NRF/CNC Proteins Mediate Ferroptosis in Diseases
Yue Huang1, Jijie Zhan2, Tian Li2 and Changlin Zhang2*
1School of Medicine, Sun Yat-Sen University, Shenzhen, China
2Department of Gynecology, The Seventh Affiliated Hospital of Sun Yat-sen University, Shenzhen, China
Submission:December 12, 2023; Published:January 04, 2024
*Corresponding author:Changlin Zhang, Department of Gynecology, The Seventh Affiliated Hospital of Sun Yat-sen University, Shenzhen, China
How to cite this article:Yue H, Jijie Z, Tian L, Changlin Z. NRF/CNC Proteins Mediate Ferroptosis in Diseases. Curr Trends Biomedical Eng & Biosci. 2024; 22(2): 556081. DOI:10.19080/CTBEB.2024.22.556081
Abstract
Ferroptosis is a novel form of cell death characterized by an imbalance of iron homeostasis and lipid peroxidation. It has been found to play a key role in guiding the fate of cells, and is closely related to the occurrence and development of a variety of diseases. One of the most important characteristics of ferroptosis is that it is unique not only in terms of morphology and biochemistry but also has a complex and diverse regulatory pathway network. NRF/CNC proteins, which act as important transcriptional activators, have recently been found to have a pivotal regulatory effect on ferroptosis through inhibition. Therefore, further research on the regulatory pathway of ferroptosis through NRF proteins could help to understand the development of various diseases and contemplate new ideas for corresponding treatments. This paper aims to summarize the regulatory roles played by NRF proteins, mainly NRF1 and NRF2, in the onset and development of ferroptosis and to describe the regulatory relationships and research prospects of ferroptosis in diseases..
Keywords:Ferroptosis; Nrf-2; Nrf-1; ROS; Iron metabolism; Lipid peroxidation; Cell death
A new form of programmed cell death: Ferroptosis
Ferroptosis is a novel form of programmed cell death (PCD) that can occur due to internal and external factors, such as oxidative stress. Ferroptosis morphologically and biochemically differs from any previously described type of PCD, including apoptosis and autophagy. Morphologically, ferroptosis does not possess the typical manifestations of apoptosis and other forms of PCD, such as karyopyknosis and karyorrhexis, but shows mitochondrial atrophy with the reduction or disappearance of mitochondrial cristae [1-3]. Biochemically, ferroptosis does not involve the typical reactions that occur during apoptosis or other forms of PCD, such as caspase activation [4,5]. Ferroptosis is closely associated with iron metabolism and lipid peroxidation. Certain factors disrupt intracellular iron homeostasis and elevate the intracellular labile iron pool (LIP), resulting in the formation of reactive oxygen species (ROS) and lipid peroxidation through the Fenton reaction, which ultimately leads to ferroptosis. Ferroptosis is also associated with a decrease in the antioxidant capacity of certain substances such as GSH/GPX4 and related pathways. Many signaling molecules and regulatory pathways are also involved in ferroptosis, forming a tight and complex regulatory network.
Overview of Ferroptosis
Researchers were first exposed to ferroptosis-related phenomena in 2003, when Dolma et al. [6] while screening for genotype-selective antitumor agents in human tumor cells, found that both erastin and camptothecin (CPT) had cytotoxic effects on cells expressing RAS/ST. Although the genetic selectivity of the two compounds was similar, CPT induced a typical apoptotic pathway, whereas cell death induced by erastin did not show the typical features of apoptosis, such as the observation of karyopyknosis, karyorrhexis, or caspase-3 activation. They also reported the loss of mitochondrial membrane potential under the influence of erastin. They experimentally confirmed that the erastin-induced cell death was non-apoptotic and not caused by cell detachment. In 2007, Yagoda et al. [2] revealed that erastin induces non-apoptotic cell death by targeting mitochondrial voltage-dependent anion channels (VDACs) and affects outer mitochondrial membrane permeability. The Ras-Raf-MEK pathway has been reported to be involved in non-apoptotic cell death. In 2008, Yang and Stockwel [3] identified RAS-selective lethal (RSL) and erastin as inducers of non-apoptotic type of cell death. RSL5, similar to erastin, is reported to act on VDACs, whereas RSL3 has a different target. More importantly, iron chelators have been shown to inhibit the pro-cell death effects of the RSL family, linking this newly discovered form of cell death to iron. In 2012, Dixon et al. [1] formally introduced the concept of ferroptosis, stating that it is associated with iron metabolism and ROS accumulation, which are sensitive to an iron chelator deferoxamine. The formation of ROS was reported in the mitochondrial electron transport chain and the NADPH oxidase pathways. They conclusively showed that ferroptosis can be induced via the inhibition of system Xc- (a glutamate/cysteine antiporter; cystine is the oxidized form of cysteine) by erastin. The complete mechanism of ferroptosis associated with system Xc- was reported in 2014 [7]. The suppression of system Xc- because of the multiple factors leads to decreased GSH levels and reduced GPX4 activity, resulting in ROS production and lipid peroxidation, which eventually results in ferroptosis. We also confirmed that the target of RSL3 is GPX4.
Role of iron metabolism in ferroptosis
Iron metabolism is inextricably linked to iron homeostasis in the interior milieu of cells. Numerous studies have demonstrated that dysregulation of iron homeostasis is closely associated with various diseases. The induction of ferroptosis, an iron-dependent form of PCD, is closely associated with the dysregulation of iron metabolism and disruption of iron homeostasis. In the cellular metabolism of iron, TFR1 is a key receptor for the transport of iron ions into cells. Transferrin binds to TFR1 on the surface of the cell membrane and transports Fe3+ into the cytosol by endocytosis. The endosome has a proton pump that assists the entry of H+, and in this environment, Fe3+ is reduced to Fe2+ by the action of STEAP3 and is transported by DMT1 and other iron transporters into the cytoplasm to form the LIP. After entering the cytoplasm, Fe2+ can participate in the formation of the intracellular electron transport chain through various receptors, including MFRN-1. Fe2+ can be stored in the cytoplasm in the form of ferritin or excreted outside the cell through ferroportin [8,9]. The aforementioned basic processes are mainly responsible for iron metabolism and homeostasis and are affected by internal and external factors during ferroptosis, ultimately leading to an imbalance in iron homeostasis, elevated LIP, and cell death.
Following the first report on the association of ferroptosis with iron in 2008 through the discovery that iron chelators inhibit the pro-cell death effect of the RSL family [3], the critical role of iron metabolism dysfunction in ferroptosis was again validated in 2015 through the discovery that silencing TERC (the gene encoding TFR1) inhibits erastin-induced ferroptosis [10]. Moreover, in 2015, Gammella et al. [11] pointed out that reducing the intracellular LIP concentration by increasing ferritin expression inhibits ferroptosis, highlighting the role of elevated LIP in ferroptosis. Elevated LIP results in an imbalance in iron homeostasis, because of which Fe2+ undergoes the Fenton/Haber–Weiss reaction with the involvement of iron sulfur clusters, leading to ROS production and lipid peroxidation and subsequently ferroptosis [12-14].
Lipid metabolism involved in ferroptosis
Lipid peroxidation is the key process in ferroptosis, and polyunsaturated fatty acids (PUFAs) are highly susceptible to lipid peroxidation because of the increased ROS production via various ferroptosis pathways. Biosynthesis and remodeling of phosphatidylethanolamine (PE)-containing arachidonic acid (AA) occurs by activating PUFAs by ACSL4 and LPCAT, resulting in lipid peroxidation through a series of reactions transitioning through various chemical forms, including PE-AA, PE-AA-OOH, and PE-OH [15-17]. In addition, AA and its derivative, epinephrine, play an important role in lipid peroxidation [18].
Signaling molecules and the pathways of ferroptosis
Various signaling molecules and pathways are involved in the regulation of ferroptosis. The regulation of ferroptosis by P53, which has dual functions, is of particular interest. Wild-type P53 can inhibit cystine uptake by suppressing the expression of SLC7A11 (a component of system Xc-) [19] and inducing ferroptosis in an ROS-dependent manner, such as through the P53-SAT1-ALOX15 pathway [19-22], the P53-P21 axis [23-25], or the DPP4-related pathway [23,26]. These effects are also involved in tumorigenesis and the survival and death of normal cells. These dual effects on ferroptosis are mainly related to numerous molecules involved in the downstream signaling of P53, including USP7 [27], PTGS228, Parkin [28], and PVT1 [29,30] and their related pathways, each of which can promote or inhibit ferroptosis [31]. Additionally, P53 regulates the expression of the afore mentioned genes at the transcriptional level, whereas P53 itself is mainly regulated at the post-translational level, particularly via ubiquitination [31] (Figure 1).
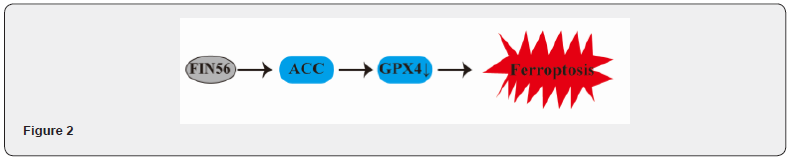
Unlike P53, FIN56 induces ferroptosis by downregulating GPX4 primarily through acetyl-CoA carboxylase (ACC) or CoQ10 by binding to SQS, whereas FINO2 induces ferroptosis indirectly by affecting GPX4 enzyme activity and directly by oxidizing ferrous ions, independent of ALOXs [32,33]. The CoQ10-related ferroptosis pathway is also involved in this process. The key amino acid for GPX4 activity is selenocysteine, and its insertion into GPX4 requires selenocysteine tRNA. The mevalonate pathway affects GPX4 activity by influencing the maturation of this tRNA, thereby participating in ferroptosis. The mevalonate pathway includes two important components: isopentenyl diphosphate and CoQ10 [34-36] (Figure 2).
These signaling molecules and pathways are widely involved in the regulation of ferroptosis and other forms of PCD, allowing the interaction of apoptosis, autophagy, and ferroptosis, indicating the involvement of a complex and sophisticated regulatory network behind these PCDs. Members of the HSP family are key molecules responsible for interconnecting these pathways. HSP90, for example, has been shown to be involved in multiple forms of PCD, including apoptosis, autophagy, and ferroptosis, as well as in a variety of diseases. However, in tumor cells, HSP90 is primarily a negative regulator that facilitates tumorigenesis, and some HSP90 inhibitors have been successfully used to induce various forms of PCDs in cancer cells [37,38]. HSP90 links various forms of PCDs in several ways. Importantly, it depletes GPX4 levels through the chaperone-mediated autophagy pathway, thereby promoting ferroptosis and depleting substances such as GAPDH, which affect cellular homeostasis [39].


Moreover, there are many other signaling molecules and related pathways involved in the regulation of ferroptosis, including the NK-κB pathway [40], trans-sulphuration pathway [41], glutamine metabolic pathways [10], and ATG5-ATG7-NCOA4 pathway [42]. In the recent years, the important regulatory role of the NRF/CNC family in ferroptosis has been revealed, which exhibits potential implications in regulating ferroptosis.
NRF/CNC proteins
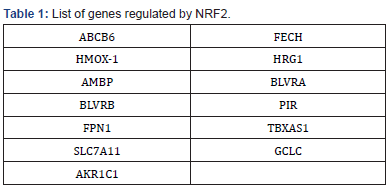
The NRF/CNC family is a class of evolutionarily conserved transcription factors (a family of basic leucine zipper transcription factors) that mainly includes P45, NFE2, and the NFE2-related factors NRF1, NRF2, and NRF3 [43-50]. The vast majority of CNC family members are transcriptional activators, and only a few isoforms or cleaved forms (caspase-cleaved) act as repressors, including BACH1 and BACH2 [51-56]. One of the most intensively studied members is NRF2, which was earlier thought to be closely associated with regulatory responses to cellular stress [57]. NRF2 functions through the antioxidant pathway. In the presence of conditions such as oxidative stress, KEAP1, a direct negative regulator of NRF2, undergoes a conformational change [58-64], thereby relieving its inhibitory effect on NRF2. This allows NRF2 to enter the nucleus and form a dimer with the MAF oncogene product, which eventually binds to an antioxidant-responsive element (ARE). This in turn promotes the expression of genes associated with antioxidants (KEAP1-NRF2-ARE pathway) [65-68], including GCL, GST, and HO1, and has different manifestations in different types of tissues [69-73]. However, in these tissues, multiple metabolic enzymes and antioxidant and antitoxic proteins show increased expression under NRF2 regulation [74]. Notably, there is some disagreement on the KEAP1-NRF2 interaction models involved in the KEAP1-NRF2-ARE pathway, including the "hinge and latch" model [63,75]. Moreover, in addition to its effects on ARE, NRF2 can exert its antioxidant effects by affecting specific gene targets, including synergistic action with D3T to induce the expression of genes involved in the recognition and repair of damaged proteins, a pathway closely linked to cellular antitumor effects [76]. Furthermore, genes regulated by NRF2 contribute to the maintenance of iron homeostasis and also have regulatory functions in calcium homeostasis, growth factors, and signaling molecules, leading to the resistance of many aspects of ferroptosis and its involvement in anti-inflammatory, anti-aging, and stem cell functions [65,77-80]. Similar to NRF2, NRF1 and NRF3 activate ARE [81], which in turn confers resistance to ferroptosis. However, while full-length NRF1 activates ARE, the naturally occurring truncated NRF1 isoform acts as an inhibitor [56]. Interestingly, since genes encoding KEAP1, NRF2, and MAF can be induced by AREs in their regulatory sequences, the members of CNC, such as NRF1 and NRF3, can enhance NRF2 transcription by acting on ARE to achieve cross-regulation [82-84].
Role of NRF2 in Mediating Ferroptosis
NRF2 mediates iron homeostasis
In 1997, Moi et al. [49] identified NRF2 for the first time in K562 cells, where it acted on the sequences of the transcription factors AP-1/NF-E2, which are located at hypersensitive site 2 in the beta-globin locus control region. NRF2 plays an important role in erythropoiesis by inducing the expression of transcription factors. The involvement of NRF2 in the synthesis and catabolism of ferroheme in erythrocytes involves iron utilization. Therefore, the normal effects of NRF2 on the transcriptional expression of various downstream molecules ensure the normal synthesis and catabolism of ferroheme and erythrocytes and maintain intracellular iron homeostasis. NRF2 may influence the biochemical synthesis of ferroheme involving intracellular divalent iron utilization by affecting the expression of genes such as ABCB6 and FECH. In 2009, Hübner et al. [85-86] while studying the smoking-responsive genes regulated by NRF2, reported that NRF2 upregulates ABCB6 expression. ABCB6 is essential for the translocation of porphyrins, including coproporphyrinogen III, from the cytosol to the mitochondria and is ultimately involved in ferroheme synthesis [87]. The NRF2-ABCB6 axis is also activated under a variety of abnormal conditions, such as exposure to inorganic arsenic. Whereas, an increased expression of ABCB6 is observed when the NRF2 pathway is activated [88]. This relationship was previously verified in hepatocytes [89]. In a recent study, it was found that, in addition to increased NRF2 expression, the expression of ABCB6 also increased during the inhalation of conventional tobacco and e-cigarettes. [90] Therefore, the involvement of the NRF2-ABCB6 axis in heme synthesis and maintenance of iron homeostasis was ascertained [91]. In addition, FECH, a ferro-chelatase is involved in the final step of heme production [92]. Wu et al. [91] found that FECH is a target of NRF2 and can promote NRF2 expression.
As only a small fraction of the daily source of iron in the body and cells comes from intestinal absorption (1-2 mg/day) and the vast majority comes from the metabolism of senescent erythrocytes (30 mg/day) [93], the maintenance of iron homeostasis is particularly important in erythrocyte catabolism. NRF2 is not only involved in the intracellular synthesis of ferroheme using divalent iron but also maintains iron homeostasis by regulating various pathways to facilitate iron transfer. The target genes involved in this process include HMOX-1, HRG1, AMBP, BLVRA, and BLVRB. Among these, the NRF2-HMOX-1 axis is the most representative one together with its research value on peroxidation damage. In macrophages, senescent erythrocytes are degraded to release ferroheme, which is then degraded into free iron ions and biliverdin by the HMOX-1 enzyme (HO-1). Subsequently, the free iron ions enter the LIP and are utilized, or transported out of the cell. The aforementioned reactions mediated by HO-1 are enhanced in the presence of oxidative stress [94]. However, because HO-1 increases the production of free intracellular iron ions, it negatively regulates ferroptosis. HO-1 inhibits intracellular iron accumulation and induces peroxidative damage. In 2021, Tang et al. [95] determined the specific inhibitory effect of HO-1 overexpression on ferroptosis by directly knocking out HO-1 or using the HO-1 inhibitor ZnPP in a retinal pigment epithelium (RPE) degeneration model and suggested that HO-1 could inhibit the onset of ferroptosis by suppressing intracellular iron accumulation and ROS production. In 2022, Ma et al. [84] found that HO-1 could inhibit ferroptosis by a similar mechanism in a model of Mycobacterium-infected macrophages, and it could also induce increased expression of GPX4 to inhibit ferroptosis. In addition, HO-1 inhibits ferroptosis by upregulating FHC to increase iron ion storage and resists peroxidative damage [96]. Notably, BACH1, a member of the CNC family, suppresses the expression of HMOX1, which is detrimental to iron homeostasis, whereas the product of BLVRA, another target gene of NRF2, suppresses the effect of BACH1 [97]. However, HO-1 has dual regulatory effects on ferroptosis, as it increases the intracellular iron concentration and releases CO. Yuan et al. [98] found that nimodipine application reduced HO-1 levels while maintaining iron homeostasis. Han et al. [99] found that the natural monomer luteolin could trigger ferroptosis through excessive upregulation of HO-1 expression and activation of LIP in clear cell renal cell carcinoma. In summary, recent studies have been controversial regarding the regulatory effects of HO-1 on ferroptosis [100-107], suggesting that the regulatory role of HO-1 in ferroptosis and related pathways is complex. NRF2 facilitates the maintenance of intracellular iron homeostasis and prevents ferroptosis by upregulating HO-1 expression.
We have previously reported that NRF2 is involved in the metabolic utilization of iron by affecting the expression of various genes, therefore maintaining iron homeostasis. LIP is a central concept in iron homeostasis and ferroptosis. In response to various factors, LIP levels are elevated, which increases ROS production and ultimately leads to lipid peroxidation and ferroptosis. Moreover, NRF2 is involved in multiple pathways that regulate LIP levels, including FTL, FTH1, and FPN1. NRF2 can also affect the expression of PIR, and the regulation of LIP by this pathway involves crosstalk with the NF-κB pathway. FTL and FTH1 are the light and heavy chains of ferritin, respectively, and are the key components of ferritin [108]. Ferritin is important for the intracellular storage and sequestration of iron, and increased ferritin synthesis facilitates the inhibition of LIP elevation, reduction of ROS production, and inhibition of ferroptosis [109]. When ferritin is degraded, such as through ferritin phagocytosis involving various pathways, the opposite effects occur [110-113]. NRF2 regulation of FTL and FTH1 expression was first discovered in 2003 by Pietsch et al. [114] found that in the presence of elevated LIP, NRF2 upregulated FTL and FTH1 expression, interfering with ROS production by synthesizing more ferritin to deplete the free iron pool. Selvakumar et al. [115] reported that the application of 1-methyl-4-phenylpyridinium to microglia obtained from individuals with Parkinson's disease promoted the entry of NRF2 into the nucleus, which eventually led to ferritin activation and reduced ROS production. Recent studies have shown that ferritin plays a negative regulatory role. Sze et al. [116] found that ferritin inhibits estradiol biosynthesis in ovarian granulosa cells in vitro by upregulating NF-κB and inducible nitric oxide synthase (iNOS) and reported that NRF2 downregulation can lead to a decrease in GPX4, suggesting that a more complex regulatory mechanism is involved. In contrast, FPN1 is an important molecule for intestinal iron absorption and the translocation of intracellular iron out of the cell and is expressed in intestinal epithelial cells, macrophages, and hepatocytes [117]. It has been well established that FPN1 maintains iron homeostasis and resists ferroptosis during oxidative stress. Lu et al. [118] found that this mechanism inhibits ferroptosis in nucleus pulposus cells and ameliorates intervertebral disc degeneration. Similarly, the use of USP35 to maintain FPN1 protein stability has an anti-ferroptotic effect [119]. FPN1 expression is closely related to the CNC family through the joint participation of heme and HO-1; BACH1 mainly represses its transcription, whereas NRF2 plays an activating role. The intrinsic mechanism may involve the binding of BACH1 to the related DNA sequence to repress transcription; upon exposure to heme, BACH1 is replaced by NRF2, BACH1 is inactivated, and FPN1 is activated [120-122]. As described above, cells maintain iron homeostasis and resist ferroptosis through NRF2-regulated ferritin storage and extracellular transfer of FPN1 in response to elevated LIP. Under the regulation of NRF2, elevation of LIP can synergistically trigger other pathways, including the NF-κB pathway, to maintain iron homeostasis. While identifying NRF2-regulated genes responsiveness to smoking, Hübner et al. [85] found that NRF2 upregulates the expression of PIR. PIR is a highly evolutionarily conserved non-heme iron-containing member of the cupin superfamily, whose oxidized form (bound to Fe3+ ions) binds to the RelA (P65) subunit and enhances the activity of the transcription factor NF-κB [123]. The NF-κB pathway is also important for inhibiting ferroptosis [40,124].
Moreover, new discoveries have recently been made on NRF2 target genes related to iron metabolism. In 2023, Anandhan et al. [125] found that HERC2 expression decreased after knockout of NRF2. As HERC2 acts as an E3 ubiquitin ligase for both FBXL5 and NCOA4, a decrease in HERC2 affects the HERC2-FBXL5-IRP1/2-FTH1/FTL pathway, resulting in reduced ferritin synthesis and increased levels of free intracellular iron ions, ultimately causing ferroptosis. TBXAS1 is also considered an NRF2 target gene [86]. TBXAS1 was initially thought to be the product of TXAS, which produces TXA2 in the AA cascade [126]. However, Minami et al. [127] found that TBXAS1 expression is important for maintaining iron homeostasis, and in malignant mesothelioma cells, the downregulation of TBXAS1 expression can cause an imbalance in iron homeostasis or tumorigenesis. Importantly, NRF2 has recently been suggested as a molecular sensor of iron-induced oxidative stress. NRF2 drives the expression of BMP6 in liver sinusoidal endothelial cells, thereby increasing hepcidin synthesis [128-130], which is essential for maintaining iron homeostasis. Besides blocking the extracellular export of iron via FPN1, it is involved in crosstalk with the IRE/IRP pathways to regulate FPN1 expression [131] and binds to iron-bound FPN1 for targeted degradation [132]. The NRF2-hepcidin-FPN1 axis is recognized as an iron homeostasis maintenance pathway. In summary, NRF2 is involved in the maintenance of intracellular iron homeostasis, including heme- and iron-related synthesis, catabolism and the maintenance of low LIP levels. Under normal conditions, NRF2 prevents iron disorders and ferroptosis.
NRF2 mediates system Xc--GSH/GPX4
Ferroptosis is not only closely related to iron metabolism but is also inextricably linked to the antioxidant capacity of the system Xc--GSH-GPX4 axis. System Xc- is an antiporter system consisting of SLC7A11 and SLC3A2 [133,134], which transports glutamate out of the cell in a 1:1 ratio while transporting cystine (the oxidized form of cysteine) into the cell. After entering the cell, cystine can also be converted to cysteine by TRR1 or GSH by GCL. Cysteine and glutamate can form γ-glutamyl cysteine glutamate, from which glutathione (GSH) [135] is ultimately formed in the presence of glycine (Gly) and GS. GLS and GS regulate the glutamate–glutamine cycle [136]. GSH is oxidized by GPX4 in a reaction that consumes intracellular free radicals. Therefore, system Xc--GSH-GPX4 is an important constituent of the antioxidation pathway and was considered an important target for oxidative damage in earlier studies on ferroptosis. Dixon et al. [1] introduced the concept of ferroptosis [1] in 2012 and reported that compounds that induce ferroptosis, such as erastin, could cause a decrease in the activity of system Xc-, leading to an increase in ROS levels. Friedmann et al. [7] proposed that system Xc--G-GSH-GPX4 is one of the key pathways in the onset of ferroptosis and that different ferroptosis-inducing substances act on various targets in this pathway, leading to a decreased ability of the cell to scavenge free radicals and resist oxidation, increased ROS production, and lipid peroxidation, which ultimately leads to ferroptosis.
System Xc-, the initiating cellular membrane transporter of the system Xc--GSH/GPX4 axis, plays a critical upstream role in combating oxidative stress and ferroptosis. NRF2 is important for enhancing and maintaining the expression of system Xc-. SLC7A11 is one of the important gene encoding system Xc- that was reported to be a target gene of NRF2 [137]. Upon the induction of NRF2 by H2O2, an increase in SLC7A11 mRNA and protein levels and an increase in system Xc- expression were observed, resulting in increased intracellular GSH levels, resistance to oxidative stress, and ferroptosis.
Importantly, NRF2 is the main transcription factor that regulates GSH metabolism. GCL is the rate-limiting enzyme involved in GSH biosynthesis, which has two subunits: GCLC and GCLM. The regulation of GCLC by NRF2 has been confirmed at the promoter level. NRF2 acts on ARE4 after binding to proteins such as JUN or MAF [138,139]. ARE4, one of the key promoter regions of GCLC [140], is located approximately 3.1 kb upstream of the distal transcription start site of the ARE element [141], and activation of ARE4 eventually enhances the transcription of GCLC. Interestingly, ARE4 activation can also induce the expression of naphthoflavone, which can transactivate GCLC expression [139,141]. Importantly, GCLC activation is associated with NRF2, but NRF1 also plays an important role in promoting GCLC transcription. The knockout of either NRF1 or NRF2 results in decreased GCLC expression, making cells more susceptible to peroxidative damage [142,143], whereas GCLC transcription is elevated when NRF1 or NRF2 is overexpressed [138,144]. Yang et al. [145] reported that the induction of GCLC expression by NRF1 and NRF2 is dependent on both AP-1 and NF-κB [145], and the inducible effect of NRF1 and NRF2 on GCLC was lost in AP-1 and NF-κB knock-out. NRF2 can also play a similar role in enhancing transcription through GCLM, which is an another subunit of GCL [138]. Moinova and Mulcahy [146] noted that NRF2 promotes the transcriptional activation of GCLM by recognizing and binding to the GCS l EpRE sequence. Moreover, Chen et al. [147] found that the induction of GCLM expression by 15d-PGJ2 requires the involvement of NRF2. GS is a key enzyme involved in GSH biosynthesis, and its transcription is regulated by NRF2. Using genetic knockout mice and chemical induction, McMahon et al. [72] reported that NRF2 controls the inducible expression of GS in a gene-specific manner. This regulation is also dependent on transcriptional regulation of the GS promoter, which leads to increased GS transcription upon NRF2 overexpression. NRF1 is also involved in this process, although the binding sites of these two proteins differ [148,149]. In summary, NRF2 promotes the expression of various enzymes involved in GSH synthesis, including GCL and GS, thereby enhancing GSH production to counter oxidative stress and ferroptosis [150].
The antioxidant elimination of free radicals by GSH also requires the participation of GPX4, which is why a decrease in GPX4 activity is one of the factors responsible for the onset of ferroptosis. Osburn et al. [151] were the first to identify the GPX4 region as a target of NRF2. Moreover, NRF2 inhibits ferroptosis by increasing GPX4 levels [152,153]. Based on the important role of ferroptosis in chronic obstructive pulmonary disease (COPD), Zhang et al. [154] reported that NRF2 inhibits ferroptosis and delays the progression of inflammation in COPD. In conclusion, NRF2 increases and maintains the activity of the system Xc--GSH/GPX4 axis at the transcriptional level, which actively resists peroxidative damage and inhibiting ferroptosis.
Other pathways
In addition to the aforementioned pathways, recent studies have identified other pathways through which NRF2 can resist ferroptosis. Zhang et al. [155] found that the expression of PPARG/PPARγ, a lipid metabolism-related transcription factor involved in the development of multiple diseases, was repressed during endoplasmic reticulum stress in MIN6 cells, which promoted the accumulation of lipid peroxides leading to ferroptosis. Moreover, NRF2 has previously been shown to maintain and increase PPARG expression, which was significantly suppressed in NRF2 knock-out mice [156]. Li et al. [157] demonstrated that defective NRF2 would lead to reduced PPARG expression and is closely associated with hepatic steatosis [149]. Interestingly, when the expression of PPARG is increased, it also increases the expression of NRF2 and enhances the ability of the cells to resist peroxidative damage [158]. In conclusion, NRF2 can inhibit ferroptosis by increasing PPARG expression and may be involved in balancing lipid metabolism and inhibiting lipid peroxide accumulation.
Moreover, Huang et al. [159] observed increased intracellular divalent iron ions and lipid peroxide levels and decreased levels of GPX4 in AKR1C1 (a member of the aldo-keto reductase family) knock-out non-small-cell lung cancer cells. This demonstrates that AKR1C1 expression has an anti-ferroptotic effect and is closely associated with tumor cell proliferation and metastasis. In 2022, Zuo et al. [160] demonstrated that in corneal epithelial cells, activated NRF2 increased AKR1C1 expression and successfully prevented ferroptosis due to peroxidative damage in a model of dry eye disease. Wohlhieter et al. [161] observed an increase in AKR1C1 expression because of the increased NRF2 activity in KEAP1 knock-out (Table 1).
Roles of NRF1 in mediating ferroptosis
NRF2 is a molecular sensor of iron-induced oxidative stress and is activated via the KEAP1-NRF2 pathway after recognizing oxidative and electrophilic molecules. However, unlike NRF2, NRF1 is primarily localized in the endoplasmic reticulum membrane, is released and translocated to the nucleus only under stressed endoplasmic reticulum conditions, and is independent of KEAP1 regulation [162]. Regardless of the differences between the activation mechanisms both NRF1 and NRF2 have clear antioxidant and anti-ferroptotic effects.
Although NRF1 has been reported to play a role in resisting oxidative damage [143,149,163,164], only the protective role of NRF2 in the field of ferroptosis has been studied to date. However, in recent years, there have been new research advances in the anti-ferroptotic action of NRF1. As mentioned previously, in the system Xc--GSH/GPX4 axis, NRF1 and NRF2 have similar and mutually coordinated antioxidant and anti-ferroptotic effects. NRF1 and NRF2 induce GCLC expression depending on AP-1 and NF-κB [145]. However, in 2022, Forcina et al. [165] analyzed gene expression in Control1/2N cells and NFE2L1KO1/2,N cells by RNA sequencing and found that disruption of NRF1 did not significantly reduce the expression of SLC7A11, FSP1, or several GSH synthesis-related genes (GCLC, GCLM, GS), whereas a significant decrease in GPX4 protein expression was observed by western blotting. In contrast, NRF2 was observed to reduce the protein expression of GCLM and particularly GCLC, suggesting a novel viewpoint that NRF1 and NRF2 can resist ferroptosis in a mutually independent manner, whereas NRF1 resist ferroptosis mainly by maintaining GPX4 levels. This mechanistic independence was confirmed experimentally by observing that overexpression of NRF2 could compensate for NRF1 and overexpression of NRF1 could compensate for NRF2 to resist ferroptosis. In clinical conditions, the antioxidative damage and anti-ferroptotic effects of NRF1 have also been confirmed in diseases such as primary bilateral macronodular adrenal hyperplasia [166].
NRF/CNC proteins mediate ferroptosis in the occurrence and development of related diseases
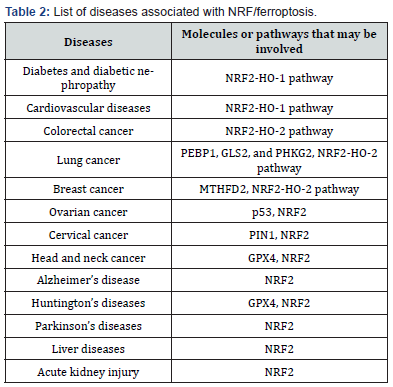
Diabetes and diabetic nephropathy
One of the important causes of diabetes is the destruction and functional impairment of pancreatic β-cells. Recent studies have shown that peroxidative damage plays an important role in pancreatic-cell damage and iron homeostasis imbalance, and ferroptosis is a key factor in the pathophysiology of this disease [167-170]. Previous studies have demonstrated the involvement of ferroptosis in the development of diabetic nephropathy (DN) [171,172]. Wang et al. [173] used salusin-β to reduce NRF2 levels and investigated the involvement of ferroptosis in DN pathogenesis and the development of diabetic nephropathy. The resulting therapeutic advances have also led to breakthroughs, as reported by Jin and Chen [174] who used umbelliferone to activate the NRF2/HO-1 pathway, inhibit ferroptosis, and successfully delay the progression of diabetic nephropathy in mice.
Cardiovascular diseases
Ferroptosis plays an important role in the development of cardiovascular diseases, including ischemia/reperfusion injury, heart failure, cardiomyopathy, and atherosclerosis [175,176]. Ferroptosis and iron homeostasis imbalance damage vascular endothelial cells, which can be protected by using ferroptosis inhibitors [177,178]. Similar effects have been observed in muscle cells, macrophages, and cardiomyocytes [179-181]. Ma et al. [182] used metformin to activate the NRF2 pathway to inhibit ferroptosis and protect muscle cells. Ferroptosis in macrophages is also highly dependent on NRF2 activation [183]. These findings suggest that the inhibition of ferroptosis can play an important role in the prevention and treatment of cardiovascular diseases. This can be achieved through the NRF2 pathway, which is considered a promising target for the treatment of cardiovascular diseases [184,185]. Song et al. [186] successfully used ulinastatin to activate NRF2 and prevent CVB3-induced acute viral myocarditis [187]. Bose et al. [187] in their study on sulforaphane reported that activation of NRF2 protected the heart from DOX toxicity, and that DOX and sulforaphane had a synergistic tumorigenic effect, suggesting that NRF2 also has a significant inhibitory effect on cardiac tumors. Importantly, NRF2 inhibition of ferroptosis improved the prognosis after myocardial infarction and ischemia/reperfusion injury [188,189] and may also relieve arrhythmias [190]. Additionally, Wang et al. [191] reported that rosuvastatin improves cardiac function and reduces myocardial hypertrophy by modulating the interaction between NRF2 and SMADs. Moreover, the NRF2-HO-1 [192] pathway has been shown to inhibit peroxidative damage and ferroptosis to delay disease progression in rheumatic arthritis (RA), suggesting that the inhibition of ferroptosis via the NRF2 pathway may have promising applications in rheumatic heart disease.
Cancers
Colorectal cancer
Colorectal cancer cells (CRC) exhibit mutations in various iron metabolism-related genes and specific tumor microenvironmental features that affect the phenotype of surrounding stromal cells. These iron metabolism- and ferroptosis-related genes have been identified successively. The immune-activated ferroptosis-related Fersig constructed by Luo et al. [193] can be used to identify the genes that can be targeted to enhance the effects of immunotherapy in patients. CRC can also activate NRF2 through pathways involving arylacetamide deacetylase expression to prevent ferroptosis in tumors [194]. This suggests that the induction of ferroptosis via the NRF2 pathway is therapeutically promising for CRC. Yang et al. [195] successfully used cetuximab to inhibit the NRF2-HO-1 axis, deactivate the classical anti-ferroptosis effect of HO-1, and promote ferroptosis in CRC. Wei et al. [192] used tagitinin C to activate NRF2 to exert the non-canonical pro-ferroptotic effect of HO-1, which kills tumor cells by promoting ferroptosis in a manner similar to that of erastin. Gao et al. [196] successfully used lysionotin to degrade NRF2 and promote ferroptosis in CRC treatment. Oxaliplatin also promotes ferroptosis in tumor cells by inhibiting the NRF2 pathway [197]. Importantly, the inhibition of the NRF2 pathway to promote ferroptosis in CRC cells could increase the sensitivity of tumors to other therapeutic approaches, including enhanced sensitivity to oxaliplatin-based chemotherapy [192].
Lung cancer
Many lung cancer-associated ferroptosis genes have now been identified, including ALOX15, PEBP1, GLS2, and PHKG2. These ferroptosis-associated genes have been used to predict the prognosis of patients with lung cancer, demonstrating the importance of ferroptosis in lung cancer [198]. Importantly, lung cancer treatment can also promote ferroptosis through NRF2-associated pathways. Hsieh et al. [199] successfully induced ferroptosis and altered the tumor microenvironment to induce immunity to the lung cancer using an NRF2 nano-modulator. A similar effect was achieved by inhibiting NRF2 using MT1DP loaded onto folate-modified liposomes [200]. This idea can also be applied to enhance the sensitivity of tumors to existing antitumor drugs. Lou et al. [201] used ginkgetin to disrupt the NRF2/HO-1 axis and enhance the chemosensitivity of NSCLC to cisplatin.
Breast cancer
Breast cancer can be subtyped prognostically using ferroptosis-related genes. Zhang et al. [202] reported that triple-negative breast cancer could be prognostically subtyped using the ferroptosis regulator, MTHFD2. Recent studies have found that activating NRF2 and utilizing the pro-ferroptotic effects of HO-1, including the use of levistilide A and ALKBH5, have a therapeutic effect on breast cancer [203,204]. Breast cancer cells can upregulate NRF2 via NR5A2 and NCOA3, resulting in inhibition of ferroptosis and enhanced resistance of breast cancer cells to BET inhibitors [205]. These results indicate the therapeutic significance and research prospects of NRF2-based pro-ferroptosis therapy for breast cancer and reveal a new mechanism of tumor drug resistance.
Ovarian cancer
Ovarian cancer has a 5-year relative survival rate of less than 50% because of its high recurrence rate and insufficient early detection methods. The standard treatment options include debulking surgery, chemotherapy, angiogenesis inhibitors, poly ADP-ribose polymerase inhibitors, and immunotherapies. However, novel therapeutic approaches are still needed, as treatment outcomes remain unsatisfactory. Ferroptosis-related therapies have emerged as promising areas of research for the treatment of ovarian cancer [204]. For the first time, Zhang et al. [206] used P53 to induce ferroptosis in ovarian cancer cells, revealing it as a potential treatment for ovarian cancer. Similar findings have been described in other reports [207], suggesting that this therapy may increase the sensitivity of ovarian cancer to chemotherapeutic agents. Liu et al. [208] reported that sustained activation of NRF2 in ovarian cancer cells exerts antioxidant and anti-ferroptotic effects and protects tumor cell survival. Accordingly, researchers have successfully promoted ferroptosis in ovarian cancer cells using various compounds to inhibit NRF2 activity, including norcantharidin and carboxymethylated pachyman [209,210]. In conclusion, the use of NRF2 inhibitors to promote ferroptosis in ovarian cancer cells would be beneficial for improving the sensitivity of ovarian cancer cells to chemotherapy and may also provide therapeutic options with better efficacy.
Cervical cancer
Patients with early-stage cervical cancer (CC), which is the fourth most common cancer in women, have good outcomes after radical surgery. Chemotherapy, particularly cisplatin-based regimens, is the primary treatment option for patients with metastatic or recurrent CC. However, such chemotherapy regimens can cause drug resistance issues, leading to worse outcomes than expected, which has become a critical issue in the treatment of advanced CC. Currently, the NRF2-ferroptosis pathway offers a potential solution for chemoresistance. Zhang et al. [211] successfully reduced NRF2 levels in CC cells by decreasing the expression of PIN1, which not only activates the ferroptosis pathway but also sensitizes the cells to the cisplatin chemotherapy regimen.
Head and neck cancer
To address the issue of drug resistance in head and neck cancer (HNC), investigators in previous studies have proposed new therapies using GPX4 inhibitors, such as RSL3 and ML-162, to induce ferroptosis; however, drug resistance issues have emerged with this treatment. Shin et al. [212] successfully reversed resistance to GPX4 inhibitor-based anti-ferroptosis therapy in a mouse model by inhibiting NRF2 activation using trigonelline. Roh et al. [213] successfully reversed cisplatin resistance in HNC cells via NRF2 inhibition.
Other cancers
NRF2 pathway-related pro-ferroptosis therapy has emerged as a promising area of research among the antitumor therapeutic approaches. Although the research is still at an early stage, excellent breakthrough results have already emerged including ferroptosis-inducing nanocomposite for the treatment of cancers. These results provide a potential solution to drug resistance in a wide range of tumors band also reveals new insights about antitumor therapy using ferroptosis to eliminate tumor cells. Such studies are not limited to the aforementioned types of tumors but also include gastric, bladder, and hepatocellular carcinoma [214-217].
Neurodegenerative Diseases
Alzheimer's disease
The critical relationship between NRF2 and ferroptosis is important in numerous neurodegenerative diseases [218], among which Alzheimer’s disease (AD) is the most common. Although degenerative changes caused by aging are important factors in the development of AD, oxidative damage and an imbalance in iron homeostasis have long been known to be involved in AD [219], and sufficient evidence has confirmed that a dysfunctional NRF2 pathway and ferroptosis are part of the pathological mechanisms underlying AD [185]. Dysfunction of the NRF2 pathway can occur by a decrease in NRF2 expression or a decrease in NRF2 nuclear translocation and action on ARE elements. Youssef et al. [220] observed a decrease in NRF2 mRNA levels in a mouse model of AD, whereas defects in NRF2 nuclear translocation and ARE binding in AD were reported in previous studies [221]. Therefore, inhibition of ferroptosis by increasing the function of the NRF2 pathway has become a new frontier in AD therapy. Blair et al. [222] successfully used the clinically approved drug dimethyl fumarate (DMF) in multiple sclerosis to enhance NRF2 expression in a clinical setting with good efficacy. Recent studies have shown that DMF may improve AD to some extent [223], suggesting that it could be a potential drug for AD. Another drug with a similar mechanism of action is 4-octyl itaconate [224]. Additionally, recent studies have found that a range of plant secondary metabolites, including carnosic acid, which has already entered clinical trials and is now considered to be milder than DMF, can activate NRF2 to inhibit ferroptosis and act as AD therapeutics [225,226]. Moreover, diacetyl-bis (4-methyl-3-thiosemicarbazonato) copper (II), an AD therapeutic currently used in phase I clinical trials, was found to favor the activation of the NRF2 pathway [227]. In summary, activating and maintaining the NRF2 pathway to inhibit ferroptosis is a novel strategy for AD treatment. Many treatment methods are under clinical trials and are expected to be implemented soon.
Huntington’s diseases
Huntington’s disease (HD) was first described by George Huntington in 1872, it is an autosomal dominant late-onset neurodegenerative disorder. The main cause of HD is polymorphic sequence of three CAG nucleotides in IT15 gene exon 1, which had been reported previously [228-230]. But now, Ferroptosis is also thought to be one of the important causes of HD [231]. According to research of Skouta et al. [232], inhibitors of ferroptosis, such as ferrostatinis and liproxstatins, are protective in model of Huntington’s diseases. In addition, similar conclusions were obtained through Leng et al. [233] GSEA as well as pathway enrichment analysis. In 2019, Mi et al. [234] provided a review of the role of ferroptosis in Huntington's disease, and they listed the manifestations of ferroptosis that have been reported in previous literature in animal models and patient cohorts. Surprisingly in patients with HD, high lipid peroxidation and low GSH plasma levels have been found [235]. Which consequently lead decrease of GPX4 [234], most importantly the imbalance in cell homeostasis leading to the process of ferroptosis [231]. However, at present, there is no effective treatment for HD except palliative care [229]. As mentioned above, the increase of ROS leads to the increase of lipid peroxidation level, further the decrease of GSH, and finally the decrease of GPX4 promotes the ferroptosis process. This series of intracellular molecule imbalance leads to the occurrence of Huntington’s disease [231,236]. Luckily, it is reported that NRF2 has the role of regulating the ferroptosis process, so it may become a new treatment scheme for HD [185].
Parkinson’s diseases
Parkinson disease (PD) is the most common neurodegenerative disease that affects movement [237], characterized by the gradual loss of dopaminergic neurons in the substantia nigra (SN), resulting in decreased dopamine secretion [238-241]. Ramsey and his colleagues [221] found elevated levels of Nrf2 in the nucleus of neurons in the substantia nigra of human Parkinson's disease, and subsequently found in numerous autopsy studies that NRF2 has a higher nuclear transposition in the brains of Parkinson's disease patients, as well as an increase in some NRF2 regulatory genes [242-244]. In 2012, NRF2-/-/α-Syn mouse model was established by Lastres-Becker et al. [245], using this mouse model, researchers also found that NRF2-/-/α-Syn mice exhibited pathological features such as increased loss of dopaminergic neurons in the substantia nigra, dendritic atrophy, protein aggregation and increased neuroinflammation, and MPTP and 6-OHDA were also confirmed in subsequent studies to induce an increase in ROS to mimic Parkinson's disease in rodents. Conversely, transplantation of NRF2-expressing astrocytes into the striatum of wild-type mice reduced the susceptibility of mice to 6-OHDA and MPTP [246-249]. It can be seen that NRF2 plays a protective role in Parkinson's disease. In addition, some studies have shown that NRF2 has the function of regulating mitochondrial quality control and balance, and moderate physical exercise can protect mice receiving 6-OHDA treatment through NRF2 neuroprotective activity, thereby facilitating the activation of mitochondrial biogenesis and preventing the development of Parkinson's disease [250-252]. Based on the study of the pathogenesis of NRF2 in Parkinson's disease, many scholars have been committed to developing activators of NRF2 to treat Parkinson's disease, including sulforaphane (SFN), a compound extracted from glucuronide precursors in brassica vegetables and its metabolite glucosinoside (ERN) [253], and halogenated vinyl sulfone (E)-3chloro-2-(2-((2-chlorophenyl)sulfonyl)vinyl Pyridine (9d) [254] and so on. These NRF2 agonists have been effective in cell experiments and animal model validation, but there is still a long way to go before they can be applied to clinical patients.
Liver diseases
Ferroptosis is thought to be involved in the pathogenesis and development of a variety of acute, chronic, malignant, and benign liver diseases. Ferroptosis-related pathways have emerged as potential new avenues for the treatment of liver diseases [255]. One of the methods of regulating ferroptosis involves the NRF2 pathway. Yang et al. [256] used maresin1 in mice to activate NRF2 to counter liver damage caused by ferroptosis and successfully achieved protection. Similarly, Pan et al. [257] have also reported delayed disease progression in liver fibrosis by activating the NRF2 pathway. Similar concepts have been supported in the studies of acute liver injury, chronic liver failure, drug-induced liver injury, and nonalcoholic fatty liver disease [176,258-261].
Acute kidney injury
Studies have suggested that ferroptosis resistance can improve the prognosis of acute kidney injury (AKI), and the activation of the NRF2 pathway is an important pathway to be exploited. Hu et al. [262] used leonurine to activate the NRF2 pathway to resist ferroptosis in cells and mice, and observed protection against cisplatin-induced AKI. Similar results have been observed in studies using DMF and melatonin [263-265]. Recent studies have used this concept to provide a prophylactic treatment for AKI. Qiongyue et al. [266] used irisin to activate the NRF2-based anti-ferroptotic pathway in mice and successfully prevented sepsis-induced AKI. This reveal the new method to utilize NRF2 pathway for inducing prophylactic resistance to ferroptosis to prevent AKI and has important clinical implications.
Other diseases
In addition to the aforementioned diseases, many other diseases can be treated with NRF2-based anti-ferroptotic therapies with good outcomes. Friedreich's ataxia is a neurodegenerative disease that lacks an effective cure. La Rosa et al. [267] successfully exploited NRF2 activation in a mouse model to counteract ferroptosis and disease progression. Moreover, the NRF2 protective function in COPD can also be activated by agents such as dihydroquercetin, suggesting the application of this pathway in cases of chronic inflammation [268]. Interestingly, the use of NRF2 against ferroptosis has been shown to have ameliorative behavioral effects on psychiatric disorders, including anxiety and depression [269]. In addition, the use of NRF2-based protection against ferroptosis has shown satisfactory progress in the study of various diseases, including ulcerative colitis, osteoarthritis, acute lung injury, and preeclampsia, both in animal models and in vitro [269-272] (Table 2).
Conclusion
Ferroptosis, a novel form of PCD, is characterized by an imbalance between iron homeostasis and lipid peroxidation. A variety of mechanisms related to the disease pathogenesis and development and a complex regulatory network underlie this form of cell death. The NRF/CNC family constitutes a regulatory network mainly centered on NRF1 and NRF2, which effect resistance to ferroptosis. They contribute to the maintenance of iron homeostasis and the antioxidant capacity of system Xc--GSH/GPX4, and function in conjunction with other regulatory pathways. This has profoundly influenced clinical research and the prospects for treating diseases, including tumors, and various acute and chronic diseases. New ideas and protocols have been developed using the NRF-ferroptosis pathway, which have been validated in cellular and animal models, and several drugs have entered clinical trials. Although the pathways regulating ferroptosis by the NRF/CNC family have not yet been fully elucidated, this mechanism is poised to become a new tool for clinicians and a new hope for patients. Future studies are needed to reveal the complex regulatory pathway behind this mechanism and to verify the importance of the NRF-ferroptosis pathway in a clinical context.
References
- Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, et al. (2012) Ferroptosis: An Iron-Dependent Form of Nonapoptotic Cell Death, Cell 149(5): 1060-1072.
- Yagoda N, Von Rechenberg M, Zaganjor E, Bauer AJ, Yang WS, et al. (2007) RAS–RAF–MEK-dependent oxidative cell death involving voltage-dependent anion channels, Nature 447: 865-869.
- Yang WS, Stockwell BS (2008) Synthetic Lethal Screening Identifies Compounds Activating Iron-Dependent, Nonapoptotic Cell Death in Oncogenic-RAS-Harboring Cancer Cells, Chem Biol 15(3): 234-245.
- Majno G, Joris I (1995) Apoptosis, Oncosis, and Necrosis 146(1): 3-15.
- Traganos F, Seiter K, Feldman E, Halicka HD, Darzynkiewicz Z (1996) Induction of Apoptosis by Camptothecin and Topotecan, Ann. N. Y. Acad. Sci 803: 101-110.
- Dolma S, Lessnick SL, Hahn WC, Stockwell BR (2003) Identification of genotype-selective antitumor agents using synthetic lethal chemical screening in engineered human tumor cells, Cancer Cell 3(3): 285-296.
- Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, et al. (2014)Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice, Nat. Cell Biol 16(12): 1180–1191.
- Bogdan AR, Miyazawa M, Hashimoto K, Tsuji Y (2016) Regulators of Iron Homeostasis: New Players in Metabolism, Cell Death, and Disease, Trends Biochem. Sci 41(3): 274-286.
- Frazer DM, Anderson GJ (2014) The regulation of iron transport: The Regulation of Iron Transport, BioFactors 40(2): 206-214.
- Gao M, Monian P, Quadri N, Ramasamy R, Jiang X (2015) Glutaminolysis and Transferrin Regulate Ferroptosis, Mol. Cell 59(2): 298–308.
- Gammella E, Recalcati S, Rybinska I, Buratti P, Cairo G (2015) Iron-Induced Damage in Cardiomyopathy: Oxidative-Dependent and Independent Mechanisms, Oxid. Med. Cell. Longev 2015: 1-10.
- Dixon SJ, Stockwell BR (2014) The role of iron and reactive oxygen species in cell death, Nat. Chem. Biol 10: 9-17.
- Doll S, Conrad M (2017)Iron and ferroptosis: A still ill‐defined liaison, IUBMB Life 69: 423-434.
- Zhang S, Xin W, Anderson GJ, Li R, Gao L, et al.(2022) Double-edge sword roles of iron in driving energy production versus instigating ferroptosis, Cell Death Dis 13: 40.
- Doll S, Proneth B, Tyurina YY, Panzilius E, Kobayashi S, et al. (2017) ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition, Nat. Chem. Biol 13: 91-98.
- Lee JY, Nam M, Son HY, Hyun K, Jang SY, (2020) Polyunsaturated fatty acid biosynthesis pathway determines ferroptosis sensitivity in gastric cancer, Proc. Natl. Acad. Sci 117(51): 32433-32442.
- Yang WS, Stockwell BR (2016)Ferroptosis: Death by Lipid Peroxidation, Trends Cell Biol 26(3): 165-176.
- Kagan VE, Mao G, Qu F, Angeli JPF, Doll S, et al. (2017) Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis, Nat. Chem. Biol 13: 81-90.
- Jiang L, Kon N, Li T, Wang SJ, Su T, et al. (2015) Ferroptosis as a p53-mediated activity during tumour suppression, Nature 520: 57-62.
- Jiang L, Hickman JH, Wang SJ, Gu W (2015) Dynamic roles of p53-mediated metabolic activities in ROS-induced stress responses, Cell Cycle 14(18): 2881-2885.
- Ou Y, Wang SJ, Li D, Chu B, Gu W (2016) Activation of SAT1 engages polyamine metabolism with p53-mediated ferroptotic responses, Proc. Natl. Acad. Sci 113(44): E6806-E6812.
- Pegg AE (2008) Spermidine/spermine-N1-acetyltransferase: a key metabolic regulator, Am. J. Physiol.-Endocrinol. Metab 294(6): E995-E1010.
- Tarangelo A, Magtanong L, Bieging-Rolett KT, Li Y, Ye J, (2018) p53 Suppresses Metabolic Stress-Induced Ferroptosis in Cancer Cells, Cell Rep 22(3): 569-575.
- Abbas T, Dutta A(2009)” p21 in cancer: intricate networks and multiple activities, Nat. Rev. Cancer 9: 400-414.
- Venkatesh D, Stockwell BR, Prives C (2020) p21 can be a barrier to ferroptosis independent of p53, Aging 12(18): 17800-17814.
- Xie Y, Zhu S, Song X, Sun X, Fan Y, et al.(2017) The Tumor Suppressor p53 Limits Ferroptosis by Blocking DPP4 Activity, Cell Rep 20(7): 1692-1704.
- Wang Y, Yang L, Zhang X, Cui W, Liu Y, (2019) Epigenetic regulation of ferroptosis by H2B monoubiquitination and p53, EMBO Rep 20(7): e47563.
- Zhang C, Lin M, Wu R, Wang X, Yang B, et al. (2011) a p53 target gene, mediates the role of p53 in glucose metabolism and the Warburg effect, Proc. Natl. Acad. Sci 108(39): 16259-16264.
- Lu J, Xu F, Lu H (2020) LncRNA PVT1 regulates ferroptosis through miR-214-mediated TFR1 and p53, Life Sci 260: 118305.
- Olivero CE, Martínez-Terroba E, Zimmer J, Liao C, Tesfaye E (2020) p53 Activates the Long Noncoding RNA Pvt1b to Inhibit Myc and Suppress Tumorigenesis, Mol. Cell 77(4): 761-774.e8.
- Liu J, Zhang C, Wang J, Hu W, Feng Z (2020) The Regulation of Ferroptosis by Tumor Suppressor p53 and its Pathway, Int. J. Mol. Sci 21(21): 8387.
- Gaschler MM, Andia AA, Liu H, Csuka JM, Hurlocker B, et al.(2018) FINO2 initiates ferroptosis through GPX4 inactivation and iron oxidation, Nat. Chem. Biol 14: 507-515.
- Shimada K, Skouta R, Kaplan A, Yang WS, Hayano M, et al. (2016) Global survey of cell death mechanisms reveals metabolic regulation of ferroptosis, Nat. Chem Biol 12: 497-503.
- Kryukov GV, Castellano S, Novoselov SV, Lobanov AV, Zehtab O, et al. (2003) Characterization of Mammalian Selenoproteomes, Science 300: 1439-1443.
- Warner GJ, Berry MJ, Moustafa ME, Carlson BA, Hatfield DL, et al. (2000) Inhibition of Selenoprotein Synthesis by Selenocysteine tRNA[Ser]Sec Lacking Isopentenyladenosine, J. Biol. Chem 275: 28110-28119.
- Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, et al. (2014) Regulation of Ferroptotic Cancer Cell Death by GPX4, Cell 156(1-2): 317-331.
- Birbo B, Madu EE, Madu CO, Jain A, Lu Y (2021) Role of HSP90 in Cancer, Int. J. Mol. Sci 22(19): 10317.
- Peng C, Zhao F, Li H, Li L, Y. Yang, et al. (2022) HSP90 mediates the connection of multiple programmed cell death in diseases, Cell Death Dis 13: 929.
- Wu Z, Geng Y, Lu X, Shi Y, Wu G, et al. (2019) Chaperone-mediated autophagy is involved in the execution of ferroptosis, Proc. Natl. Acad. Sci 116: 2996-3005.
- Li S, He Y, Chen K, Sun J, Zhang L, et al. (2021) RSL3 Drives Ferroptosis through NF-κB Pathway Activation and GPX4 Depletion in Glioblastoma, Oxid. Med. Cell. Longev 2021: 1-10.
- Zaragozá R, García C, Rus AD, Pallardó FV, Barber T, et al. (2003) Inhibition of liver trans-sulphuration pathway by propargylglycine mimics gene expression changes found in the mammary gland of weaned lactating rats: role of glutathione, Biochem. J 373: 825-834.
- Chen H, Wang C, Liu Z, He X, Tang W, et al. (2022) Ferroptosis and Its Multifaceted Role in Cancer: Mechanisms and Therapeutic Approach, Antioxid. Basel Switz 11(8): 1504.
- Andrews NC, Erdjument-Bromage H, Davidson MB, Tempst P, Orkin SH (1993) Erythroid transcription factor NF-E2 is a haematopoietic-specific basic–leucine zipper protein, Nature 362: 722-728.
- Chan YJ, Cheung MC, Moi P, Chan K, Kan Y (1995) Chromosomal localization of the human NF-E2 family of bZIP transcription factors by fluorescence in situ hybridization, Hum. Genet 95(3): 265-269.
- Chan JY, Han XL, Kan YWB (1993) Cloning of Nrf1, an NF-E2-related transcription factor, by genetic selection in yeast., Proc. Natl. Acad. Sci 90(23): 11371-11375.
- Chan JY, Han XL, Kan YW(1993) Isolation of cDNA encoding the human NF-E2 protein., Proc. Natl. Acad. Sci 90(23): 11366-11370.
- Kobayashi A, Ito E, Toki T, Kogame K, Takahashi S, et al. (1999) Molecular Cloning and Functional Characterization of a New Cap’n’ Collar Family Transcription Factor Nrf3, J. Biol. Chem 274(10): 6443-6452.
- Kobayashi M, Itoh K, Suzuki T, Osanai H, Nishikawa K, et al. (2002) Identification of the interactive interface and phylogenic conservation of the Nrf2-Keap1 system: Conserved regulation of cytoprotective genes, Genes Cell 7(8): 807-820.
- Moi P, Chan K, Asunis I, Cao A, Kan YE (1994) Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the beta-globin locus control region., Proc. Natl. Acad. Sci 91(21): 9926-9930.
- Ney PA, Andrews NC, Jane SM, Safer B, Purucker ME, et al. (1993) Purification of the Human NF-E2 Complex: cDNA Cloning of the Hematopoietic Cell-Specific Subunit and Evidence for an Associated Partner, Mol. Cell. Biol 13(9): 5604-5612.
- Dhakshinamoorthy S, Jain AK, Bloom DA, Jaiswal AK (2005) Bach1 Competes with Nrf2 Leading to Negative Regulation of the Antioxidant Response Element (ARE)-mediated NAD(P)H:Quinone Oxidoreductase 1 Gene Expression and Induction in Response to Antioxidants, J. Biol. Chem 280(17): 16891-16900.
- Motohashi H, O Connor T, Katsuoka F, Engel JD, Yamamoto M (2002) Integration and diversity of the regulatory network composed of Maf and CNC families of transcription factors, Gene 294(1-2): 1-12.
- Muto A (1998) Identification of Bach2 as a B-cell-specific partner for small Maf proteins that negatively regulate the immunoglobulin heavy chain gene 3’ enhancer, EMBO J 17(19): 5734-5743.
- Ohtsubo T, Kamada S, Mikami T, Murakami H, Tsujimoto Y (1999) Identification of NRF2, a member of the NF-E2 family of transcription factors, as a substrate for caspase-3(-like) proteases, Cell Death Differ 6(9): 865-872.
- Oyake T, Itoh K, Motohashi H, Hayashi N, Hoshino H, et al. (1996)Bach Proteins Belong to a Novel Family of BTB-Basic Leucine Zipper Transcription Factors That Interact with MafK and Regulate Transcription through the NF-E2 Site, Mol. Cell. Biol16(11): 6083-6095.
- Wang W, Kwok AM, Chan JY (2007) The p65 Isoform of Nrf1 Is a Dominant Negative Inhibitor of ARE-mediated Transcription, J. Biol. Chem 282(34): 24670-24678.
- Sykiotis GP, Bohmann D (2010) Stress-Activated Cap’n’collar Transcription Factors in Aging and Human Disease, Sci. Signal 3.
- Cullinan SB, Gordan JD, Jin J, Harper JW, Diehl JA(2004) The Keap1-BTB Protein Is an Adaptor That Bridges Nrf2 to a Cul3-Based E3 Ligase: Oxidative Stress Sensing by a Cul3-Keap1 Ligase, Mol. Cell. Biol 24(19): 8477-8486.
- Eggler AL, Liu G, Pezzuto JM, Van Breemen RB, Mesecar AD (2005) Modifying specific cysteines of the electrophile-sensing human Keap1 protein is insufficient to disrupt binding to the Nrf2 domain Neh2, Proc. Natl. Acad. Sci 102(29): 10070-10075.
- Furukawa M, Xiong Y (2005) BTB Protein Keap1 Targets Antioxidant Transcription Factor Nrf2 for Ubiquitination by the Cullin 3-Roc1 Ligase, Mol. Cell. Biol 25(1): 162-171.
- Kobayashi A, Kang MI, Okawa H, Ohtsuji M, Zenke Y, et al. (2004) Oxidative Stress Sensor Keap1 Functions as an Adaptor for Cul3-Based E3 Ligase To Regulate Proteasomal Degradation of Nrf2, Mol. Cell. Biol 24(16): 7130-7139.
- Kobayashi A, Kang MI, Watai Y, Tong KI, Shibata T, et al.(2006)Oxidative and Electrophilic Stresses Activate Nrf2 through Inhibition of Ubiquitination Activity of Keap1, Mol. Cell. Biol 26(1): 221-229.
- Tong KI, Kobayashi A, Katsuoka F, Yamamoto M (2006) Two-site substrate recognition model for the Keap1-Nrf2 system: a hinge and latch mechanism, Biol. Chem 387(10-11): 1311-1320.
- Zhang DD, Lo SC, Cross JV, Templeton DJ, Hannink M (2004) Keap1 Is a Redox-Regulated Substrate Adaptor Protein for a Cul3-Dependent Ubiquitin Ligase Complex, Mol. Cell. Biol 24(24): 10941-10953.
- Lee JM, Calkins MJ, Chan K, Kan YW, Johnson JA (2003) Identification of the NF-E2-related Factor-2-dependent Genes Conferring Protection against Oxidative Stress in Primary Cortical Astrocytes Using Oligonucleotide Microarray Analysis, J. Biol. Chem 278(14): 12029-12038.
- Itoh K, Chiba T, Takahashi S, Ishii T, Igarashi K, et al. (1997) An Nrf2/Small Maf Heterodimer Mediates the Induction of Phase II Detoxifying Enzyme Genes through Antioxidant Response Elements, Biochem. Biophys. Res. Commun 236(2): 313-322.
- Motohashi H, Katsuoka F, Engel JD, Yamamoto M (2004) Small Maf proteins serve as transcriptional cofactors for keratinocyte differentiation in the Keap1–Nrf2 regulatory pathway, Proc. Natl. Acad. Sci 101(17): 6379-6384.
- Yamamoto M, Kensler TW, Motohashi H (2018) The KEAP1-NRF2 System: a Thiol-Based Sensor-Effector Apparatus for Maintaining Redox Homeostasis, Physiol. Rev 98(3): 1169-1203.
- Hayes JD, Chanas SA, Henderson CJ, McMahon M, Sun C, et al. (2000) The Nrf2 transcription factor contributes both to the basal expression of glutathione S-transferases in mouse liver and to their induction by the chemopreventive synthetic antioxidants, butylated hydroxyanisole and ethoxyquin, Biochem. Soc. Trans 28(2): 33-41.
- Kansanen E, Kuosmanen SM, Leinonen H, Levonen AL(2013) The Keap1-Nrf2 pathway: Mechanisms of activation and dysregulation in cancer, Redox Biol 1(1): 45-49.
- Lee J, Li J, Johnson DA, Stein TD, Kraft AD (2005) Nrf2, a multi‐organ protector? FASEB J. 19(9): 1061-1066.
- McMahon M, Itoh K, Yamamoto M, Chanas SA, Henderson CJ, et al. The Cap ‘n’ Collar Basic Leucine Zipper Transcription Factor Nrf2 (NF-E2 p45- related Factor 2) Controls Both Constitutive and Inducible Expression of Intestinal Detoxification and Glutathione Biosynthetic Enzymes, (n.d.).
- Motohashi H, Yamamoto M (2004) Nrf2–Keap1 defines a physiologically important stress response mechanism, Trends Mol. Med 10(11): 549-557.
- Thimmulappa RK, Mai KH, Srisuma S, Kensler TW, Yamamoto M, et al. Identification of Nrf2-regulated Genes Induced by the Chemopreventive Agent Sulforaphane by Oligonucleotide Microarray, (n.d.).
- Lo SC, Li X, Henzl MT, Beamer LJ, Hannink M (2006) Structure of the Keap1:Nrf2 interface provides mechanistic insight into Nrf2 signaling, EMBO J 25(15): 3605-3617.
- Kwak MK, Wakabayashi N, Itoh K, Motohashi H, Yamamoto M, et al. (2003) Modulation of Gene Expression by Cancer Chemopreventive Dithiolethiones through the Keap1-Nrf2 Pathway, J. Biol. Chem 278: 8135-8145.
- Kobayashi EH, Suzuki T, Funayama R, Nagashima T, Hayashi M, et al. (2016) Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription, Nat. Commun 7: 11624.
- Lee JM, Shih AY, Murphy TH, Johnson JA (2003) NF-E2-related Factor-2 Mediates Neuroprotection against Mitochondrial Complex I Inhibitors and Increased Concentrations of Intracellular Calcium in Primary Cortical Neurons, J. Biol. Chem 278(39): 37948-37956.
- Murakami S, Suzuki T, Harigae H, Romeo PH, Yamamoto M (2017) NRF2 Activation Impairs Quiescence and Bone Marrow Reconstitution Capacity of Hematopoietic Stem Cells, Mol. Cell Biol 37(19): e00086-17.
- Wati SM, Matsumaru D, Motohashi H(2020) NRF2 pathway activation by KEAP1 inhibition attenuates the manifestation of aging phenotypes in salivary glands, Redox Biol 36: 101603.
- Sankaranarayanan K, Jaiswal AK (2004) Nrf3 Negatively Regulates Antioxidant-response Element-mediated Expression and Antioxidant Induction of NAD(P)H:Quinone Oxidoreductase1 Gene, J. Biol. Chem 279(49): 50810-50817.
- Katsuoka F, Motohashi H, Engel JD, Yamamoto M (2005) Nrf2 Transcriptionally Activates the mafG Gene through an Antioxidant Response Element, J. Biol. Chem 280(6): 4483-4490.
- Kwak MK, Itoh K, Yamamoto M, Kensler TW(2002) Enhanced Expression of the Transcription Factor Nrf2 by Cancer Chemopreventive Agents: Role of Antioxidant Response Element-Like Sequences in the nrf2 Promoter, Mol. Cell. Biol 22(9): 2883-2892.
- Lee OH, Jain AK, Papusha V, Jaiswal AK (2007) An Auto-regulatory Loop between Stress Sensors INrf2 and Nrf2 Controls Their Cellular Abundance, J. Biol. Chem 282(50): 36412-36420.
- Hübner RH, Schwartz JD, De BP, Ferris B, Omberg L, et al. (2009) Coordinate Control of Expression of Nrf2-Modulated Genes in the Human Small Airway Epithelium Is Highly Responsive to Cigarette Smoking, Mol Med 15: 203-219.
- Campbell MR, Karaca M, Adamski KN, Chorley BN, Wang X, et al. (2013) Novel Hematopoietic Target Genes in the NRF2-Mediated Transcriptional Pathway, Oxid. Med. Cell. Longev 20131-12.
- Krishnamurthy PC, Du G, Fukuda Y, Sun D, Sampath J, et al.(2006) Identification of a mammalian mitochondrial porphyrin transporter, Nature 443: 586-589.
- Córdova EJ, Martínez-Hernández A, Uribe-Figueroa L, Centeno F, Morales-Marín M, et al. (2014) The NRF2-KEAP1 Pathway Is an Early Responsive Gene Network in Arsenic Exposed Lymphoblastoid Cells, PLoS ONE 9: e88069.
- Chavan H, Oruganti M, Krishnamurthy P (2011) The ATP-Binding Cassette Transporter ABCB6 Is Induced by Arsenic and Protects against Arsenic Cytotoxicity, Toxicol. Sci 120(2): 519-528.
- Kaisar MA, Sivandzade F, Bhalerao A, Cucullo L (2018) Conventional and electronic cigarettes dysregulate the expression of iron transporters and detoxifying enzymes at the brain vascular endothelium: In vivo evidence of a gender-specific cellular response to chronic cigarette smoke exposure, Neurosci. Lett 682: 1-9.
- Wu KC, Cui JY, Klaassen CD (2011) Beneficial Role of Nrf2 in Regulating NADPH Generation and Consumption, Toxicol. Sci 123: 590-600.
- Khan AA, Quigley GJ(2011) Control of intracellular heme levels: Heme transporters and heme oxygenases, Biochim. Biophys. Acta BBA - Mol. Cell Res 1813: 668-682.
- Pantopoulos K, Porwal SK, Tartakoff A, Devireddy L (2012) Mechanisms of Mammalian Iron Homeostasis, Biochemistry 51: 5705-5724.
- Ma C, Wu X, Zhang X, Liu X, Deng G (2022) Heme oxygenase-1 modulates ferroptosis by fine-tuning levels of intracellular iron and reactive oxygen species of macrophages in response to Bacillus Calmette-Guerin infection, Front. Cell. Infect. Microbiol 12: 1004148.
- Tang Z, Ju Y, Dai X, Ni N, Liu Y, et al. (2021)HO-1-mediated ferroptosis as a target for protection against retinal pigment epithelium degeneration, Redox Biol 43: 101971.
- Cheng HT, Yen CJ, Chang CC, Huang KT, Chen KH, et al. (2015) Ferritin heavy chain mediates the protective effect of heme oxygenase-1 against oxidative stress, Biochim. Biophys. Acta BBA - Gen. Subj 1850(12): 2506-2517.
- Tudor C, Lerner-Marmarosh N, Engelborghs Y, Gibbs PEM, Maines MD (2008) Biliverdin reductase is a transporter of haem into the nucleus and is essential for regulation of HO - 1 gene expression by haematin, Biochem J 413(3): 405-416.
- Yuan Y, Guo JZ, Zhou QX (2008) The homeostasis of iron and suppression of HO-1 involved in the protective effects of nimodipine on neurodegeneration induced by aluminum overloading in mice, Eur. J. Pharmacol 586: 100-105.
- Han S, Lin F, Qi Y, Liu C, Zhou L, et al.(2022) HO-1 Contributes to Luteolin-Triggered Ferroptosis in Clear Cell Renal Cell Carcinoma via Increasing the Labile Iron Pool and Promoting Lipid Peroxidation, Oxid. Med. Cell. Longev 2022: 1-26.
- Fang X, Wang H, Han D, Xie E, Yang X, et al. (2019) Ferroptosis as a target for protection against cardiomyopathy, Proc. Natl. Acad. Sci 116: 2672-2680.
- Adedoyin O, Boddu R, Traylor A, Lever JM, Bolisetty S, et al. (2018) Heme oxygenase-1 mitigates ferroptosis in renal proximal tubule cells. Am J Physiol Ren Physiol 314(5): F702-F714.
- Stockwell BR, Jiang X, Gu W (2020) Emerging Mechanisms and Disease Relevance of Ferroptosis. Trends Cell Biol 30(6) 478-490.
- Totsuka k, Ueta T, Uchida T, Roggia MF, Nakagawa S, et al. (2019) Oxidative stress induces ferroptotic cell death in retinal pigment epithelial cells. Exp Eye Res 181: 316-324.
- Kwon MY, Park E, Lee SJ, Chung SW (2019) Heme oxygenase-1 accelerates erastin-induced ferroptotic cell death. Oncotarget 6(27): 24393-24403.
- Li R, Zhang J, Zhou Y, Gao Q, Wang R, et al. (2020) Transcriptome Investigation and In Vitro Verification of Curcumin-Induced HO-1 as a Feature of Ferroptosis in Breast Cancer Cells. Oxid Med Cell Longev 2020: 1-18.
- Dong H, Qiang D, Chai D, Peng J, Xia Y, et al. (2020) Nrf2 inhibits ferroptosis and protects against acute lung injury due to intestinal ischemia reperfusion via regulating SLC7A11 and HO-1. Aging 12(13): 12943-12959.
- Consoli V, Sorrenti V, Pittalà V, Greish K, D’Amico AG, et al. (2022) Heme Oxygenase Modulation Drives Ferroptosis in TNBC Cells. Int J Mol Sci 23(10): 5709.
- Zhang N, Yu X, Xie J, Xu H (2021) New Insights into the Role of Ferritin in Iron Homeostasis and Neurodegenerative Diseases. Mol Neurobiol 58(6): 2812-2823.
- Mesquita G, Silva T, Gomes AC, Oliveira PF, Alves MG, et al. (2020) H-Ferritin is essential for macrophages’ capacity to store or detoxify exogenously added iron. Sci Rep 10: 3061.
- Borkowska A, Popowska U, Spodnik J, Herman-Antosiewicz A, Woźniak M, et al. (2020) JNK/p66Shc/ITCH Signaling Pathway Mediates Angiotensin II-induced Ferritin Degradation and Labile Iron Pool Increase, Nutrients 12 (3): 668.
- Feng J, Li C, Xu R, Li Y, Hou Q, R et al. (2020) DpdtC-Induced EMT Inhibition in MGC-803 Cells Was Partly through Ferritinophagy-Mediated ROS/p53 Pathway. Oxid Med Cell Longev 2020: 9762390.
- Li L, Li H, Li Y, Feng J, Guan D, et al. (2021) Ferritinophagy-Mediated ROS Production Contributed to Proliferation Inhibition, Apoptosis, and Ferroptosis Induction in Action of Mechanism of 2-Pyridylhydrazone Dithiocarbamate Acetate. Oxid Med Cell Longev 2021: 5594059.
- Lu R, Jiang Y, Lai X, Liu S, Sun L, et al. (2021) A Shortage of FTH Induces ROS and Sensitizes RAS-Proficient Neuroblastoma N2A Cells to Ferroptosis. Int J Mol Sci 22(16): 8898.
- Pietsch EC, Chan JY, Torti FM, Torti SV (2003) Torti, Nrf2 Mediates the Induction of Ferritin H in Response to Xenobiotics and Cancer Chemopreventive Dithiolethiones. J Biol Chem 278(4): 2361-2369.
- Selvakumar GP, Ahmed ME, Raikwar SPR, Thangavel R, Kempuraj D, et al. (2019) CRISPR/Cas9 Editing of Glia Maturation Factor Regulates Mitochondrial Dynamics by Attenuation of the NRF2/HO-1 Dependent Ferritin Activation in Glial Cells. J Neuroimmune Pharmacol 14(4): 537-550.
- Sze SCW, Zhang L, Zhang S, Lin K, Tzi Bun Ng, et al. (2022) Aberrant Transferrin and Ferritin Upregulation Elicits Iron Accumulation and Oxidative Inflammaging Causing Ferroptosis and Undermines Estradiol Biosynthesis in Aging Rat Ovaries by Upregulating NF-Κb-Activated Inducible Nitric Oxide Synthase: First Demonstration of an Intricate Mechanism. Int J Mol Sci 23(20): 12689.
- Donovan A, Lima CA, Pinkus JL, Pinkus GS, Zon LI, et al. (2005) Andrews, the iron exporter ferroportin/Slc40a1 is essential for iron homeostasis. Cell Metab 1(3): 191-200.
- Lu S, Song Y, Luo R, Li S, Li G, et al. (2021) Ferroportin-Dependent Iron Homeostasis Protects against Oxidative Stress-Induced Nucleus Pulposus Cell Ferroptosis and Ameliorates Intervertebral Disc Degeneration In Vivo. Oxid Med Cell Longev 2021: 6670497.
- Tang Z, Jiang W, Mao M, Zhao J, Chen J, et al. (2021) Deubiquitinase USP35 modulates ferroptosis in lung cancer via targeting ferroportin. Clin Transl Med 11(4): e390.
- Marro S, Chiabrando D, Messana E, Stolte J, Turco E, et al. (2010) Heme controls ferroportin1 (FPN1) transcription involving Bach1, Nrf2 and a MARE/ARE sequence motif at position -7007 of the FPN1 promoter. Haematologica 95(8): 1261-1268.
- Hintze KJ, Katoh Y, Igarashi K, Theil EC (2007) Bach1 Repression of Ferritin and Thioredoxin Reductase1 Is Heme-sensitive in Cells and in Vitro and Coordinates Expression with Heme Oxygenase1, β-Globin, and NADP(H) Quinone (Oxido) Reductase1. J Biol Chem 282 (47): 34365-34371.
- Ogawa K (2001) Heme mediates derepression of Maf recognition element through direct binding to transcription repressor Bach1. EMBO J 20 (11): 2835-2843.
- Zhang Y, Knatko EV, Higgins M, Dayalan Naidu S, Smith G, et al. (2022) Pirin, an Nrf2-Regulated Protein, Is Overexpressed in Human Colorectal Tumors, Antioxidants. 11(2): 262.
- Gao J, Luo T, Wang J (2021) Gene interfered-ferroptosis therapy for cancers. Nat Commun 12(1): 5311.
- Anandhan A, Dodson M, Shakya A, Chen J, Liu P, et al. (2023) NRF2 controls iron homeostasis and ferroptosis through HERC2 and VAMP8. Sci Adv 9(5): eade9585.
- Shen RF, Tai HH (1998) Thromboxanes: Synthase and receptors. J Biomed Sci 5(3): 153-172.
- Minami T, Takigawa N, Kato Y, Kudo K, Isozaki H, et al. (2015) Downregulation of TBXAS in an iron‐induced malignant mesothelioma model. Cancer Sci 106(10): 1296-1302.
- 128 Lim PJ, Duarte TL, Arezes J, Garcia-Santos D, Hamdi A, et al. (2019) Nrf2 controls iron homoeostasis in haemochromatosis and thalassaemia via Bmp6 and hepcidin. Nat Metab 1(5): 519-531.
- Duarte TL, Talbot NP, Drakesmith H (2021) NRF2 and Hypoxia-Inducible Factors: Key Players in the Redox Control of Systemic Iron Homeostasis. Antioxid Redox Signal 35(36): 433-452.
- Kautz L, Meynard D, Monnier A, Darnaud V, Bouvet R, et al. (2008) Iron regulates phosphorylation of Smad1/5/8 and gene expression of Bmp6, Smad7, Id1, and Atoh8 in the mouse liver. Blood 112(4): 1503-1509.
- Charlebois E, Fillebeen C, Katsarou A, Rabinovich A, Wisniewski K, et al. (2022) A crosstalk between hepcidin and IRE/IRP pathways controls ferroportin expression and determines serum iron levels in mice. ELife 11: e81332.
- Billesbølle CB, Azumaya CM, Kretsch RC, AS Powers, Gonen S, et al. (2020) Structure of hepcidin-bound ferroportin reveals iron homeostatic mechanisms. Nature 586 (7831): 807-811.
- Sato H, Tamba M, Ishii T, Bannai S (1999) Cloning and Expression of a Plasma Membrane Cystine/Glutamate Exchange Transporter Composed of Two Distinct Proteins. J Biol Chem 274 (17): 11455-11458.
- Sato H, Tamba M, Kuriyama-Matsumura K, Okuno S, Bannai S (2000) Molecular Cloning and Expression of Human xCT, the Light Chain of Amino Acid Transport System x c. Antioxid. Redox Signal 2(4): 665-671.
- Lewerenz J, Hewett SJ, Huang Y, Lambros M, Gout PW, et al. (2013) The Cystine/Glutamate Antiporter System x c − in Health and Disease: From Molecular Mechanisms to Novel Therapeutic Opportunities. Antioxid Redox Signal 18(5): 522-555.
- Dringen R, Brandmann M, Hohnholt MC, Blumrich EM (2015) Glutathione-Dependent Detoxification Processes in Astrocytes, Neurochem Res 40(12): 2570-2582.
- Habib E, Linher-Melville K, Lin HX, Singh G (2015) Expression of xCT and activity of system xc- are regulated by NRF2 in human breast cancer cells in response to oxidative stress. Redox Biol 5: 33-42.
- Wild AC, Moinova HR, Mulcahy RT (1999) Regulation of γ-Glutamylcysteine Synthetase Subunit Gene Expression by the Transcription Factor Nrf2. J Biol Chem 274 (47): 33627-33636.
- Erickson AM, Nevarea Z, Gipp JJ, Mulcahy RT (2002) Identification of a Variant Antioxidant Response Element in the Promoter of the Human Glutamate-Cysteine Ligase Modifier Subunit Gene. J Biol Chem 277 (34): 30730-30737.
- Langston JW, Li W, Harrison L, Tak Yee Aw (2011) Activation of promoter activity of the catalytic subunit of γ-glutamylcysteine ligase (GCL) in brain endothelial cells by insulin requires antioxidant response element 4 and altered glycemic status: Implication for GCL expression and GSH synthesis, Free Radic Biol Med 51(1): 1749-1757.
- Mulcahy RT, Wartman MA, Bailey HH, Gipp JJ (1997) Constitutive and β-Naphthoflavone-induced Expression of the Human γ-Glutamylcysteine Synthetase Heavy Subunit Gene Is Regulated by a Distal Antioxidant Response Element/TRE Sequence. J Biol Chem 272(11): 7445-7454.
- Chan K, Han XD, Kan YW (2001) An important function of Nrf2 in combating oxidative stress: Detoxification of acetaminophen. Proc Natl Acad Sci 98 (8): 4611-4616.
- Chen L, Kwong M, Lu R, Ginzinger D, Lee C, et al. (2003) Nrf1 Is Critical for Redox Balance and Survival of Liver Cells during Development. Mol Cell Biol 23(13): 4673-4686.
- Myhrstad MCW, Husberg C, Murphy P, Nordström O, Blomhoff R, et al. (2001) TCF11/Nrf1 overexpression increases the intracellular glutathione level and can transactivate the γ-glutamylcysteine synthetase (GCS) heavy subunit promoter, Biochim. Biophys Acta 1517(2): 212-219.
- Yang H, Magilnick N, Lee C, Kalmaz D, Ou X, et al. (2005) Nrf1 and Nrf2 Regulate Rat Glutamate-Cysteine Ligase Catalytic Subunit Transcription Indirectly via NF-κB and AP-1. Mol Cell Biol 25(14): 5933-5946.
- Moinova HR, Mulcahy RT (1999) Up-Regulation of the Human γ-Glutamylcysteine Synthetase Regulatory Subunit Gene Involves Binding of Nrf-2 to an Electrophile Responsive Element. Biochem Biophys Res Commun 261(3): 661-668.
- Chen ZH, Yoshida Y, Saito Y, Sekine A, Noguchi N, et al. (2006) Induction of Adaptive Response and Enhancement of PC12 Cell Tolerance by 7-Hydroxycholesterol and 15-Deoxy-Δ12,14-Prostaglandin J2 through Up-regulation of Cellular Glutathione via Different Mechanisms. J Biol Chem 281(20): 14440–14445.
- Lee TD, Yang H, Whang J, Lu SC (2005) Cloning and characterization of the human glutathione synthetase 5′-flanking regions. Biochem J 390(2): 521-528.
- Kwong M, Kan YW, Chan JY (1999) The CNC Basic Leucine Zipper Factor, Nrf1, Is Essential for Cell Survival in Response to Oxidative Stress-inducing Agents. J Biol Chem 274(52): 37491-37498.
- Lu SC (2013) Glutathione synthesis. Biochim Biophys 1830(5): 3143-3153.
- Osburn WO, Wakabayashi N, Misra V, Nilles T, Biswal S, et al. (2006) Nrf2 regulates an adaptive response protecting against oxidative damage following diquat-mediated formation of superoxide anion. Arch Biochem Biophys 454(1): 7-15.
- Dodson M, Castro-Portuguez R, Zhang DD (2019) NRF2 plays a critical role in mitigating lipid peroxidation and ferroptosis. Redox Biol 23: 101107.
- Ma H, Wang X, Zhang W, Li H, Zhao W, et al. (2021) Melatonin Suppresses Ferroptosis Induced by High Glucose via Activation of the Nrf2/HO-1 Signaling Pathway in Type 2 Diabetic Osteoporosis. Oxid Med Cell Longev 1-18.
- Zhang Z, Fu C, Liu J, Sai X, Qin C, et al. (2021) Hypermethylation of the Nrf2 Promoter Induces Ferroptosis by Inhibiting the Nrf2-GPX4 Axis in COPD. Int J Chron Obstruct Pulmon Dis 16: 3347-3362.
- Zhang X, Jiang L, Chen H, Wei S, Yao K, et al. (2022) Resveratrol protected acrolein-induced ferroptosis and insulin secretion dysfunction via ER-stress- related PERK pathway in MIN6 cells. Toxicology 465: 153048.
- Cho HY, Gladwell W, Wang X, Chorley B, Bell D, et al. (2010) Nrf2-regulated PPARγ Expression Is Critical to Protection against Acute Lung Injury in Mice. Am J Respir Crit Care Med 182(2): 170-182.
- Li L, Fu J, Liu D, Sun J, Hou Y, et al. (2020) Hepatocyte-specific Nrf2 deficiency mitigates high-fat diet-induced hepatic steatosis: Involvement of reduced PPARγ expression. Redox Biol 30: 101412.
- Li Z, Liu T, Feng Y, Tong Y, Jia Y, et al. (2022) PPARγ Alleviates Sepsis-Induced Liver Injury by Inhibiting Hepatocyte Pyroptosis via Inhibition of the ROS/TXNIP/NLRP3 Signaling Pathway. Oxid Med Cell Longev 2022: 1269747.
- Huang F, Zheng Y, Li X, Luo H, Luo L (2021) Ferroptosis-related gene AKR1C1 predicts the prognosis of non-small cell lung cancer. Cancer Cell Int 21(1): 567.
- Zuo X, Zeng H, Wang B, Yang X, He D, et al. (2022) AKR1C1 Protects Corneal Epithelial Cells Against Oxidative Stress-Mediated Ferroptosis in Dry Eye. Investig Opthalmology Vis Sci 63(10): 3.
- Wohlhieter CA, Richards AL, Uddin F, Hulton CH, Quintanal-Villalonga À, et al. (2020) Concurrent Mutations in STK11 and KEAP1 Promote Ferroptosis Protection and SCD1 Dependence in Lung Cancer. Cell Rep 33(9): 108444.
- Wang W, Chan JY (2006) Nrf1 Is Targeted to the Endoplasmic Reticulum Membrane by an N-terminal Transmembrane Domain. J Biol Chem 281(28): 19676-19687.
- Leung L, Kwong M, Hou S, Lee C, Chan JY (2003) Deficiency of the Nrf1 and Nrf2 Transcription Factors Results in Early Embryonic Lethality and Severe Oxidative Stress. J Biol Chem 278(48): 48021-48029.
- Radhakrishnan SK, Lee CS, Young P, Beskow A, Chan JY, et al. (2010) Transcription Factor Nrf1 Mediates the Proteasome Recovery Pathway after Proteasome Inhibition in Mammalian Cells. Mol Cell 38(1): 17-28.
- Forcina GC, Pope L, Murray M, Dong W, Abu-Remaileh M, et al. (2022) Ferroptosis regulation by the NGLY1/NFE2L1 pathway. Proc Natl Acad Sci 119(11): e2118646119.
- Cavalcante IP, Rizk-Rabin M, Ribes C, Perlemoine K, Hantel C, et al. (2022) Tumor suppressor gene ARMC5 controls adrenal redox state through NRF1 turnover. Endocr Relat Cancer 29(11): 615-624.
- Elumalai S, Karunakaran U, Moon JS, Won KC (2022) Ferroptosis Signaling in Pancreatic β-Cells: Novel Insights & Therapeutic Targeting. Int J Mol Sci 23(22): 13679.
- Masuda Y, Ichii H, Vaziri ND (2013) At pharmacologically relevant concentrations intravenous iron preparations cause pancreatic beta cell death. Am J Res 6(1): 64-70.
- Huang J, Jones D, Luo B, Sanderson M, Soto J, et al. (2011) Iron Overload and Diabetes Risk: A Shift from Glucose to Fatty Acid Oxidation and Increased Hepatic Glucose Production in a Mouse Model of Hereditary Hemochromatosis. Diabetes 60(1): 80-87.
- Cooksey RC, Jones D, Gabrielsen S, Huang J, Simcox JA, et al. (2010) Dietary iron restriction or iron chelation protects from diabetes and loss of β-cell function in the obese ( ob/ob lep -/-) mouse. Am J Physiol Endocrinol Metab 298(6): E1236-E1243.
- Wang Y, Bi R, Quan F, Cao Q, Lin Y, et al. (2020) Ferroptosis involves in renal tubular cell death in diabetic nephropathy. Eur J Pharmacol 888: 173574.
- Li S, Zheng L, Zhang J, Liu X, Wu Z (2021) Inhibition of ferroptosis by up-regulating Nrf2 delayed the progression of diabetic nephropathy. Free Radic Biol Med 162: 435-449.
- Wang WJ, Jiang X, Gao CC, Chen WZ (2021) Salusin‑β participates in high glucose‑induced HK‑2 cell ferroptosis in a Nrf‑2 ‑dependent manner. Mol Med Rep 24(3): 674.
- Jin T, Chen C (2022) Umbelliferone delays the progression of diabetic nephropathy by inhibiting ferroptosis through activation of the Nrf-2/HO-1 pathway Food Chem Toxicol 163: 112892.
- Guo Y, Lu C, Hu K, Cai C, Wang W (2022) Ferroptosis in Cardiovascular Diseases: Current Status, Challenges, and Future Perspectives. Biomolecules 12(3): 390.
- Wang M, Liu CY, Wang T, Yu HM, Ouyang SH, et al. (2020) (+)-Clausenamide protects against drug-induced liver injury by inhibiting hepatocyte ferroptosis, Cell Death Dis 11(9): 781.
- Vinchi F, Porto G, Simmelbauer A, Altamura S, Passos ST, et al. (2020) Atherosclerosis is aggravated by iron overload and ameliorated by dietary and pharmacological iron restriction. Eur Heart J 41(28): 2681-2695.
- Wang Y, Tang M (2019) PM2.5 induces ferroptosis in human endothelial cells through iron overload and redox imbalance. Environ Pollut 254: 112937.
- Sampilvanjil A, Karasawa T, Yamada N, Komada T, Higashi T, et al. (2020) Cigarette smoke extract induces ferroptosis in vascular smooth muscle cells. Am J Physiol Heart Circ Physiol 318(3): H508-H518.
- Hu X, Cai X, Ma R, Fu W, Zhang C, et al. (2019) Iron‐load exacerbates the severity of atherosclerosis via inducing inflammation and enhancing the glycolysis in macrophages. J Cell Physiol 234(10): 18792-18800.
- Li W, Feng G, Gauthier JM, Lokshina I, Higashikubo R, et al. (2019) Ferroptotic cell death and TLR4/Trif signaling initiate neutrophil recruitment after heart transplantation. J Clin Invest 129(6): 2293-2304.
- Ma WQ, Sun XJ, Zhu Y, Liu NF (2021) Metformin attenuates hyperlipidaemia-associated vascular calcification through anti-ferroptotic effects. Free Radic Biol Med 165: 229-242.
- He R, Liu B, Xiong R, Geng B, Meng H, Lin W, et al. (2022) Itaconate inhibits ferroptosis of macrophage via Nrf2 pathways against sepsis-induced acute lung injury. Cell Death Discov 8: 43.
- Ge ZD, Lian Q, Mao X, Xia Z (2019) Current Status and Challenges of NRF2 as a Potential Therapeutic Target for Diabetic Cardiomyopathy. Int Heart J 60(30): 512-520.
- Abdalkader M, Lampinen R, Kanninen KM, Malm TM, Liddell JR (2018) Targeting Nrf2 to Suppress Ferroptosis and Mitochondrial Dysfunction in Neurodegeneration. Front Neurosci 12: 466.
- Song F, Kong F, Zhang H, Zhou Y, Li M (2018) Ulinastatin Protects against CVB3-Induced Acute Viral Myocarditis through Nrf2 Activation. Inflammation 41(3): 803-810.
- Bose C, Awasthi S, Sharma R, Beneš H, Hauer-Jensen M, et al. (2018) Sulforaphane potentiates anticancer effects of doxorubicin and attenuates its cardiotoxicity in a breast cancer model, Plos One 13(3): e0193918.
- Zhang M, Zhu J, Qin X, Zhou M, Zhang X, et al. (2019) Cardioprotection of Tetrahedral DNA Nanostructures in Myocardial Ischemia-Reperfusion Injury. ACS Appl Mater Interfaces 11(34): 30631-30639.
- Chen W, Chen XY, Wang Y, Wang HY, Zhou WJ, et al. (2019) Mechanism of Emulsified Isoflurane Postconditioning–Induced Activation of the Nrf2–Antioxidant Response Element Signaling Pathway During Myocardial Ischemia-Reperfusion: The Relationship With Reactive Oxygen Species. J Cardiovasc Pharmacol 73(5): 265-271.
- Enayati A, Yassa N, Mazaheri Z, Rajaei M, Pourabouk M, et al. (2018) Cardioprotective and anti-apoptotic effects of Potentilla reptans L. root via Nrf2 pathway in an isolated rat heart ischemia/reperfusion model. Life Sci 215: 216-226.
- Wang P, Luo L, Shen Q, Shi G, Mohammed A, et al. (2018) Rosuvastatin improves myocardial hypertrophy after hemodynamic pressure overload via regulating the crosstalk of Nrf2/ARE and TGF-β/ smads pathways in rat heart. Eur J Pharmacol 820: 173-182.
- Wei R, Zhao Y, Wang J, Yang X, Li S, et al. (2021) Tagitinin C induces ferroptosis through PERK-Nrf2-HO-1 signaling pathway in colorectal cancer cells. Int J Biol Sci 17(11): 2703-2717.
- Luo W, Dai W, Li Q, Mo S, Han L, et al. (2022) Ferroptosis-associated molecular classification characterized by distinct tumor microenvironment profiles in colorectal cancer. Int J Biol Sci 18(5): 1773-1794.
- Sun R, Lin Z, Wang X, Liu L, Huo M, et al. (2022) AADAC protects colorectal cancer liver colonization from ferroptosis through SLC7A11-dependent inhibition of lipid peroxidation. J Exp Clin Cancer Res 41(1): 284.
- Yang J, Mo J, Dai J, Ye C, Cen W, et al. (2021) Cetuximab promotes RSL3-induced ferroptosis by suppressing the Nrf2/HO-1 signalling pathway in KRAS mutant colorectal cancer. Cell Death Dis 12(11): 1079.
- Gao Z, Jiang J, Hou L, Ji F (2022) Lysionotin Induces Ferroptosis to Suppress Development of Colorectal Cancer via Promoting Nrf2 Degradation. Oxid Med Cell Longev 2022: 1-18.
- Liu B, Wang H (2022) Oxaliplatin induces ferroptosis and oxidative stress in HT29 colorectal cancer cells by inhibiting the Nrf2 signaling pathway, Exp. Ther. Med. 23(6): 394.
- Tabnak P, HajiEsmailPoor Z, Soraneh S (2021) Ferroptosis in Lung Cancer: From Molecular Mechanisms to Prognostic and Therapeutic Opportunities. Front Oncol 11: 792827.
- Hsieh CH, Hsieh HC, Shih FH, Wang PW, Yang LX, et al. (2021) An innovative NRF2 nano-modulator induces lung cancer ferroptosis and elicits an immunostimulatory tumor microenvironment. Theranostics 11(14): 7072-7091.
- Gai C, Liu C, Wu X, Yu M, Zheng J, et al. (2020) MT1DP loaded by folate-modified liposomes sensitizes erastin-induced ferroptosis via regulating miR-365a-3p/NRF2 axis in non-small cell lung cancer cells. Cell Death Dis 11: 751.
- Lou JS, Zhao LP, Huang ZH, Chen XY, Xu JT, et al. (2021) Ginkgetin derived from Ginkgo biloba leaves enhances the therapeutic effect of cisplatin via ferroptosis-mediated disruption of the Nrf2/HO-1 axis in EGFR wild-type non-small-cell lung cancer. Phytomedicine 80: 153370.
- Zhang H, Zhu S, Zhou H, Li R, Xia X, Xiong H (2023) Identification of MTHFD2 as a prognostic biomarker and ferroptosis regulator in triple-negative breast cancer. Front Oncol 13: 1098357.
- Li W, Huang G, Wei J, Cao H, Jiang G (2023) ALKBH5 inhibits thyroid cancer progression by promoting ferroptosis through TIAM1–Nrf2/HO-1 axis. Mol Cell Biochem 478(4): 729-741.
- Jing S, Lu Y, Zhang J, Ren Y, Mo Y, et al. (2022) Levistilide a Induces Ferroptosis by Activating the Nrf2/HO-1 Signaling Pathway in Breast Cancer Cells. Drug Des Devel Ther 16: 2981-2993.
- Qiao J, Chen Y, Mi Y, Jin H, Huang T, et al. (2020) NR5A2 synergizes with NCOA3 to induce breast cancer resistance to BET inhibitor by upregulating NRF2 to attenuate ferroptosis, Biochem. Biophys. Res. Commun. 530(2): 402-409.
- Zhang Y, Xia M, Zhou Z, Hu X, Wang J, et al. (2021) p53 Promoted Ferroptosis in Ovarian Cancer Cells Treated with Human Serum Incubated-Superparamagnetic Iron Oxides. Int J Nanomedicine 16: 283-296.
- Gentric G, Kieffer Y, Mieulet V, Goundiam O, Bonneau C, et al. (2019) PML-Regulated Mitochondrial Metabolism Enhances Chemosensitivity in Human Ovarian Cancers. Cell Metab 29(1): 156-173.
- Liu N, Lin X, Huang C (2020) Activation of the reverse transsulfuration pathway through NRF2/CBS confers erastin-induced ferroptosis resistance. Br J Cancer 122: 279-292.
- Zhu X, Chen X, Qiu L, Zhu J, Wang J (2022) Norcantharidin induces ferroptosis via the suppression of NRF2/HO‑1 signaling in ovarian cancer cells. Oncol Lett 24(4): 359.
- Jing T, Guo Y, Wei Y (2022) Carboxymethylated pachyman induces ferroptosis in ovarian cancer by suppressing NRF1/HO‑1 signaling, Oncol. Lett. 23(5): 161.
- Zhang Z, Hu Q, Ye S, Xiang L (2022) Inhibition of the PIN1-NRF2/GPX4 axis imparts sensitivity to cisplatin in cervical cancer cells. Acta Biochim Biophys Sin 54(9): 1325-1335.
- Shin D, Kim EH, Lee J, Roh JL (2018) Nrf2 inhibition reverses resistance to GPX4 inhibitor-induced ferroptosis in head and neck cancer. Free Radic Biol Med 129: 454-462.
- Roh JL, Kim EH, Jang H, Shin D (2017) Nrf2 inhibition reverses the resistance of cisplatin-resistant head and neck cancer cells to artesunate-induced ferroptosis. Redox Biol 11: 254-262.
- Fu D, Wang C, Yu L, Yu R (2021) Induction of ferroptosis by ATF3 elevation alleviates cisplatin resistance in gastric cancer by restraining Nrf2/Keap1/xCT signaling. Cell Mol Biol Lett 26(1):26.
- Guan D, Zhou W, Wei H, Wang T, Zheng K, et al. (2022) Ferritinophagy-Mediated Ferroptosis and Activation of Keap1/Nrf2/HO-1 Pathway Were Conducive to EMT Inhibition of Gastric Cancer Cells in Action of 2,2 ′ -Di-pyridineketone Hydrazone Dithiocarbamate Butyric Acid Ester, Oxid. Med. Cell. Longev. 2022: 1-15.
- Xiang Y, Chen X, Wang W, Zhai L, Sun X, et al. (2021) Natural Product Erianin Inhibits Bladder Cancer Cell Growth by Inducing Ferroptosis via NRF2 Inactivation. Front Pharmacol 12: 775506.
- Wang Q, Bin C, Xue Q, Gao Q, Huang A, et al. (2021) GSTZ1 sensitizes hepatocellular carcinoma cells to sorafenib-induced ferroptosis via inhibition of NRF2/GPX4 axis. Cell Death Dis. 12(5): 426.
- Song X, Long D (2020) Nrf2 and Ferroptosis: A New Research Direction for Neurodegenerative Diseases. Front Neurosci 14: 267.
- Barnham KJ, Masters CL, Bush AI (2004) Neurodegenerative diseases and oxidative stress. Nat Rev Drug Discov 3(3): 205-214.
- Youssef P, Chami B, Lim J, Middleton T, Sutherland GT, et al. (2018) Evidence supporting oxidative stress in a moderately affected area of the brain in Alzheimer’s disease. Sci Rep 8(1): 11553.
- Ramsey CP, Glass CA, Montgomery MB, Lindl KA, Ritson GP, et al. (2007) Expression of Nrf2 in Neurodegenerative Diseases. J Neuropathol Exp Neurol 66(1): 75-85.
- Blair HA (2019) Dimethyl Fumarate: A Review in Relapsing-Remitting MS. Drugs 79(18): 1965-1976.
- Majkutewicz I, Kurowska E, Podlacha M, Myślińska D, Grembecka B, et al. (2018) Age-dependent effects of dimethyl fumarate on cognitive and neuropathological features in the streptozotocin-induced rat model of Alzheimer’s disease. Brain Res 1686: 19-33.
- Liu H, Feng Y, Xu M, Yang J, Wang Z, et al. (2018) Four-octyl itaconate activates Keap1-Nrf2 signaling to protect neuronal cells from hydrogen peroxide. Cell Commun Signal 16(1): 81.
- Lipton SA, Rezaie T, Nutter A, Lopez KM, Parker J, et al. (2016) Therapeutic advantage of pro-electrophilic drugs to activate the Nrf2/ARE pathway in Alzheimer’s disease models. Cell Death Dis 7(12): e2499-e2499.
- Fakhri S, Pesce M, Patruno A, Moradi SZ, Iranpanah A, et al. (2020) Attenuation of Nrf2/Keap1/ARE in Alzheimer’s Disease by Plant Secondary Metabolites: A Mechanistic Review. Molecules 25(21): 4926.
- Srivastava S, Blower PJ, Aubdool AA, Hider RC, Mann GE, et al. (2016) Cardioprotective effects of Cu(II)ATSM in human vascular smooth muscle cells and cardiomyocytes mediated by Nrf2 and DJ-1. Sci Rep 6(1): 7.
- Gellera C, Meoni C, Castellotti B, Zappacosta B, Girotti F, et al. (1996) Errors in Huntington disease diagnostic test caused by trinucleotide deletion in the IT15 gene. Am J Hum Genet 59(2): 475-477.
- Wexler NS (2012) Huntington’s Disease: Advocacy Driving Science. Annu Rev Med 63: 1-22.
- HUNTINGTON’S chorea, Med J Aust 1 (1946) 308.
- Reichert CO, De Freitas FA, Sampaio-Silva J, Rokita-Rosa L, De Barros PL, Levy D, et al. (2020) Ferroptosis Mechanisms Involved in Neurodegenerative Diseases. Int J Mol Sci 21(22): 8765.
- Skouta R, Dixon SJ, Wang J, Dunn DE, Orman M, et al. (2014) Ferrostatins inhibit oxidative lipid damage and cell death in diverse disease models. J Am Chem Soc 136(12): 4551-4556.
- Leng J, Xing Z, Li X, Bao X, Zhu J, et al. (2023) Assessment of Diagnosis, Prognosis and Immune Infiltration Response to the Expression of the Ferroptosis-Related Molecule HAMP in Clear Cell Renal Cell Carcinoma. Int J Environ Res Public Health 20(2): 913.
- Mi Y, Gao X, Xu H, Cui Y, Zhang Y, et al. (2019) The Emerging Roles of Ferroptosis in Huntington’s Disease. Neuromolecular Med 21(2): 110-119.
- Klepac N, Relja M, Klepac R, Hećimović S, Babić T, et al. (2007) Oxidative stress parameters in plasma of Huntington’s disease patients, asymptomatic Huntington’s disease gene carriers and healthy subjects : a cross-sectional study. J Neurol 254(12): 1676-1683.
- Johnson WM, Wilson-Delfosse AL, John Mieyal J(2012) Dysregulation of Glutathione Homeostasis in Neurodegenerative Diseases. Nutrients 4(2): 1399-1440.
- Dawson Ted M, Valina Dawson L (2003) Molecular Pathways of Neurodegeneration in Parkinson’s Disease. Science 302(5646): 819-822.
- Kim US, De Vellis J (2005) Microglia in health and disease. J Neurosci Res 81(3): 302-313.
- McGeer PL, McGeer EG (2008) Glial reactions in Parkinson’s disease. Mov Disord 23(4): 474-483.
- Rappold PM, Tieu K (2010) Astrocytes and therapeutics for Parkinson’s disease. Neurother J Am Soc Exp Neurother 7(4): 413-423.
- Vila M, Jackson-Lewis V, Guégan C, Wu DC, Teismann P, et al. (2001) The role of glial cells in Parkinson’s disease. Curr Opin Neurol 14(4): 483-489.
- Van Muiswinkel FL, de Vos RAI, Bol JGJM, Andringa G, Jansen Steur ENH, et al. (2004) Expression of NAD(P)H:quinone oxidoreductase in the normal and Parkinsonian substantia nigra. Neurobiology Aging 25(9): 1253-1262.
- Perry TL, Yong VW (1986) Idiopathic Parkinson’s disease, progressive supranuclear palsy and glutathione metabolism in the substantia nigra of patients. Neurosci Lett 67(3): 269-274.
- Schipper HM, Liberman A, Stopa EG (1998) Neural Heme Oxygenase-1 Expression in Idiopathic Parkinson’s Disease. Exp Neurol 150(1): 60-68.
- Lastres-Becker I, Ulusoy A, Innamorato NG, Sahin G, Rábano A et al. (2012) α-Synuclein expression and Nrf2 deficiency cooperate to aggravate protein aggregation, neuronal death and inflammation in early-stage Parkinson’s disease, Hum. Mol. Genet 21(14): 3173-3192.
- Pei-Chun C, Marcelo RV, Amar KP, Richard JS, Delinda Johnson A (2009) Nrf2-mediated neuroprotection in the MPTP mouse model of Parkinson’s disease: Critical role for the astrocyte. Proc Natl Acad Sci USA 106(8): 2933-2938.
- Jakel RJ, Townsend JA, Kraft AD, Johnson JA (2007) Nrf2-mediated protection against 6-hydroxydopamine. Brain Res 1144: 192-201.
- Nadia GI, Agnieszka J, Ana IR, Concepción García, Fernández-Ruiz J (2010) Different Susceptibility to the Parkinson’s Toxin MPTP in Mice Lacking the Redox Master Regulator Nrf2 or Its Target Gene Heme Oxygenase-1 | PLOS One 5(7): e11838.
- Jakel RJ, Kern JT, Johnson DA, Johnson JA (2005) Induction of the Protective Antioxidant Response Element Pathway by 6-Hydroxydopamine In Vivo and In Vitro. Toxicol Sci 87(1): 176-186.
- Dinkova-Kostova AT, Abramov AY (2015) The emerging role of Nrf2 in mitochondrial function. Free Radic Biol Med 88: 179-188.
- Aguiar AS, Duzzioni M, Remor AP, Tristão FSM, Matheus FC, et al. (2016) Moderate-Intensity Physical Exercise Protects Against Experimental 6-Hydroxydopamine-Induced Hemiparkinsonism Through Nrf2-Antioxidant Response Element Pathway. Neurochem Res 41: 64-72.
- Burton NC, Kensler TW, Guilarte TR (2006) In vivo modulation of the Parkinsonian phenotype by Nrf2. Neuro Toxicology 27(6): 1094-1100.
- Morroni F, Sita G, Djemil A, D’Amico M, Pruccoli L, et al. (2018) Comparison of Adaptive Neuroprotective Mechanisms of Sulforaphane and its Interconversion Product Erucin in in Vitro and in Vivo Models of Parkinson’s Disease. J Agric Food Chem 66(4): 856-865.
- Choi JW, Kim S, Yoo JS, Kim HJ, Kim Ji H, et al. (2021) Development and optimization of halogenated vinyl sulfones as Nrf2 activators for the treatment of Parkinson’s disease. Eur J Med Chem 212: 113103.
- Capelletti MM, Manceau H, Puy H, Peoc’h K (2020) Ferroptosis in Liver Diseases: An Overview. Int J Mol Sci 21(14): 4908.
- Yang W, Wang Y, Zhang C, Huang Y, Yu J, et al. (2022) Maresin1 Protect Against Ferroptosis-Induced Liver Injury Through ROS Inhibition and Nrf2/HO-1/GPX4 Activation. Front Pharmacol 13: 865689.
- Pan Q, Luo Y, Xia Q, He K (2021) Ferroptosis and Liver Fibrosis. Int J Med Sci 18(15): 3361-3366.
- Huang S, Wang Y, Xie S, Lai Y, Mo C, et al. (2022) Hepatic TGFβr1 Deficiency Attenuates Lipopolysaccharide/D-Galactosamine–Induced Acute Liver Failure Through Inhibiting GSK3β–Nrf2–Mediated Hepatocyte Apoptosis and Ferroptosis. Cell Mol Gastroenterol Hepatol 13(6): 1649-1672.
- Wu J, Xue R, Wu M, Yin X, Xie B, et al. (2022) Nrf2-Mediated Ferroptosis Inhibition Exerts a Protective Effect on Acute-on-Chronic Liver Failure, Oxid. Med. Cell. Longev. 2022: 1-23.
- Cai X, Hua S, Deng J, Du Z, Zhang D, et al. (2022) Astaxanthin Activated the Nrf2/HO-1 Pathway to Enhance Autophagy and Inhibit Ferroptosis, Ameliorating Acetaminophen-Induced Liver Injury. ACS Appl Mater Interfaces 14(38):
- 42887-42903.
- Gao G, Xie Z, Li E, Yuan Y, Fu Y, et al. (2021) Dehydroabietic acid improves nonalcoholic fatty liver disease through activating the Keap1/Nrf2-ARE signaling pathway to reduce ferroptosis. J Nat Med 75(3): 540-552.
- Hu J, Gu W, Ma N, Fan X, Ci X (2022) Leonurine alleviates ferroptosis in cisplatin‐induced acute kidney injury by activating the Nrf2 signalling pathway. Br J Pharmacol 179(15): 3991-4009.
- Yang Y, Cai F, Zhou N, Liu S, Wang P, et al. (2021) Dimethyl fumarate prevents ferroptosis to attenuate acute kidney injury by acting on NRF2. Clin Transl Med 11(4): e382.
- Huang Y, Jiang L, Liu X, Wang X, Gao L, et al. (2022) Melatonin Alleviates Acute Kidney Injury by Inhibiting NRF2/Slc7a11 Axis-Mediated Ferroptosis. Oxid Med Cell Longev 2022: 1-24.
- Qiu W, An S, Wang T, Li J, Yu B, et al. (2022) Melatonin suppresses ferroptosis via activation of the Nrf2/HO-1 signaling pathway in the mouse model of sepsis-induced acute kidney injury. Int Immunopharmacol 112: 109162.
- Qiongyue Z, Xin Y, Meng P, Sulin M, Yanlin W, et al. (2022) Post-treatment With Irisin Attenuates Acute Kidney Injury in Sepsis Mice Through Anti-Ferroptosis via the SIRT1/Nrf2 Pathway. Front Pharmacol 13: 857067.
- La Rosa P, Petrillo S, Turchi R, Berardinelli F, Schirinzi T et al. (2021) The Nrf2 induction prevents ferroptosis in Friedreich’s Ataxia. Redox Biol 38: 101791.
- Liu X, Ma Y, Luo L, Zong D, Li H, et al. (2022) Dihydroquercetin suppresses cigarette smoke induced ferroptosis in the pathogenesis of chronic obstructive pulmonary disease by activating Nrf2-mediated pathway. Phytomedicine 96: 153894.
- Dang R, Wang M, Li X, Wang H, Liu L, et al. (2022) Edaravone ameliorates depressive and anxiety-like behaviors via Sirt1/Nrf2/HO-1/Gpx4 pathway. J Neuroinflammation 19(1): 41.
- Guo Z, Lin J, Sun K, Guo J, Yao X, et al. (2022) Deferoxamine Alleviates Osteoarthritis by Inhibiting Chondrocyte Ferroptosis and Activating the Nrf2 Pathway. Front Pharmacol 13: 791376.
- Wang Y, Chen D, Xie H, Jia M, Sun X, et al. (2022) AUF1 protects against ferroptosis to alleviate sepsis-induced acute lung injury by regulating NRF2 and ATF3. Cell Mol Life Sci 79(5): 228.
- Liao T, Xu X, Ye X, Yan J (2022) DJ-1 upregulates the Nrf2/GPX4 signal pathway to inhibit trophoblast ferroptosis in the pathogenesis of preeclampsia. Sci Rep 12: 2934.






























