Can Multi-Detector Computed Tomography (MDCT) Help in Differentiation of Neoplastic Parotid Lesions?
Enas Ahmed Abdel-Gawad MD, Mohamed Aboul-fotouh E Mourad MD* and Mona Abdel-Kafy A Wahab
Radiology Department, Minia University, Egypt
Submission: March 29, 2017; Published: May 04, 2017
*Corresponding author: Mohamed Aboul-fotouh E Mourad, Radiology Department, Minia University, Minia, Egypt, Email: Mohamed.mourad@mu.edu.eg
How to cite this article: Enas A A-G MD, Mohamed A-f E M M, Mona Abdel-K A W. Can Multi-Detector Computed Tomography (MDCT) Help in Differentiation of Neoplastic Parotid Lesions?. Curr Trends Clin Med Imaging. 2017; 1(4): 555572. DOI: 10.19080/CTCMI.2017.01.555572
Abstract
Background: Parotid lesions are uncommon issue in clinical practice with wide differential diagnosis based upon multiple pathological varieties and limited clinical values. Computed tomography (CT) try to solve this issue as possible.
Objectives: To evaluate the diagnostic reliability of multi-detector computed tomography (MDCT) in characterization of benign and malignant parotid lesions using a systematic approach in comparison to final pathological data.
Patients & Methods: A retrospective study includes136 patients diagnosed pathologically as parotid tumors underwent multi-detector computerized tomography (MDCT) examination using 64-row multi-detector computerized tomography (CT) scanner. All the images were evaluated for certain multi-detector computerized tomography (MDCT) criteria in a systematic manner.
Results: Our study revealed primary malignant tumors in 88 patients, secondary metastatic lesions in 3 patients and benign tumors in 45 patients. Five multi-detector computerized tomography (MDCT) criteria were statistically significant in differentiating between benign and malignant parotid lesions, they were; lesion margin (p=0.001), location (p=0.05), enhancement pattern (p=0.001), central necrosis (p=0.001) and para-pharyngeal space extension(p=0.001).
Conclusion: Multi-detector computerized tomography (MDCT) can be valuable in differentiating between benign and malignant parotid lesions and leading the management strategy aiming for best surgical outcome.
Keywords: Parotid; MDCT; Benign; Malignant; Tumors
Key Points
a) Parotid lesions show unsatisfactory discriminating imaging criteria to guide the surgical approach.
b) Rapid advances of MDCT techniques allow optimum parotid lesions characterization.
c) Imaging is forming a crucial triad with clinical data and surgical approach.
Introduction
Parotid lesions are uncommon in clinical practice with unsatisfactory discriminating imaging criteria to guide the surgical approach. They account for 1% of head and neck tumors and about 80-85% of parotid neoplasm are benign, 15-20% of parotid lesions are malignant [1,2]. The clinical challenge of parotid lesions is that, they grow slowly over years before the patients seek medical consultation, only few cases may have represented by facial nerve palsy. Also the fine needle aspiration results may be inaccurate and non-conclusive in many cases [3]. The availability and low cost of computed tomography (CT) in conjunction with rapid advance of CT technology including dual source, prospective triggering and high pitch helical acquisition should be applied for better characterization of various parotid lesions. All benign and malignant parotid lesion are indicated for surgery and our rule is to get well preoperative differentiation between benign and malignant lesions and leading the surgical approach to be as conservative as possible especially for facial nerve and lymph node dissection as well as improve the postoperative management strategy [4-6].
Patients and Methods
Patients
This retrospective study was approved by the ethics committee of our institution during the period between May 2014 and February 2016. It included 136 patients who were pathologically diagnosed by biopsy or fine needle aspiration biopsy (FNAB) as parotid neoplasm.
MDCT technique
Multi-detector CT examination of the parotid region was performed for all the 136 patients included in this study using 64-row multi-detector CT scanner (Aquilion 64; Toshiba Medical Systems Corporation, Otawara, Japan), which is a 64x0.5mm collimation scanner with a gantry rotation speed of 400ms/ rotation. The examination was performed with the patient supine in quite respiration. A pad is placed beneath the patient scapula produces mild hyperextension of the neck and provide consistent images perpendicular to long axis of the neck, that minimizing dental artifacts. The scanning range will be individually adapted and included from the external auditory canal to the level of the hyoid bone. Contrast material is injected through an 18-20 gauge catheter in to the ante-cubital vein with flow rate 4ml/ sec. The contrast used according to body weight 1.5ml/kg. A non ionic contrast material (Ultravist 370) was used. A timing bolus tracking technique was employed. The acquisition parameters were 120kvp, 350mAs, a helical pitch of 0.983: 1,7.5 second scan time, 7.5 second total exposure time, 1.25mm helical slice thickness, and 1.2 5mm reconstruction interval with a large FOV.
Post-processing and image reconstructions
The axial source images with a 1.25mm slice were transferred to an external work station (Vitrea) to perform multi-planar reformatted images (MPR) images in coronal and sagittal planes with a section thickness of 3-5mm.
Image analysis
The MDCT images were interpreted by two expert radiologists in head and neck imaging 5 and 10 years of experience who were blinded to results of each other. All the MDCT images were interpreted for the MDCT criteria of the parotid lesions that included the following items; size, margin, location, enhancement pattern, multiplicity, central necrosis, para pharyngeal fat extension and calcifications as well as associated lymph node enlargement.
Statistical analysis
Data entry was done by SPSS version 11. Frequency distribution, descriptive statistics and correlation analysis were done using Chi2 and Fisher exact tests for qualitative data. The probability (p value) of less than 0.05 is used as a cut off point for all significant tests.
Pathological examination
Biopsy was performed for 79 lesions, fine needle aspiration biopsy (FNAB) for 57 lesions and 96 patients had surgical excision. Histo-pathologic examination was performed for the entire biopsied specimen.
Results
The patient sages of the studied group ranged from 5 to 77 years old with a mean age of 44.4± 22.2. They were 46 males (57.5%) and 34 females (42.5%). Our biased study shows higher rate of malignant parotid lesions with lower incidence of benign tumors. Table 1 listed the pathologic types of the parotid lesions according to final pathological examination. The benign lesions were detected in 45 (33.1%) out of 136 patients. The most common benign lesion was pleomorphic adenoma which represents 55.6% of all benign lesions. The malignant lesions were further classified into primary and metastatic lesions. The primary lesions were detected in 88(64.7%) out of 136 patients, the most common primary malignant lesion was mucoepidermoid carcinoma which represents 28.4%. The metastatic lesions were detected in 3(2.2%) out of 136 patients, 2 of them were metastasis from malignant scalp lesion and 1 of them was metastasis from nasopharyngeal squamous cell carcinoma.
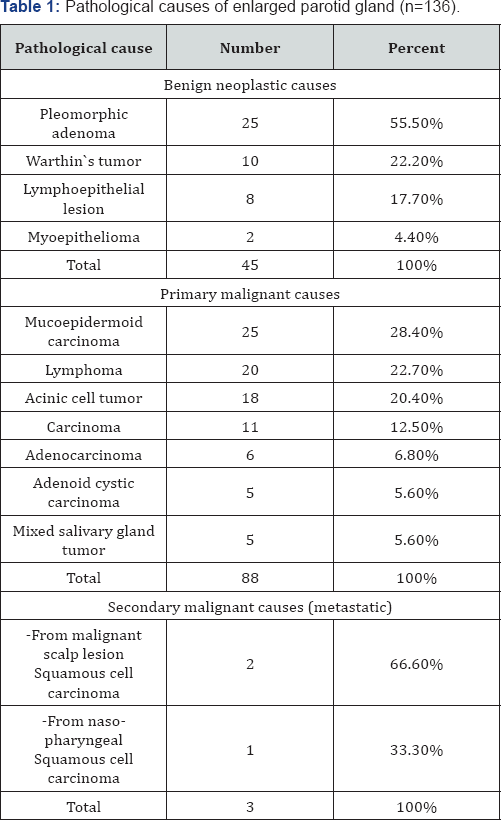
The examined MDCT criteria for benign and malignant parotid lesions were shown in Table 2. The size criteria were determined by measuring the longest dimension of the lesion, consider, the lesions were classified accordingly to small lesions if their size is less than 2 cm, intermediate size if they were from 2-4cm and large lesions if their sizes were more than 4cm. This classification was based on National Comprehensive Cancer Network [7]. The margins of enlarged parotid gland in this study were either sharp well-defined that can be delineated along its all margins or ill-defined which means ill definition of one point at least. Thirty-nine out of 45 benign lesions shows well-defined margins, while 54 out of 91 of malignant lesions show ill-defined margins. The parotid lesions were located either in the superficial lobe, deep lobe or both lobes. Most parotid lesions that involved both lobes were malignant which found in 44(50%) and most of the parotid lesions that involve the superficial lobe were benign which found in 27 patients (60%).
Most of benign lesions were single and detected in 30 patients and most of malignant lesions were multiple and detected in 59 patients. The enhancement pattern of the lesions was varying from no enhancement, homogenous enhancement, and heterogeneous enhancement to peripheral or marginal enhancement. It was found that the most common enhancement pattern in the benign lesions were homogenous pattern, as it was determined in 29 out of 45 patients. Most of the malignant parotid lesions exhibited heterogeneous pattern, as it was determined in 57 out of 91 patients [8-10].
Central necrosis was present in 8 benign lesions and in 33 malignant lesions and was highly significant. Another significant MDCT criterion was the extension of the parotid lesions to the para-pharyngeal fat space which was determined in 48 out of 91 malignant lesions and in 20 out of 45 benign lesions. Calcifications were detected in 4 benign lesions and in 12 malignant lesions. The lastly evaluated MDCT criterion was the associated lymph node enlargements which were detected in association with 52malignant lesions and 9 benign lesions.
Each one of the MDCT criteria was statistically tested for its reliability for diagnosis and for differentiating between benign and malignant causes using the pathological diagnosis as a gold standard. In this study, the most reliable MDCT criteria that show statistical significance for diagnosis of parotid gland lesions (significant p values) were the margin (0.001), central necrosis (0.001), enhancement pattern (0.001) and extra-parotid extension (0.001) as well as lesion location (0.05) (Table 2).
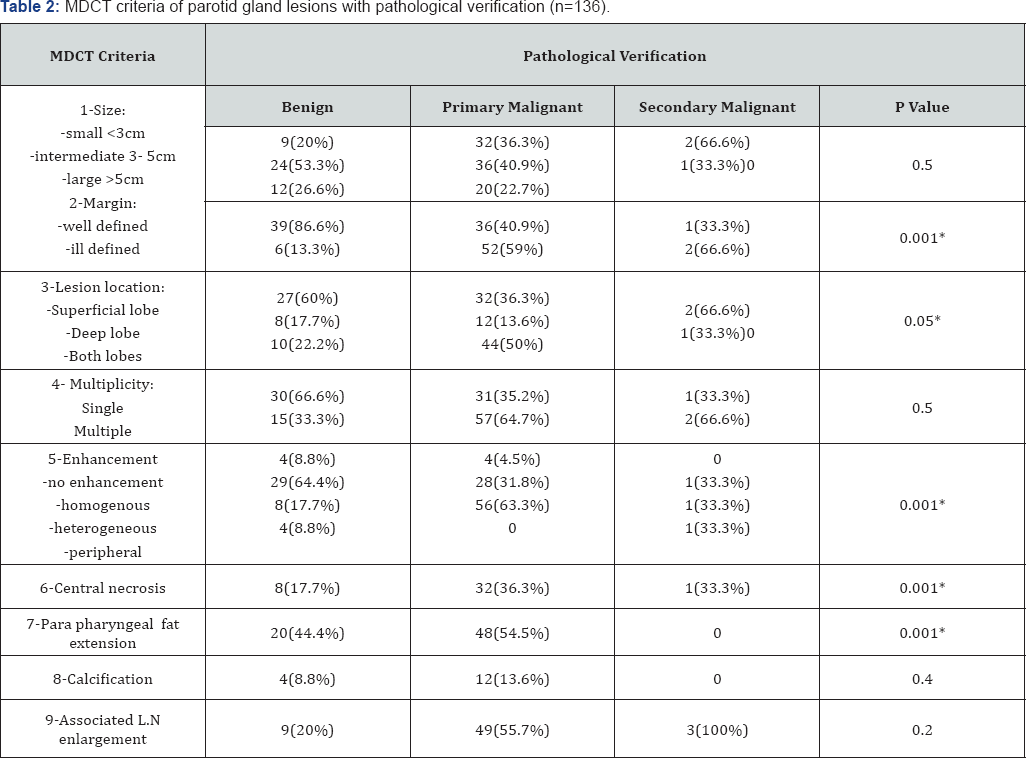
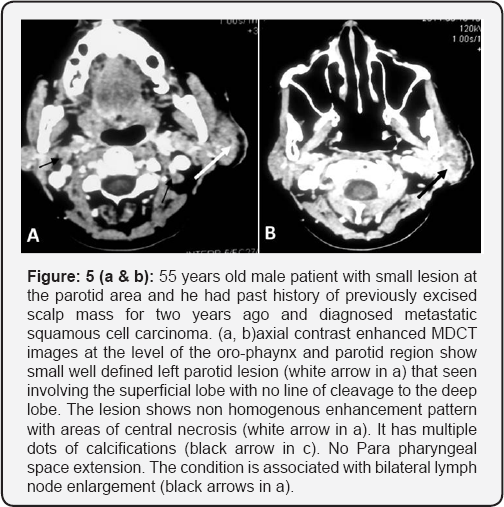
The associated lymph node (LN) enlargements were unilateral and multiple, and most of the enlarged LN (52) that interpreted for their number and bi-laterality. Most of the associated malignant parotid lesions were bilateral and multiple enlarged LN (9) that associated benign parotid lesions were (Table 3). Illustrative cases Figure 1-5.
p value is considered significant if less than 0.05.
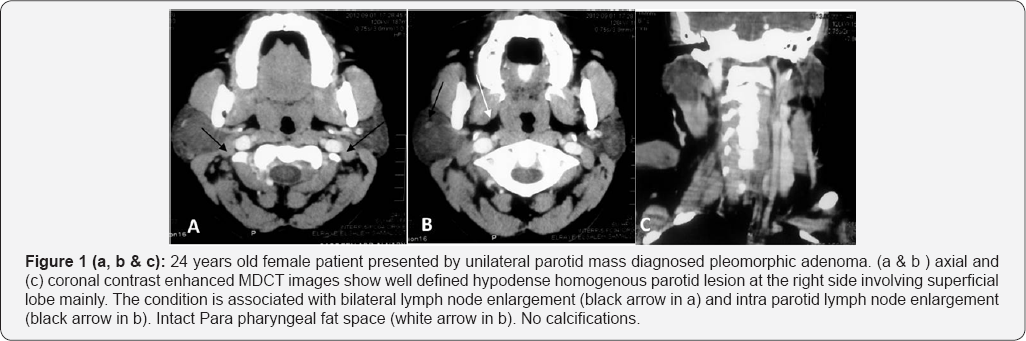
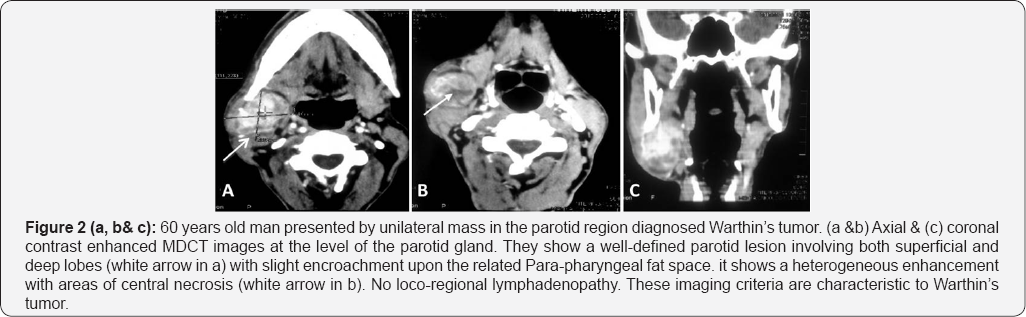
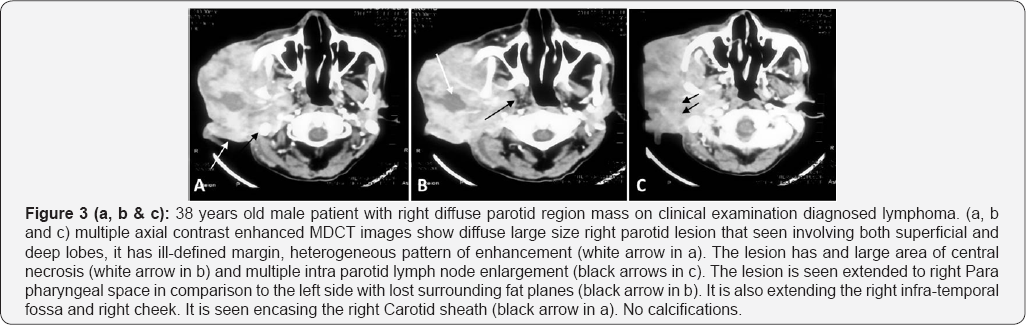
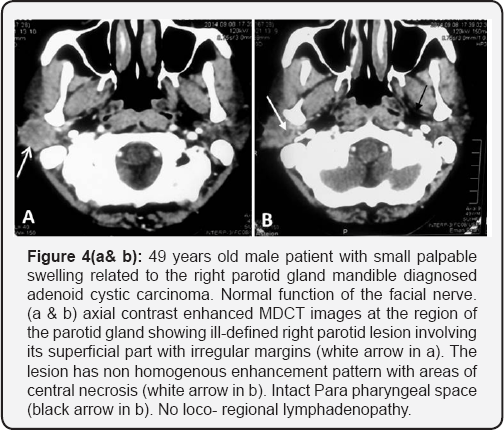
Discussion
Parotid lesions are a major challenge for radiologists especially those of neo plastic etiologies. Parotid masses in general have relative infrequency reaching up to 1% of head and neck tumors [1] and show no definite specific clinical signs with indeterminate imaging criteria. Many modalities are used including conventional X-ray, sialography, CT and MRI to reach the clinical diagnosis, helping better surgical approach and optimum postoperative outcome.
In the past, the role of CT was limited to evaluation of salivary gland calculi and obstructive or inflammatory disease [3]. So, in our study we try to re-evaluate and update the role of multi-detector CT in diagnosing neo plastic parotid lesions by assessment of various MDCT criteria of parotid lesions that routinely included within patient's report aiming to allow helpful clues to differentiate between benign and malignant neo plastic lesions and optimize the surgical outcome.
Owing to the multiple advantages of MDCT such as the enhanced speed of scan acquisition, the high spatial resolution, the precise timing of multiphasic imaging and the multi-planar reformations (MPR), the anatomic details and localization of parotid gland and its intra and extra-glandular lesions become more sharp and accurate. It also allows good visualization of deep parotid lesions as well as reliable assessment of their size, location, margins, and areas of necrosis, hemorrhage, calcification, and extension to adjacent structures as well as determination of perineural spread especially by curved planar reconstruction images (CPR) [3].
In our study, the mean age of the patients (44.4±22.2) as well as the prevalence of the most common benign and malignant parotid tumors; pleomorphic adenoma and mucoepidermoid carcinoma respectively were in concordance with Rastogi et al. [11], who reported that in patients older than 40 years, the most causes of enlarged parotid gland are neo plastic lesions either benign or malignant. The most common benign lesions are the pleomorphic adenoma and Warthin's tumor and the most common malignant tumor are the mucoepidermoid carcinoma and adenoid cystic carcinoma.
Our patients were collected from oncology center, all studied groups proved pathologically to have parotid neo plastic lesions. The benign neoplasms were detected in 45(33%) patients, 88(64.7%) patients had primary malignant tumors and 3(2.2%) patients had secondary malignant tumors. These values are not matched with other studies [1,3,12] due to the fact that the study is biased based only upon neo plastic group of patients. Other factors may be responsible such as increased prevalence of the disease that may be related to the ethnicity; however this needs further studies which are out of the scope of our research. In this study the MDCT criteria of all parotid lesions were evaluated individually. Regarding to the size criterion, the parotid lesions were classified into small, intermediate or large sizes. However despite of these variations in tumors size, there is no statistically significant difference between the size of benign and malignant lesions. Our results are matching with Brunese et al. [13], who reported no significant differentiation between benign and malignant lesions by the lesion size.
Regarding to the margin of the parotid lesions, the margin was classified into well-defined and ill-defined on both pre- and post-contrast imaging. When the margin was blurred or ill-defined at only one point, the whole margin was deemed ill- defined. Most of benign tumors have well defined margins. The margins of malignant parotid tumors may be confusing; low grade malignancy may have well-defined margins that mimicking the benign mixed tumors where the high-grade malignancy usually has invasive margins with irregular outlines. Our results are matching with Lu et al. [14], who evaluate the margin of benign and malignant parotid lesions of 133 patients. This imaging point is highly conclusive in our study to differentiate between benign and malignant parotid lesions.
Most of the benign parotid tumors were located within the superficial lobe while most of the malignant lesions were located within both parotid lobes. Our results were matching with to Lu YC et al (14), who suggest that, a parotid tumor located in the superficial lobe, with a round or oval contour and sharp margin, is more likely to be a benign tumor; otherwise, it might be a malignant tumor. Our study is also matching with A. Christe et al. [15], who postulated that, the predilection of deep lobe involvement was only indicative but not significant for malignancy, because inflammatory disease and some benign tumors, like schwannoma and Warthin tumors, also affected the deep lobe. However, involvement of both lobes was significant for malignant lesions, like the superficial lobe affinity seen with benign tumors.
The extra-glandular extension of parotid lesions is a grave prognostic finding, in this study it was considered when the parotid lesions extend to the prestyloid parapharyngeal space. Accordingly, parapharyngeal fat extension was detected in 48 malignant lesions and 20 benign lesions. This MDCT criterion shows highly significant diagnostic reliability in our study. The number of the parotid lesions was determined in each patient and we found that most of benign tumors were single while most of malignant tumors were multiple. However, this MDCT finding shows no statistical significant difference between the benign and malignant lesions. This sign is matched with Thoeny et al. [3], who postulated that multiple parotid lesions may be benign or malignant, and the most common multiple parotid gland tumors are the Warthin's tumor followed by acinic cell carcinoma.
The enhancement pattern of the lesion is another important MDCT criterion for assessment of the parotid lesions. The enhancement ofthe lesion seems to imply increased its vascularity and suggests a wide differential diagnosis. In this study, most of the malignant parotid lesions exhibited heterogeneous enhancement pattern, as it was determined in 57 out of 91 patients, while most of the benign lesions showed homogenous enhancement pattern, as it was determined in 29 out of 45 patients. The heterogeneous enhancement can be explained by central necrosis and vascularity. This study is nearly symmetrical to that of DC Howlett et al. [16]. Enhancement pattern is a very helpful imaging criterion in our study to differentiate between benign and malignant parotid lesions.
One ofthe important MDCT criteria that help in differentiating benign from malignant parotid lesions in this study was the central necrosis, it was found in 33 out of 91patients with malignant lesions and in 8 out of 45 patients with benign lesions. Allen et al. [17], discussed five cases of pleomorphic adenoma and conclude that central necrosis is not necessary specified to malignant lesions especially in cases of pleomorphic adenoma and Warthin's tumors which have high prevalence of cystic/necrotic regions in benign lesions. This study did not agree with the results of our study which may be explained by recent advances in MDCT techniques including high spatial resolution with thin collimation that leading to better tissue characterization and picking central necrosis more accurately.
Other MDCT criteria was the calcification of the parotid lesions which was only demonstrated in 16 (11.7%) out of 136 patients. Izum et al. [18], reported that calcifications of the neck are uncommon and mainly seen in cases of inflammatory lesions and Sjogren's syndrome.
The lastly evaluated MDCT criterion was the associated lymph node enlargement that was found in 61out of 136 cases. The majority of patients (49 patients) with lymph node enlargement had primary malignant lesions and all the 3 patients with secondary metastases also had lymph node involvement. These results were in agreement with Stennert et al. [19], who reported high incidence of lymph node metastasis in major salivary gland cancer with importance of neck dissection in surgical approach.
Limitations
The major limitation of this study is the biased nature of the study because we were concerning about imaging of neo plastic parotid group of patients who were directed to tertiary oncology center for treatment. It shows different results than literature regard the prevalence of benign and malignant parotid lesions. Another limitation of this study is that the MDCT criteria of all parotid lesions were evaluated individually, combined MDCT criteria assumed to have a powerful diagnostic reliability in distinguishing benign from malignant parotid lesions, further study is recommended.
Conclusion
Rapid advances of MDCT techniques giving rise to get an optimum parotid lesion characterization in a simple rapid and low cost maneuver. Multiple promising MDCT imaging criteria can be valuable in differentiation between benign and malignant parotid tumors. Imaging is forming a crucial triad with clinical data and surgical approach to get the satisfactory operative outcome.
References
- Bastakis JG (1979) Tumors of the head and neck. (2nd edn), Baltimore: Williams & Willkines, Myrland, pp. 2-75.
- Vermey A (1989) Benigne epithelialeparotistumoren (with English summary). Assen/Masstricht: Van Gorcum.
- Thoeny HC (2007) Imaging of salivary gland tumours. Cancer Imaging 7(1): 52-62.
- Wong DS, Li GK (2000) The role of needle aspiration cytology in the management of parotid tumors: a critical clinical appraisal. Head Neck 22(5): 469-473.?
- Rajwanshi A, Gupta K, Gupta N, Shukla R, Srinivasan R, et al. (2006) Fine-needle aspiration cytology of salivary glands: diagnostic pitfalls- revisited. DiagnCytopathol 34(8): 580-584.
- Al-Khafaji BM, Nestok BR, Katz RL (1998) Fine-needle aspiration of 154 parotid masses with histologic correlation: ten-year experience at the University of Texas M. D. Anderson Cancer Center. Cancer 84(3): 153-159.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Head and Neck Cancers. V 1.2015.
- Yousem DM, Kraut MA, Chalian AA (2000) Major salivary gland imaging. Radiology 216(1): 19-29.
- Parker GD, Harnsberger HR (1991) Clinical-radiologic issues in perineural tumor spread of malignant diseases of the extracranial head and neck. Radiographics 11(3): 383-399.
- Barakos JA, Dillon WP, Chem WM (1991) Orbit, skull base, and pharynx: contrast-enhanced fat suppression MR imaging. Radiology 179(1): 191-198.
- Rastogi R, Bhargava S, Mallarajapatna GJ, Singh SK (2012) Pictorial essay: Salivary gland imaging. Indian J Radiol Imaging 22(4):325-333.
- Choi DS, Na DG, ByunSH, Ko YH, Kim CK, et al. (2000) Salivary gland tumors: Evaluation with two-phase helicalCT. Radiology 214(1): 231236.
- Brunese L, Ciccarelli R, Fucili S, Romeo A, Napolitano G, et al. (2008) Pleomorphic adenoma of parotid gland: delayed enhancement on computed tomography. Dentomaxillofacial Radiology 37(8): 464-469.
- Lu YC, Fan WJ, Shen JX (2007) CT features of parotid tumors: an analysis of 133 cases. Ai Zheng 26(11): 1263-1267.
- Christe A, Waldherr C, Hallett R, Zbaeren P, Thoeny H, et al. (2011) MR Imaging of Parotid Tumors: Typical Lesion Characteristics in MR Imaging Improve Discrimination between Benign and Malignant Disease. AJNR Am J Neuroradiol 32(7): 1202-1207.
- Howlett DC, Kesse KW, Hughes DV, Sallomi DF (2002) Pictorial Review The Role of Imaging in the Evaluation of Parotid Disease. Clinical Radiology 57: 692-701.
- Allen CM, Damm D, Neville B, Rodu B, Page D, et al. (1994) Necrosis in benign salivary gland neoplasms, Not necessarily a sign of malignant transformation. Oral Surc Oral Med Oral Pathol 78(4): 455-461.
- Izum M, Kise Y, Murata K (2013) Multiple calcifications within the parotid gland of patients with Sjogren's syndrome. Oral Science International 10: 28-32
- Stennert E, Kisner D, Jungehuelsing M, Guntinas-Lichius O, Schröder U, et al. (2003)High Incidence of Lymph Node Metastasis in Major Salivary Gland Cancer. Arch Otolaryngol Head Neck Surg 129(7): 720-723.