Genetics and Imaging Aspects of Joubert’s Syndrome (JS)
Cherif Mohamadou Aidara*, Abdoulaye Dione Diop, Madeleine Diop, Delphine Diop, Abdoulaye Ndoye Diop and Sokhna Ba Diop
Radiology Office of Fann University Hospital, Switzerland a
Submission: February 23, 2017; Published: March 27, 2017
*Corresponding author: Cherif Mohamadou Aidara, Radiology Office of Fann University Hospital, Cheikh Anta Diop Street, BP 5035 Dakar Senegal, Switzerland, Email: matoouz@gmail.com
How to cite this article: Cherif M Aidara, A Dione Diop, Madeleine D, Delphine D, Abdoulaye N D, et al. Genetics and Imaging Aspects of Joubert’s Syndrome (JS). Curr Trends Clin Med Imaging. 2017; 1(2): 555559. 10.19080/CTCMI.2017.01.555559
Commentary
JS was described for the first time by Dr. Marie Joubertfour decades ago. It is an autosomal recessive neurodevelopmental genetic disorder. It is estimate dapproximatively to affect 1 in 100,000 newborns without sex predominance [1].
JS involves the normal development of the cerebellum, the vermis and the brainsteam. This,explains significantly the clinical features during the course of the disease. In the neonatal period, axial hypotonia, abnormal ocular movements and respiratory disorders are more frequently observed. Axial hypotonia is important. Intensity of symptoms is variable but tend to decrease with age [1,2]. Developemental delay and mental retardation are also variable from one individual to another, which can be responsible for a difficulty in learning or a significant delay in schooling. Oculomotor disorders appear to be characteristic in this condition. It is an oculomotorapraxia associated with a nystagmus. Strabismus or ptosis may also be observed. Autistic abnormalities are also described. However, penetrance of JS is variable with an almost normal development for some children [2,3].
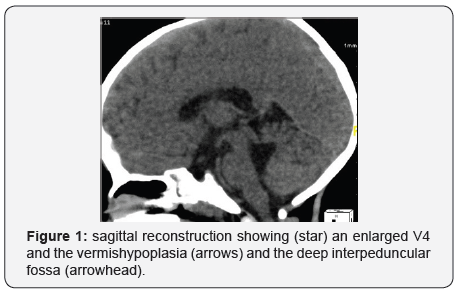
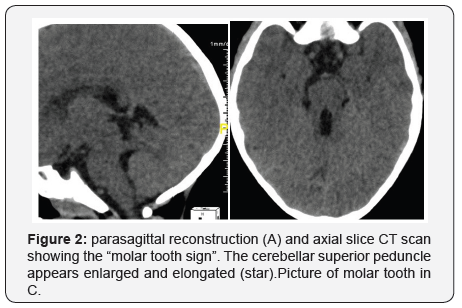
CT scan and MRI imaging can both show the pathognomonic sign that is called the “molar tooth” sign. It results from a midbrain-hindbrain malformation characterized by thickened and elongated superior cerebellar peduncle and an abnormally deep interpeduncular fossa (Figures 1 & 2). However, MRI is the current gold standard technique to confirm the diagnosis and to look for any other associated malformation [2,3].
In addition to abnormalities of the vermis (dysplasia and hypoplasia) and the brain steam which are constant, Poretti et al described other abnormalities such as cephalocele, polymicrogyria, periventricular heterotopia, ventriculomegaly, hippocampal malrotation, hypoplasia or even agenesis of the corpus callosum and agenesis of the septum pellucidum. These findings show the usefullness of neuro imaging in this condition
[2,3]. In our so called developing countries MRI is not often available and CT scan must be used efficiently.
Identification of responsible genes is important for genetic counseling especiallyin a level of restricted population or an identified group of individuals. At least 11 genes have been identified. Mutations are described on chromosomes 2, 6 or 12 among others. But their role in brain Developement is unclear [2,4].
Prognosis of this condition is favorable in the moderate form and depends initially on the intensity of respiratory disorders and other visceral abnormalities that are possibly associated; renal disorders (cystic dysplasia and/or nephronophthisis), liver disorders (hepatic fibrosis). Eye disorders may be associated with retinopathy. The mental retardation and Developemental delay may require care in a specialized center depending on the specific demand of each child [1].
References
- Joubert syndrome.
- Louie CM, Gleson J (2005) Genetic basis of Joubert syndrome and relateddesorders of cerebellardéveloppement. Human Molecular Genetics 2: R235-R242.
- Poretti A, Huisman TA, Scheer I, Boltshauser E (2011) Joubert Syndrome and Related Desorders. Spectrum of Neuroimaging Findings in 75 patients. AJNR Am J Neuroradiol 32(8): 1459-1463.
- Srour M, Hamdan FF, McKnight D, Davis E, Mandel H, et al. (2015) JoubertSyndrom in French Canadians and Identification of Mutation in CEP104. The American Journal of human Genetics 97(5): 744-753.