhibitor and the first in its class to receive FDA approval. The PARP enzyme repairs single stranded DNA mismatches, which may promote incorporations of mutations in patients with deleterious BRCA mutations (and other DNA repair deficiencies). Inhibition of PARP results in apoptosis of the cancer cell. Thus, there is much clinical research being done to combine PARP inhibitors with DNA-strand breaking therapies (platinum chemotherapy, radiation) as well as use in cancer populations harboring gained or germ line mutations associated with impaired DNA repair mechanisms [1].
Olaparib received FDA approval in 2014 based on the phase 2 trial performed by Domchek and colleagues [2] in which 137 ovarian cancer patients harboring a germ line BRCA mutation following 3 or more lines of chemotherapy were administered olaparib capsules at 400 mg twice daily (BID) as a single agent [2]. Olaparib was associated with an ORR of 34% and duration of response of ~8 months. Major adverse drug events included nausea, fatigue, vomiting, and anemia.
The original approval of olaprarib was the capsule form which is commercially available in 50 mg capsules requiring a patient on full dose therapy to take eight capsules by mouth BID (16 capsules/day) due poor solubility of the active agent. While it is a common strategy of manufacturers of oral oncology drugs to provide daily doses in multiple tablets or capsules for convenient dose reduction, the capsule size of olaparib were not consistent with this paradigm given the recommended dose reductions at 200 mg (four capsules) and 100 mg (two capsules) BID dose levels [3].
In 2017, the tablet form of olaparib was approved with a new indication as the result of the SOLO2 and Study 19 trials [4,5]. The SOLO2 trial was a phase 3, randomized, double-blind trial comparing olaparib tablets at 300 mg BID to placebo as maintenance therapy in patients with ovarian cancer and BRCA1/2 mutation following treatment with 1-2 lines a of platinum-based chemotherapy regimen. Olaparib maintenance was associated with an improvement in PFS of 19.1 versus 5.5 months in the placebo arm.
Dosage Form Switch
Mateo and colleagues [6] performed a dose escalation and dose expansion cohort study comparing the pharmacokinetic, adverse event profiles, and disease response between the approved capsule dosage form and the more recently developed meltextrusion tablets (designed for improved bioavailability) [6]. Thirty-one patients with advanced solid tumors (majority ovarian, breast, and colorectal cancers) were included in the dose escalation phase, which compared pharmacokinetic profiles of single administration and continuous dosing of the capsule (dose range 50-400 mg) and tablet dosage forms (dose range 25250 mg). The study authors found approximately linear dose to Cmax and AUC values with dose increases of both dosage forms and slightly faster Tmax associated with the tablet (0.5-2 hours) compared to the capsule (1-3 hours). Overall, the capsule and tablet dosage forms were not found to be bioequivalent.?
In the dose expansion phase, eight different dosing groups were compared which included cohorts with tablet and capsule formulations, cross-over study designs, and intermittent dosing regimens. The authors discovered that the 450 mg tablet BID was intolerable due to a high incidence of grade > 3 cytopenias. Also, the 400 mg BID tablet cohort experienced more anemia and thrombocytopenia than the 300 mg BID tablet cohort. The authors concluded that the 300 mg BID (as two 150 mg tablets) tablet dosing was most equivalent to the originally approved capsule dosage form at 400 mg BID and would be the recommended dose for future studies (Table 1).
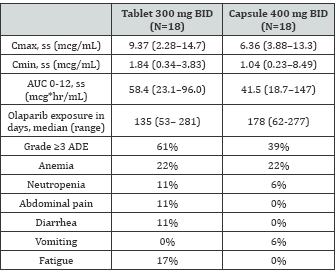
ADE: Adverse Drug Event
Discussion
When comparing the tablet and capsule groups in the dose expansion phase of the study, a few differences should be noted. First, the pharmacokinetic parameters of the capsule form were consistently higher than the tablet. Second, a corresponding trend suggests decreased tolerability of the tablet form when reviewing overall exposure and relevant adverse events. Importantly, there was no difference in decrease in tumor volume in ovarian cancer patients with known BRCA mutation comparing these two formulations and dosing regimens.
The impetus for the development of the SOLO2 trial was based on the results of Study 19, which included patients in a similar clinical setting using the capsule dosage form at 400 mg BID [5]. However, Study 19 included patients regardless of BRCA mutation status and PFS for the general population, while improved over placebo, was not as impressive as the BRCA mutated subgroup. While it is difficult to make general conclusions across these two studies given the different populations and response rates, it is interesting to note more grade 3/4 anemia (19% vs. 5.1%) and neutropenia (5% vs. NR) in the study using tablets than capsules which the SOLO2 authors conclude was due to longer olaparib exposure and not due to the change in dosage form.
Conclusion
Regulatory approval of the olaparib tablet dosage form was based on the SOLO2 trial, which stemmed from prior pharmacokinetic and dose finding studies. With this approval was the expansion of the tablet dosage form for use in BRCA mutated ovarian cancer patients with 3-4 prior lines of chemotherapy, despite adequate clinical data supporting the use in this setting. Given both the pharmacokinetic and clinical differences highlighted above, the expansion of this approval is premature and may put more heavily treated metastatic cancer patients at risk of adverse events in an unproven setting.
Subsequent to the tablet approval, the capsule form of olaparib is being phased out and is currently only available through a single specialty pharmacy associated with the manufacturer. However, from a safety standpoint this approach may be beneficial as medication errors would surely increase with both formulations being available through similar distribution routes. It is important for physicians, nurses, and pharmacists to be aware of the changes in dosage form and to counsel patients and caregivers effectively and thoroughly. Hopefully, the tablet dosage form, at 4 tablets per day, will improve treatment adherence and pill burden.
Given the differences discussed between the two dosage forms, clinicians should be cautious of the change in dosage form, especially in patients already tolerating the capsule formulation. It is possible to obtain the capsules through the manufacturer program, though potentially less convenient. Furthermore, caution for those designing clinical trials or applying early phase trials to direct patient care on an off-protocol basis should be taken when assigning treatment dose as the dosage form of the agents is not bioequivalent.
Conflict of Interest
None.






























