Dose Escalation with Brachy therapy in Nasopharyngeal Cancers- an Under-Utilized Tool
*Ritika Harjani Hinduja
Gujarat University, India
Submission: April 03, 2017; Published: April 17, 2017
*Corresponding author: Ritika Harjani Hinduja, The Gujarat Cancer and Research institute, M.P.Shah Cancer Hospital New Civil Hospital Campus, Ahmedabad 380016, India, Tel: 917259749641; Email: ritikaharjani@gmail.com
How to cite this article: Ritika H H. Dose Escalation with Brachy therapy in Nasopharyngeal Cancers- an Under-Utilized Tool. Canc Therapy & Oncol Int J. 2017; 4(4).555642. DOI: 10.19080/CTOIJ.2017.04.555642
Abstract
Treatment of nasopharyngeal cancers with radiotherapy shows a dose response relationship. Dose escalation is an attractive way to improve outcomes. Brachy therapy and newer modalities like Intensity Modulated Radiation Therapy and Stereotactic Radiation Therapy can achieve high doses to the naso pharynx, sparing the organs at risk. Brachy therapy is the highest form of conformal therapy and its use in nasopharyngeal cancers dates back to 1920s. It's an easy to perform and economical procedure and has evidence in support of its use in early stage nasopharyngeal cancer. There are studies that highlight its indication in large primary tumors. Intra-cavitary brachy therapy using the Rotterdam Nasopharyngeal Applicator (RNA) is the most commonly and recommended technique. Treatment planning can be done by marking tumor related and organ at risk points on orthogonal radiographs or by using computerized tomography images. The technique, indications, planning, doses and toxicity are discussed in the review.
Introduction
Radiation therapy remains the cornerstone of management in nasopharyngeal carcinomas. Treatment with radiotherapy alone results in local control rates of 85-90% for early stage disease (T1-2, N0) and 44-71% for advanced stage disease (T3-4, N+) [1]. Use of concurrent and adjuvant cisplatin based chemotherapy has improved the local control, reduced the number of distant relapses and thereby, has resulted in an improvement in the progression free survival and overall survival. Hence, in recent times, radiation is routinely supplemented with chemotherapy [2]. However, treatment with chemo-radiation for nasopharyngeal carcinoma is still associated with a significant local failure rate. It has been well documented that lack of local control is an independent prognostic factor for development of distant metastases [3]; hence, improvement of local control remains to be an effective way of improving overall outcomes in patients with nasopharyngeal carcinoma. Escalation of the radiation dose is an attractive option for improving local control of primary disease.
Evidence in Support of Dose-Escalation
A significant dose response relationship has been observed in majority of the retrospective studies. The dose response relationship was described by Yan et al. [4] who noted an improvement in the local control in tumors that were treated beyond the conventional 70 Gy (Gray). In their retrospective analysis, tumors boosted with reduced fields beyond 70 Gy had significantly higher local control and survival rates. Additionally, Lee et al. [5] proposed that there was a 9% decrease in local control rate for each Gray below the conventional 66 to 70 Gy. Marks et al. [6] showed that local control was significantly improved in patients who received greater than 67Gy to the tumor target [6].
A significant radiation dose-effect relationship was also found by Levendag et al. [7] for the physical dose, the biological effective dose (BED) 10, and the dose corrected for the overall treatment time (OTT;BEDcor10). The steepest slope in dose- effect was seen for the BEDcor10. He suggested that, on an average, a 15% fall in local failure rate can be obtained with every additional 10 Gy BEDcor10 in excess of 60 Gy [7].
In a study by Perez et al. [8], nasopharyngeal tumor control rate was 80% in the patients who received 6600 to 7000 cGy and100% in those who received over 7000 cGy. However, these results were limited to patients with T1, T2, and T3 tumors. In T4 lesions, the tumor control rate did not rise above 55% even for doses over 7000 cGy [8].
These finding prove without doubt the benefit of dose escalation on tumor control in T1-3 lesions. But the evidence is not completely conclusive for T4 tumors. T4 tumors comprise of tumors with intra-cranial extension or involvement of the cranial nerves, hypo pharynx, orbit, infra-temporal fossa or masticator space [9]. Literature suggests that besides consideration of the prescribed dose, the problem of insufficient coverage has to be overcome in these advanced tumors.
These studies have paved way for the standard treatment regimen for nasopharyngeal carcinoma which comprises radiation up to a dose of 70 Gy to the gross disease over seven weeks, along with 50 to 60 Gy elective irradiation of areas with subclinical disease.
Difficulty
Although nasopharyngeal cancers are relatively radiosensitive tumors, they are one of the most technically difficult sites within the head and neck region to treat. The dose to which radiation can be safely delivered is limited by the critical location of the naso pharynx. Naso pharynx is located in the midline just below the base of skull. It is difficult to cover the entire planning target volume (PTV) by the prescription iso dose along with respecting the tolerance of surrounding critical structures like brain, brainstem, optic apparatus, pituitary gland, amongst others [10]. As these organs at risk are mostly serial organs, a balance is generally sought between coverage of tumor and the normal tissue tolerance.
As a result, there has been a need to use methods that would achieve higher doses to the tumor alone, sparing the critical structures. Brachy therapy and stereotactic therapy are two such modalities that provide conformal dose distribution facilitating dose escalation.
Brachy Therapy
Brachy therapy is the most conformal means of radiotherapy Its rapid dose fall off and very high dose near the source accounts for a high therapeutic ratio, making it an effective way of dose escalation. Applying this modality to naso pharynx is feasible as it is accessible to applicator positioning.
History
In 1920s, Pierquin and Richard were among the first persons to employ brachy therapy in the treatment of nasopharyngeal carcinomas [11]. In the Christie hospital at Manchester, Peterson used a 15 mg (milligrams) radium tube inserted in a cork with a diameter of 15 to 20 mm. The dose prescribed was 80 rads in seven days to a depth of 0.5 cm. Peterson described this technique as an useful alternative to small field X-ray technique but not superior to the use of X-rays. At the same time, several other brachy therapy techniques were also developed by other authors, notably Cade who used a linear array of radium tubes inside catheters and Paris where radium needles packed in gauze were used for brachy therapy. However the bulky size of the applicators, inconsistant dosimetry and discomfort associated with prolonged application made this form of brachy therapy unpopular.
Indications
According to GEC-ESTRO [The Groupe Europeen de Curietherapie (GEC) and the European Society for Radiotherapy & Oncology (ESTRO)] guidelines [12],
Brachy therapy is indicated in the following scenarios;
A. Boost areas of minimal residual local disease after external beam irradiation
B. As salvage therapy for well-circumscribed and superficial local recurrences limited to the nasopharyngeal cavity.
The indication discussed in this review is primarily with the intent of boosting the tumor site (gross tumor volume- GTV) to a higher dose. Suitable patients include whose depth of the target volume is within 10 mm (millimetres). Consequently, only superficial tumors or those exhibiting sufficient shrinkage after external beam radiotherapy and/or chemo radiation and not involving the underlying bone or deeply invading the infra temporal space are suitable for brachy therapy. The T1-2 subset of patients have shown the maximum benefit
Evidence
A large number of studies [5,7,13-18] have shown the benefit of brachy therapy especially in early T stages (T1,2).
Dose escalation in advanced T stage tumors failed to produce improved tumor outcomes. A study by Ozyar et al. [19] of patients with T1- 4 tumors treated with High Dose Rate (HDR) brachy therapy boost of 12 Gy in 3 fractions did not show improvement over External beam Radiation alone (3 yr L-FFR (local freedom from relapse), 86% vs 94%; p- 0.23). Rosenblatt et al. [20] in a study by International Atomic Energy Agency (IAEA), Vienna, Austria evaluated patients treated with induction chemotherapy followed by concurrent chemo-radiation. He compared patients receiving External beam radiation to a dose of 70 Gy with patients receiving same external RT (Radiation Therapy) dose followed by brachy therapy boost (11Gy Low Dose Rate (LDR) or 9Gy/3Fr (Fractions) HDR to Nasopharyngeal tumor tissue (TT) point). Overall survival, local relapse free survival and disease free survival were not significantly different between treatment arms.
Levendag et al. [21] performed a pooled analysis including the patients in the above study (n = 274) with two additional cohorts of patients with NPC treated in Rotterdam (n = 94) and Amsterdam (n = 89). The pool comprised a total of 457 patients. Thirty three patients were excluded because of having early (T1- T2 N0) disease. No significant difference in local recurrences were observed for the T3-T4 N+ tumors.
However, for T1-T2 N+ tumours, significant differences in local control were found between patients treated with or without a brachy therapy boost in the pooled analysis, thus confirming previous studies for patients with early local disease.
Types of Nasopharyngeal Brachy therapy
A. Mould brachy therapy
B. Intracavitary or endocavitary brachy therapy
C. Interstitial brachy therapy
Mould brachy therapy

In the customised mould technique, two to four sagittally oriented plastic tubes (Figure 1) are fixed on the surface of a rigid acrylic applicator, made from an individual impression of the nasopharyngeal cavity under neuroleptic anesthesia. The radiation oncologist determines the position of the sources with respect to the topography of the tumor, as it appears imprinted on the applicator. Boundaries of the tumor are highlighted in ink and source position that will provide optimal coverage of the lesion are indicated with sagittal lines. Spacing between sources varies from 10 to 15 mm. In an averaged sized naso pharynx, two sources can be placed on one lateral wall, three sources on two walls, for example the roof and one lateral wall or four sources on three walls [22].
Interstital brachy therapy
It can also be done. Sharp needles are inserted into the naso pharynx and the para pharyngeal space [23].
Endo cavitary brachy therapy
With the development of easy to introduce endo cavitary nasopharyngeal applicator, it has become the most common device to perform nasopharyngeal brachy therapy. GEC- ESTRO recommends the use of endocavitary techniques [12] as the naso pharynx is deeply situated and surrounded by bone, vessels and nerves. The same technique has been endorsed by American Brachy therapy Society (ABS) [24]. The same has been discussed in detail in this article.
Application
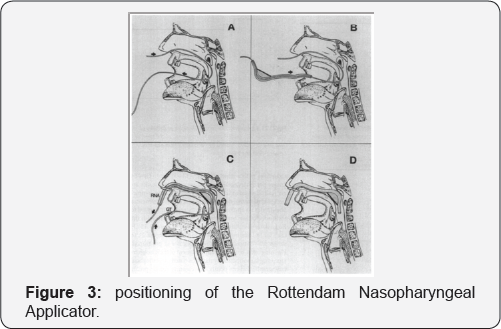
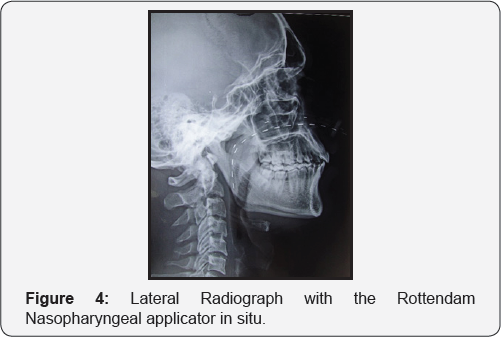
The Rotterdam Nasopharyngeal Applicator (RNA) (Figure 2) was designed by Peter Levendag [7]. It has an outer diameter of 5.5mm and an inner diameter of 3.5mm; and is made of silicone and thus is flexible and closely conforms to the curvature of the naso pharynx. After decongestion and topical anesthesia (R/Concaine hydrochloride 7%) of the nasal mucosa and naso pharynx, guide tubes (outer diameter 2 mm) are introduced through the nose and exit through the mouth. The Rotterdam Naso pharynx Applicator (RNA) is guided intra orally over the guide tubes (GT) by pulling on the nasal part of the guide tubes. The applicator is finally placed in situ into the naso pharynx and nose. To facilitate positioning of the applicator into the naso pharynx, gently pushing the oral parts of the guide tubes intra orally using some standard type of forceps can sometimes be of additional help. By using a silicone flange, the Rotterdam Nasopharynx Applicator is secured in the correct position for the duration of the treatment (Figure 3) [25]. Figure 4 shows a lateral radiograph with applicators in situ.

Treatment Planning
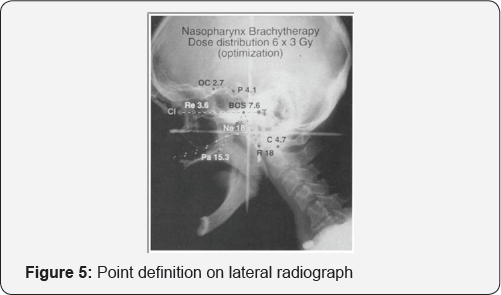
Dosimetric calculations can be done using orthogonal films or Computerized Tomography (CT) scans. Tumor points and normal-tissue points, which will be used for dose optimization, are indicated on the lateral radiograph (Figure 5), and then the points are transferred to the antero posterior (AP) radiograph at a prescribed distance from the midline. Points are selected which should receive the reference dose. Usually, these are the naso pharynx points (Na) and the Rouviere node (R) [7]. The dose distribution is optimized such that these points receive the prescribed dose. A check of doses received is made to the normal tissue points namely, pituitary (Pi), cord (C), retina (Re), optic chiasma (OC) and palate (Pa).
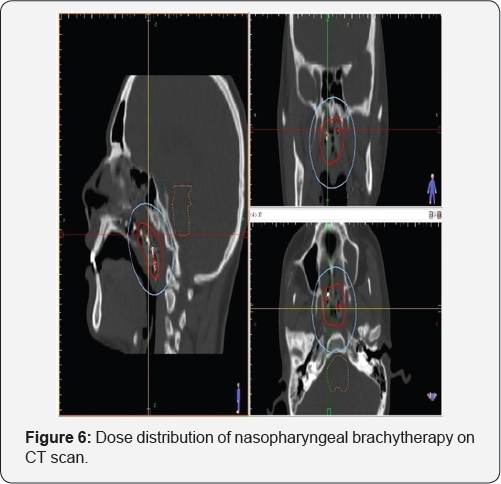
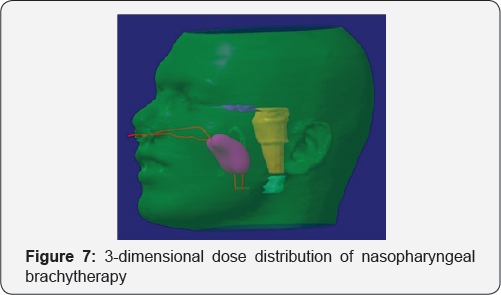
Alternatively, dose can be prescribed to CT scan [with or without MRI (Magnetic Resonance Imaging) fusion] based volume. The Clinical Target Volume (CTV) is defined based on the information from endoscopy, CT scan and MRI. However, as it is difficult to delineate tumor extension and to define the depth of the GTV precisely, a large safety margin is recommended around the gross disease. On CT slices, the dose is usually prescribed to an isodose covering the surface of the underlying bone, which is situated at 5-10 mm from the mucosal surface. Dose distribution is then optimized when possible so that the reference iso dose follows the bone surface [12]. Dose distribution on CT scan images and 3-dimensional view in a patient who received brachytherapy boost is shown in (Figure 6) and (Figure 7) respectively.
Dose
According to Levendag et al [7], the recommended dose for patients with a T1 tumor is 60 Gy of external beam radiotherapy in 30 fractions and 6 weeks, followed, after a rest period of 1-2 weeks, by HDR brachy therapy, 18 Gy in 6 fractions over 3 days (2 fractions per day, 6 h apart), and for T2-4 tumors (if performed for T3-4), 70 Gy of external beam radiotherapy followed, after a rest period of 1-2 weeks, by HDR brachy therapy, 12 Gy in 4 fractions. The same recommendations have been reinforced by the GEC-ESTRO and ABS working groups [7,12,24]. When HDR is given twice a day, the ABS recommends that the interval between the HDR fractions be as long as possible, with the minimum being 6 hours [24].
Toxicity
Naso pharyngeal brachy therapy has been reported to have an acceptable tolerance across most studies. The most common complication is mucosal ulceration, causing synechiae and crusting. Levendag et al. [7] noted that three patients experienced significant grade 3latetoxicity, ie, synechiae of the nasal mucosal linings because of the addition of Brachy therapy [7]. This complication, however, was easily correctable with minor surgery in all cases. To minimize synechiae, ABS recommends that paraffin-impregnated gauze should be inserted into the nasal cavity after removal of the applicator. It should remain in situ for approximately 1week [24].
Owing to higher toxicity rates when doses exceeded 75Gy (cumulative ExtRT and Brachy therapy), Chang et al. [15] suggested that an optimal dose for Nasopharyngeal cancers is between 72.5 to 75 Gy. He encountered toxicities such as perforation of the palate and/or sphenoid sinus floor. This pattern of toxicity contrasts markedly from other studies. A number of findings may explain this. First, Chang et al. [15] prescribed the dose at 2 cm from the source axis, which thereby resulted in much higher doses at the surface of the applicator than is the case with Levendag et al. [7], who prescribed to a fixed anatomic (NP) point, which is generally closer (1 cm) to the source axis. Most other studies have prescribed at 1 cms off axis and thus have had lesser toxicities than that observed by Chang et al. [15] Also, a system of routinely optimizing dose distributions with respect to a large number of dose points that depict critical surrounding normal tissues will reduce the likelihood of toxicity. A smaller fraction size will also reduce the toxicities.
Concomitant Chemotherapy
Concomitant chemotherapy during brachy therapy is not recommended by GEC-ESTRO for the treatment of primary tumors outside clinical trials, unless relevant clinical data are available [12].
Limitation
Nasopharyngeal brachy therapy has failed to improve outcome when used as a boost modality for advanced primary tumors. Stereotactic therapy boost serves as a better tool for dose escalation in such patients.
Levendag et al. [7], in another study divided the patients on the basis of tumor response; i.e. LD (T1,2a) with Limited Disease, i.e. disease confined to naso pharynx), LRD (T2b, with Limited Residual Disease), ERD (T2b, with Extensive Residual Disease), or patients initially diagnosed with T3,4 tumors [26]. Dose distributions for endocavitary brachy therapy were compared to parallel-opposed three-dimensional conformal radiation therapy, intensity modulated radiation therapy (IMRT) and stereotactic radiotherapy (SRT).
For T2b ERD and T3-4 tumors, brachy therapy failed to produce adequate coverage sparing normal tissue doses. Whereas, stereotactic therapy and IMRT based plans produced better conformal target coverage and optimal sparing of organs at risk (OARs).
Conclusion
Nasopharyngeal brachy therapy is an easy to perform procedure and is an effective tool to escalate the dose and hence facilitate better outcomes in early stage nasopharyngeal tumors.
References
- Schantz SP, Harrison LB, Forastiere AA. Tumors of the nasal cavity and paranasal sinuses, nasopharynx, oral cavity, and oropharynx. In: Cancer: principles and practice of oncology. (6th edn). Edited by DeVita VT, Hellman S, Rosenberg SA, (Eds.), Lippincott, Philadelphia,USA, 2001: 824-832.
- Al-Sarraf M, LeBlanc M, Shanker PG, Giri PG, Fu KK, et al. (1998) Chemo radio therapy versus radiotherapy in patients with advanced nasopharyngeal cancer: Phase III randomized Intergroup study 0099. J Clin Oncol 16(4): 1310-1317.
- Kwong DL, Sham JS, Choy D (1994) The effect of loco-regional control on distant metastatic dissemination in carcinoma of the naso pharynx: an analysis of 1301 patients. Int J Radiat Oncol Biol Phys 30: 10291036.
- Yan JH, Qin DX, Hu YH, Cai WM, Xu GH, et al. (1989) Management of local residual primary lesion of nasopharyngeal carcinoma: Are higher doses beneficial? Int J Radiat Oncol Biol Phys 16: 1465-1469.
- Lee AW, Poon YF, Foo W, Law SC, Cheung FK, et al. (1992) Retrospective analysis of 5037 patients with nasopharyngeal carcinoma treated during 1967-1985: Overall survival and patterns of failure. Int J Radiat Oncol Biol Phys 23: 261-270.
- Marks JE, Bedwinek JM, Lee F (1982) Dose response analysis for nasopharyngeal carcinoma. Cancer 50(6): 1042-1050.
- Levendag PC, Schmitz PI, Jansen PP, Eijkenboom WM, Visser AG, et al. (1998) Fractionated high dose rate brachy therapy in primary carcinoma of the naso pharynx. J ClinOncol 16(6): 2213-2220.
- Perez CA, Devineni VR, Marcial-Vega V (1992) Carcinoma of the naso pharynx: Factors affecting prognosis. Int J Radiat Oncol Biol Phys 23(2): 271-280.
- Pharynx. In: AJCC Cancer Staging Manual. (7th edn) Edge SB, Byrd DR, Compton CC, (Eds.), New York, NY: Springer, 2010 p. 41-56.
- Marks LW, Yorke ED, Jackson A, Ten Haken RK, Constine LS, et al. (2010) Use of normal tissue control probability models in the clinic. Int J Radiation Oncology Biol Phys 76(3): 10-19.
- Pierquin J, Richard G, Pierquin B (1952) Radium therapy of adenoid growths of the naso pharynx; criticism of Crowe's method; our personal method. Journal de radiologie, d'electrologie & archives d'electricitemedicale. 33(7-8): 404-407.
- Mazeron JJ, Ardiet JM, HaieMeder C, Kovacs G, Levendag P, et al. (2009) GEC-ESTRO recommendations for brachytherapy for head and neck squamous cell carcinomas. Radiotherapy and Oncology 91: 150-156.
- Teo PML, Leung SF, Fowler J (2000) Improved local control of early T-stage nasopharyngeal carcinoma-a tale of two hospitals. Radio ther Oncol 57(2): 155-166.
- Leung TW, Wong VYW, Sze W-K (2008) High dose rate intra cavitary brachy therapy boost for early T stage nasopharyngeal carcinoma. Int J Radiat Oncol Biol Phys 70(2): 361-367.
- Chang JTC, See LC, Tang SG (1996) The role of brachy therapy in early-stage nasopharyngeal carcinoma. Int J Radiat Oncol Biol Phys 36(5):1019-1024.
- Yeo R, Fong KW, Hee SW (2009) Brachy therapy boost for t1/t2 nasopharyngeal carcinoma. Head & Neck 31(12): 1610-1618.
- Malde R, Agarwal JP, Laskar SG (2005) High dose rate brachy therapy boost for primary nasopharyngeal carcinoma: preliminary results of an ongoing prospective study. Bull Cancer 92 (7-8): 45-50.
- Lu J, Shakespeare T, Tan L, Goh BC, Cooper JS (2004) Adjuvant fractionated high dose rate intra cavitary brachy therapy after external beam radiotherapy in T1 and T2 nasopharyngeal carcinoma. Head Neck 26(5): 389-395.
- Ozyar E, Yildz F, Akyol FH, Atahan IL (2010) Adjuvant high-dose-rate brachy therapy after external beam radiotherapy in nasopharyngeal carcinoma. International Journal of Radiation 52(1):101-108.
- Rosenblatt E, Wahab MA, El- Gantiry M, Elattar I, Bourque JM, et al. (2014) Brachy therapy boost in loco-regionally advanced nasopharyngeal carcinoma: a prospective randomized trial of the International Atomic Energy Agency. Radiat Oncol 9: 67.
- Levendag PC, Keskin-Cambay F, Teguh DN, El-Gantiry M, Wildeman MA, et al. (2011) Brachytherapy, a Highly Focused Technique for Applying High Booster Doses of Radiation. Can it be of Value in Reducing the Local Relapse Rate in Advanced Cancer of the Nasopharynx? Int J Radiat Oncol Biol Phys 81(2): 510-511.
- Jean Jacques Mazeron, Erik Van Limbergen. Nasopharynx.
- Ren YF, Gao YH, Cao XP, Ye WJ, Teh BS (2010) 3D-CT implanted interstitial brachy therapy for T2b nasopharyngeal carcinoma. Radiat Oncol 5: 113.
- Nag S, Cano ER, Demaned DJ (2001) The American Brachy therapy Society recommendations for high dose rate brachy therapy for hean- and-neck carcinoma. Int J Radiat Oncol Biol Phys 50: 1190-1198.
- Joslin CAF, Flynn A, Hall EJ (2001) Principles and Practice of Brachy therapy using after loading systems. Great Britian: Arnold publishers.
- Levendag PC, Lagerwaard FJ, Pan CD, Noever I, Nimwegen AV, et al. (2002) High-dose, high-precision treatment options for boosting cancer of the nasopharynx. Radiotherapy and Oncology 63: 67-74.






























