Antibody-Drug Conjugates as Novel and Potent Cancer Therapeutics
*Veneela KR Vasigala
Rangaraya Medical College, NTR University of Health Sciences, Kakinada, AP, India
Submission: August 30, 2017; Published: September 06, 2017
*Corresponding author: Veneela KR Vasigala, Rangaraya Medical College, NTR University of Health Sciences, Kakinada, AP, India-533001, Tel: , Email: veneela.vk@gmail.com
How to cite this article: Veneela KR V. Antibody-Drug Conjugates as Novel and Potent Cancer Therapeutics. Canc Therapy & Oncol Int J. 2017; 6(5): 555698. DOI: 10.19080/CTOIJ.2017.06.555698
Abstract
Antibody drug conjugates (ADCs) are an emerging class of anti-cancer therapeutics that combines the selectivity of monoclonal antibodies with the potency of cytotoxicity drugs to target specific antigen-expressing tumor cells. ADCs use monoclonal antibodies as vehicles and deliver cytotoxic drugs to tumor cells for specific killing. The three structural components of ADCs; monoclonal antibody, linker, and cytotoxic drug need to be carefully selected in the context of target antigen to achieve high efficacy, selectivity, and safety. US Food and Drug Administration (FDA) approval of ADCs such as Mylotarg®, and Adcetris® for treating hematological cancer, and Kadcyla® for treating breast cancer tumors has provided the much-needed momentum to the field. Here, we review the key characteristics of monoclonal antibody, target antigen, linker, and cytotoxic drug needed for the clinical success of ADCs in cancer therapy.
Keywords: Antibody drug conjugate; ADC; Antibody; Cancer; Cytotoxic Payload; Linker; Immunotherapy
Abbreviations: ADCs: Antibody Drug Conjugates; FDA: Food and Drug Administration; MAb: Monoclonal Antibody; DAR: Drug Antibody Ratio; ALCL: Anaplastic Large Cell Lymphoma; HL: Hodgkin Lymphoma; ASCT: Autologous Stem Cell Transplantation; MMAE: Monomethyl Auristatin E; MBC: Metastatic Breast Cancer
Introduction
Conventional chemotherapeutic drugs to treat cancer suffers from the lack of selectivity towards the tumor site and thus causing damage to even healthy tissues. Drug dose escalation to therapeutically active levels is often impossible with chemotherapeutic drugs due to this toxicity. The selectivity of the treatment was improved with the introduction of monoclonal antibody drugs that are directed against targets that are selectively expressed in tumor cells [1-5]. Because antibodies bind specifically to their cognate antigen on the surface of cells, they can also be used as vehicles to selectively deliver the cytotoxic drug to the cancer cells. The class of therapeutic drug products where monoclonal antibody is conjugated with cytotoxic drug (pay load) are called Antibody drug conjugates (ADCs) [6]. ADCs are powerful cancer therapeutics which provides high selectivity and efficacy towards cancer cells, and reduced toxicity to healthy cells [6,7].
Specific antibodies used in ADC guide the ADC towards tumor tissue and thus can reduce cytotoxicity to normal tissues. ADCs bound to antigen on surface of tumor cells are internalized into the tumor cell where cytotoxic drug is released and kill the tumor cells through their established cytotoxicity mechanism [8,9]. Alternatively, cytokines can be fused directly to antibodies and these cytokines recruit immune cells while in circulation and kill tumor cells indirectly by immune mediated cytotoxicity. Structurally ADCs consists of three components
- Monoclonal antibody(MAb),
- Pay load (cytotoxic drug),
- Linker that connects MAb and pay load (Figure 1).
Mechanistically ADCs are designed in such a way that the molecules are stable in circulation, and release the cytotoxic drug intracellular after initial antigen specific binding and internalization. An in-depth understanding of monoclonal antibody, linker, cytotoxic drug, and the conjugation mechanism is essential for the successful development of ADCs for cancer treatment.
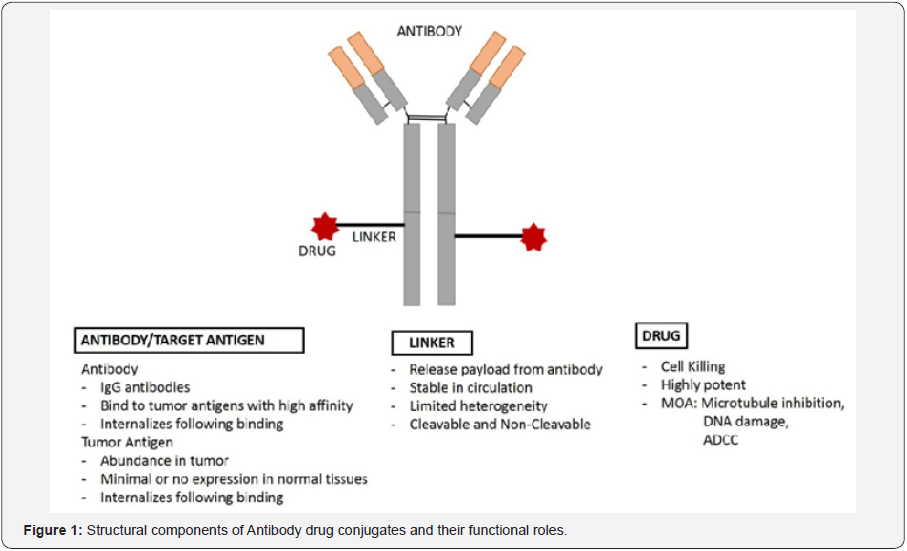
MAb and Target Binding
Binding of MAb to its cognate antigen is the first step of the ADC mechanism and thus a proper selection of the MAb and target antigen plays a critical role in the overall efficiency of the ADC treatment. MAbs used in the current ADCs are in IgG format. The IgG molecules have a long half-life in blood, and can extravasate out of blood vessels and diffuse into the tumor mass. Since IgG antibodies bear an intact Fc region, full IgGs bound to tumor cells may also recruit complement, natural killer cells, and initiate antibody-dependent cell cytotoxicity. Although full IgG has many benefits, for certain types of payloads (cytokines that acts extracellularly) it is preferable to use smaller antibody fragments to avoid excessive inflammation due to prolonged half-life.
The target antigen selected for MAb binding must be abundantly expressed on surface of tumor cells and should be accessible to ADC via blood stream for efficient ADC binding [10- 12]. To ensure high selectivity and low toxicity, the target antigen with minimal or no expression in normal cells is preferable. The MAb and antigen must have high affinity to each other, allowing for potential internalization of the pay load. During the initial ADC development, MAbs must be tested for efficient internalization since non-internalizing ADCs would be less effective. Antigens that show heterogeneous expression on tumor cells and antigens that shed from cells into circulation should be avoided as it impacts the overall efficacy of ADC treatment. The selection of the right antigen is further complicated by the evolutionary pressure on the cancer cell due to cancer treatment. Ideally,cancer cell surface antigens whose expression is down regulated during cancer treatment must be avoided to ensure successful binding of ADC to surface antigens.
Conjugation of MAb to payload via Linkers
A variety of Linkers can connect MAb to the drug of our choice. Linker conjugation mechanism influences stability of ADCs and drug antibody ratio (DAR) of the ADC therapeutics. In general, linkers used in ADCs should be stable and avoid prerelease of the payload during circulation in the blood. At the same time linkers should be amenable to release the payload after internalization into tumor cells [13]. The number of drug molecules bound to antibody (DAR) and their position would greatly influence the ADCs binding affinity to antigen, aggregation, efficacy, and even safety [14].
The most commonly used linkers conjugate the drug to antibody via a covalent bond with either thiol group of cysteines or eplison amine group of lysine on antibody. The presence of four inter-chain disulphide bridges in IgG antibody provides eight maximum possible conjugation sites [15]. Therefore, a drug conjugated via cysteine can have a DAR (No of drug molecules/antibody as measured by chromatography and mass spectrometry) up to 8 depending on the number of cysteine sites conjugated with payload [16-18]. In contrast to cysteine conjugation site, conjugation at lysine would generate highly heterogenous mixtures of ADCs with variable DAR ratio since antibodies have up to 30 lysine amino acids available for conjugation. Site-specific antibody engineering methods have been adapted to insert reactive cysteine or lysine sites to generate homogeneous mixture of ADCs with predictable DAR ratios sufficient for cell cytotoxicity [19].
Linkers can be broadly categorized into cleavable and noncleavable linkers depending on their mechanism of drug release. Cleavable linkers release the drug from MAb by hydrolysis or enzymatic cleavage where as non-cleavable linkers release drug by MAb degradation after internalization. Cleavable linkers are sub characterized into peptide (amide) linkers, hydrazone linkers, and disulphide linkers. Peptide linkers are usually accommodated with protease cleavage sites and are sensitive to proteases in the host cells. A valine-citrullinepeptide linker with protease cathepsin D cleavage site is one successful example used in clinically approved brentuximab vedotin ADC for treatment of relapsed or refractory Hodgkin lymphoma (HL) and systemic anaplastic large cell lymphoma (ALCL) [20].
Hydrazone linkers are susceptible to acidic pH conditions in endosomes and lysosomes, and release the drug by hydrolysis. Disulphide linkers are susceptible to reductive environment in the tumor cells due to high levels of glutathione produced in tumor cells [21]. Non-cleavable linkers release drug after ADC complex degradation post internalization. The antibody portion of ADC gets degraded in lysosomal and endosomal compartments releasing a linker bound drug molecules [22]. It is considered that non-cleavable linkers contribute to high stability in blood stream, longer half-life, and reduced toxicity to ADCs [23,24]. In contrast to cytotoxic drugs that require chemical conjugation to antibodies, antibody cytokine conjugates can be developed as fusion proteins.
Payloads
The payload is usually a potent cytotoxic drug that kills the target tumor cells post release from MAb. The first-generation ADCs employed cancer chemotherapy drugs such as doxorubicin, 5-fluorouracil, and methotrexate with well-known acceptable toxicity profile. However, these agents are not successful in the clinic due to poor cytotoxicity and poor selectivity with only 1-2% drug reaching tumor cells. Subsequently, very potent cytotoxic drugs are employed to ensure tumor cell cytotoxicity even at low levels of ADC. Mechanisms such as inhibition of microtubule polymerization (monomethyl auristatin E, maytansinoid DM1, tubulysin) [25-28] and DNA damage (Pyrrolobenzodiazepine, Calicheamicin) [29] were chosen as the primary targeting mechanisms. Payloads targeting microtubule polymerization not only arrest tumor cell division and may also prevent tumor cell protein trafficking [30]. DNA damaging agents such as Pyrrolobenzodiazepine bind as dimers to DNA minor grooves and prevent DNA replication [29]. By binding to DNA minor groove, these agents also get unrecognized by DNA repair mechanisms. Other agents under investigation include the RNA polymerase II binding α-amanitin which can inhibit DNA transcription and induce apoptosis [31].
Alternative to the above types of payloads that act intracellularly, agents such as cytokines bound to antibodies that act extracellularly are also being investigated as payloads [32]. These cytokines modulate immune cell activity at picomolar concentrations by activating complement system, recruiting natural killer cells, and cytotoxic T lymphocytes and thus and kill tumor cells indirectly. Bacterial toxins, antibodies, and kinase inhibitors are also being tested as payloads to further advance the ADC potential to treat cancer, infectious diseases, and cardio vascular diseases, respectively [33-40].
FDA approved ADC drugs
So far three ADCs have received US Food and Drug Administration (FDA) approval for a variety of haematological and solid tumors. The first drug approved was gemtuzumab ozogamicin (Mylotarg®) in 2000 as a monotherapy against CD33 positive acute myeloid leukemia (AML) elderly patients (above age 60) that are not candidates for further chemotherapy due to observed toxicity at initial treatment [41]. The ADC is comprised of CD33 antibody conjugated to DNA damaging agent calicheamicin. This drug was subsequently with drawn in 2010 as post-approval phase III with ADC and chemo combination therapy showed high toxicity and no clinical improvement compared to chemotherapy alone [42]. The high toxicity was later attributed to poor stability of linker leading to pre-release of calicheamicin and poor selectivity to tumor site [43].
In 2011, Brentuximab vedotin received accelerated approval for treating patients with Hodgkin lymphoma (HL) with prior failure of autologous stem cell transplantation (ASCT) or patients that showed relapse after two chemotherapies and not eligible for ASCT [20]. Brentuximab vedotin is comprised of an anti- CD30 chimeric antibody attached to tubulin inhibitor payload monomethyl auristatin E (MMAE) via a protease-cleavable linker. The FDA approval was based on single arm phase II study that showed an objective response rate of 75%, complete remission in 34% patients, and median duration of 20.5 months [44].
In 2013, ado-trastuzumab-emtansine (T-DM1, Kadcyla) received FDA approval for treatment of HER-2 positive metastatic breast cancer (MBC) who previously received trastuzumab or taxane chemotherapies [23]. T-DM1 is comprised of trastuzuMAbMAb liked to may tansinoid DM1 via a non-cleavable linker. T-DM1 FDA approval was based on a phase III study that showed a progression-free survival of 9.6 months with T-DMI versus 6.4 months with lapatinib plus capecitabine [45]. Research into ADCs as cancer therapies gained much needed attention with the approval of these drugs. More than 100 clinical trials testing different ADCs are presently underway against multiple haematological and solid tumors [7]. The emerging clinical data from these trials will provide further insights into this evolving technology.
Conflict of Interest
Author declares no conflict of interest.
Funding
The work received no specific grant from any funding agency in public, private or non-profit agencies.
Acknowledgement
I would like acknowledge Dr. Veerendra Koppolu (Scientist, AstraZeneca, USA) for his helpful comments and suggestions in the preparation of this manuscript.
References
- Shuptrine CW, Surana R, Weiner LM (2012) Monoclonal antibodies for the treatment of cancer. Semin Cancer Biol 22(1): 3-13.
- Scott AM, Wolchok JD, Old LJ (2012) Antibody therapy of cancer. Nat Rev Cancer 12(14): 278-87.
- Koppolu V, Vasigala V (2016) CTLA-4 antibodies in cancer immunotherapy. MOJ Immunol 3(3): 00092.
- Koppolu V, Pack C, Selvaraj P (2015) Treatment of triple negative breast cancer using a tumor membrane based immunotherapy. 7th Asia-Pacific Biotech Congress, Beijing, China: J Biotechnol Biomater, pp. 112.
- Koppolu V, Vasigala VK (2017) Cancer Immunotherapy Arsenal: Current and Promising New Approaches. Cancer Therapy & Oncology International Journal 5(3): 1-5.
- Sievers EL, Senter PD (2013) Antibody-drug conjugates in cancer therapy. Annu Rev Med 64: 15-29.
- Parslow AC, Parakh S, Lee FT, Gan HK, Scott AM (2016) Antibody-Drug Conjugates for Cancer Therapy. Biomedicines 4(3): 14.
- Diamantis N, Banerji U (2016) Antibody-drug conjugates--an emerging class of cancer treatment. Br J Cancer 114(4): 362-367.
- Jaracz S, Chen J, Kuznetsova LV, Ojima I (2005) Recent advances in tumor-targeting anticancer drug conjugates. Bioorg Med Chem 13(7): 5043-5054.
- Teicher BA (2009) Antibody-drug conjugate targets. Curr Cancer Drug Targets 9(8): 982-1004.
- Shefet-Carasso L, Benhar I (2015) Antibody-targeted drugs and drug resistance-challenges and solutions. Drug Resist Updat 18: 36-46.
- Parakh S, Parslow AC, Gan HK, Scott AM (2016) Antibody-mediated delivery of therapeutics for cancer therapy. Expert Opin Drug Deliv 13(3): 401-419.
- Senter PD (2009) Potent antibody drug conjugates for cancer therapy. Curr Opin Chem Biol 13(3): 235-244.
- Hamblett KJ, Senter PD, Chace DF, Sun MM, Lenox J, et al. (2004) Effects of drug loading on the antitumor activity of a monoclonal antibody drug conjugate. Clinical cancer research: an official journal of the American Association for Cancer Research 10(20): 7063-7070.
- Sochaj AM, Swiderska KW, Otlewski J (2015) Current methods for the synthesis of homogeneous antibody-drug conjugates. Biotechnol Adv 33(1): 775-784.
- Chen T, Chen Y, Stella C, Medley CD, Gruenhagen JA, et al. (2016) Antibody-drug conjugate characterization by chromatographic and electrophoretic techniques. J Chromatogr B Analyt Technol Biomed Life Sci 1032: 39-50.
- Bobaly B, Fleury-Souverain S, Beck A, Veuthey JL, Guillarme D, et al. (2017) Current possibilities of liquid chromatography for the characterization of antibody-drug conjugates. J Pharm Biomed Anal.
- Mulagapati S, Koppolu V, Raju TS (2017) Decoding of O-Linked Glycosylation by Mass Spectrometry. Biochemistry-Us 56(9): 1218- 1226.
- Panowski S, Bhakta S, Raab H, Polakis P, Junutula JR (2014) Site-specific antibody drug conjugates for cancer therapy. MAbs 6(1): 34-45.
- Senter PD, Sievers EL (2012) The discovery and development of brentuxiMAb vedotin for use in relapsed Hodgkin lymphoma and systemic anaplastic large cell lymphoma. Nat Biotechnol 30(7): 631- 637.
- Kigawa J, Minagawa Y, Kanamori Y, Itamochi H, Cheng X, et al. (1998) Glutathione concentration may be a useful predictor of response to second-line chemotherapy in patients with ovarian cancer. Cancer 82(4): 697-702.
- Doronina SO, Mendelsohn BA, Bovee TD, Cerveny CG, Alley SC, et al. (2006) Enhanced activity of monomethylauristatin F through monoclonal antibody delivery: effects of linker technology on efficacy and toxicity. Bioconjug Chem 17(1): 114-124.
- LoRusso PM, Weiss D, Guardino E, Girish S, Sliwkowski MX (2011) TrastuzuMAb emtansine: a unique antibody-drug conjugate in development for human epidermal growth factor receptor 2-positive cancer. Clinical cancer research: an official journal of the American Association for Cancer Research 17(20): 6437-6447.
- Girish S, Gupta M, Wang B, Lu D, Krop IE, et al. (2012) Clinical pharmacology of trastuzuMAb emtansine (T-DM1): an antibody-drug conjugate in development for the treatment of HER2-positive cancer. Cancer Chemother Pharmacol 69(5): 1229-1240.
- Dumontet C, Jordan MA (2010) Microtubule-binding agents: a dynamic field of cancer therapeutics. Nat Rev Drug Discov 9(10): 790-803.
- Oroudjev E, Lopus M, Wilson L, Audette C, Provenzano C, et al. (2010) Maytansinoid-antibody conjugates induce mitotic arrest by suppressing microtubule dynamic instability. Mol Cancer Ther 9(10): 2700-2713.
- Sachdev J, Edelman M, Harb W, Matei D, Nguyen B, et al. (2015) P1.08A phase 1 study of 3 different schedules of the folic acid-tubulysin smallmolecule drug conjugate EC1456 in pts with advanced solid tumors. Annals of Oncology 26(2): 18.
- Valasani KR, Kuruva CS, Koppolu V, Vangavaragu JR, Victor DW (2017) Synthetic and Biological Applications of Benzothiazole Phosphonates. Heterocyclic Compounds and Biological Applications: Science Publishing Group, New York, USA.
- Hartley JA (2011) The development of pyrrolobenzodiazepines as antitumour agents. Expert opinion on investigational drugs 20(6): 733-44.
- Poruchynsky MS, Komlodi-Pasztor E, Trostel S, Wilkerson J, Regairaz M, et al. (2015) Microtubule-targeting agents augment the toxicity of DNA-damaging agents by disrupting intracellular trafficking of DNA repair proteins. Proceedings of the National Academy of Sciences of the United States of America 112(5): 1571-1576.
- Moldenhauer G, Salnikov AV, Luttgau S, Herr I, Anderl J, et al. (2012) Therapeutic potential of amanitin-conjugated anti-epithelial cell adhesion molecule monoclonal antibody against pancreatic carcinoma. J Natl Cancer Inst 104(8): 622-34.
- List T, Casi G, Neri D (2014) A chemically defined trifunctional antibody-cytokine-drug conjugate with potent antitumor activity. Mol Cancer Ther 13(11): 2641-2652.
- Lorberboum-Galski H (2011) Human toxin-based recombinant immunotoxins/chimeric proteins as a drug delivery system for targeted treatment of human diseases. Expert Opin Drug Deliv 8(5): 605-621.
- Pillow TH (2017) Novel linkers and connections for antibody-drug conjugates to treat cancer and infectious disease. Pharm Pat Anal 6(1): 25-33.
- Lehar SM, Pillow T, Xu M, Staben L, Kajihara KK,et al. (2015) Novel antibody-antibiotic conjugate eliminates intracellular S. aureus. Nature 527(7578): 323-328.
- Liu R, Wang RE, Wang F (2016) Antibody-drug conjugates for nononcological indications. Expert Opinion on Biological Therapy 16(5): 591-593.
- Koppolu V, Osaka I, Skredenske JM, Kettle B, Hefty PS, et al. (2013) Small-molecule inhibitor of the Shigella flexneri master virulence regulator VirF. Infect Immun 81(11): 4220-4231.
- Skredenske JM, Koppolu V, Kolin A, Deng J, Kettle B, et al. (2013) Identification of a small-molecule inhibitor of bacterial AraC family activators. J Biomol Screen 18(5): 588-598.
- Koppolu V (2013) Molecular Mechanisms and Inhibition of Transcription Activation by Bacterial AraC Family Activator Proteins. University of Kansas, Lawrence, USA, pp. 1-202.
- Koppolu V, Vasigala VK (2016) Role of Escherichia coli in biofuel production. Microbiology insights 9: 29-35.
- Tsimberidou AM, Giles FJ, Estey E, O’Brien S, Keating MJ, et al. (2006) The role of gemtuzuMAb ozogamicin in acute leukaemia therapy. Br J Haematol 132(4): 398-409.
- Petersdorf SH, Kopecky KJ, Slovak M, Willman C, Nevill T, et al. (2013) A phase 3 study of gemtuzuMAb ozogamicin during induction and postconsolidation therapy in younger patients with acute myeloid leukemia. Blood 121(24): 4854-4860.
- Ten Cate B, Bremer E, de Bruyn M, Bijma T, Samplonius D, et al. (2009) A novel AML-selective TRAIL fusion protein that is superior to GemtuzuMAb Ozogamicin in terms of in vitro selectivity, activity and stability. Leukemia 23(8): 1389-1397.
- Younes A, Gopal AK, Smith SE, Ansell SM, Rosenblatt JD, et al. (2012) Results of a pivotal phase II study of brentuxiMAb vedotin for patients with relapsed or refractory Hodgkin’s lymphoma. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 30(18): 2183-2189.
- Verma S, Miles D, Gianni L, Krop IE, Welslau M, et al. (2012) TrastuzuMAb emtansine for HER2-positive advanced breast cancer. The New England journal of medicine 367(19): 1783-1791.






























