Diversity of Climbing Plants in an Urban Forest Fragment
Flora Magdaline Benitez Romero1,2,3*, José Henrique Costa e Penha Júnior1, Rosana Barbosa de Castro Lopes1, Julia Pereira de Vasconcelos1, Rodrigo Moreira da Silva1 and Guido Hernán Vásquez Colomo3
1National Institute of Amazonian Research (INPA), Av. André Araújo, 2936, Manaus, Amazonas, CEP 69067-375, Brazil
2Department of Forest Sciences, Faculty of Agricultural Sciences, Federal University of Amazonas (UFAM), Amazonas, Manaus, CEP 69077-000, Brazil
3Faculty of Biological and Natural Sciences, Universidad Amazónica de Pando (UAP), Cobija 11, Pando
Submission:October 18, 2023; Published:November 22, 2023
*Corresponding author:Flora Magdaline Benitez Romero, National Institute of Amazonian Research (INPA), Av. André Araújo, 2936, Manaus, Amazonas, CEP 69067-375, Brazil
How to cite this article:Flora Magdaline Benitez Romero, National Institute of Amazonian Research (INPA), Av. André Araújo, 2936, Manaus, Amazonas, CEP 69067-375, Brazil. Curr Trends Biomedical Eng & Biosci. 2023; 22(1): 556079. DOI:10.19080/CTBEB.2023.22.556079
Summary
Climbers are herbaceous or woody plants that germinate in the soil and remain rooted throughout their lives, needing other plants to support their development. The aim of this study was to analyze the edge effect on the climbing plant community in the forest fragment of the Federal University of Amazonas. Fifty plots were set up for sampling. The species were grouped and phytosociological parameters were calculated. Six hundred and thirteen (613) individuals were recorded, represented by 13 families found inside and on the edge of the UFAM forest fragment. The family with the highest ecological importance value (VI) was Fabaceae. The genera Bauhinia and Derris had the highest ecological importance value at the edge and in the interior of the forest. The scandent climbing mode was the most important and was observed in 56 species. This study confirmed the diversity of climbing plants, although there were more climbing plants at the edge of the forest than in the interior, due to the greater luminosity, and the scandent climbing habit was abundant in both environments. Climbing plants integrate the native vegetation of the forest fragment with their richness and diversity. They also contribute ecologically by providing food and shelter for living organisms, organic matter, among other components.
Keywords: Phytosociological; Genera Bauhinia; Amazonas; Ecologically; Herbaceous; Microhabitats; Mechanical; Autotrophic; Fabaceae; Bignoniaceae; Dilleniaceae; Bignoniaceae; Malpighiaceae
Introduction
A structural component of relevance in “forest fragments are the vines, which connect the treetops creating passages for animals living in the canopy, their flowers, fruits and leaves serve as food for animals of various groups” [1,2]. Another benefit is related to their foliage, as they help maintain the stability of the microclimate in the cold, dry season, a period when most canopy trees lose their leaves [3,4], “generating improved conditions for germination and seedling establishment”[5]. Climbers are herbaceous or woody plants that germinate in the soil and remain rooted throughout their lives, needing other plants to provide support for their development [6].
However, vines can also interact in a negative way, for example, “a high density of vines can generate an excessively heavy load, breaking the branches of the trees, deforming their canopies, reducing their leaf area and fecundity [7,8]. “In addition, vines end up reproducing very quickly and, as they compete for light, water and space with other trees, they end up being detrimental to the biodiversity of the site in large numbers, in which case it is necessary to manage these species to keep the vegetation more balanced [7,8].
A very important characteristic of climbing plants is that “they grow rapidly compared to tree species and can often dominate the crowns of supporting and neighboring trees” [9]. “Knowledge of this group of plants is of fundamental importance for the study of forest dynamics and management, due to their diversity and ecological importance” [10]. “Although their presence is more beneficial than harmful, a very high abundance of vines may even interfere with the natural dynamics of these forest fragments [11]. Studies on the importance of climbing plants to the forest community are still incipient” [12]. This lack of data may be related to the very difficulties of collecting in the forest canopy [13] “and may also be associated with problems in identifying this diverse group”[12]. In this context, the study aims to determine the horizontal structure of the creepers in order to characterize their occurrence at the edge and in the interior of the forest fragment, as well as to classify their climbing mechanisms.
Materials and Methods
The study was carried out in the forest fragment of the Arthur Virgílio Filho University Campus at UFAM (Federal University of Amazonas) (03º04’34” South latitude and 59º57’30” West longitude), located in the Western Amazon, in the municipality of Manaus, Amazonas, Brazil. The area of the UFAM fragment totals 6.7 million square meters, with a perimeter of 16.9km of land. “This area is the third largest natural fragment in an urban area in the world and the first in the country, which contributed to the creation of the Environmental Protection Area - APA UFAM”. According to Duarte [15] “there are five types of vegetation cover: alluvial dense ombrophilous forest, submontane dense ombrophilous forest, open ombrophilous forest, grassland and anthropized area”. In addition, the same author classifies it as a preserved forest area of the Federal University of Amazonas, which is an Environmental Unit (UNA) of the Municipality created by decree of Municipal Law No. 321 of December 20, 1995, with a large proportion of intact areas covered by forest and altered areas that have been used for the construction of buildings, pavements and parking lots.

The UFAM fragment has various types of plant formations, which produce a variety of microhabitats that guarantee the establishment of various species of flora and fauna. However, the forest is under constant threat from occupation and misuse in its surroundings by neighborhoods.
Sample design
In this study the term climber was used as proposed by” [15], to designate autotrophic, vascular plants that germinate in the soil and maintain contact with it throughout their life cycle, which lose the ability to self-support as they grow and require mechanical support for their development. To sample the individuals, 50 plots measuring 10m x 20m (10,000m²) were set up inside the forest, 100m from the edge, 5m apart horizontally between plots. Similarly, 50 plots measuring 10m x 20m (10,000m²) were set up at the edge of the forest, 20m from the road.
Measurement characteristics and identification of climbing plants
Climbing plants with stems ≥ 1 cm DBH (Diameter at Breast Height - 1.30m) were included in the sampling. For the measurements, protocols proposed by” [16] were adopted, and the DBH values and climbing mechanisms observed for each individual were recorded. In this way, the compositional analyses will cover the inner part of the forest, where measurements are taken at 1.30cm from the main rooting point. To determine the phytosociological parameters of the creepers, the following were calculated: Absolute density (AD) and relative density (RD), absolute frequency (AF) and relative frequency (RF) [17]. Excel software and the R program were used to process and analyze the data [18]. Species identification was carried out by a parabotanist, followed by verification using the book Fora da reserva Ducke”[19], and subsequent confirmation in the Herbarium of the Federal University of Amazonas.
Analysis of climbing mechanisms
For each individual, the main form of ascent to the canopy was observed. Adding these observations to data from the literature, the species were grouped into three categories: voluble vines, prehensile vines and climbing vines. “Voluble plants were those that wrapped themselves around the support by means of branches, stems and/or petioles; prehensile plants were those that reached the canopy by attaching themselves to the support by means of modified structures such as tendrils, and scandent plants were those that climbed by leaning passively on a support, according to the protocol of” [16].
Results and Discussion
Six hundred and thirteen (613) individuals were cataloged, representing 13 families found inside and on the edge of the UFAM forest fragment. The family with the highest ecological importance value (VI) was Fabaceae in both the interior (21.1%) and the edge (21.1%). It should be noted that the VI of the families was affected by the number of individuals found in the plots in both environments. Therefore, the occurrence of families can happen randomly as detailed in Table 1.
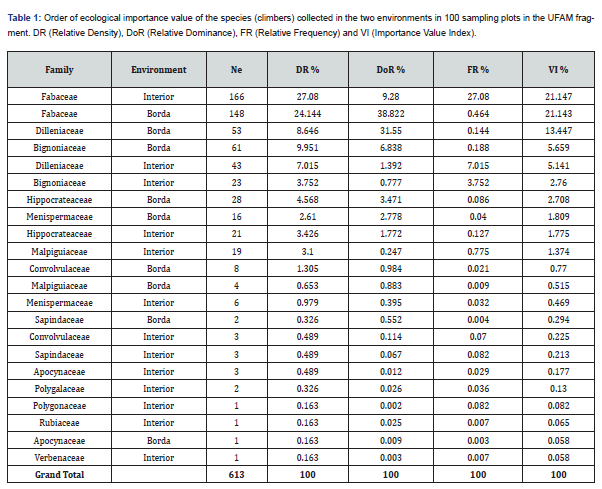
The Fabaceae family had the highest VI both inside and on the edge of the forest in the UFAM fragment. These results, when compared with other studies in western Amazonia, show the same behavior and predominance of this Ducke family [20,21]. However, they differ in the forest environments. In the present study, Fabaceae was found on terra firme, while in the study by Oliveira et al. [21] this family was found on plateaus and slopes. Fabaceae has various types of plant habits [20], which leads us to interpret that this family can adapt to different environments, as observed in this study.
The other families such as Dilleniaceae, Bignoniaceae, Apocynaceae, Malpighiaceae and Sapindaceae contribute almost half of the genera and species”[1]. Peixoto & Gentry [22] also “indicated the Bignoniaceae as one of the richest and most common families in Neotropical forests, especially in dry forests”. However, at the species level, 120 were found, totaling six hundred and thirteen (613) individuals found in 100 plots in the forest fragment. Of these, 50 plots were evaluated in each environment (interior and edge).
Ecological importance value of the species collected in the interior and at the edge
In the interior of the forest, seventy-nine (79) species were found in the 50 plots in the fragment, totaling two hundred and ninety-two (292) individuals. The species Bauhinia guianensis (7.449%), Derris floribunda (6.830%) and Bauhinia sp.1 (5.539%) had the highest ecological importance value, followed by the other species detailed in Annex 1. Oliveira [23] also recorded the genus Bauhinia sp. with the highest VI. Similarly, Maia24, evaluating the photosiological aspects of lianas on terra firme in the Manaus region, recorded the species Bauhinia sp. as one of the species with the highest importance value. Therefore, the genus and species Bauhinia guianensis is the most representative within the forest fragment. In general, the genus Bauhinia had greater dominance, which expresses the proportion of its size or coverage in relation to the community space.
Forty-one (41) species of vines were found in 50 plots, totaling three hundred and twenty-one (321) individuals. The species Derris floribunda (16.728%), Doliocarpus dentatus (10.377%) and Dalbergia multiflora (7.591%) showed the best results for the phytosociological parameters, with the Derris floribunda and Doliocarpus dentatus species having the highest VI results in the environment. In the UFAM fragment, a greater number of climbing plants were found at the edge of the forest than in its interior. However, in order to guarantee the balance and functionality of the forest in this fragment, the margins must be protected, as a means of avoiding infestation of vines in its interior and preventing greater impacts on all local biodiversity.
The number of individuals was higher at the edge of the forest than in the interior, which is justified by the shading that occurs in the interior. According to Hegarty [24] “canopy closure and shading control liana infestation. According to Ribeiro [25], “edge effects involve changes in the abundance and distribution of species caused by abiotic factors in the vicinity of edges, such as increased plant density due to increased solar radiation” [26]. In this way, interspecific competition is expected to be greater in these environments, which according to” [27]. Leads populations to present increasingly aggregated spatial patterns. The total abundance of vines, according to Laurance et al. [28] “is known to be positively associated with forest edges and areas of disturbance”. In large tracts of forest, vines (lianas) act as bandages that mend the fall of a tree and also seal the forest edge [29].
However, the vines in the fragment are indicators of natural disturbance. However, if a fragment is subjected to anthropogenic actions, such as new fragmentation, the vines that take advantage of spaces and light (clearings) become more numerous, and they can penetrate deeper into the interior of these forests. In addition, they can disturb environments and kill trees or cause them to store less carbon, and their presence can also affect the diversity of tree species in the forest. In this way, they transform the habitat of local animals and organisms. In other words, changes in the growth and number of lianas can cause fundamental changes in the structure of a forest and the way it functions, highlighting the importance of protecting forest margins [29].
Types of Climbing Mechanisms Inside and on the Edge of the Fragment
In terms of climbing habit, the scandent climbing mode was the most important, observed in 56 species. The prehensile mode was observed in 12 species and the voluble mode in 11 species. In the forest interior of the fragment, 70.89% of the climbers had the scandent climbing mechanism, 15.19% prehensile and 13.52% voluble. At the edge, 60.98% were scandent, 31.71% prehensile and 7.32 voluble. The species with the highest number of individuals in the different climbing methods at the edge of the forest were Derris floribunda, Bauhinia guianensis and Doliocarpus dentatus, representing a greater number of scandent species with 33 individuals, prehensile species with 14 individuals and voluble species with 12 individuals. The species that had the highest number of individuals in the different climbing methods in the interior were Derris floribunda, Mimosa guilandinae and Acacia multipinnata, representing a greater number of scandents with 53 individuals, voluble with 20 and prehensile with 17 individuals.
In Villagra’s study [30] all three types of mechanism were found, and in the same study the most common mechanism was voluble”. However, in our study it was scandent in both environments. This could mean that the climbers create their own way of climbing depending on the environment. Climbing mechanisms are evolutionary strategies of climbing plants, considering that they need, in their development and maturity phase, to get as close as possible to the treetops in search of one of the essential resources for their survival, light, so trees are the faithful hosts of this plant group so peculiar to our forests.
Conclusion
The diversity of climbing plants was confirmed in the Ufam fragment, although there were more individuals of climbing plants at the edge of the forest than in the interior. This is because there is more light at the edge of the forest and consequently the number of individuals increases. As they are indicators of disturbance in forest fragments, it is necessary to guarantee the balance and functionality of the forest in this fragment, as a means of avoiding infestation of creepers in its interior and preventing greater impacts on the entire biodiversity of tree species. On the other hand, the climbing habit was abundant in both environments, with greater representation in the Fabaceae family, and this form of climbing was the richest in the forest fragment’s climbing community. From a phytosociological point of view, climbing plants play an essential role in the diversity and richness of plant species in the forest fragment of the Federal University of Amazonas. In balance with the forest, climbing plants also contribute ecologically by providing food and shelter for wildlife and other living organisms, as well as organic matter, among other components.
References
- Gentry AH (1991) The distribuition and evolution of climbing plants. In: PUTZ, F.E. The biology of vines. Cambridge University Press, Cambridge pp. 3-49.
- Putz Fe (2005) Ecologia das Trepadeiras. Ecology Info 24: 1-15.
- Morellato LPC, Leitão Filho HF (1996) Reproductive phenology of climbers in a southeastern Brazilian forest. Biotropica 28(2): 180191.
- Schnitzer SA, Bongers F (2002) The ecology of lianas and their role in forests. Trends in Ecology & Evolution 17: 5.
- Parren MPE, Bongers F (2005) Management of climbers in the forests of West Africa. Forests Climbing Plants of West Africa: Diversity, Ecology and Management. Wallingford: CABI Publishing.
- Darwin C (1897) On the movements and habits of climbing plants. Journal of the Linnean Society ser Botany 9: 1-118.
- Richards PW (1996) The tropical rain Forest: na ecological study. (2nd edition). Cambridge University Press, Cambridge, pp. 575.
- Hegarty EE, Caballé G (1991) Distribution and abundance of vines in Forest communities. In: FE Putz & HA Mooney (eds.), The Biology of Vines. Cambridge University Press, Cambridge, United Kingdom, 313-335.
- Campbell EJF, Newbery DM (1993) Ecological relationships between lianas and trees in lowland rain forest in Sabah, East Malaysia. Journal of Tropical Ecology 9: 469-490.
- Putz FE (1984) How trees avoid and shed lianas. Biotropica 16: 19-23.
- Engel VL, Fonseca RCB, Oliveira RE (1998) Ecologia de lianas e o manejo de fragmentos florestais. Série Técnica IPEF, Piracicaba 12(32): 43-64.
- Udulutsch RG, Assis MA, Picchi DG (2010) Florística de trepadeiras numa floresta estacional semidecídua, Rio Claro, Araras, Estado de São Paulo, Brasil. Revista Brasileira de Botânica 27(1): 125-134.
- Villagra BLP, Neto RS (2010) Florística de trepadeiras no Parque Estadual das Fontes do Ipiranga, São Paulo, SP, Brasil. Revista Brasileira de Biociências 8(2): 186-200.
- Duarte ACOC (2000) Comunidade de Anuros em um fragmento de Floresta Urbano:Unidade Ambiental-UNA da Universidade do Amazonas - UA. Dissertação (Mestrado em Biologia Tropical e Recursos Naturais) INPA/UFAM. Manaus p60.
- Weiser VL (2007) Árvores, arbustos e trepadeiras do cerradão do Jardim Botânico Municipal de Bauru, SP. 100 f. Tese (Doutorado em Ecologia) - Instituto de Biologia, Universidade Estadual de Campinas, Campinas.
- Gerwing JJ et al (2006) A standard protocol for liana censures. Biotropica 38(2): 256261.
- Schnitzer SA, Rutishauser S, Aguilar S (2008) Supplemental protocol for liana censuses. Forest Ecology and Management 255: 1044-1049.
- Müller-Dombois D, Ellemberg H (1974) Aims and methods of vegetation ecology. New York, John Wiley.
- Ribeiro JELS, Pereira EDC, Silva CFD, Mesquita MR, Procopio LC, et al.(1999) Flora da Reserva Ducke: guia de identificação das plantas vasculares de uma floresta de terra-firme na Amazônia Central. INPA.
- Ducke A, Black GA (1954) Notas sobre a fitossociologia da Amazônia Brasileira. Boletim Técnico do Instituto Agronômico Norte 29: 3-48.
- Oliveira AN, Amaral IL, Ramos MBP, Formiga KM (2008) Aspectos florísticos e ecológicos de grandes lianas em três ambientes florestais de terra firme na Amazônia Central. Acta Amazonica 38(2): 421-430.
- Peixoto AL, Gentry AH (1990) Diversidade e composição florística da mata de tabuleiro na reserva Florestal de linhares (espírito santo, Brasil). Revista Brasileira de Botânica 13: 19-25.
- Maia LMA (1991) Aspectos fitossociológicos de lianas em mata de terra firme, Manaus-Amazonas.
- Hegarty EE (1991b) Vine-host interactions. In: Putz FE & Mooney HA (eds.), The biology of vines. Cambridge: Cambridge University Press, United Kingdom, pp. 357-375.
- Ribeiro Msl (2007) Efeitos de borda sobre a vegetação e estruturação populacional em fragmentos de Cerradão no Sudoeste Goiano, Brasil.
- Murcia C (1995) Edge effects in fragmented forests: implications for conservation. Trends in Ecology and Evolution 10: 58-62.
- Greig-Smith MAP (1964) Quantitative plant ecology. (2nd edition), London, Buther Worths.
- Laurance WF, Perez-Salicrup D, Delamonica P, Fearnside PM, D Angelo S, et al.(2001) Rain forest fragmentation and the structure of Amazonian liana communities. Ecology 82: 105-116.
- Campbell MJ, Edwards W, Magrach A, Alamgir M, Porolak G, et al. (2018) disturbance drives liana abundance increase and alteration of liana–host tree interactions in tropical forest fragments. Ecol Evol 8: 4237-4251.
- Villagra BLP (2008) Diversidade florística e estrutura da comunidade de plantas trepadeiras no Parque Estadual das Fontes do Ipiranga, São Paulo, SP, Brasil / Berta Lúcia Pereira Villagra - São Paulo.






























