Identification and Estimation of Total Tannins from Citrus limon l. Burm. F. (lemon) and its endophytes
Rathod Zalak R, Sarita Sharma and Saraf Meenu S*
Department of Microbiology and Biotechnology, University School of Sciences, Gujarat University, India
Submission:May 09, 2022;Published:May 24, 2022
*Corresponding author:Department of Microbiology and Biotechnology, University School of Sciences, Gujarat University, Ahmedabad- 380009, Gujarat, India
How to cite this article:Rathod Z R, Sarita S, Saraf Meenu S. Identification and Estimation of Total Tannins from Citrus limon l. Burm. F. (lemon) and its endophytes. Curr Trends Biomedical Eng & Biosci. 2022; 20(5): 556049. DOI:10.19080/CTBEB.2022.20.556049
Abstract
Citrus limon (Lemon) is a member of the Rutaceae family and it is also known as Kagdi Limbu or Lime. The purpose of the study was to determine the total tannin content of Citrus limon plant and endophytes isolated from the leaves, steam, root, seed, and peel. By using Bromocresol green approach and HPLC with Gallic acid and tannic acid as standards, the total tannin content (TTC) was determined spectrophotometrically. Tannins present in excess amount. Leaf extricates had the highest tannin content of the various sections studied. TLC, spectrophotometric, FTIR, HPLC, and GCMS tests were used to determine the total tannins content of different Endophytes isolated from whole Citrus limon plant vs extract of Citrus limon plant. Intriguingly, a close examination of all out tannins in Endophytes was completed, which can be used.
Keywords: Citrus limon (Lemon); GC-MS; HPLC; Tannins; Tannic acid
Introduction
Plants are a source of a variety of chemical substances that can be used in medicine and other uses. The compounds are found in plant parts such as leaves, flowers, bark, seeds, fruits, stems, and roots. Alkaloids, steroids, tannins, glycosides, volatile oils, fixed oils, resins, and other bioactive compounds Flavonoids and phenols. Because of their importance in the preceding environment, preliminary phytochemical tests are important. Plant screening is highly motivated by a desire to discover and develop novel medicinal compounds. increased efficacy Such investigations have also been reported by several study groups around the world. There is one an increasing understanding of the relationship between a medicinal plant’s phytochemical elements and its effectiveness. Pharmacological activity is a term that refers to the ability of a substance to the discovery of novel drugs has resulted from the screening of active chemicals found in plant pharmaceuticals [1,2].
Medicinal plants are a rich source of innovative medications that are used in traditional medicine, modern medicine, pharmaceutical intermediates, and lead compounds in the pharmaceutical industry. Synthetic drugs the fact that they contain antibiotics is the rationale for their use as medicine. Components of therapeutic value in chemical form These substances can be found in nature. Therapeutic herbs, leaves, vegetables, and roots with defence mechanisms and antimicrobial properties different illnesses Plants have medical benefit because of certain chemical compounds (typically flavonoids). secondary metabolites in the human body that have a specific physiological impact. In Plant research has gotten a lot of attention recently all across the world, and there’s a lot of data to back it up. Evidence has accumulated to suggest that therapeutic plants applied in diverse ways have enormous potential traditional [3].
Plant secondary metabolites exhibit a variety of biological and pharmacological properties, including antioxidative, anti-allergic, anti-inflammatory, antibacterial, and hypoglycemic properties. and anti-carcinogenic. Phenols and polyphenols are important antioxidants. Secondary metabolites are compounds found in plants. They’ve done it. Multiple biological impacts have been described, including Antioxidant and antibacterial properties. They are necessary. source in the plant for regular growth and defence against pathogens Infection and trauma are also potential consequences. The bioactive substance was isolated and identified. Plant chemicals have always been a difficult problem for scientists and researcher’s Preliminary phytochemical quality assessment The presence of biomolecules was assessed using a variety of methods. Anthraquinone, alkaloids, catechol, flavonoids, and phenolic acids are among examples. saponins, steroids, tannins, and triterpenoids are all examples of chemicals respectively [4]. Natural medicines are currently in demand, and their popularity is growing. Because a large amount of crude material is effectively available, WHO recommends and encourages the use of traditional spices or treatments in the medical field. Plants are extremely complicated creatures [5]. Ordinary substances extracted from plants belonging to the Rutaceae family are used to treat a variety of ailments all around the world. Citrus spp. is an important species in this family, with over 600 species found in tropical and subtropical regions [6]. Its medicinal potential is demonstrated by the presence of phytochemical substances in the plants. Alkaloids have antibacterial, anticancer, antiarrhythmic, and pain-relieving properties. Steroids operate as signaling atoms and are antagonistic to cardiac tonic activity [7]. Endophytes are microorganisms that survive inside plant tissues without leaving any visible signs of their presence, and their interaction with the host shifts from saprobe to idle microbe to communalistic/ mutualistic symbiont. In addition to boosting the host plant’s development and yield, endophytes play an important role in combating abiotic and biotic stresses [8,9]. There is a great amount of compound communication going on inside the plant structure, which is interrupted by an arrangement of sign molecules that has mostly gone unexplored.
Tannins are high molecular weight polyphenolic chemicals found in plants, foods, and drinks that are soluble in water and polar organic solvents [10,11]. Tannins are divided into two categories based on their chemical structure and biological activity: hydrolysable and condensed tannins. Both forms of tannins could create strong compounds with certain proteins, slowing down their digestion. Tannins can link to bacterial enzymes or create indigestible complexes with cell wall carbohydrates, lowering the digestibility of the cell wall. In addition, tannins have been shown to be effective regulators of lipid peroxidation in cardiac mitochondria and to have antifibrotic properties. Tannins can be applied to cure a variety of ailments and improve human health because of their therapeutic characteristics. As a result, the current study will review the following pharmacological activities that have recently been investigated. Tannins were found in the leaves of the Citrus limon plant and their endophytes, according to phytochemical screening. Tannins are antioxidants in nature [12-14].
Materials and Methods
The entire Citrus limon plant was obtained from AAU Anand’s Department of Horticulture in Gujarat, India. Citrus limon fruits and leaves were also completely cleaned and surface sterilized with sterile distilled water. Plant extraction was carried out utilising a steam distillation approach with a variety of solvents in a souxlate apparatus. MSZLd and MSZLe bacterial endophytes were identified from Citrus limon leaf and seed, respectively. The bacteria were extracted using a solvent-solvent cold extraction technique. The root, the stem, and the leaf Tannins were extracted from Citrus limon plant and Endophytes and assessed as phytochemicals with a good source of secondary metabolites [15,17].
Reagents and Chemicals
All reagents and chemicals used for testing were analytical grade, SRL, Himedia and Sigma brand.
Instruments used
Electric microscope (Lawrence and Mayo), Laminar air flow (Bright), Soxhlet unit (SENTWIN), rotary vacuum evaporator (Nutronics DTC201), Cooling centrifuge (Remi 421LAG), Refrigerator (Kelvinator Master cool Deluxe), Deep freezer (Blue star), Orbital shaker (Ssi) and Autoclave (EQUITRON), Spectrophotometer (Shimadzu UV-1800), FTIR (BRUKER), HPLC (Shimadzu), GC (Shimadzu).
Extraction: Solvent extraction using Souxlate apparatus were done
Qualitative analysis: 1ml of the concentrate was mixed with 0.5 mg of crude extract was boiled in 20 ml of water in test tubes then filtered. Addition of few drops of 0.1% ferric chloride produced brownish green or blue or black coloration indicating presence of tannins [18,19].
Thin layer Chromatographic analysis: To assess the number of chemicals, present in the plant crude extract, TLC was done on pre-coated silica gel TLC plates grade F254 (E-Merck, Darmstadt, Germany). Using capillary tubes, a total of 5 l (10 mg/ml) of sample was spotted 1 cm from the bottom of silica gel plates. Metabolites were profiled using a variety of solvents in varied combinations and concentrations. The chromatogram was created in closed tanks where the environment was saturated with eluent vapor by soaking the filter paper lining. Anisaldehyde in sulphuric acid and Ferric chloride solution (5 percent) reagent spray were used to visualize the chromatogram. The following formula was used to compute the Rf values of the compounds.
Preparative TLC for compound isolation: Preparative TLC with chloroform identified two compounds: ethyl acetate: methanol (5: 3: 2). Scrape the chemicals from the plate, dissolve in methanol, and concentrate to dryness [20,21].
Rf = distance travelled by the compound/ distance travelled by the solvent front
Total tannin contents: The tannin content was determined using a modified version of the Broadhurst et al., 1978 method, which used catechin as a reference component. A volume of 400μL of extract is added to 3 mL of a solution of vanillin (4% in methanol) and 1.5 mL of concentrated hydrochloric acid. The absorbance was measured at 500 nm after 15 minutes of incubation. UV-Vis Spectrophotometry was done using Shimadzu UV 1800 Double beam Spectrophotometer. Total spectrum analysis from 4000 nm to 400 nm was done as it facilitates to understand various peaks arising due to multiple components present in the extract. By this we identified the main constituent present in methanolic extract.
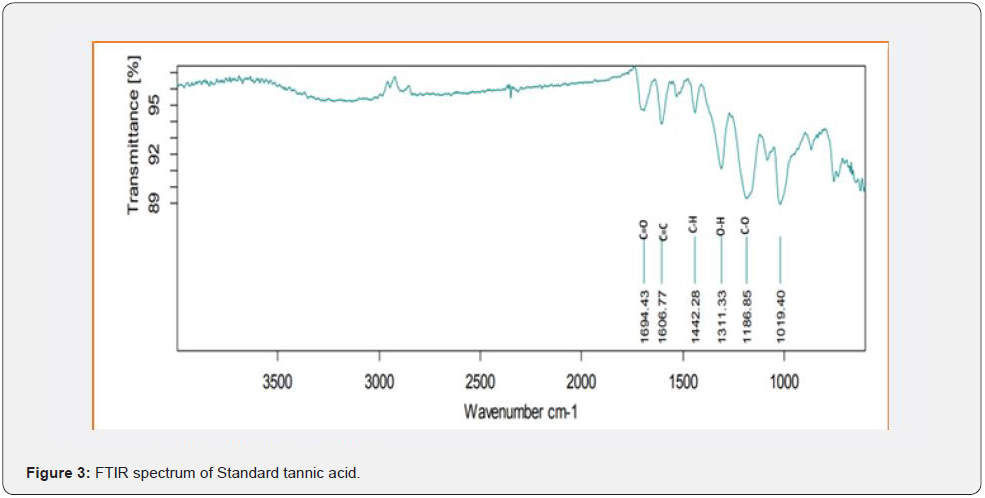
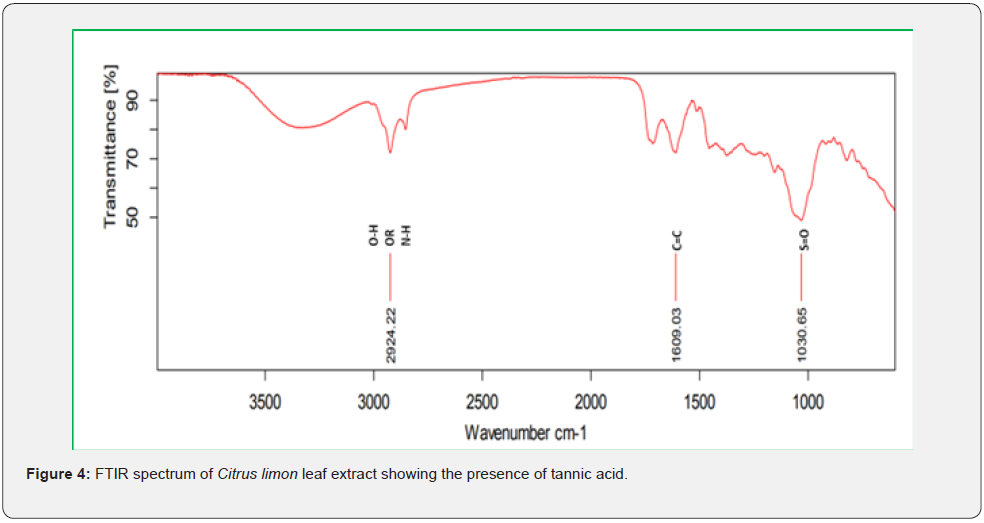
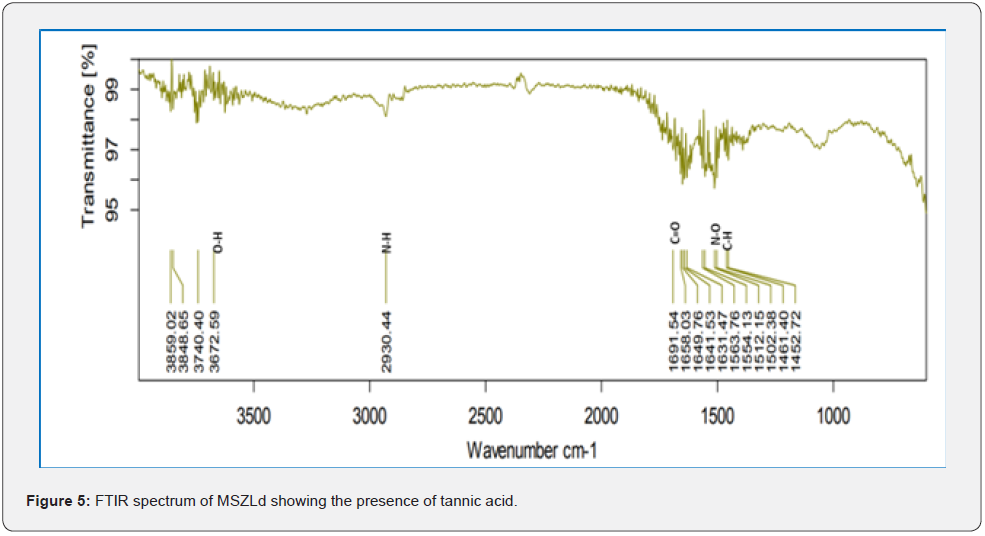
FTIR analysis: After KBr pelleting, Sigma brand stander of tannic acid and chloroform methanolic extract of samples were applied. The sample was put into a Shimadzu IR Affinity Japan FTIR spectroscope with a wavelength ranging of 400-4000 cm-1 [22,23].
HPLC determination of tannin: A 100-ml flat-bottom flask with ground-glass joint was filled with 50 ml of a 40 percent aqueous ethanol solution and a precisely weighed amount (approximately 2 g) of comminuted roots and rhizomes of garden burnet. The contents of the flask were weighed precisely (to within 0.01 g), connected to a reflux cooler, heated to boiling for 1 hour, then chilled. The contents of the flask were then weighed again, and the original weight was restored by adding the same 40% aqueous ethanol solution. To obtain solution A, the mass was filtered via a paper filter (the first 10-ml filtrate portion was rejected). A 2-l aliquot of solution A was applied to a Milikhrom-5 chromatograph column (Orel, Russia) equipped with a 280 nm UV detector. Diasorb C16 was used to fill the stainless-steel column (80 mm-2.0 mm) (or an analogous sorbent with a 5 m particle size). The analysis was carried out at room temperature in the isocratic elution mode using a mobile phase of acetonitrile, water and glacial acetic acid (4: 6: 0.1). The flow rate of the mobile phase was 40 l”min. The analysis lasted 12 minutes. Preliminary experiments led to the selection of these ideal circumstances [24].
GC-MS analysis: To determine the numerous bioactive chemicals, present, the extract was submitted to GC MS analysis. The sample was analysed using the programme Turbomass 5.2 version on a Perkin Elmer Clarus-600 instrument. A non-polar capillary column (30 m X 0.25 mm, 0.25 mm film thickness) was utilized. The volume of the injected specimen was 1 l of methanol extract, the injector temperature was 220 oC, and the split ratio of the carrier gas Helium was 25:1. Initial temp 60 oC for 5 min, ramp 7 oC /min to 300 oC, hold 15 min, solvent delay=3.00 min, source temperature 180 oC, oven temperature programme initial temp 60 oC for 5 min, ramp 7 oC/min to 300 oC, hold 15 min In the electronic ionisation mode, scan: 50 to 600 Da, ionisation energy 70 eV. Computer matching of mass spectra with those of standards was used to identify chemicals (Mainlib, Replib and NIST library) [25-27].
Data analysis
Spectrophotometric data analysed by analysis of variance (ANOVA) and their further confirmation done with FTIR photometry of functional group. Retention time and concentration of tannins was distinguished by utilizing HPLC programming (Spectra research laboratory - Himatnagar) and Identification was done with GCMS instrument (Sicart lab - Aanad). Data obtained
Results and Discussion

Microbial assistance metabolites have made significant contributions to the advancement of a variety of drugs, including antibiotics, metabolic inhibitors, immunomodulatory experts, cell fortifications, and anticancer trained professionals. In the previous decade, endophytic bacteria and fungi have also opened avenues for the investigation and exploitation of new constituent components created during the plant-microorganism connection for both medication and green applications. Three distinct Bacillus spp. isolated from the Citrus limon plant were chosen for tannin analysis. The total tannin content (TTC) of six extracts of Citrus limon L. Burm. f., three from plant extracts and three from bacterial endophytes extracts, were determined. Qualitative tests determine with FeCl3 yielded positive results for all six extracts. Figure 1 shows the quantitative analysis of these six extracts, it was also confirmed on thin layer chromatography and that Rf value was calculated (Table 1). TAC was given in milligrams of tannic acid (mg TAE). The MSZLe leaf endophyte extract of Citrus limon has the highest Tannins concentration (7.4±0.14 mg TAE), followed by Citrus limon leaf extract (5.9±0.20 mg TAE). Citrus limon root has the least amount of tannins concentration (4.8±0.18 mg TAE). The leaves and their endophytes had the highest tannin concentration (Table 2). The tannin content of the components was determined using a UV spectrophotometer (Shimadzu UV- 1800) at 470nm (Figure 2). The Dalbergia sissoo showed highest value (1.59±0.31 mg/g DW) as compared to other two medicinal plants and the moderate concentration in Tephrosia purpurea (0.77±0.05 mg/g DW) whereas lowest source was examined to be Delonix regia (0.52±0.15 mg/g DW).


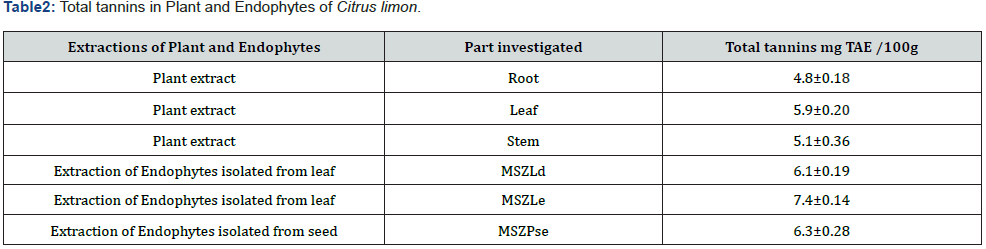
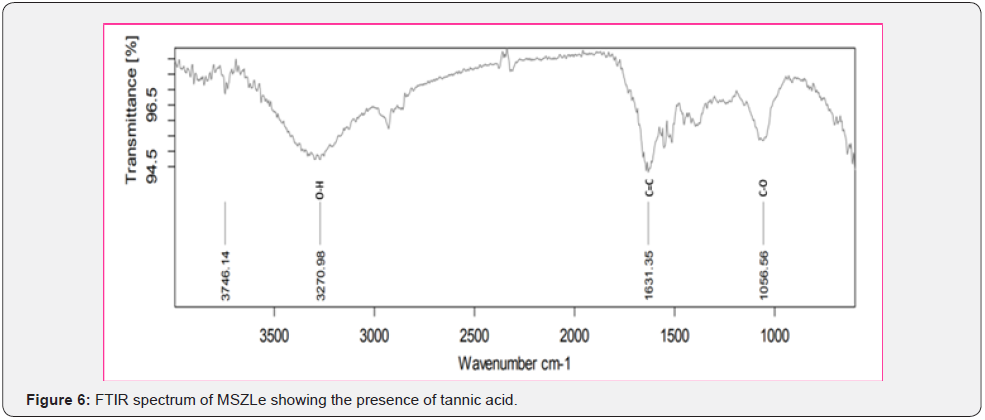
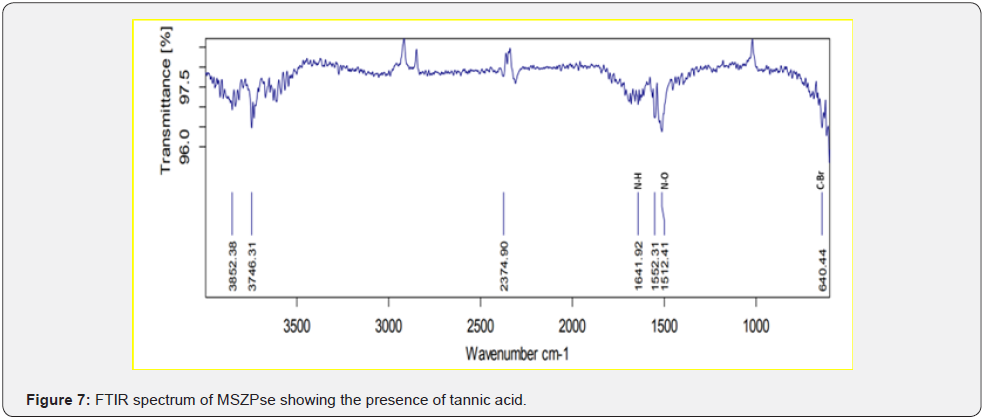
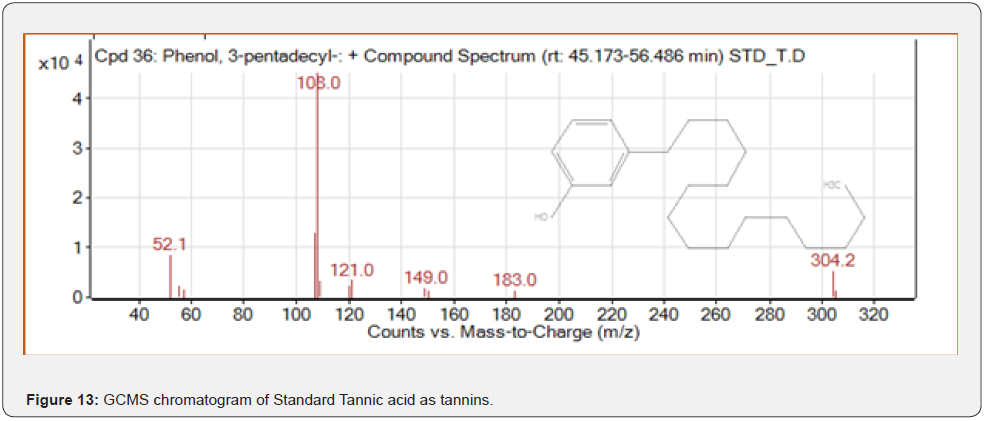
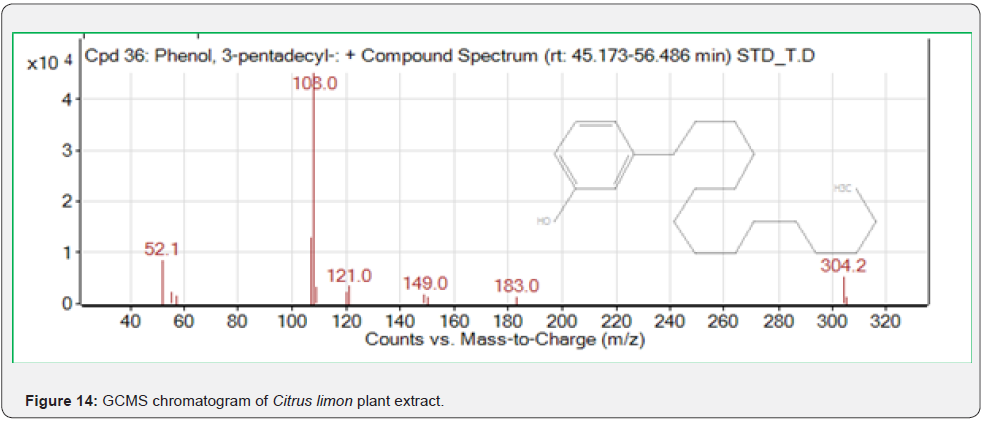
Based on peak values in the infrared region, FTIR analysis was performed to identify the functional group of active components. The FTIR spectrum was investigated, and the FTIR spectrum profile results were shown in Figures 4-7 also reported in Table 3. The extract of Citrus limon leaf MSZLd, MSZLe and MSZPse shows the presence of tannic acids as secondary metabolites. The functional groups discovered were matched with the tannin’s moiety in the range of 1000-1500 cm-1, according to FTIR spectroscopic data with compare to stander tannic acid (Figure 3)[28-30].

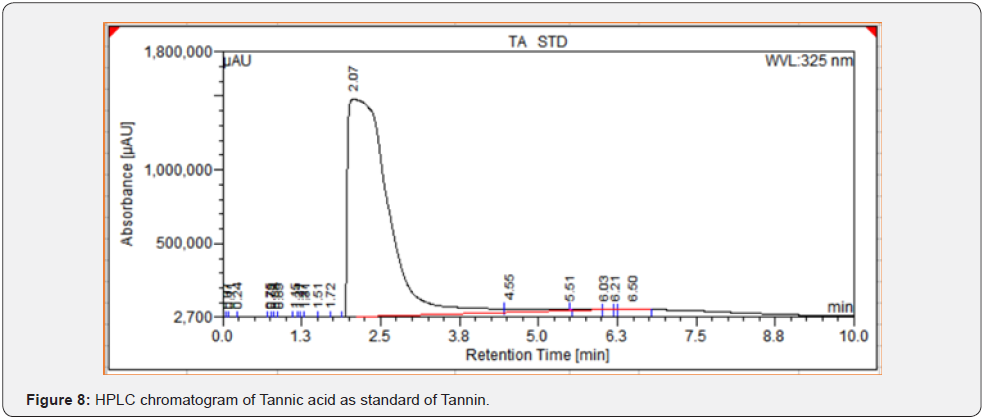
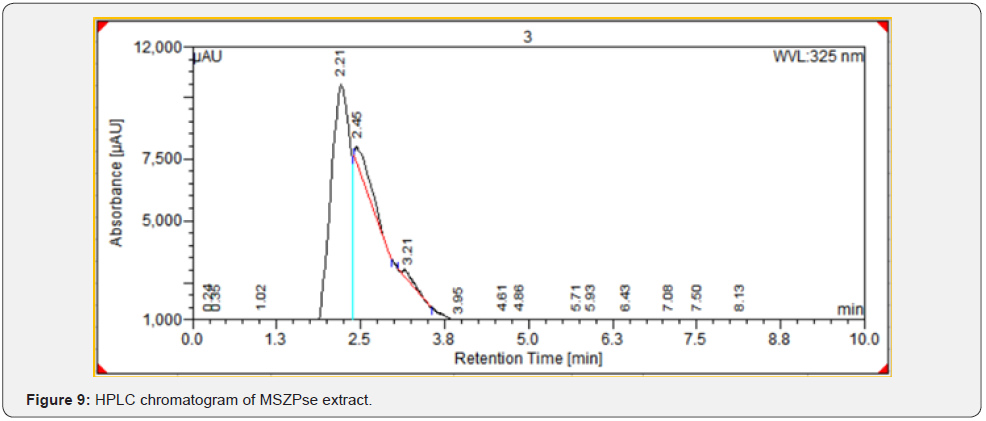
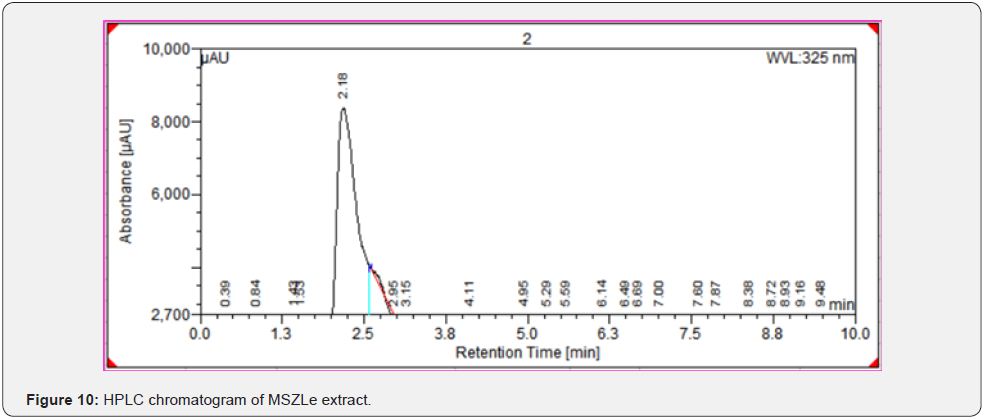
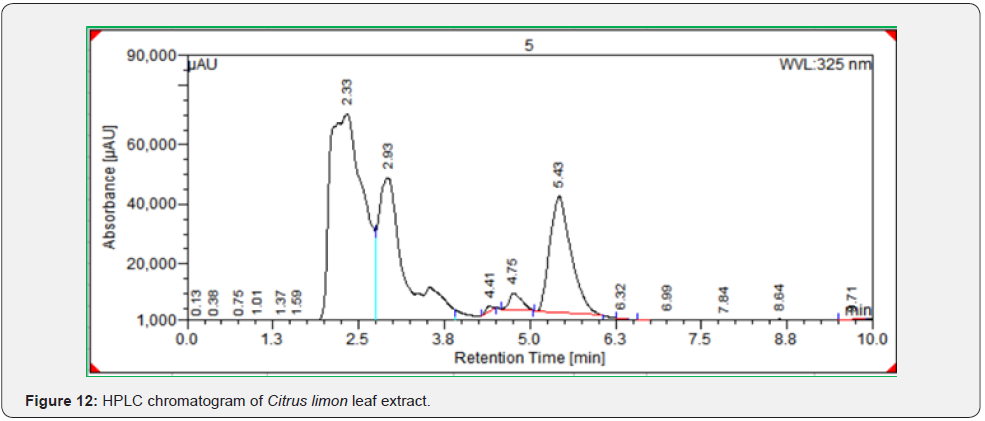
As per the paraoptometric analysis Plant leaf and all three bacterial endophytes producing in good number of tannins which were further tested with HPLC analysis. The standard Tannic acid HPLC chromatogram shows the compound revealed at 2.07min Figure 8. The highest peaks were present in seed endophyte MSZPse of Citrus limon in Figure 9, then leaf endophytes MSZLe of Citrus limon in Figure 10 and then MSZLd of Citrus limon in Figure 11. HPLC profile of Citrus limon leaf reveled 6 peaks of different phytochemical compounds with retention time 2.33min, 2.93min, 4.41min, 4.75min, 5.43min and 6.32min respectively in Figure 12. All extractions detected under UV detection at 325nm. Citrus limon plant HPLC data showing leaf extract and endophytic bacteria which isolated from leaf are produced such higher amount of tannin production. It may be considered that Citrus limon plant produced tannins due the presence of their inner microorganism, endophytes shown In-situ production of phytochemicals.

Conclusion
The tannin substance of various sections and endophytes of Citrus limon, which are restoratively vital plants, were discovered in the current study. The crucial report for these plant endophytes is a relative assessment of all out-tannin content. MSZLd, MSZLe, and MSZPse are bacterial endophytes isolated from Citrus limon leaves and seeds. Bacillus Spp. was identified using 16S rRNA nucleotide sequencing. Bacillus subtilis MK592771 was found in isolate MSZLd, Bacillus vallismortis MN533948 was identified in isolate MSZLe, and Bacillus licheniformis MN160317 was identified in isolate MSZPse. The NCBI gene bank contains all bacterial nucleotide gene sequences. The peak pattern of chromatograms obtained directly from HPLC and FTIR analysis may typically identify the source of various parts from Citrus limon and their endophytes. Similarly, GC analysis will reveal all the chemicals in the leaf extract and bacterial extract through specific qualitative and quantitative patterns. Citrus limon has a wide range of biological features that can be used to treat a variety of ailments. The chemicals discovered by the initial qualitative and GCMS analyses have a wide range of applications in the medical field. Each of the compounds discovered has a distinct property that can be used to treat a variety of ailments. More research is needed to uncover its significance in various fields to adequately treat disorders. This study, above provide information on Citrus limon identity, chemical contents, and physicochemical characteristics, indicating that the plant’s own endophytes produce more secondary metabolites than the plant itself. This is the first time that such a research of plant endophytes with phytochemicals as secondary metabolites has been described without triggering any genes. These inspections assist in the identification and approval of plant material. The right distinct verification and quality insistence of the starting components is a primary requirement for ensuring the reproducibility of local drugs, which will increase their success and appropriateness.
Acknowledgement
I am grateful to UGC RGNF fellowship for financial support, guide and Head of the Department of Microbiology and Biotechnology, Gujarat University for providing guidance and Spectra research laboratory - Himatnagar and Sicart laboratory - Anand for providing research facilities to conducting the research work.
Conflict of interest
The authors have no beyond reconciliation circumstance in arranging of this article.
References
- Ajiboye BO, Ibukun EO, Edobor G, Ojo AO, Onikanni SA (2013) Qualitative and quantitative analysis of phytochemicals in Senecio biafrae leaf. Int J Inv Pharm Sci 1(5): 428-432.
- Ezeonu CS, Ejikeme CM (2016) Qualitative and quantitative determination of phytochemical contents of indigenous Nigerian softwoods. New J Sci p. 9.
- Gnanaraja R, Prakash V, Peter S, Mahendraverman M (2014) Qualitative and quantitative phytochemicals analysis of selected fabaceae medicinal plants from Allahabad region. Pharma Innov J 3(7): 53-56.
- Saxena R, Sharma R, Nandy B, Jasuja DN (2014) Qualitative and quantitative estimation of bioactive compounds in Mimosa hamata. Int J Pharm Pharm Sci 6(6): 72-75.
- Rathod ZR, Meenu SS (2021) Plant as an inexhaustible medicinal source of Secondary metabolites. Vidya A J Gujarat Univ 1(1): 64-78.
- Rathod ZR, Meenu SS (2021) Effect of endophytic consortium on Pisum sativum and Vigna angularis plant analyst with ftir spectroscopy. Acta Sci Microbiol 4(12): 79-84.
- Rathod ZR, Meenu SS (2021) Spectrophotometric and HPLC estimation of total alkaloids of Citrus limon L Burm f (Lemon) and its Endophytes. Acta Sci Biotechnol 2(3): 21-26.
- Rathod ZR, Jain R, Sarita S, Shah RK, Lucie KM, et al. (2020) To Determine the minimum inhibitory concentrations and minimum bactericidal concentrations of the effective plants extract and endophytes of Citrus limon. Biotechnol Comm 13(1): 210-215.
- Saxena S (2021) Biologically active secondary metabolites from endophytic alternaria species. Endophytes pp. 1-20.
- Shad MA, Nawaz H, Rehman T, Ahmad HB, Badar H, Hussain M (2012) Optimization of extraction efficiency of tannins from Cichorium intybus : Application of response surface methodology. J Med Plants Res 6(28): 4467-4474.
- Yuliana P, Laconi EB, Wina E, Jayanegara A (2014) Extraction of tannins and saponins from plant sources and their effects on in vitro methanogenesis and rumen fermentation. J Indonesian Trop Anim Agric 39(2): 91-97.
- Harborne JB (1973) Phytochemicals methods. London: Hapman and Hall Ltd. 49-188.
- Ingle KP, Padole DA, Khelurkar VC (2017) Preliminary phytochemical screening of methanolic extract from different parts Of Jatropha curcas (L.). Multilogic Sci 6(19): 111-115.
- Madhubala M, Santhi G (2019) Phytochemical and GC-MS analysis on leaves of selected medicinal plants in Boraginaceae family Cordia dichotoma Pramana Res J 9: 2249-2276.
- Rathod ZR, Meet AD, Sarita S, Baldev VP, Meenu SS (2020) A review on phytochemical screening of secondary metabolites. Vidhya A J Gujarat Univ 15(2): 175-190.
- Rathod ZR, Sonal KM, Venisha VC, Meenu SS (2021) Pharmacognostic and phytochemical evaluation of Citrus limon (Lemon) fruit and leaf. Int J Inn Res Sci Engg Tech 10(2): 115-119.
- Rathod ZR, Venisha VC, Sonal KM, Meenu SS (2021) Preliminary phytochemical investigation of extraction from Citrus limon. Int J Sci Engg Tech 9(1): 1-5.
- Rathod ZR, Meet AD, Amita YM, Baldev VP, Meenu SS (2020) Book chapter: a review on extraction, tools and techniques of phytochemicals. PLANTA Research Book Series. 1: 129-140.
- Rathod ZR, Meet AD, Amita YM, Baldev VP, Meenu SS (2020) A review on phytochemical analysis of secondary metabolites and extraction techniques. Int Engg J Res Develop 5(5): 1-10.
- Rathod ZR, Meet AD, Amita YM, Baldev VP, Meenu SS (2020) A review article: Citrus as salt tolerance under plant tissue culture. Int J Engg Res Develop 7(8): 34-44.
- Sonam M, Singh RP, Pooja S (2017) Phytochemical screening and TLC profiling of various extracts of Reinwardtia indica. Int J Pharmacogn Phytochem Res 9(4): 523-527.
- Rathod ZR, Meet AD, Amita YM, Ritika J, Baldev VP, et al. (2020) Compatibility studies on different endophytes of Citrus limon antagonistic to bacterial with plant pathogen. Adv Biores 11(3): 126-129.
- Rathod ZR, Venisha VC, Meenu SS (2021) Assessment of endophytic Bacillus spp as exopolysaccharide producers with reference of FTIR analysis. Acta Sci Biotechnol 2(7): 05-12.
- John B, George S, Reddy V (2014) Spectrophotometric estimation of total alkaloids in selected Justicia species. Int J Pharm Pharmaceutic Sci 6(5): 647-648.
- Rathod ZR, Sarita S, Meenu SS (2022) Isolation and screening of multivariant enzyme producing endophytic bacteria from Citrus limon. Acta Sci Microbiol 5(2): 38-42.
- Mukherjee PK (2002) An approach to evaluation of Botanicals. In: Quality control of herbal drugs. Business Horizons, New Delhi, India, pp. 248.
- Rathod ZR, Shah D, Shukla P, Meghani S, Patel C (2022) Citrus limon as an abundant source of phytochemicals their evaluation and antioxidant analysis with bacterial endophytes. Curr Trends Biomedical Eng & Biosci 20(4): 1-11.
- Vijay S (2017) Evaluation on anti-diabetic effect of ethanolic extract of whole plant of Nymphaea Pubescens on streptozotocin induced diabetes in wistar rats.oral dissertation, Masters thesis, C.L.Baid Metha College of Pharmacy, Chennai, India.
- Saxena M, Jyoti S, Neema R, Singh D, Guptha A (2013) Phytochemistry of medicinal plants. J Pharmacognosy Phytochem 1(6): 168-182.
- Rathod ZR, Axita PV, Parth RM, Prachi KP, Payal KG, et al. (2022) In Vitro evaluation of protein and antioxidant activity of different probiotic and endophytic bacteria Curr Trends Biomedical Eng & Biosci 20(4): 556042.






























