What was the Effect of Taxol in Non-Metastatic Non-Small Cell Lung Cancer Patients in Mansoura University Hospital?
Manal Mostafa Saleh Elghareeb, Hend M Hamdey Rashed Elkalla*, Mohamed Saad Elashry and Ahmed Elshahat
Clinical Oncology and Nuclear Medicine Department, Mansoura University Hospital, Egypt
Submission: August 04, 2017; Published: August 10, 2017
*Corresponding author: Hend M Hamdey Rashed Elkalla, Lecturer at Faculty of Medicine, Mansoura University Hospital, Clinical Oncology and Nuclear Medicine department, Egypt, Tel: 00201210200602; Email: dr.hend1113@gmail.com
How to cite this article: Manal M S E, Hend M H R E, Mohamed S E, Ahmed E. What was the Effect of Taxol in Non-Metastatic Non-Small Cell Lung Cancer Patients in Mansoura University Hospital?. Canc Therapy & Oncol Int J. 2017; 6(4): 555691. DOI: 10.19080/CTOIJ.2017.06.555691
Abstract
Introduction: Lung cancer has been turned from a rare disease to a worldwide problem. Non-small cell lung cancer (NSCLC) is the most commonly diagnosed type of lung cancer forming about 85% of all cases. Measures for treatment in NSCLC include: surgical resection, radiotherapy, chemotherapy and targeted therapy. They can be used as solo modality or in combination. Taxanes as paclitaxel and docetaxel are important classes of chemotherapeutic agents.
Aim of the work: The aim of our retrospective study is to evaluate the role of taxol based chemotherapy in treatment of non-metastatic NSCLC.
Patient & methods: Between January 2008 and December 2013, thirty five patients with newly diagnosed, non- metastatic NSCLC were included for this analysis. Between January 2008 and December 2013, thirty five patients with newly diagnosed, non- metastatic NSCLC were included for this analysis. The patients received paclitaxel containing regimens. All were combined either with carboplatin (33 patients) or with cisplatin (2 patients).
Results: As regard response rate only one patient achieved complete response, partial response was detected in 18 patients, stationary disease was demonstrated in 12 patients and 4 patients had progressive disease. The most common toxicity was the hematological toxicity. The median PFS was 8 months (95% Confidence Interval, 6.774 -9.226 months). The median OS was 13 months (95% Confidence Interval, 10.533 –15.467 months). Conclusion: Taxol is active class of chemotherapeutic agents in NSCLC and well tolerated.
Keywords: Lung cancer; Taxanes; NSCLC; Chemo-radiotherapy; Taxol
Abbreviations: NSCLC: Non-Small Cell Lung Cancer; ECOG: Eastern Cooperative Oncology Group; NCI-CTCAE: Common Terminology Criteria for Adverse Events; SPSS: Statistical Package for Social Sciences; PFS: Progression Free Survival; OS: Overall Survival; RR: Response Rate; CR: Complete Response; PR: Partial response; SD: Stationary Disease
Introduction
Lung cancer has been turned from a rare disease to a worldwide problem. The deaths in USA were 160,340 in 2012 due to lung cancer. In USA, it was responsible for 28% of all cancer mortality. Changing the location does not affect markedly the global lung cancer mortality, with 43% of deaths occurring in more developed countries and 57% occurring in the developing regions Ridge et al. [1]. Non-small cell lung cancer (NSCLC) is the most commonly diagnosed type of lung cancer forming about 85% of all cases. It grows up and spreads much slowly than small cell lung cancer Barzi & Pennell [2]. At diagnosis, more than 80% have locally advanced stage III or stage IV tumor and are not candidate for curative surgery. Five year survival is less than 10% in this patient population Treat et al. [3]. The one year survival rate for lung cancer increased from 35% in between 1975 and 1979 to 42% in 1988 up to 2008. Five year survival rate for lung cancer is poor. The overall 5-year survival rate for lung cancer of all stages was 16.8% in 2004 and increased slightly over time, in comparison with a 13.3% 5-year survival rate in 1982 Ridge et al. [1].
Measures for treatment in NSCLC include: surgical resection, radiotherapy, chemotherapy and targeted therapy. They can be used as solo modality or in combination depending on the disease status Ravenel [4]. Treatment options for resectable locally advanced NSCLC include radical chemo-radiotherapy and induction neo-adjuvant therapy followed by surgery Thomas et al. [5]. Combination chemotherapy is the standard of care for patients with locally advanced non-resectable NSCLC and a performance status score of 0 or 1 Ramalingam & Belani [6]. Taxanes as paclitaxel and docetaxel are important classes of chemotherapeutic agents McGrogan et al. [7]. The aim of our retrospective study is to evaluate the role of taxol based chemotherapy in treatment of non-metastatic NSCLC in the term of response rate (RR) and toxic profile then to assess progression free survival (PFS) and overall survival (OS).
Patients and Methods
Patients Inclusion Criteria
- Patients were aged 18 to70 years.
- Performance status ≤ 2.
- Pathologically proven non-small cell lung cancer
- Non metastatic disease.
- Adequate bone marrow, liver, and kidney functions (haemoglobin ≥ 9g/dl, platelet ≥100,000/l, WBCs ≥3000/l provided that absolute neutrophil count (ANC) ≥1500/l and serum creatinine ≤1.5 upper limit of normal).
- No prior treatment with chemotherapy.
This study was approved by the Ethical Committee of Faculty of Medicine, Mansoura University. The collected data from patient’s sheets included the history from all patients as age, sex and smoking habit. General examination was done including weight, height and surface area. Performance status was assessed according to Eastern Cooperative Oncology Group (ECOG). Tumor staging for all patients were documented according to TNM staging system. Investigations included pathological, radiological and laboratory investigations.
Treatment Regimens
Thirty five patients were included in this study. The patients received paclitaxel containing regimens. All were combined either with carboplatin (33 patients) or with cisplatin (2 patients). In most of patients the schedule of paclitaxel used was ranged from 175 -200 mg/m2 IV infusion over 3 h every 3weeks ,only 4 patients received paclitaxel 75 mg/m2 IV infusion weekly. The number of cycles ranged from 4 to 8 and the median number was 6. All patients were received pre-medications as recommended. Symptoms and signs of toxicities were assessed either hematological toxicities or non-hematological toxicities. Toxicities were reported according to the Common Terminology Criteria for Adverse Events (NCI-CTCAE) version 4.0. The primary end point in our research was to assess the RR and the toxic profile and the secondary end points were to assess PFS and OS.
Statistical Analysis
The data was collected, coded, processed and analyzed using SPSS (statistical package for social sciences) version 16. Qualitative data were described using number and percent. Associations between categorical variables were tested using Chi-square test (c2). Monte Carlo test was used whenever there was one or more cell counted 5 or less. Continuous variables were presented as mean ± SD (standard deviation) and Median (min – max). The two groups were compared with Student t test (parametric data) P-value considered significant if ≤ 0.05. The survival functions were tested using Kaplan-Meier.
Results
Patient and Tumor profile
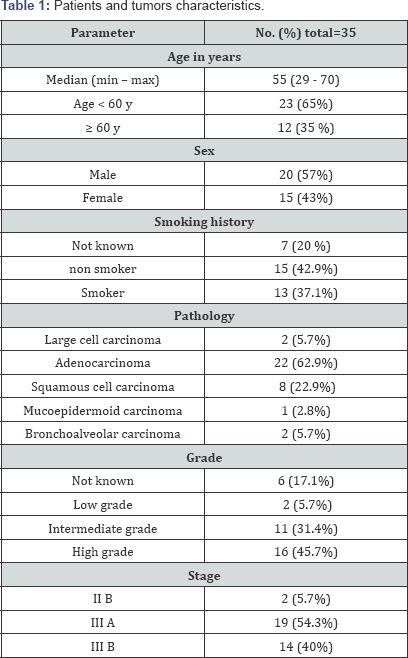
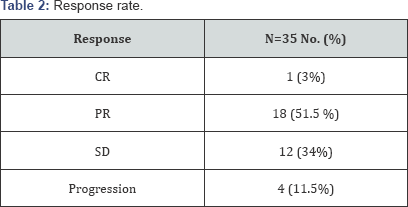
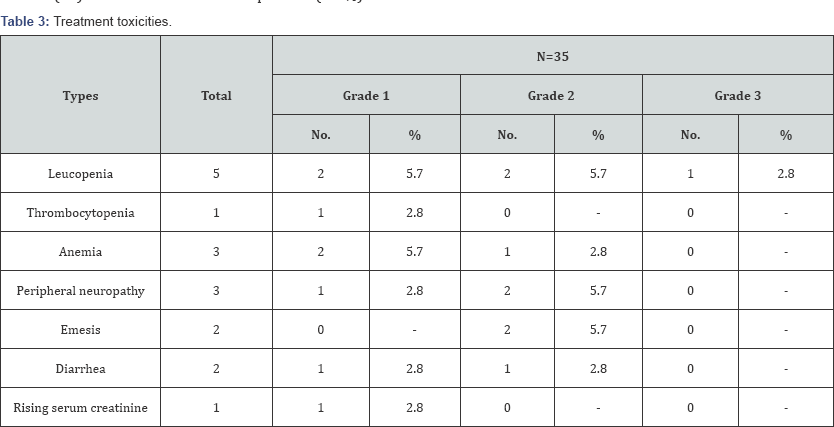
Between January 2008 and December 2013, thirty five patients with newly diagnosed, non- metastatic NSCLC were included for this analysis. Patient’s characteristics and tumors features were mentioned in Table 1. As regard response rate only one patient (3%) achieved complete response (CR), partial response (PR) was detected in 18 patients (51.5%), stationary disease (SD) was demonstrated in 12 patients (34 %) and 4 patients (11.5%) had progressive disease as summarized in Table 2. The most common toxicity was the hematological toxicity. Leucopenia was detected in 5 patients (14%); (grade I was 5.7 %), (grade II was 5.7 %), and (grade III was 2.8 %). Thrombocytopenia was found only in one patient (2.8%) and was grade I. Three patients (8.5%) developed anemia during therapy, 2 patients (5.7 %) was grade I anemia and 1 patient (2.8%) was grade II. Other adverse effects as peripheral neuropathy was observed in 3 patients (8.5%), grade I was in one patient and grade II was detected in 2 patients (5.7 %). Two patients (5.7 %) suffered from grade II emesis. Also diarrhea was noticed in 2 patients (5.7 %). Less common toxicities were observed as rising serum creatinine as shown in Table 3. Survival analysis was done for this study showed that median PFS was 8 months (95% Confidence Interval, 6.774 -9.226 months) as shown in Figure 1. The median OS was 13 months (95% Confidence Interval, 10.533 –15.467 months) as shown in Figure 2.


Discussion
Our study was a retrospective study included 35 patients with non-metastatic NSCLC received taxol based chemotherapy. It showed male predominance 57% and female percentage was 43% in comparable to the study of Liao WY et al. [8] at 2013 with male 52% and female percentage48% Liao et al. [8]. In our study 94 % of patients had stage III disease. Complete response (CR) was achieved in one patient(3%) and 20 patient (51.5 %) showed partial response (PR) with overall response rate (ORR) (54.5%) , while Kocher F and his colleagues at 2014 evaluated response after induction of docetaxeland cisplatin in 78 chemonaïve patients with NSCLC stage II, IIIA and IIIB. Partial response to induction therapy was observed in 43 patients (55%) Kocher et al. [9]. Liao WY and his colleagues at 2013demonstrated the efficacy of taxanes among 62 patients with stage III N2 NCSLC, CR was detected in one patient (2%) and PR was achieved in 25 patients (40%) with ORR (42%) Liao et al. [8]. Other studies as Chen YM et al. [10], Stathopoulos GP et al. [11], Inoue A et al. [12] showed ORR 40%, 42.86%, 45% respectively. This difference could be attributed to larger sample size in other studies.
The median OS in our study was 13 months (95% Confidence Interval, 10.533 -15.467 months) which cope with Bonomi P et al. [13] results. Their study included 599 patients to Compare OS and life quality in advanced non-small-cell lung cancer patients treated paclitaxel combined with cisplatin versus etoposide with cisplatin. The median survival duration for the stage IIIB subgroup in paclitaxel arm was13.1 months. A randomized phase II study compare weekly paclitaxel versus vinorelbine in combination with cisplatin against inoperable chemo-naïvenonsmall- cell lung cancer included 140 patients.
The median survival time was 11.7 months in the paclitaxelcisplatin arm Y Chen et al. [14]. Also the results obtained in our series are better than Fossella F et al. [15], Sandler A et al. [16], Lynch TJ et al. [17] and Zhu J et al. [18] with OS 11.3, 10.3, 8.38 , 8.9 months respectively. This difference could be attributed to larger sample size in these studies; also these studies included stage III and IV disease, so their OS lower than the OS in our study. Regarding PFS in our study was 8 months and were higher than Chen YM et al. [10], Mok TS et al. [19], Mitsudomi T et al. [20] and Niho S et al. [21] that reported median PFS of 6, 5.8, 6.3, 5.9 months respectively.
The commonest toxicity in our study was the hematological toxicity. Other adverse effects as peripheral neuropathy was observed in 3 patients (7.5%), 2 patients (5%) suffered from grade II emesis and diarrhea was noticed in 2 patients (5%) only. On the other hand; Kosmidis P et al. [22] demonstrated mild toxicities in paclitaxel and carboplatin combinations for the treatment of advanced non-small-cell lung cancer. Grades ¾ neutropenia, thrombocytopenia, and anemia were seen in (15%) and (15%), (2%) respectively Kosmidis et al. [22]. Inoue A et al. [12] assessed toxicities of weekly paclitaxel combined with carboplatin in elderly patients with advanced NSCLC.
The most Common toxicities were hematological; Grades ¾ leucopenia, thrombocytopenia, and anemia were observed in (30%), (15%), (13%) respectively, while peripheral neuropathy and nausea were detected only in 3% of patients. The higher hematological toxicities in this study could be explained by the old age that may be liable for more adverse events Inoue et al. [12]. Scagliotti GV and his colleagues reported at 2010 the adverse effects of carboplatin and paclitaxel in chemotherapynaïve patients with advanced NSCLC. Neutropenia was 7%, thrombocytopenia was 3%, Anemia was 9%, peripheral neuropathy was 13%, vomiting was 7%, diarrhea was 13 % and oral mucositis was 2% Scagliotti et al. [23].
Conclusion
In conclusion in our study; taxol are active class of chemotherapeutic agents in NSCLC and well tolerated. This agent exhibit clinical differences that can affect the choice of agents. However, further studies with longer follow up are needed for better orientation and comparisons with other agents.
Disclosure
The authors have no conflicts of interest to disclose.
References
- Ridge CA, McErlean AM, Ginsberg MS (2013) Epidemiology of lung cancer. Paper presented at the Semin Intervent Radiol 30(2): 93-98.
- Barzi A, Pennell NA (2010) Targeting angiogenesis in non-small cell lung cancer: agents in practice and clinical development. European Journal of Clinical and Medical Oncology 2(1): 31-42.
- Treat J, Gonin R, Socinski M, Edelman M, Catalano R, et al. (2010) A randomized, phase III multicenter trial of gemcitabine in combination with carboplatin or paclitaxel versus paclitaxel plus carboplatin in patients with advanced or metastatic non-small-cell lung cancer. Ann Oncol 21(3): 540-547.
- Ravenel JG (2012) Evidence-based imaging in lung cancer: a systematic review. Journal Thorac Imaging 27(5): 315-324.
- Thomas M, Rübe C, Hoffknecht P, Macha HN, Freitag L, et al. (2008) Effect of preoperative chemoradiation in addition to preoperative chemotherapy: a randomised trial in stage III non-small-cell lung cancer. The lancet oncology 9(7): 636-648.
- Ramalingam S, Belani C (2008) Systemic chemotherapy for advanced non-small cell lung cancer: recent advances and future directions. The Oncologist 13(1): 5-13.
- McGrogan BT, Gilmartin B, Carney DN, McCann A (2008) Taxanes, microtubules and chemo resistant breast cancer. Biochimica Biophysica Acta 1785(2): 96-132.
- Liao WY, Chen JH, Wu M, Shih JY, Chen KY, et al. (2013). Neoadjuvant Chemotherapy with Docetaxel-Cisplatin in Patients With Stage III N2 Non–Small-Cell Lung Cancer. Clin Lung cancer 14(4): 418-424.
- Kocher F, Pircher A, Mohn-Staudner A, Romeder F, Duller W, et al. (2014). Multicenter phase II study evaluating docetaxel and cisplatin as neoadjuvant induction regimen prior to surgery or radio chemotherapy with docetaxel, followed by adjuvant docetaxel therapy in chemo naive patients with NSCLC stage II, IIIA and IIIB (TAX-AT 1.203 Trial). Lung Cancer 85(3): 395-400.
- Chen YM, Perng RP, Lee YC, Shih JF, Lee CS, et al. (2002) Paclitaxel plus carboplatin, compared with paclitaxel plus gemcitabine, shows similar efficacy while more cost-effective: a randomized phase II study of combination chemotherapy against inoperable non-small-cell lung cancer previously untreated. Annals of oncology 13(1): 108-115.
- Stathopoulos G, Veslemes M, Georgatou N, Antoniou D, Giamboudakis P, et al. (2004) Front-line paclitaxel–vinorelbine versus paclitaxel– carboplatin in patients with advanced non-small-cell lung cancer: a randomized phase III trial. Ann Oncol 15(7): 1048-1055.
- Inoue A, Usui K, Ishimoto O, Matsubara N, Tanaka M, et al. (2006) A phase II study of weekly paclitaxel combined with carboplatin for elderly patients with advanced non-small cell lung cancer. Lung Cancer 52(1): 83-87.
- Bonomi P, Kim K, Fairclough D, Cella D, Kugler J, et al. (2000) Comparison of survival and quality of life in advanced non–smallcell lung cancer patients treated with two dose levels of paclitaxel combined with cisplatin versus etoposide with cisplatin: Results of an Eastern Cooperative Oncology Group trial. Journal of Clinical Oncology 18(3): 623-631.
- Chen Y, Perng R, Shih J, Lee Y, Lee C, et al. (2004) A randomised phase II study of weekly paclitaxel or vinorelbine in combination with cisplatin against inoperable non-small-cell lung cancer previously untreated. British journal of cancer 90(2): 359-365.
- Fossella F, Pereira JR, Von Pawel J, Pluzanska A, Gorbounova V, et al. (2003) Randomized, multinational, phase III study of docetaxel plus platinum combinations versus vinorelbine plus cisplatin for advanced non–small-cell lung cancer: The TAX 326 study group. J Clin Oncol 21(16): 3016-3024.
- Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, et al. (2006) Paclitaxel–carboplatin alone or with bevacizumab for non–small-cell lung cancer. New England Journal of Medicine 355: 2542-2550.
- Lynch TJ, Patel T, Dreisbach L, McCleod M, Heim WJ, et al. (2010) Cetuximab and first-line taxane/carboplatin chemotherapy in advanced non–small-cell lung cancer: Results of the randomized multicenterphase III trial BMS099. Journal of Clinical Oncology 28(6): 911-917.
- Zhu J, Sharma DB, Gray SW, Chen AB, Weeks JC et al. (2012) Carboplatin and paclitaxel with vs without bevacizumab in older patients with advanced non–small cell lung cancer. JaMa, 307(15): 1593-1601.
- Mok T, Wu Y, Thongprasert S, Yang C, Chu D, et al. (2009) Initial treatment of pulmonary adenocarcinoma with gefitinib or carboplatin/ paclitaxel. N Engl J Med 361: 947-957.
- Mitsudomi T, Morita S, Yatabe Y, Negoro S, Okamoto I, et al. (2010) Gefitinib versus cisplatin plus docetaxel in patients with non-smallcell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised phase 3 trial Lancet oncol 11(2): 121-128.
- Niho S, Kunitoh H, Nokihara H, Horai T, Ichinose Y, et al. (2012) Randomized phase II study of first-line carboplatin-paclitaxel with or without bevacizumab in Japanese patients with advanced nonsquamous non-small-cell lung cancer. Lung Cancer 76(3): 362-367.
- Kosmidis P, Mylonakis N, Nicolaides C, Kalophonos C, Samantas E, et al. (2002) Paclitaxel plus carboplatin versus gemcitabine plus paclitaxel in advanced non–small-cell lung cancer: a phase III randomized trial. Journal of Clinical Oncology 20(17): 3578-3585.
- Scagliotti G, Novello S, Von Pawel J, Reck M, Pereira JR, et al. (2010). Phase III study of carboplatin and paclitaxel alone or with sorafenib in advanced non–small-cell lung cancer. Journal of Clinical Oncology 28(11): 1835-1842.






























