Acceptability and Usability of Colli-Pee UAS for First Void Urine Self Sampling
Stephanie Jordaens1,2*, Alejandra Ríos Cortés2, Wiebren Tjalma1,3, Patrick Pauwels1, Vanessa Vankerckhoven2,4 and Koen Beyers2
1Center for Oncological Research (CORE), Integrated Personalized & Precision Oncology Network (IPPON), University of Antwerp, Wilrijk, Belgium
2Novosanis NV, Wijnegem, Belgium
3Multidisciplinary Breast Clinic, Gynecological Oncology Unit, Department of Obstetrics and Gynecology, Antwerp University Hospital (UZA), Edegem, Belgium
4Vaccine & Infectious Disease Institute (VAXINFECTIO), Center for Evaluation of Vaccination (CEV), University of Antwerp, Wilrijk, Belgium
Submission:January 12, 2024; Published:January 23, 2024
*Corresponding author:Stephanie Jordaens, Center for Oncological Research (CORE), Integrated Personalized & Precision Oncology Network (IPPON), University of Antwerp, Wilrijk, Novosanis NV, Wijnegem, Belgium
How to cite this article:Stephanie J, Alejandra Ríos C, Wiebren T, Patrick P, Vanessa V, et al. Acceptability and Usability of Colli-Pee UAS for First Void Urine Self Sampling. Curr Trends Biomedical Eng & Biosci. 2024; 22(2): 556083. DOI:10.19080/CTBEB.2024.22.556083
Abstract
First-void urine as sample type has shown great potential for cancer detection, screening and monitoring. It is an emerging liquid biopsy with broad applicability in different analytes and cancers. The aim of this feasibility study was to investigate the experiences of healthy and diseased cohorts of first-void urine collection and preservation using the Colli-Pee® UAS™ devices (Novosanis NV), both for collection during doctors visit as well as home-sampling. Questionnaires from 98 participants enrolled in the URODETECT studies were analyzed. Participants reported that Colli-Pee® was clear (90%) and comfortable (68%) to use. Most respondents (82%) rated their general experience of Colli-Pee® as easy to very easy, while only 2% indicated it was difficult. Participants also reported their intention to use Colli-Pee® again (87%) or to recommend Colli-Pee® to their friends and/or family (88%). A subset of participants (n=60) collected urine samples at home using Colli-Pee® UAS™ devices and returned their samples to the laboratory. Most participants’ responses were positive (very easy, 35%, easy 40%) about the entire self-sampling process. Urine collection, at home or in the clinic, using Colli-Pee® was clearly accepted by all participants. Furthermore, self-sampling could offer the possibility to increase at home cancer screening coverage and provide more comfort for individuals.
Keywords:Cancer detection; Colli-Pee®; Biopsy; Surgical pathology; Clinical diagnostics; Human Papillomavirus
Abbreviations:BMI: Body mass index; HPV: Human Papillomavirus; IFU: Instructions for use
Introduction
In past decades, liquid biopsy samples have gained momentum in several disease fields. In cancer research, liquid biopsy has some advantages over tissue biopsy as it may better capture intratumor heterogeneity and, can be used as a screening tool for early cancer detection, monitoring of treatment response, and detection of recurrence [1-4]. Different body fluids have considerable potential as liquid biopsies for different diseases, like blood which is considered minimally invasive or urine which is considered non-invasive [3]. Urine has added advantages over blood as it is easy to collect, allows for at home sampling, and there is no need for a health care practitioner. Furthermore, urine allows for serial sampling, multiple tests can be performed on the same sample, and it offers possibilities for multi-omic analysis [5,6]. Urine as liquid biopsy has shown potential to improve the detection and monitoring of cancers such as prostate cancer [7,8] and breast cancer [9-11]. The predicted global cancer burden is expected to increase significantly. By 2040, 30.2 million new cancer cases are estimated, compared to 19.3 million cases reported worldwide in 2020 [12]. Early detection of cancer can increase the chances of survival and improve the overall quality of life of a patient. Therefore, research into screening methods and sample types enabling screening gained interest in the last two decades. Urine has been proposed as a promising self-sampling method for disease screening. However, guidelines around pre-analytical parameters including collection, preservation, and storage are not yet defined, even though several studies have already showed the need for urine preservation for oncology applications [13-15].
Several commercial products are already available for nucleic acid preservation, intended for preserving either DNA and/or RNA. Therefore, Novosanis (Wijnegem, Belgium) and DNA Genotek (Ottawa, Canada) developed a urinary preservative that prevents the lysis of bacterial and human cells, thereby blocking the release of unwanted nucleic acids into the biological sample [16-18]. Currently, the UAS™ preservative (DNA Genotek, Ottawa, Canada) is validated for preventing bacterial growth, preserving cfDNA and maintaining the integrity of host cells for a minimum of 7 days at room temperature [19]. The Colli-Pee® devices (Novosanis NV, Wijnegem, Belgium) on the other hand are developed for the standardized and volumetric collection of first-void (first-catch or first-pass) urine, typically the first part of urine flush collected at any time of the day [20,21]. When the UAS™ preservative is integrated into the Colli-Pee® devices, these enable standardized volumetric collection and immediate preservation of first-void urine for home-based sampling. The aim of this feasibility study was to investigate the experiences and preferences of healthy and diseased cohorts for the collection and preservation of first-void urine using the Colli-Pee® UAS™ devices (Novosanis NV, Wijnegem, Belgium) [20-22], both for collection during a doctor’s visit as well as home-sampling. This study did not aim to perform any downstream analysis on the collected urine samples.
Methods
Study population
The questionnaires to assess experiences and preferences were collected from participants in the URODETECT studies. These are prospective studies, aimed to investigate the potential of urine as liquid biopsy for cancer detection (ClinicalTrials.gov identifiers: NCT05453604, NCT05454371, NCT05453591). Study NCT05453604 recruited healthy female and male adult volunteers, breast and prostate cancer patients, and pregnant women, while study NCT05454391 and NCT0545371 recruited healthy female adult volunteers and breast cancer patients or healthy male adult volunteers and prostate cancer patients, respectively. Healthy female and male adult volunteers, breast and prostate cancer patients, and pregnant women were recruited between June 2020 and December 2022 at the Antwerp University Hospital (Belgium) and the University of Antwerp (Belgium). All participants understood Dutch and provided written informed consent.
Participants received a package with a participant information form, a consent form, Colli-Pee® UAS™ device(s) for the collection and preservation of first-void urine (Novosanis, Wijnegem, Belgium), instructions for use (IFU) for the devices, and a questionnaire to be completed online or on paper. Generally, the participant received one Colli-Pee® UAS™ FV-5040 device. Only the subset of participants (n=60) recruited for the home-sampling part received three variants of the device, Colli-Pee® UAS™ FV-5010 (collecting approximately 10 mL), Colli-Pee® UAS™ FV-5020 (collecting approximately 20 mL) and Colli-Pee® UAS™ FV-5040 (collecting approximately 40 mL). Participants were asked to collect a urine sample using the Colli-Pee® UAS™ device and subsequently complete the questionnaire. The samples, the questionnaire, and the written informed consent were either provided to the nursing staff or mailed to the laboratory of surgical pathology of the Antwerp University Hospital. After arrival in the laboratory, samples were stored to allow for testing on specific urinary biomarkers (not part of this paper). The collected samples were not analyzed as part of this manuscript, as we only aimed to assess usability and acceptability. Ethical approval for the URODETECT studies was provided by the Ethical Committee of the Antwerp University Hospital (no 20/10/115 and B3002021000030). All studies were conducted in accordance with the Declaration of Helsinki.
Questionnaires
Questionnaires were created in Qualtrics® (Seattle, Washington, USA) online survey software, allowing the possibility to provide a paper or an online questionnaire to the study participants. The questionnaires consisted of a maximum of three categories: general information, Colli-Pee® performance, and home-sampling. These categories were further divided into eight sections: general – collection, general – sexual, general – health information, before collection – preparation of Colli-Pee®, during collection – performance of Colli-Pee®, after collection – disassembly of Colli-Pee®, feedback on overall usage of Colli-Pee®, and home-sampling. The questionnaire had open, multiple choice and rating questions. Rating responses were gathered with a scale ranging from zero, the most negative result, to 100, the most positive result. For the assessment questions, if a given step received a score between 80 and 100, it was classified as in full agreement with the proposed statement. Not all participants answered all questions, however the number of received answers is clearly indicated throughout the manuscript. The original Dutch questionnaire was translated to English and added as Appendix 1.
Statistical analysis
Online questionnaire responses were automatically captured in the Qualtrics® online survey software. Questionnaire responses of paper forms were manually entered in the Qualtrics® online survey software, to create one database with all responses. The questions with open answers were manually analyzed by reading the responses and group them based on similar terms (e.g. for question H2 the terms postal, packaging, shipment was combined in one group). Contingency for Fisher and chi-square tests were used to analyze the categorial variables: the population characteristics (gender, education, profession), the replies to the yes-no, multiple choice and the home-sampling questions. While frequency distribution analyses were performed for the continuous variables: the analysis of population characteristics (age, BMI) and the assessment questions (scores between 0 and 100). All analyses were conducted with GraphPad Prism (version 9.4.1, GraphPad Software Inc., La Jolla, California, USA).
Results
Participant characteristics
Between the 25th of June 2020 and the 31st of December 2022, 100 participants were enrolled in the URODETECT studies. Questionnaires were obtained and analyzed from a total of 98 participants: 59 women and 39 men. The participants’ ages ranged from 20 to 83 years old, with a median age of 36 years. Most (84%) of the participants were between 20 and 60 years. Additionally, other relevant characteristics were investigated, including BMI, education, and profession. Most respondents had a higher level of educational (college, university or PhD; 76%). A total of eight breast cancer patients (8%), twenty-eight healthy female volunteers (29%), thirty-one healthy male volunteers (32%), twenty-three pregnant women (23%) and eight prostate cancer patients (8%) completed the questionnaires. Overall participant characteristics are compiled in Table 1.
Ɨ, two missing values; ǂ, one missing value.
Evaluation of Colli-Pee® UAS™ use
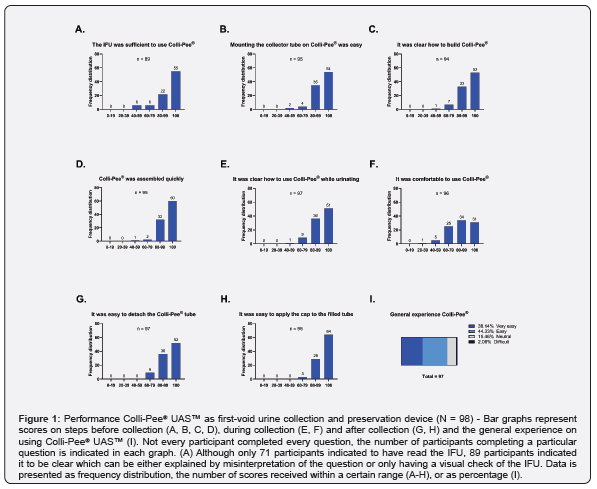
Most participants (72%) reported to have used the IFU, while 26% reported not doing so and 2% did not complete the question (Table 2). Approximately 91% responded to the question about the clarity of the IFU, of these 87% indicated that the IFU was sufficient to use Colli-Pee® UAS™ (Figure 1A). Although only 71 participants indicated to have read the IFU, 89 participants indicated it to be clear which can be either explained by misinterpretation of the question or only having a visual check of the IFU. A total of 89% rated the assembly of the collector tube on Colli-Pee® UAS™ as easy (Figure 1B). Additionally, participants reported that it was clear (91%) and quick (94%) to assemble the Colli-Pee® UAS™ device before use (Figure 1C, D).
Evaluating the use of Colli-Pee® UAS™, approximately 58% reported that Colli-Pee® was held against the body during urination, while 38% did not (Table 2). Most participants (90%) scored Colli-Pee® clear in use (i.e., participants clearly understood how to use the device and take a self-sample), and 68% scored Colli-Pee® comfortable to use (Figure 1E, F). Looking at spillage, most respondents did not experience any spillage of urine during (73%) and after (80%) collection (Table 2). Spillage was clearly correlated to the female gender (p=0.01) as this was reported by 20 women out of 25 participants who reported spilling. Approximately 40% stated ‘moving the Colli-Pee® during collection’ as a reason for spillage occurring during collection. While for spillage occurring after collection, most respondents indicated that urine droplets came from the floater (39%) or housing (44%) of Colli-Pee®. No respondent reported a device malfunction as the reason for the urine spillage (data not shown). The majority of participants scored detaching and subsequent capping of the tube as easy (detaching, 91%; capping, 97%) (Figure 1G, H). Finally, most respondents (82%) rated their general experience of Colli-Pee® as easy to very easy to handle, while only 2%indicated it was difficult (Figure 1I).
Participant preferences and future considerations
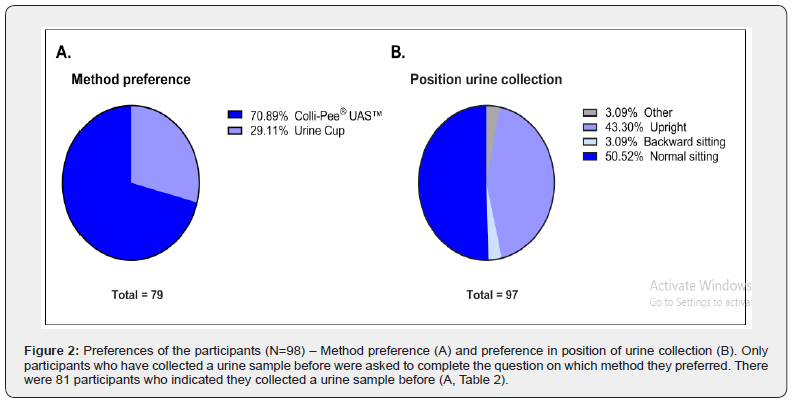
The pie charts in Figure 2 show the preferred method of urine collection method (A) and the choice of urinating position for urine collection (B). Prior experience with urine collection (e.g., urine cup) was reported by 82% of participants (Table 2). A total of 71% of these would choose to collect a urine sample using Colli-Pee® and 29% would use a urine cup. There were no significant correlations found between the preferred method and gender, education, BMI, or spillage (data not shown). However, there was a significant correlation between the preferred method and age (p=0.016), with a higher portion of younger participants preferring a urine cup (26-37 years: 70%). Interestingly, seven of these young participants were pregnant women (44%). Furthermore, the two most used urinating positions to collect urine using Colli-Pee® were normal sitting (51%) and standing upright (43%). Participants also reported their intention to use Colli-Pee® again in the future (87%) or to recommend Colli-Pee® to their friends and/or family (88%). Additionally, 96% indicated that they had the impression that urine collection was performed correctly using Colli-Pee® (Table 2).

Home-sampling experiences
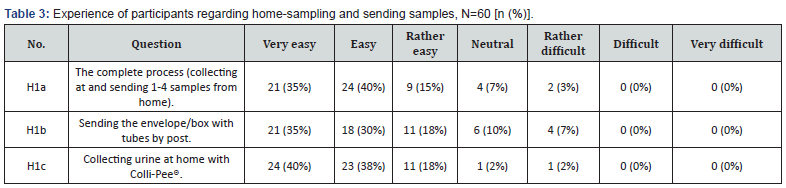
A subset of all study participants (n =60) was requested to collect first-void urine samples at home using Colli-Pee® UAS™ device variants and return their samples back to the laboratory via postal mail. These participants were then asked to complete a specific question on home-sampling and sending samples via postal mail, as well as to freely provide any further feedback. The responses to questions H1a-H1c are summarized in Table 3. Most participants were positive (very easy, 35%; easy, 40%) about the entire process of collecting urine samples at home and sending these back to the laboratory. The home-sampling experience was scored very positively (very easy, 40%; easy, 38%), while postal mailing of the samples was rated slightly lower (very easy, 21%; easy, 30%). Participants reporting ‘rather difficult’ were limited for the full process (3%), sending the samples (7%) and home-sampling (2%). None of the participants rated any of these questions as difficult or very difficult. About 19 participants (32%) provided additional feedback. Most feedback was related to the size of the envelope or box, which did not fit well or easily in official postal mailboxes in Flanders, Belgium.
Discussion
Urine sampling offers many advantages as it is easy, non-invasive, and suitable for at home collection. Home-sampling allows individuals to use the comfort and privacy of their home environment to collect a sample without the need to visit a health care institution. Furthermore, home-sampling reduces the pressure on healthcare workers and protective materials required for clinic-based testing [23]. However, the implementation of home-sampling has been slow in clinical trials and in routine clinical diagnostics. Concerns have been raised on the quality and stability of the collected samples, as well as comparability of results to standard sampling methods [24]. To maximize the potential of self-sampling and at home collection, the entire process (i.e., from collection to return of samples to the laboratory) requires optimization. Therefore, sampling devices are needed to allow for maximal ease-of-use. Additionally, trends in digital health require more efficient and reliable communication channels to be developed and used, to provide psychological support to a patient, especially when envisioning virtual patient-doctor meetings or when a test result needs to be communicated [23].
Colli-Pee® UAS™ devices are developed for self-sampling of first-void urine and contain the UAS™ preservative, which allows the storage of first-void urine prior to analysis, whether due to home collection, sample shipment or biobanking. This study shows that first-void urine collection using Colli-Pee® UAS™ is considered easy to very easy even in a home-based setting. Participants reported that Colli-Pee® UAS™ was clear and comfortable in use. We noticed that 26% did not use the IFU before collecting the urine sample. Some patients and pregnant women did ask verbally how the device worked upon receipt at the clinic, which could have impacted their assessment of usability in the questionnaire as they received more information compared to those that collected at home. Most participants preferred urine collection using Colli-Pee® UAS™ over urine collection using a regular urine cup. We noticed a significant correlation between the preferred method to collect a urine sample and age (p=0.016), with a higher portion of younger participants preferring a urine cup (26-37 years: 70%). Interestingly, of these younger participants, seven were pregnant women (44%). Based on verbal feedback, some participants were unsure about aiming the Colli-Pee® device towards the toilet because they lost sight of the device. Similar verbal feedback has been given in a study using Colli-Pee® devices with participants with a higher BMI (unpublished data). When self-sampling at home, most participants were very positive about their experience.
Other devices have been developed to collect of first-void urine, such as First Burst (DRW, Cambridge, UK) [25] and Peezy First Stream (Forte Medical, London, UK) [26], and/or to enable the collection of specific volume, such as First Burst (DRW, Cambridge, UK) [25], Monovette (Sarstedt, Nümbrecht, Germany), Vacuette (BD, Franklin Lakes, New Jersey, USA) [27] and Uriswab (Copan, Brescia, Italy) [28]. However, Monovette (Sarstedt, Nümbrecht, Germany) and Vacuette (BD, Franklin Lakes, New Jersey, USA) [27] only allow volumetric collection of a sub-sample after the collection by the individual. In contrast, Colli-Pee® (Novosanis, Wijnegem, Belgium) [20] allows for standardized volumetric collection of first-void urine immediately when the patient collects the sample. The standardized volumetric collection of the Colli-Pee® device was validated previously and therefore not part of this study.
Optimized self-sampling and at home collection processes could improve participation in urine-based liquid biopsy screening programs. For example, cervical cancer, for which major progress in screening coverage has been made in the past decades via urine self-sampling. A meta-analysis showed an overall 2.14-fold increase in screening coverage due to self-samples [29]. Also, a study in France selected 5000 hard to reach women (40-65 years) who had not had a cervical cytology (PAP) smear examination over the past three years and send them a urine home-sampling kit. They received 771 urine samples by postal mail, highlighting that urinary Human Papillomavirus (HPV) testing may be useful to reach women who do not regularly have cervical smears done to find high-grade cervical lesions [30]. Studies have also shown that self-samples provided equivalent results to physician-collected samples for the detection of HPV [31]. For first-void urine, several studies already showed its value and analytical performance [32-35]. Additionally, previous studies have shown urine as the most preferred sampling method for cervical cancer screening [34,36-40]. These studies also showed more confidence of women for collecting a urine sample compared a cervicovaginal self-sampling [39,40]. The World Health Organization and Human Reproduction Program have even suggested in their new guidelines that self-collected samples, such as urine, can be used when providing HPV DNA testing [41].
Limitations of the study
Our study had some limitations. First, the purpose of our study was to investigate the acceptability and usability of the Colli-Pee® UAS™ device and therefore the actual suitability of purpose for downstream analysis was not assessed. Secondly, the standardized volumetric collection of the Colli-Pee® UAS™ devices was previously validated by Novosanis NV (Wijnegem, Belgium) and therefore not re-evaluated as part of this study. Thirdly, since the patient recruitment was hampered during the COVID-19 pandemic (2020-2022) there is a slight bias towards healthy volunteers and pregnant women. However, age, education level, profession and BMI distribution allows drawing parallels between healthy volunteers and cancer patients. Additionally, the patient sample size was very limited as we considered this more as a feasibility assessment. The research can be considered as highly innovative, as it will ultimately explore the feasibility of biomarker detection in urine Conclusion
Conclusion
Urine is an emerging liquid biopsy with broad applicability in different analytes and cancer research. In this study evaluating Colli-Pee® UAS™ enabled first-void urine collection in both healthy and cancer participants, the device was rated positive for its usability. Participants indicated they would choose Colli-Pee® for their next urine sample, recommend using it to friends and/or family and, had the impression urine collection was performed correctly. Importantly, the self-sampling method was well received, and this could offer the possibility to increase screening coverage and provide more comfort for individuals during screening and patients during their disease monitoring in a home-setting or doctor’s visit.
Acknowledgement
First, we would like to thank all the cancer patients, pregnant women and healthy volunteers who took part in the URODETECT studies and completed the questionnaires. We would also like to thank Leen Stulens from HappyAging (Heverlee, Belgium) for her collaboration and support in recruiting healthy volunteers. Additionally, we would like to thank Nette Meers and Jhana Hendrickx from Novosanis for their close collaboration and support.
References
7 Fujita K, Nonomura N (2018) Urinary biomarkers of prostate cancer. Int J Urol 25(9): 770-779.
20 Van Damme P (2013) Liquid sampler, kit of parts, and method for assembly.
21 Beyers K (2021) Small volume liquid sampler.
22 Arora A (2021) Stabilizing composition and method for preserving a bodily fluid.
25 Smetham GTL, SPJ, Hazlewood S, Nadala ECB, Wisniewksi CA (2004) Liquid Sampler and Method.
26 GFL, F (2018) Urine Collection Device.
27 Ealovega GD, Hatz E (2014) Urine-Specimen Collection, Storage and Testing Device.
28 Triva D, Rennie RP (2006) Container for Urine Sample.
41 WHO (2021) WHO guidelines for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention WHO Guidelines Approved by the Guidelines Review Committee.






























