How Efficacious is Ziverel® for Symptomatic Relief of Acute Radiation-Induced Esophagitis? Retrospective Study of Patients Receiving Oncologic Treatment
*Carrasco E, López-Campos F, Sastre-Gallego S, Barrionuevo P, Martín M, Vallejo C, Hernanz R and Sancho S
Radiation Therapy Oncology Service, Ramón y Cajal University Hospital, Spain
Submission: October 23, 2017; Published: November 10, 2017
*Corresponding author: Eliseo Carrasco Esteban, Radiation Oncology Service. University Hospital Ramón y Cajal. Ctra. Colmenar Viejo, Km. 9,100. 28034 Madrid, Spain, Tel: +34 913 368 222; Fax: +34 913 368 202; Email: eliseo.carrasco.esteban@gmail.com
How to cite this article: Carrasco E, López-Campos F, Sastre-Gallego S, Barrionuevo P, Martín M, Vallejo C, Hernanz R, Sancho S. How Efficacious is Ziverel® for Symptomatic Relief of Acute Radiation-Induced Esophagitis? Retrospective Study of Patients Receiving Oncologic Treatment. Canc Therapy & Oncol Int J. 2017; 7(5): 555724. DOI: 10.19080/CTOIJ.2017.07.555724
Abstract
Objective: To evaluate the efficacy of ZIVEREL® for symptomatic relief in a retrospective cohort of patients with acute radiation-induced esophagitis receiving oncologic treatment with radiotherapy alone or radiochemotherapy.
Introduction: ZIVEREL® is a new oral medical device composed of hyaluronic acid, chondroitin sulfate, and poloxamer 407. Radiation- induced esophagitis is a dose-limiting toxicity in oncologic treatment with radiotherapy or radiochemotherapy, and sometimes a limiting factor for treatment.
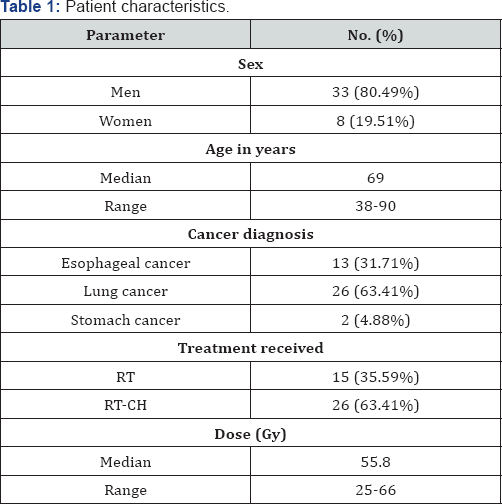
Material and Methods: Between February 2016-July 2017, we evaluated 41 patients (33 men and 8 women) treated with ZIVEREL®, with a diagnosis of lung cancer (63.41%), gastric cancer (31.71%), and esophageal cancer (4.88%) who developed acute radiation-induced esophagitis (CTCAE grade 1 [60.98%] and grade 2 [39.02%]) during treatment with radiotherapy alone (36.59%) or radiochemotherapy (63.41%). The median age was 69 years (range, 38 to 90 years).
Results: Of the total number of patients, 38 (92.68%) experienced an improvement of their symptoms; 13 of these patients (34.21%) had previously received support treatment, according to usual clinical practice, compared with 3 patients (7.32%) in whom ZIVEREL® did not lead to an improvement in symptoms. ZIVEREL® was prescribed as initial treatment (41.46%), after initiation of support treatment (34.15%), or together with support treatment (24.39%). Of the 41 patients treated, 39 patients (95.12%) completed the oncologic treatment satisfactorily, and it was necessary to interrupt oncologic treatment in only 2 cases (4.88%) of total number of patients.
Conclusion: ZIVEREL® is well tolerated and plays a key role in the symptomatic relief of radiation-induced esophagitis resulting from oncologic treatment.
Keywords: Acute esophagitis; Chemotherapy; Chemoradiotherapy; Chondroitin sulfate; Hyaluronic acid; Poloxamer 407; Radiotherapy; Radiochemotherapy; ZIVEREL®
Abbreviations: COX-2: Cyclooxygenase-2; CTCAE: Common Terminology Criteria for Adverse Events; DNA: Deoxyribonucleic Acid; ECM: Extracellular Matrix, Gy: Gray; IL-1β: Interleukin 1 Beta; NFκB: Nuclear Factor Kappa Beta; PGE2: Prostaglandin E2; RT: Radiotherapy; RT-CH: Chemoradiotherapy; TNF-α: Tumor Necrosis Factor Alpha
Introduction
The esophagus is a tube composed of muscle and membranous tissue. It extends from the pharynx (upper border of the cricoid cartilage, 15 cm from the incisors) to the stomach and is closed at the ends by the contraction of 2 sphincter muscles. It serves to transport food from the mouth to the stomach and has no major secretary function. In terms of anatomy, the esophagus comprises 3 sections: the cervical section (5-6 cm), the thoracic section (20 cm), and the abdominal section (2-3 cm). In terms of histology, it is composed of 4 functional layers:
i. The mucosa, which is composed of stratified squamous epithelium;
ii. The submucosa, which is formed by loose connective tissue that supports the mucosal layer;
iii. The muscularis propria, which is in turn formed by a circular layer and a longitudinal layer and is responsible for voluntary and involuntary movements of the esophagus;
iv. The adventitia, which is composed of highly vascularized supportive connective tissue and is surrounded by mesothelium in the abdominal portion [1].
In the context of oncologic treatments, the fact that the esophagus covers 3 anatomical regions means that it can be irradiated in various clinical situations and is sometimes difficult to protect. Both palliative treatment and curative treatment with ionizing radiation between C6 and T12 are associated with partial irradiation of the esophagus.
Radiation-induced esophagitis is an acute, dose-limiting toxicity in oncologic treatment. Since it can lead to interruption of treatment, hospitalization, and even death, clinicians should be aware of this condition as an adverse effect of oncologic therapies, in order to prevent interruption of treatment, as it has been associated with reduced survival [2,3]. The pathogenesis of radiation- induced esophagitis is complex and not well defined. However, there is sufficient evidence that the symptoms are caused by lesions affecting the structure of the epithelium and that they are induced by the action of ionizing radiation on the esophageal mucosa, with rupture of double-stranded DNA [4], formation of reactive oxygen species [5], over expression of proinflammatory cytokines [6,7] and bacterial overgrowth on ulcerated lesions [8]. Clinically, acute esophagitis includes dysphagia, odynophagia, nausea, anorexia, and retrosternal pain. If these symptoms are severe, they can lead to dehydration, malnutrition, and weight loss [9,10].
ZIVEREL® is a new, over-the-counter medical device that comes as an oral solution in a 10-ml stick pack. It is composed of hyaluronic acid, chondroitin sulfate, and poloxamer 407 and plays a role in the symptomatic relief of esophagitis induced by radiotherapy alone or radiochemotherapy in oncologic patients. ZIVEREL® acts naturally and exerts a protective action by reinforcing the effect of the elements that make up the extracellular matrix, where the connective tissue provides the architecture, proteoglycans maintain the fluid-electrolyte balance, and glycoproteins maintain the intracellular substrate responsible for cell-cell reactions and cell-matrix reactions.
Hyaluronic acid is a glycosaminoglycan formed by glucuronic acid and N-acetylglucosamine disaccharide units. It is found mainly in the extracellular matrix of the loose connective tissue and is associated with several key processes, including cell signaling and repair, as well as with tissue generation, morphogenesis, and structural organization of the extracellular matrix itself [11]. In clinical terms, its role is well known in conditions such as mouth ulcers, where its barrier effect relieves symptoms [12,13]. Chondroitin sulfate forms part of the glycosaminoglycan group in the extracellular matrix, which is in turn formed by D-glucuronic acid and N-acetylgalactosamine. It has been shown to protect the epithelium of the esophageal mucosa by shielding the epithelial areas damaged by acid, thus diminishing catabolic activity and inhibiting proteolytic enzymes (e.g., metalloproteases, collagenase, or elastase). Chondroitin sulfate also regulates several inflammatory mediators (TNF-α, IL-1β, COX-2, PGE2, and NFĸB) and reduces the synthesis of nitric oxide, which is involved in the inflammatory cascade [14,15]. Polaxamer 407 is a bioadhesive component that acquires the consistency of gel at body temperature, thus ensuring that the active ingredients of ZIVEREL® adhere to the damaged mucosa and are not dragged away by ingestion of food and liquids [16].
The objective of this study was to evaluate whether administration of ZIVEREL® diminishes the grade of acute radiation-induced esophagitis and the incidence of severe esophagitis in oncologic patients treated with radiotherapy or radiochemotherapy.
Material and Methods
Between February 2016 and July 2017, we retrospectively evaluated 41 patients diagnosed with cancer (lung, gastric and esophageal cancer). The patients had received radiotherapy alone (RT) or radiochemotherapy (RT-CH) and developed acute radiation-induced esophagitis grade 1 or 2 according to the most recent CTCAE criteria [10]. Their support treatment included ZIVEREL®. The patients were evaluated weekly during treatment with RT or RT-CH and until 2 weeks after the end of treatment.
Results
RT: radiotherapy; RT-CH: radiochemotherapy; Gy: Gray
We analyzed data from 41 patients (33 men [80.49%] and 8 women [19.51%]), of whom 26 (63.41%) were diagnosed with lung cancer, 13 (31.71%) were diagnosed with esophageal cancer, and 2 (4.88%) were diagnosed with gastric cancer. The median age was 69 (range, 38-90) years. A total of 15 patients received radiotherapy alone (36.59%) and 26 received radiochemotherapy (63.41%). The median dose was 55.8 Gy (range, 25-66 Gy) (Table 1). Of the 41 patients who received treatment with ZIVEREL®, 16 (39.02%) started treatment with ZIVEREL® when they developed grade 2 acute radiation-induced esophagitis and 25 (60.98%) when they developed grade 1 esophagitis. ZIVEREL® was administered daily during treatment once a day (10 mL [4.88%]), twice a day (20 mL [58.54%]), or 3 times a day (30 mL [36.58%]), depending on the usual clinical practice of the prescribing physician, for a minimum of 4 weeks (Table 2).
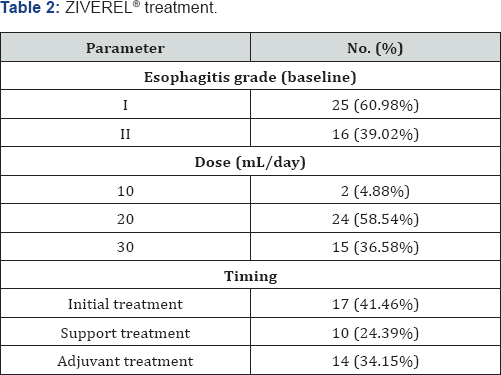
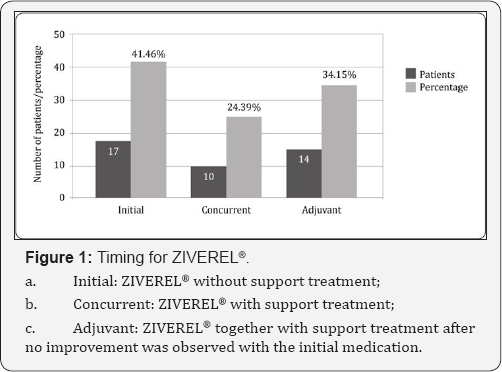
Support treatment was indicated according to usual clinical practice, and, depending on the intensity of the symptoms, the patients received proton pump inhibitors, anti-inflammatory drugs, analgesics, anti-H2 antagonists, antacids, and/or opioids. Of the 41 patients studied, symptoms improved in 38 patients (92.68%) and did not improve in 3 (7.32%) after treatment with ZIVEREL®. Of the patients whose symptoms improved, 13 (34.21%) had started support treatment before administration of ZIVEREL®. ZIVEREL® was the first product used for symptomatic relief of grade 1 and 2 esophagitis in 17 patients (41.46%) and was administered without further support treatment. It was prescribed with support treatment in 10 patients (24.39%) according to the usual clinical practice of the prescribing physician. In 14 patients (34.15%), ZIVEREL® was used as an adjuvant to the initial support treatment after no improvement was observed with the initial medication (Figure 1).
Of the 17 patients (41.46%) who received ZIVEREL® as their initial treatment, 7 did not require subsequent support treatment. Five of the 7 patients had a diagnosis of acute grade 1 esophagitis when the drug was indicated, and ZIVEREL® was prescribed in the remaining 2, who had a diagnosis of grade 2 esophagitis. Of the total number of patients, 39 (95.12%) completed radiotherapy and radiochemotherapy satisfactorily. Oncologic treatment had to be interrupted in only 2 cases (4.88%) owing to acute toxicity, which took the form of grade 3 esophagitis, although in both cases, a curative dose for the type of tumor treated was reached. Three of the 41 patients (7.32%) studied had to be admitted to hospital with acute grade 3 radiation-induced esophagitis. No adverse reactions to ZIVEREL® were reported. The drug was well tolerated by all those patients it was prescribed to.
Discussion
As stated above, acute esophagitis caused by oncologic treatment is a common finding and a dose-limiting factor for oncologic treatment. Therefore, clinicians should be aware of this adverse effect in order to prevent interruption of radiotherapy or radiochemotherapy, as this has been associated with reduced overall survival, because the patient cannot receive the intended treatment optimally [2,3].
Appropriate diagnosis of the symptoms of acute radiation- induced esophagitis is hampered by several factors, including infection of the esophagus, whose symptoms mimic those of esophagitis and which can be affected by oncologic treatment. Infection can lead the esophagus to lose its barrier function, thus facilitating local invasion by commensal microorganisms and pathogens and gastro esophageal reflux before initiation of treatment [8]. Therefore, correct evaluation of the patient before support treatment is prescribed is essential in order to rule out esophageal candidiasis and oropharyngeal mucositis, which can lead to an erroneous initial diagnosis. In our study, all patients were exhaustively assessed in order to identify those who had concurrent infection, oropharyngeal mucositis, gastroesophageal reflux, or any other condition that could mask or mimic the symptoms of acute radiation-induced esophagitis.
In our study, most of the patients analyzed (95.12%) completed their oncologic treatment satisfactorily. As explained above, the timing for ZIVEREL® varied, and the product was indicated as initial treatment in 41.46% of patients, together with support treatment in 24.39%, and as an adjuvant to initial treatment in 34.15% (Figure 1). Similarly, dosing of ZIVEREL® differed depending on the usual practice of the prescribing physician (once a day [10 mL] in 4.88%, twice a day [20 mL] in 58.54%, and 3 times a day [30 mL] in 36.58%). Limited experience with this product in our setting was one of the reasons why dosing of ZIVEREL® varied from patient to patient. According to results from patients with gastroesophageal reflux the optimal dose is 4 sachets per day (40 mL/day) [12], although we found that symptoms improved with a lower dose in 63.42% of patients. It remains unclear if ZIVEREL®, whether taken alone or in combination with support treatment and administered according to usual clinical practice, helps to ensure completion of the oncologic treatment prescribed, which is clearly associated with better outcomes and increased survival [17,18]. Therefore, determination of the optimal dose for symptomatic relief in these patients would provide relevant data and thus make it possible to establish the best dosing regimen for the control of the symptoms of acute esophagitis induced by oncologic treatment, regardless of the support treatment prescribed. Similarly, nonsteroidal anti-inflammatory drugs are a cornerstone of symptomatic treatment, although these are contraindicated in many patients owing to comorbid conditions or drug interactions; therefore, they should be avoided where possible [19]. Thus, it would be interesting to determine the role of ZIVEREL® as a support treatment for these groups of patients with acute esophagitis after radiotherapy or radiochemotherapy.
As mentioned above, the esophagus covers 3 anatomical regions. Therefore, ionizing radiation, whether palliative or curative, administered between C6 and T12 could partially affect the esophageal mucosa. The patients we studied were diagnosed with lung cancer (63.41%), esophageal cancer (31.71%), and gastric cancer (4.88%). Radiotherapy-related factors that play a role in acute toxicity include volume of irradiated tissue, total dose received, daily dose, total treatment time, irradiation technique, and concurrent systemic therapy [20,21]. When systemic therapy is administered concomitantly with radiotherapy, the symptoms of acute esophagitis appear a week earlier than when treatment is administered sequentially Furthermore, a greater percentage of patients are unable to complete their initially planned treatment, and the risk of associated death is greater [22].
The radiotherapy dosing parameters that best define onset of acute esophagitis remain unclear. Several studies have evaluated the effect of the dose-volume histogram (DVH) on the probability of acute and late complications affecting the esophagus. The most widely studied dosing parameters include the absolute volume, mean dose (Dmean), the percentage of volume that receives the reference dose (Vdose ), or the maximum dose (Dmax) delivered to the esophagus. A systematic review of the literature [23] showed that the best studied predictors, and those for which most evidence is available with respect to radiation-induced esophagitis, were Dmean , V20 , V30 , V40 , V45 , and V50. Improved radiotherapy technology has led to a reduction in the incidence and severity of esophagitis, thus demonstrating that older techniques based on 2D planning led to increased acute and chronic adverse effects [24]. New techniques have made it possible to treat the target volume more accurately, thus limiting the dose that reaches healthy tissue, including the esophagus [25]. Given that our study was performed at a single center, the radiotherapy technology available was homogeneous for all the patients included, with the only differences being in the dose per fraction and the total dose administered. Therefore, all of the study patients were susceptible to developing acute esophagitis induced by oncologic treatment, since treatment planning included a considerable volume of the esophagus within the treatment field.
Similarly, adding chemotherapy to the radiotherapy schedule increases the frequency of esophagitis by approximately 5-fold [26], whereas treatment with radiotherapy alone with curative intent leads to significantly lower rates of grade 3 or higher esophagitis [27]. We found that 15 patients (35.59%) received treatment with radiotherapy alone and 26 patients (63.41%) received treatment with radiochemotherapy with curative intent. All of the patients who were admitted to hospital (3 patients, 7.32%) or whose oncologic treatment was interrupted (2 patients, 4.88%) owing to acute associated toxicity, were receiving treatment with radiochemotherapy. This observation leads us to ask whether patients who received radiotherapy and chemotherapy concurrently would benefit from early administration of support therapy or intensification thereof and thus be able, where possible, to complete the prescribed treatment appropriately.
The role of radio protectors in the development of esophagitis induced by oncologic treatment has been studied in recent years. The role of amifostine in particular is controversial, and the drug has been evaluated in several studies, with contradictory results. Some authors reported a reduced frequency of acute esophagitis with this therapy [28], whereas others found no benefit of combining amifostine with radiochemotherapy [29]. Amifostine was also a poorly tolerated and highly emetogenic. At present, data are insufficient to recommend daily use of amifostine for prevention of esophagitis induced by ionizing radiation. However, other agents (e.g., glutamine) have proven successful as prophylaxis of radiation-induced esophagitis [30], and a significant reduction in the incidence of acute radiation-induced esophagitis has been shown in patients who received treatment with glutamine [31]. In our study, we have not observed any side effects secondary to the administration of ZIVEREL®. Therefore, we can conclude that ZIVEREL® is a safe and well-tolerated product that may have an important role in the management of acute esophagitis in patients receiving oncologic treatment with radiotherapy alone or radiochemotherapy.
Conclusion
ZIVEREL® is a well-tolerated and safe product that plays a major role in the symptomatic relief of patients with acute esophagitis induced as a consequence of oncologic treatment. Initial data must be confirmed in new, well-designed prospective studies in which ZIVEREL® is uniformly administered in a homogeneous cohort of patients.
Conflict of Interest
No potential conflicts of interest relevant to the present article were reported.
References
- Czito BG, DeNittis AS, Palta M, Christopher G Willett CG (2013) Esophageal cancer. In: Halperin EC, Perez CA, Brady LW (Editors),Perez and Brady's Principles and Practice of Radiation Oncology (6th edn), Philadelphia, USA, pp. 3431-3434.
- Roberts SA, Hendry JH (1993) The delay before onset of accelerated tumour cell repopulation during radiotherapy: a direct maximum- likelihood analysis of a collection of worldwide tumour-control data. Radiother Oncol 29(1): 69-74.
- Fowler JF, Lindsfrom MJ (1992) Loss of local control with prolongation in radiotherapy. Int J Radiat Oncol Biol Phys 23(2): 457-467.
- Sonis ST, Elting LS, Keefe D, Peterson DE, Schubert M, et al. (2004) Perspectives on cancer therapy-induced mucosal injury: pathogenesis, measurement, epidemiology, and consequences for patients. Cancer 100(9): 1995-2025.
- Gate L, Paul J, Ba GN, Tew KD, Tapiero H (1999) Oxidative stress induced in pathologies: the role of antioxidants. Biomed Pharmacother 53(4): 169-180.
- Epperly MW, Gretton JA, DeFilippi SJ, Greenberger JS, Sikora CA, et al. (2001) Modulation of radiation-induced cytokine elevation associated with esophagitis and esophageal stricture by manganese superoxide dismutase-plasmid/liposome (SOD2-PL) gene therapy. Radiat Res 155(1): 2-14.
- Gabay C, Kushner I (1999) Acute-phase proteins and other systemic responses to inflammation. N Engl J Med 340: 448-454.
- Khan SA, Wingard JR (2001) Infection and mucosal injury in cancer treatment. J Natl Cancer Inst Monogr 29: 31-36.
- Baker S, Fairchild A (2016) Radiation-induced esophagitis in lung cancer. Lung Cancer 7: 119-127.
- National Cancer Institute (2009) Common Terminology Criteria for Adverse Events (CTCAE); Version 4.0. National Institutes of Health, US Department of Health and Human Services, USA, pp. 196.
- Dicker KT, Gurski LA, Pradhan-Bhatt S, Witt RL, Farach-Carson MC, et al. (2014) Hyaluronan: A simple polysaccharide with diverse biological functions. Acta biomater 10(4): 1558-1570.
- Savarino V, Pace F, Scarpignato C (2017) Randomised clinical trial: mucosal protection combinedwith acid supression in the treatment of non-erosive reflux disease - efficacy of Esoxx, a hyaluronic acid- chondroitin sulphate based bioadhesive formulation. Aliment Pharmacol Ther 45(5): 631-642.
- Aya KL, Stern R (2014) Hyaluronan in wound healing: rediscovering a major player. Wound Repair Regen 22(5): 579-593.
- Lauder RM (2009) Chondroitin sulphate: a complex molecule with potential impacts on a wide range of biological systems. Complement Ther Med 17(1): 56-62.
- du Souich P, García AG, Vergés J, Montell E (2009) Immunomodulatory and anti- inflammatory effects of chondroitin sulphate. J Cell Mol Med 13(8a): 1451-1463.
- Kim SY, Chae SW, Lee J (2014) Effect of Poloxamer 407 as a carrier vehicle on rotator cuff healing in a rat model. J Orthop Surg Res 9(1): 12.
- Shaikh T, Handorf EA, Murphy CT, Mehra R, Ridge JA, et al. (2016). The impact of radiation treatment time on survival in patients with head and neck cancer. Int J Radiat Oncol Biol Phys 96(5): 967-975.
- Withers HR, Taylor JM, Maciejewski B (1988) The hazard of accelerated tumor clonogen repopulation during radiotherapy. Acta Oncol 27(2): 131-146.
- van Dijk KN, Plat AW, van Dijk AA, Piersma-Wichers M, de Vries-Bots AM, et al. (2004) Potential interaction between acenocoumarol and diclofenac, naproxen and ibuprofen and the role of CYP2C9 genotipe. Thromb Haemost 91(1): 95-101.
- Fairchild A (2013) Chapter 5: Side effects of palliative radiation therapy. In: Lutz S, Chow E, Hoskin P (Editors). Radiation Oncology in Palliative Cancer Care. West Sussex, Wiley- Blackwell, UK, p. 43-60.
- Spiro S, Douse J, Read C, Janes S (2008) Complications of lung cancer treatment. Semin Respir Crit Care Med 29(3): 302-317.
- Howell D (2006) The role of radiation therapy in the palliation of gastrointestinal malignancies. Gastroenterol Clin North Am 35(1): 125-130.
- Rose J, Rodrigues G, Yaremko B, Lock M, D'Souza D (2009) Systematic review of dose- volume parameters in the prediction of esophagitis in thoracic radiotherapy. Radiother Oncol 91(3): 282-287.
- Morgan GW, Breit SN (1995) Radiation and the lung: a reevaluation of the mechanisms mediating pulmonary injury. Int J Radiat Oncol Biol Phys 31(2): 361-369.
- Fraass BA, Moran JM (2012) Quality, technology and outcomes: evolution and evaluation of new treatments and/or new technology. Semin Radiat Oncol 22(1): 3-10.
- O'Rourke N, Roque IFM, Farre Bernado N, Macbeth F (2010) Concurrent chemoradiotherapy in non-small cell lung cancer. Cochrane Database Syst Rev 16(6): CD002140.
- Byhardt RW, Scott C, Sause WT, Emami B, Komaki R, et al. (1998). Response, toxicity, failure patterns, and survival in five Radiation Therapy Oncology Group (RTOG) trials of sequential and/or concurrent chemotherapy and radiotherapy for locally advanced non- small-cell carcinoma of the lung. Int J Radiat Oncol Biol Phys 42(3): 469-478.
- Antonadou D, Coliarakis N, Synodinou N, Athanassiou H, Kouveli A, et al. Randomized phase III trial of radiation treatment ± amifostine in patients with advanced-stage lung cancer. Int J Radiat Oncol Biol Phys 51(4): 915-922.
- Werner-Wasik M, Paulus R, Curran WJ Jr, Byhardt R (2011) Acute esophagitis and late lung toxicity in concurrent chemoradiotherapy trials in patients with locally advanced non- small-cell lung cancer: analysis of the radiation therapy oncology group (RTOG) database. Clin Lung Cancer 12(4): 245-251.
- Algara M, Rodriguez N, Vinals P, Lacruz M, Foro P, et al. (2007) Prevention of chemoradiotherapy-induced esophagitis with glutamine: results of a pilot study. Int J Radiat Oncol Biol Phys 69(2): 342-349.
- Topkan E, Yavuz MN, Onal C, Yavuz AA (2009) Prevention of acute radiation-induced esophagitis with glutamine in non-small cell lung cancer patients treated with radiotherapy: evaluation of clinical and dosimetric parameters. Lung Cancer 63(3): 393-399.






























