Complications in Postoperative Evolution After Resection for Rectal Cancer and The Time of Postoperative Chemotherapy
Lorant Kiss*, Roland Kiss and Castilia Dobra
Emergency Academic Hospital Sibiu, University Lucian Blaga Sibiu, Romania
Submission: April 12, 2019; Published: May 14, 2019
*Corresponding Address:Lorant Kiss, Emergency Academic Hospital Sibiu, Ist Surgical Clinic, University Lucian Blaga Sibiu, Romania
How to cite this article:Lorant K, Roland K, Castilia D. Complications in Postoperative Evolution After Resection for Rectal Cancer and The Time of Postoperative Chemotherapy. Canc Therapy & Oncol Int J. 2019; 13(4): 555868. DOI:10.19080/CTOIJ.2019.13.555868
Abstract
Background: Same studies found that postoperative complications after rectal cancer resection influence the timing of chemotherapy. The relevance of severity of postoperative complications after rectal cancer resection with curative intent on survival/recurrence rates is a matter of controversy.
The aim: To investigate the import of postoperative complications on survival after curative resection for rectal cancer. The second objective was to show the risk factors for delays in chemotherapy and the consequence of delayed therapy on long –term survival.
Material and Methods: We investigated 177 patients with curative resection for rectal cancer between 2005-2015, operated in Ist surgical clinic. This study was a retrospective review with univariate analysis to identify risk factors for delayed chemotherapy. In the present study were included the patient’s category who underwent resection with primary curative intent were excluded all other cases with proctectomy for rectal cancer.
Results: Delays in chemotherapy > 8 weeks after resection were associated with postoperative complications worse local and distant recurrences rates and worse overall survival were observed in patients with chemotherapy > 8 weeks. In present study there was no significant difference between patients without complications, minor complications and major complications in terms of overall disease –free survival. Our study is retrospective and was performed at a single institution, and this is a possible limitation of this study
Conclusion: After rectal resection with curative intent, the severity of postoperative complications according to Clavien classification system does not have significant effect of overall and disease – free survival. Our results shown that the presence of postoperative complications are risk factors for delayed chemotherapy
Introduction
In rectal cancer surgical resection is accepted to be the most effective treatment for localized malignant tumors but with considerable postoperative morbidity and mortality [1-3]. Colorectal cancer is the third most common causes of cancer –related mortality [4]. The postoperative complication rates after rectal cancer resection ranging from 18% to 38% [5-10]. In the recent study Hendren and colleagues [5] showed as association between postoperative complications and omission in chemotherapy in stage III colon cancer patients. In this study the authors demonstrated that patients with complications have a significantly delay in chemotherapy > 60 days. Multiple patients’ factors have been found in same studies to contribute to delays chemotherapy: older age, the preoperative comorbidities [6-11]. Multiple studies have demonstrated a negative effect in a delayed chemotherapy, but in the current literature, risk factors for delayer postoperative therapy in patients with colorectalcancer have not been were described. Same publications showed am improved survival in patients who have adjuvant chemotherapy within 8 weeks after resection [12-14,15]. In others studies a decrease in overall survival were observed in delayed postoperative chemotherapy (>12 weeks) [10,11,16,17].
Postoperative complications after resection for rectal cancer are associated with higher reoperation rate, prolonged hospital stay and with lower oncologic long-term outcome [18-20]. Decision recording adjuvant therapy after potentially curative surgery most be based on information about resected specimen and p TNM stabilization, together with pathological tumor characteristics. The aim of present study was to evaluate the effect of postoperative complications on survival in patients with rectal cancer and establish were there is a difference between minor and major complications following resection.
Patients and Methods
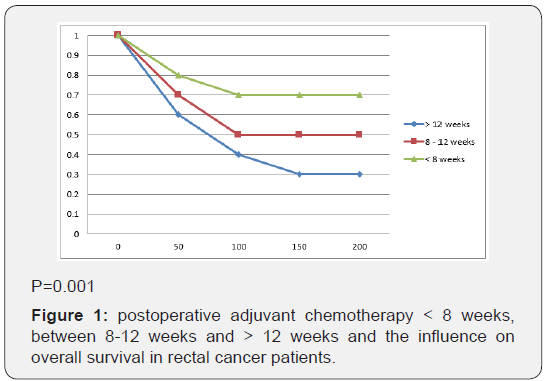
We reviewed patients with rectal cancer who underwent rectal resection between 2000-2015 in our surgical clinic. Rectal carcinoma was defined as tumors located between aval verge and 16 cm above it. The lower third rectum carcinoma were located between the aval verge and 8 cm, above between 12-16 cm upper third [15,21]. All patients of this study underwent curative resection (R0 according to the nice AJCC [22,23], with macroscopically or microscopically no residual tumor. All surgical operations were open surgery, including Dixon resection, low anterior resection, abdominao - perineal excision and occasionally local excision in special situation (Tis, or contraindications for general anaesthesis in T1 tumors). In all tumors located in low or midl resection were performed a total mesorecatl excision (TME) and hight ligation of inferior mesenteric artery. In all tumors located at the upper rectal third we performed a partial mesorectal excision with a distal resection margin of 5 cm surrounded by mesorectum. In very low tumors and in presence of splincher invasion of the tumors the abdomino-perineal excision were indicated. Indications for chemotherapy was defined as CT image with lymph node involvement on histopathologicaly lymph node involvement, or the presence of tumor distant metastases. We analyzed the timing to postoperative chemotherapy, and we observed significant difference between patients with therapy. Delayer past 8 weeks (Figure 1).
In the statistical analysis were included: sex, age, tumor diameter , macroscopic tumor type, localization in rectal lumen, nice stage, pT, pN stage, differenced with groding, the presence of lymph node invasion, blood vessel invasion, in special vein invasion, neo- and adjuvant therapy, incidence of anastomotic leakage and the complications grad according to Clavien. In this study we recorded the thirty –day postoperative complications: all surgical infections with or without worent dehiscence, pulmonary embolia, reintervention, respiratory failure, acute renal failure, urinary tract infections, coma, strake delirium, cardiac arrest, myocardial infraction, deep vein thrombosis, postoperative bluding, blood transfusion, sepsis data for 30-ty day readmission, reoperation within 30 days, mortality.
Results
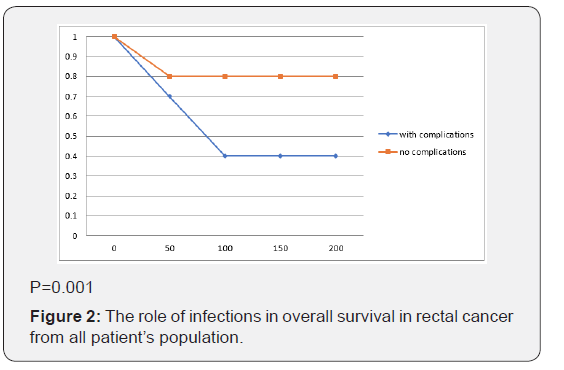
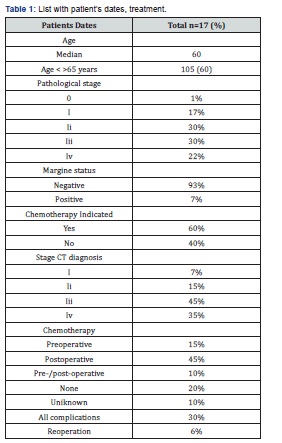
We analyzed 177 patients who underwent proctectomy for rectal cancer between 2000-2015. In table 1 there is shown the list with patient’s dates, treatment. Overall 78% of the patients presented with stage III, IV locally advanced rectal cancer andwere considering for chemotherapy. The overall complications rate was 30%. No difference was observed in complication rate between neoadjuvant chemotherapy and postoperative chemotherapy (31%). In 51% of complication surgical infections was the most common complications in patients with chemotherapy < 8 weeks and in patients reclived chemotherapy after 8 weeks, the infections rates were 54% of complications. In patients with delayed chemotherapy the most frequent infection was the deep organ surgical infection. In Figure 2 postoperative complications were associated with overall survival in all patients (177 cases). P= 0.012 In patients treated with chemotherapy and having postoperative complications, the medin survival was 120 months (Figure 3).


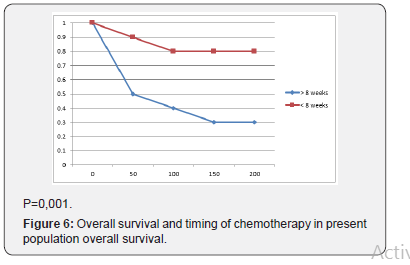
P= 0.014 Curative resection was performed in 67% op the resections. Minor complications (Clavien I+II) were identified in 20% of the patients with 13 % of major complications (Clavien III + IV), within the first no days post surgery 3,5 % patients died (Clavien V). In 8,5 % of the resected patients we recorded anastomotic dehiscence (Figure 4). Univariate Cox regression analysis indicated age, tumors factors, adjuvant therapy, as significant factors to overall survival and disease-free survival. The mortality risk for patients is different in relation with age, 45% in patients older than 80 years, and in youngers than 65 years 72%. The incidence of local recurrence and postoperative chemotherapy at < 8 weeks, >8 weeks following surgery is shown in figure 5. At patients that received adjuvant chemotherapy on time the median time to local recurrence was196 months in comparation with 141 months in patients with delayed chemotherapy. The overall survival in patients having chemotherapy <8 wees and > 8 weeks shown a lower survival in patients with delayed chemotherapy: 76 months (Figure 6). These data shown worse disease-free and overall survival in patients with delayed chemotherapy >8 weeks.

Discussion
Some studies shown that postoperative complications following rectal cancer surgery may not only affect short – term survival but also long-term evolution [18,20,24,25]. Law et al. [18] & Khuri et al. [19], shown that the postoperative complications after colorectal surgery are one independent factor associated with survival. Nespoli et al described the influence of postoperative infections on long-term survival in resected colorectal cancer patients [24]. The aim of present study was to analyze the postoperative complications and the timing of chemotherapy in resected rectal carcinoma. We see a higher incidence of postoperative complications in patients with delayed chemotherapy >8 weeks after curative resection of rectal cancer. The most frequent complications after colorectal surgery in considered anastomotic leakage and the influence on long –term survival is discussed controversially [25,26]. Tumorfree survival following postoperative anastomotic leakage is low, this result was reported by Kube et al. [25]. After beginning of the anastomotic leakage, the patients developed MSOF syndrome, cardio-pulmonary and renal complications and in some cases thromboembolic accidents. The study published by Kube etal. [25] suggest that presence of anastomotic leakage after colon resection have severe complications caused by diffuse peritonitis, leading to SIRS with inflammatory cascade, release of proinflammatory cytokines and vascular growth factors and may promote tumor growth and dissemination [27,28].
The clinical manifestations and evolution in anastomotic leakage after rectal surgery is different in comparations with colon resection, because such severe sepsis is at times not seen in patients with leakage after rectal resection and the most frequent response is the localized inflammatory response [20,28-30]. Law et al. [18] demonstrated on influence of tumorspecific factors on survival rates, but not anastomotic lea on 5-year cancer-specific survival after rectal cancer resection. It has been were established in the same studies that delaying chemotherapy in postoperative time, more than 8 to 12 weeks has worse overall survival after colorectal cancer resection [10,14]. Timmress et al. [31] reported on long-term survival after surgery that the need for intervine care therapy reflecting the presence of postoperative complications, bad a negative effect on long-term survival. Timmress et al. identified that the need for dialysis and APACHE II score, strongly influence by sever sepsis and are negative predicting factors
In concordance with these results [31] our results the overall survival may be influence by the pathophysia logical sequela of sepsis in patients with Clavien III/IV complications rates. Of patients with postoperative complications those with delayed chemotherapy bad worse overall survival showing that delay of this therapy affects longer-term survival. Our results in concordance with other publications say that complications did not significantly affect overall survival at patients who received chemotherapy, but postoperative complications indirectly influence outcomes by leading to delays in chemotherapy. One limitation of our study is that represent a cohort study from a single center, another limitation could be the retrospective grading of complications. Our overall and disease – free survival rates compare were with others publishing studies [18, 20] reflecting a certain generalizability of our data. From our study and evaluation of the preoperative comorbidities, we revalue to show the influence of survival after resection. In the other board Ouellette et al. [32] reported that comorbidities have on real import the survival of patients with colorectal cancer, but in other authors research the presence of comorbidities was not influenced significantly the survival [33].
Conclusion
Our study demonstrated an association between delayed chemotherapy > 8 weeks and postoperative complications at patients with curative rectal cancer and the influence on longterm / evolution in these cases
References
- World Health Orsa Mization (2012) Cancer Fact sheet No 297 2012.
- Alves A, Panis Y, Mathieu P, Mantion G, Kwiatkowski F, et al. (2005) Association Francaise de Chirurgie- Postoperative mortality and morbidity in French patients undergoing colorectal surgery: results of a prospective multicenter study. Arch Surg 140(3): 278-284.
- Bokey EL, Chapuis PH, Fung C, Hughes WJ, Koorey SG, et al. (1995) Postoperative morbidity and mortality following resection of the colon and rectum for cancer. Dis Colon Rectum 38(5): 480- 486.
- Siegel R, Naishadham D, Jemal A (2012) Cancer statistics 2012. CA Cancer J Clin 62(1): 10-29.
- Hendren S, Birkmeyer JD, Yin H, Banerjee M, Sonnenday C, et al. (2010) Surgical complication are associated with ornissin of chemotherapy for stage III colorectal cancer- Dis. Colon Rectum 53(12): 1587-1593.
- Cohen ME, Bilimoria KY, Ko CY, Hall BL, et al. (2009) Development of an American College of Surgeons National Surgery Quality Improvement program: morbidity and mortality risk calculator for colorectal surgery- J Ami Coll Surg 208(6): 1009-1016.
- Winget M, Hossain S, Yasui Y, Scarfe A (2010) Characteristics of patients with stage III Colon adenocarcinoma who fail to receive guideline-recommended treatment. Cancer 116(20): 4849-4856.
- Ahmed S, Ahmad I, Zhu T, Arnold FP, Faiz Anan G, et al. (2010) – Early discontinuation but not the timing of adjuvant therapy affects survival of patients with high – risk colorectal cancer: a population-based study. Dis Colon Rectum 53(10): 1432-1438.
- Cree M, Tonita J, Turner D, Nugent Z, Alvi R, et al. (2009) – Comparison of treatment received versus long –standing guidleines for stage III colon cancer and stage II/ III rectal cancer patients – Clin. Colorectal Cancer 8(3): 141-145.
- Lima IS, Yasui Y, Scarfe A, Winget M, et al. (2011) Association between receipt and timing of adjuvant chemotherapy and survival for patients with stage III colon cancer in Alberta Canada- Cancer 117(6): 3833-3840.
- Cheung WY, Neville BA, Earle CC (2009) Etiology of delays in the initiation of adjuvant chemotherapy and their import on outcomes for stage II and III rectal cancer – Dis. Colon Rectum 52(6): 1054-1064.
- Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, et al. (2009) The Clavien- Dindo classification of surgical complications: five years’ experience. Ann Surg 250(2): 187-196.
- Hershman D, Hall MJ, Wang X, Jacobson JS, McBride R, et al. (2006) Timing of adjuvant chemotherapy initiation after surgery for stage III colon cancer. Cancer 107(11): 2581-2588.
- Des Guetz G, Nicolas P, Perret GY, Morere JF, Uzzan B, et al. (2010)– Does delaying adjuvant chemotherapy after curative surgery for colorectal cancer impair – survival? A meta-analysis- Eur. J. Cancer 46(6): 1049-1057.
- Mrak K, Eberl T, Laske A, Jagoditsch M, Fritz J, et al. (2013) Impact of postoperative complications on long-term survival after resection for rectal cancer- Dis. Colon Rectum 56(1): 20-28.
- Tevis SE, Kohlnhofer BM, Stringfield S, Foley EF, Harms BA, et al. (2013) Postoperative complications in patients with rectal cancer are associated with delays in chemotherapy that lead to worse disease-free and overall survival. Dis. Colon Rectum 56(12): 1339-1348.
- Law WL, Poon JT, Fan JK, Lo OS, et al. (2012) Survival following laparoscopic versus open resection for colorectal cancer. Int J Colorectal Dis 27(8): 1077-1085.
- Law WL, Choi HK, Lee YM, Ho JW (2007) The import of postoperative complications on long- term outcomes following curative-resection for colorectal cancer. Ann Surg Oncol 14(9): 2559-2566.
- Khuri SF, Henderson WG, DePalma RG, Mosca C, Healey NA, et al. (2005) Determinants of long-term survival after major surgery and the adverse effect of postoperative complications. Ann Surg 242(3): 326-341.
- Ptok H, Marusch F, Meyer F, Schubert D, Gastinger I, et al. (2007) Import of anastomotic leakage on oncological outcome after rectal cancer resection. Br J Surg 94(12): 1548-1554.
- Fielding LP, Arsenault PA, Chapuis PH, Dent O, Gathright B, et al. (1991) Clinicopathological staging for colorectal cancer: an International Documentation System (IDS) and an International Comprehensive Anatomical Terminology (ICAT). J Gastroenteral Hepatol 6(4): 325-344.
- Sobinl H, Wittekin DC, UICC (2002) TNM classification of malignant tumors. Wiley, New York, USA.
- Greenfly, Balch CM (2002) AJCC cancer staging manual- Ny Springer, New York, USA.
- Nespoli A, Giantotti L, Totism (2004) Correlation between postoperative infections and long-term survival after colorectal resection for cancer. Tumors 90: 485-490.
- Kube R, Ranowski D (2010) Anastomotic leakage of ten colon cancer surgery. Eur J Surg Oncol 36: 120-124.
- Law WL, Chu KW (2004) Anterior resection for rectal cancer with mesorectal excision: a prospective evaluation of 622 patients. Ann Surg 240(2): 260–268.
- O’Dwyer PZ, Marti Ewjr (1989) Viable intraabdominal cells and local / regional tumor grow in experimental colon cancer. Ann R Call Surg Engl 71(1): 54-56.
- Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evoluation in cohort of 4336 patients of Ann Surg 240(2): 205-213.
- Berger AC, Sigurdson ER, LeVoyer T, Hanlon A, Mayer RJ, et al. (2005) l – Colon cancer survival is associated with decreasing rations of metastatic to examined lymph nodes. J Clin Oncol 23: 8706-8712.
- Eberl T, Jagoditsch M, Klingler A, Tschmelitsch J (2008) Risk factors for anastomotic leakage aften resection for rectal cancer. Ann J Surg 196: 592 - 598.
- Mansour T A Sharabiani, Francesca Fiorentino, Gianni D Angelini, Nishith N Patel, et al. (2011) Long-term survival after surgical intensive care avit admission: fifty percent die Within 10 years. Ann Surg 253: 151-157.
- Ouellette JR, Small DG, Termuhlen PM (2004) Evaluation of Charlson – Age Commorbidity index as predictor of morbidity and mortality in patients with colorectal carcinoma. J Gastrointest Surg 8(8): 1061-1067.
- Fernández-Ruiz M, Guerra-Vales JM, Colina-Ruizdelgado F (2009) Comorbidity negatively influences prognosis in patients with extra- hepatic cholangiocarcinoma. World J Gastroenterol 15(42): 5279-5286.






























