Salinity Management in Glycine Max L. Using Cytokinin from Rhizobacteria Isolated from Mines and Dump Sites
Sarita Sharma, Rathod Zalak R and Meenu S Saraf*
Department of Microbiology and Biotechnology, University School of Sciences, Gujarat University, India
Submission:April 30, 2022;Published:May 17, 2022
*Corresponding author:Meenu S Saraf, Department of Microbiology and Biotechnology, University School of Sciences, Gujarat University, Ahmedabad- 380009, Gujarat, India
How to cite this article: Sarita S, Rathod Zalak R, Meenu S S. Salinity Management in Glycine Max L. Using Cytokinin from Rhizobacteria Isolated from Mines and Dump Sites. Curr Trends Biomedical Eng & Biosci. 2022; 20(5): 556047. DOI:10.19080/CTBEB.2022.20.556047
Abstract
Salinity stress is one of the most damaging abiotic stresses, and it is quickly spreading over the world. It has a major negative impact on plant health and yield, resulting in massive losses. The current work looks at the salt tolerance of rhizobacteria SHHMZ4, SMHMZ46, and SMHMP23 isolated from mines and landfills. These isolates grew in Nutrient broth with NaCl concentrations ranging from 0% to 20% and were able to synthesis cytokinin under salty conditions (2 percent w/v NaCl) and generated 19, 23, and 20 μg/ml, respectively. To confirm cytokinin biosynthesis, TLC and FTIR analyses of isolated cytokinin and reference standard kinetin were performed. Plant investigations in vitro were also performed to establish the efficiency of the cytokinin-producing rhizobacteria. According to the findings of this study, PGPR has a significant function in enhancing salt tolerance in plants and can be utilized as a biofertilizer to promote crop growth in saline areas.
Keywords: Phytohormone; Cytokinin; Salt tolerant PGPR; Rhizobacteria; Salt stress
Introduction
Drought and salt, which create osmotic stress and impede crop growth and development, have a significant impact on crop productivity. Salinity is a negative state that occurs in arid and semi-arid areas all over the world. It is one of the most serious environmental concerns endangering the food security of the world’s growing human population, resulting in a yearly loss of 1-2 percent of arable land [1]. Plants’ cellular processes are altered by salinity, resulting in a variety of physiological, morphological, biochemical, and molecular changes [2]. The accumulation of toxic ions such as Na+ and Cl– in cells, as well as improper nutrient absorption and oxidative stress damage, all have a significant impact on plant growth and development [3,4]. When plants are exposed to high salt stress, the rhizosphere acts as a reservoir for plant growth-promoting rhizobacteria (PGPRs), which can aid plant acclimation and growth. Through both direct and indirect impacts on plant growth, PGPRs play a crucial role in promoting plant development even in stressful settings. Among the direct mechanisms include phytohormone production (for example, cytokinin), enhanced nitrogen fixation, phosphate solubilization, HCN release, and so on [5,6]. Bacterial species such as Alcaligenes, Azospirillum, Arthrobacter, Klebsiella, Acinetobacter, Bradyrhizobium, Bacillus, Burkholderia, Enterobacter, Erwinia, Flavobacterium, Pseudomonas, Serratia, and Rhizobium have already been found as plant growth promoters [7-9].
Cytokinins are phytohormones that play a significant role in the cell cycle and impact a number of developmental programmed [10]. Endogenous cytokinin levels are exceedingly low in plant tissues [11]. The ability of rhizobacteria to produce phytohormones such as cytokinin is a key role in plant growth. Cytokinin is widely distributed in higher plants, algae, and bacteria, and it is also produced by plant-associated microbes [12,13]. This hormone is vital in the development of plant cancers [14]. Cytokinin regulates cell division, cell enlargement, and tissue expansion, which improves plant growth and yield and plays an important role from seed germination to leaf and plant senescence [4,15], as well as harmonizing important physiological processes throughout the plant’s lifecycle [16,17]. The current research focuses on the discovery and characterization of cytokinin-producing rhizobacteria isolated from the Zawar mines in Udaipur, Rajasthan, and the Pirana waste site in Ahmedabad, Gujarat. We highlight the efficacy of salt-tolerant rhizobacteria for cytokinin biosynthesis, as well as their aptitude as an alternative ecofriendly bio-enhancer for enhanced crop production in salt-stressed situations, in this research.
Materials and Methods
Rhizobacterial Isolates
We obtained 51 isolates from the Zawar mines in Udaipur, Rajasthan, and 40 isolates from the Pirana waste site in Ahmedabad, Gujarat, in previous research. Three isolates (SMHMZ4, SMHMZ46, and SMHMP23) were shown to be highly resistant to heavy metals (Cd, Ni, and Pb) [18-20]. Under salt stress conditions, this rhizobacteria was tested for salt resistance and the ability to produce Phytohormones (Cytokinin).
Chemicals and Media
MgSO4, NaCl, Na2HPO4, CaCl2, and KH2PO4 were bought from Fine Chemicals (P) Ltd. in New Delhi, India. While the microbiological media viz. tryptone yeast broth, nutrient broth, agar powder, pre-coated TLC plates, casamino acid, thiamine, and biotin were obtained from SRL Pvt. Ltd, Mumbai, India, the methanol, ethyl acetate, kinetin, sodium hypochlorite, carboxymethyl- cellulose, and glycerol were obtained from Hi Media Laboratories, Mumbai, India.
Screening of salt tolerant plant growth promoting rhizobacteria
The tolerance of the rhizobacterial isolate to NaCl was examined in nutrient broth (NB) medium with different NaCl concentrations (0, 2.5, 5, 7.5, 10, 12.5, 15, 20, 22.5, and 25% (w/v)). 50 μl of overnight grown rhizobacterial culture was inoculated in 5 ml of NB medium and incubated at 30 oC in a rotary shaker (Redmi, India) at 200 rpm for 24 hours. A UV-Vis spectrophotometer was used to evaluate the optical density of the rhizobacterial cultures at 600 nm (Systronics 166). To determine the level of salt tolerance, the Minimum Inhibitory Concentration (MIC) techniques were utilized. The MIC values are the lowest salt concentrations at which rhizobacterial growth is inhibited. To determine the lowest inhibitory concentration of all selected isolates, cultures were allowed to grow on N-agar plates treated with salt (NaCl). The initial concentration of salt was 1%, and it was gradually increased by 1% until no viable colonies were observed. One loopful of activated culture was streaked onto a Nutrient agar plate containing 1% NaCl and incubated at 37 oC for 24 to 48 hours. The following day, a loop of these incubated plates was streaked into a Nutrient agar plate containing 2% NaCl and incubated for 24 hours at 37 oC.
The Minimum Inhibitory Concentration (MIC) approaches were used to assess the level of salt tolerance. The MIC values are the lowest salt concentrations that inhibit rhizobacterial growth. Cultures were allowed to grow on N-agar plates treated with salt to determine the lowest inhibitory concentration of all selected isolates (NaCl). The starting concentration of salt was 1%, and it was subsequently increased by 1% until no viable colonies were seen. One loopful of activated culture was streaked onto a Nutrient agar plate containing 1% NaCl and incubated at 37 oC for 24 to 48 hours. The next day, a loopful of these incubated plates was streaked into a Nutrient agar plate containing 2% NaCl and incubated at 37 oC for 24 hours. This process was continued until a salt concentration was reached at which no viable bacterial growth could be observed [21,22].
Determination of cytokinin synthesis by salt tolerant rhizobacteria in M9 medium
Preparation of the inoculum: An inoculum of activated culture was transferred to fresh M9 media supplemented with 0.2 percent casamino acids, 0.01 percent thiamine, and 2 pg biotin per liter and cultivated for 5 days at 282 oC at 200 rpm (Remi, India). An aliquot of 0.1 percent (v/v) activated culture turbidity with an optical density of 0.90.1 at 600 nm was used as inoculum [23]. A Systronics 166 spectrophotometer was used to measure the growth rate at 600 nm [17,24].
Cytokinin production: For cytokinin production, fresh M9 medium supplemented with 0.2 percent casamino acids, 0.01 percent thiamine, 2 pg of biotin, and 2% (w/v) was used [23]. 250 ml Erlenmeyer flask with 100 ml M9 medium inoculated with 1 ml activated inoculum of salt tolerant rhizobacteria and incubated for 5 days at 282 oC at 200 rpm (Remi, India). The experiment was conducted out three times. After 72, 96, and 120 hours, cytokinin production was quantified spectrophotometrically at 665 nm. M9 medium injected with a single virus served as the control [17,24].
Extraction of crude cytokinin: After 72, 96, and 120 hours, the rhizobacterial cells were isolated from the supernatant by centrifugation for 15 minutes at 4 oC (Remi, India). The cell free supernatant was filtered using a 0.45 μm filter (Hi-media, India). Cytokinin was extracted three times with ethyl acetate. The extracted insoluble fraction was diluted in 1 ml of HPLC grade methanol and stored at -20 oC until further analysis [17,24].
Thin layer chromatography of extracted cytokinin: Thin layer chromatographic separation was achieved with a small volume (10 l) of ethyl acetate extract (sample) and standard cytokinin (kinetin) using mobile phase n-butanol: acetic acid: water (12:3:5 v/v/v) and was observed under UV light (254 nm) using a UV transilluminator (Biorad, India) [17,24].
FTIR characterization of extracted cytokinin: FTIR spectra obtained as described by utilizing BRUCKER Alpha ECO-ATR (Attenuated Total Reflectance) with 16 scan and 4000-400 cm-1 using extracted cytokinin dissolved in HPLC grade methanol confirmed the molecular identification of cytokinin produced by rhizobacteria [17, 25].
Pot experiments on Glycine max L. with selected rhizobacteria
The formation of saline soil: The sandy loamy soil came from the agricultural areas of Jagatpur, near Gota in Ahmedabad,Gujarat, India. At a rate of 100 mM/kg, this soil was treated with salt (NaCl). A soil sample was transported to IFFCO in Gandhinagar, Gujarat, India, for measurement of soil properties. This number was determined using the average NaCl content in saline soil, which ranged from 48 to 111 mM [26,27].
Seed bacterization and Pot studies: Soya bean seeds (Glycine max L.) were used in this study. The seeds were surface sterilized for 2 minutes in 70% ethanol and 5 minutes in 2% sodium hypochlorite before being washed ten times in sterile distilled water. The selected powerful isolates SMHMZ4, SMHMZ46, and SMHMP23 were grown in M9 medium at 282 oC for 5 days. Surface sterilized seeds were soaked overnight at room temperature in culture inoculated M9 media including sterilized carboxymethyl- cellulose (1 percent CMC) as an adhesive, then air dried for further research. As a control, seeds were treated with sterile distilled water amended with CMC alone. For the pot study, 35 (D) 27.5 CM pots were used, and 1000 gm of sterile soil amendment containing 100 mM NaCl/Kg soil was sown in each pot with 10 seeds. The plant’s vegetative profile was monitored after 30 days in the pot. The experiment was carried out in triplicate [17].
Plant analysis: Plant growth parameters such as fresh weight, dry weight, root and shoot length, pigments, and proline concentration were measured after 30 days of treatment. The DW was calculated after 72 hours of drying in an 80 oC hot air oven (REMI).
Chlorophyll content estimation
The chlorophyll content was determined using Arnon’s (1949) technique. To calculate the amount of chlorophyll a and b,the following formulas were employed.
Chlorophyll a
Chlorophyll b
where, OD665, OD645, = absorbance at respective wavelengths (nm); v = final volume of the filtrate (1 ml) and w = fresh weight of the leaf (0.1 g).
Proline Content
According to Bates et al. the proline content of the leaves was determined [28]. The concentration of samples was calculated with reference to standard graph of proline prepared in the range of 10-100 μg ml-1.
Statistical Analysis
The experiment used a randomized block design. All experiments were conducted in triplicate. Each treatment’s results were assumed as an arithmetic mean with standard error. Data were subjected to one-way analysis of variance (ANOVA) followed the DMRT (Duncan’s Multiple Range Analyze) by using IBM SPSS Statistics version 22. (SPSS Inc. Chicago, USA). The Levene test was applied to evaluate variance homogeneity
Result and Discussion
Physicochemical characteristic of soil sample
A standard method was used to examine the physicochemical parameters of soil samples. Table 1 summarizes the findings. Higher pH and chloride levels suggested alkaline soil. The organic and inorganic content of soil has a significant impact on the structure of the microbial community [17].
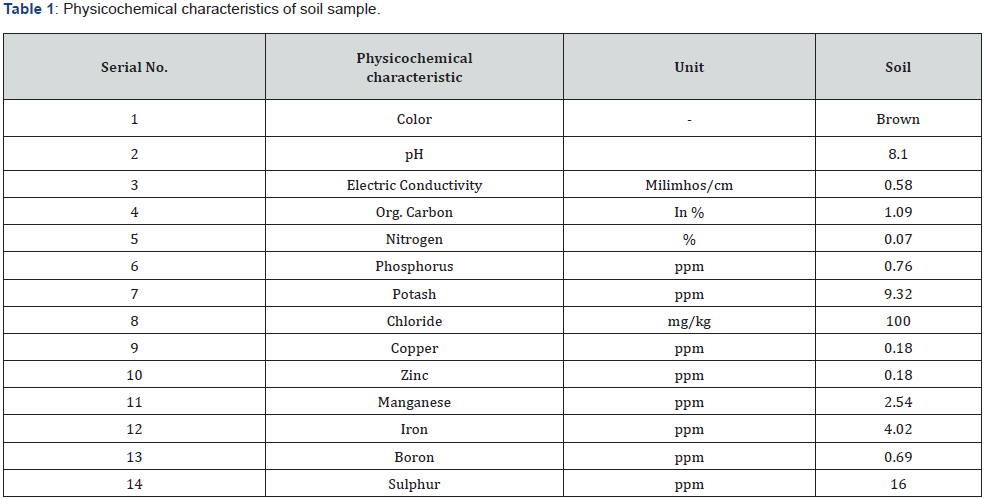
Screening of salt tolerance rhizobacteria
Salt tolerance rhizobacteria screening: All of the rhizobacterial isolates tested positive for NaCl tolerance in this study. Many scientists’ observations are consistent with the findings of our inquiry [17,26,27]. The rhizobacterial isolates SMHMZ4, SMHMZ46, and SMHMP23 grew in NB medium at NaCl concentrations ranging from 0 to 22.5 percent (Figure 1). The relative growth of these isolates in medium supplemented with varying amounts of NaCl is depicted in Figure 1. Higher salt concentrations of up to 22% were tolerated by SMHMZ4, SMHMZ46, and SMHMP23. For all isolates, the minimum inhibitory concentration was 23 percent NaCl.
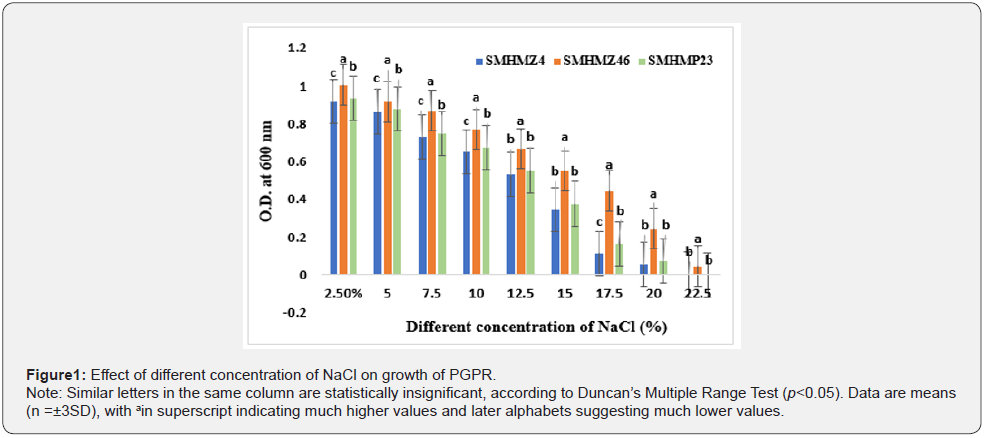
Determination of cytokinin synthesis in M9 Medium
The quantification of cytokinin secretion using a spectrophotometer, as described by [17,24]. All three isolates (SMHMZ4, SMHMZ46, and SMHMP23) produced cytokinin at 17.67, 23.23, and 18.26 μg/ml, respectively, under conventional test conditions (Figure 2). Shah et al. 2020 revealed that their microorganisms RM3 produced 18.1 μg/ml cytokinin in M9 medium under salt stress. In a peptone-rich growth medium, Karadeniz et al. found that the plant growth regulators auxin, gibberellin, cytokinin, and abscisic acid were synthesized as primary and secondary metabolites by their bacteria Proteus mirabilis, P. vulgaris, Klebsiella pneumoniae, Bacillus megaterium, B. cereus, and Escherichia coli [29].
Detection of cytokinin by thin layer chromatography
TLC plates were spotted with an ethyl acetate fraction of cytokinin and developed with an n-butanol:acetic acid:water (12:3:5 v/v/v) mobile phase. A blue speck was seen under UV light that recognized the cytokinin molecule (Figure 3). The extracted cytokinin sample in the lane had the same Rf value as the cytokinin reference standard (Kinetin). The findings are consistent with prior findings from our lab and other authors [17,24,30].

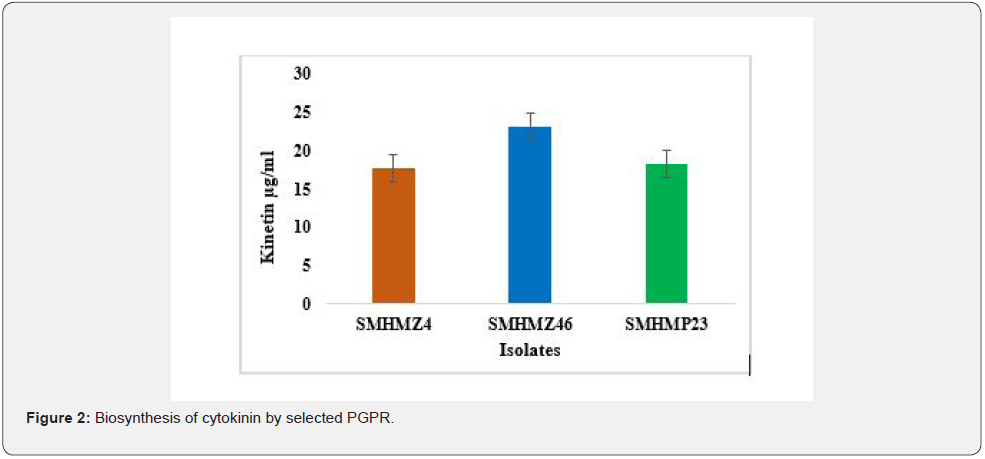
FTIR analysis of extracted cytokinin
The FTIR graph in Figure 4 indicates the presence of strong peaks in the stretching f region. The presence of cytokinin, as well as additional compounds with different functional groups created by SMHMZ46 under salt stress and capable of supporting plants in alleviating salt stress, was revealed by a full overlap of standard and sample peak locations. In a similar investigation, Shah et al. found that their strain RM3 produced cytokinin in the presence of 670 mM NaCl [17]. DS Hart and colleagues published an FTIR analysis of cytokinin in aqueous solution to determine its stability at different pH levels [31].
The Effect of salt-tolerant rhizobacteria on the growth of Glycine max L. under salinity stress
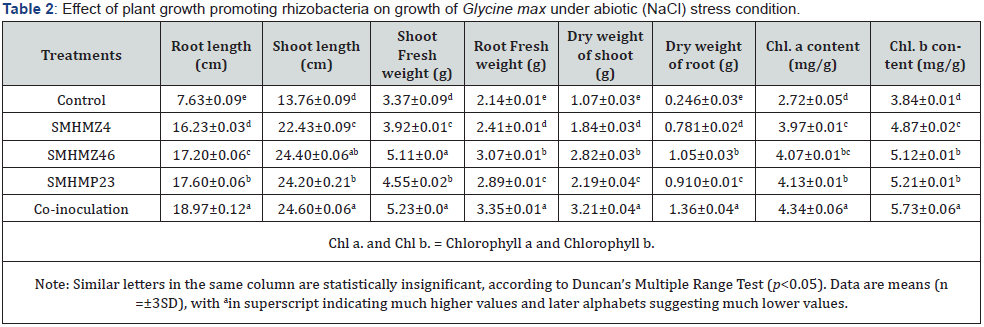
Salinity has a direct impact on the physiochemical and biological properties of soil, which has a negative impact on plant development and productivity. Because of osmotic stress, specific ion toxicity, nutritional imbalances, and/or a combination of these variables, salinity has a deleterious influence on plant growth. Several research on the application of PGPRs to alleviate salt stress in a range of crops have been reported [21,32,33]. Our findings showed that inoculating plants with NaCl-tolerant PGPR improved plant growth under salt stress conditions. This showed that rhizobacteria contact reduced NaCl stress. After being successfully treated with a cytokinin-producing bacterial culture, fresh and dry weight of shoots and roots increased, as did shoot and root length (Table 2). The results of a 30-day development cycle in a pot experiment with inoculated and untreated plant samples are shown in Figures 5 and 6. Under salt stress, un-inoculated Glycine max L. shoot and root lengths dropped significantly (p<0.05), whereas inoculations with SMHMZ4, SMHMZ46, and SMHMP23, as well as co-inoculation (all three bacterial strains), significantly enhanced their lengths.
Our results revealed that inoculated plants with NaCl-tolerant PGPR promoted superior plant growth under salt stress conditions. This demonstrated that rhizobacteria interaction alleviated NaCl stress. Successfully treated with a cytokinin-producing bacterial culture, there was an improvement in fresh and dry weight of shoots as well as roots, as well as an increase in shoot and root length (Table 2). Figures 5 and 6 illustrate the results of a 30-day growth cycle in a pot experiment with inoculated and untreated plant samples. Under salt stress, the shoot and root lengths of un-inoculated Glycine max L. decreased significantly (p<0.05), whereas inoculations with SMHMZ4, SMHMZ46, and SMHMP23, as well as co-inoculation (all three rhizobacterial strains), significantly increased their lengths. The growth-promoting and increased NaCl tolerance effects of the inoculated isolates could be attributed to Cytokinin synthesis in a NaCl-stressed environment. Sapre et al. [34] used ½ MS medium supplemented with 100 mM NaCl to study the effect of PGPR strain IG 3 (Klebsiella sp.) on the detrimental effect of NaCl on oat seedlings [21]. Root length, shoot dry weight, and root dry weight were substantially (p<0.05) higher in PGPR injected plants than in controls under stress conditions. In comparison to the control, halotolerant RM3 exhibited a favorable effect on Trigonella foenum-graecum under saline conditions, according to Shah et al. In Triticum aestivum L., the action of halotolerant consortia in boosting plant growth has also been proven [34].


Durum Wheat (Triticum turgidum subsp. durum) has also been shown to have PGPR activity from halotolerant bacteria [35]. It has been discovered that PGPR with ACC deaminase activity can protect against a wide range of abiotic stressors, including NaCl stress [36-38]. It is well understood that phytohormones, notably IAA generated by rhizobacteria, are required for root initiation and the elongation of root lengths of lateral roots and adventitious roots, hence supporting the host plant in maximum nutrient absorption [39,40]. Plants seeded with PGPR strain IG 3 and treated under NaCl stress produced significantly more biomass than negative control plants.
Proline Content
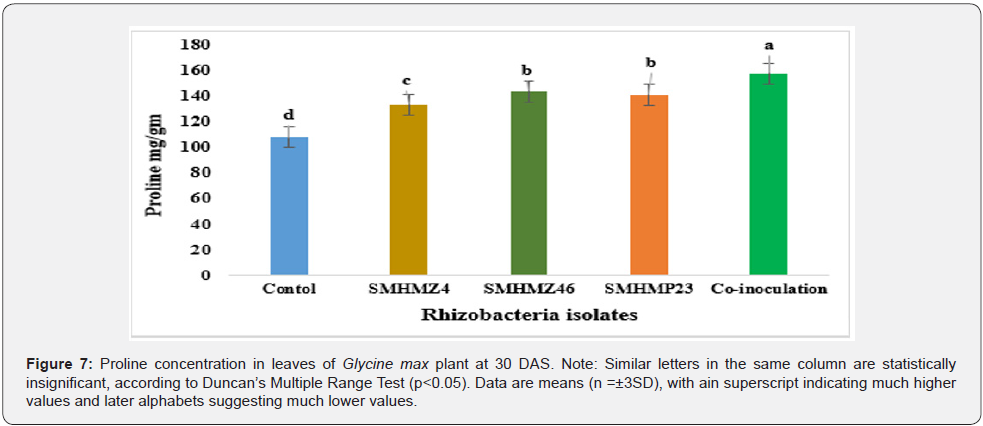
In reaction to environmental stress, plants have a defence mechanism that increases proline content. Proline could be beneficial in avoiding membrane damage [41]. In our study, rhizobacteria treatment on the plants significantly increased free proline content (p<0.05), which may contribute to the development of ecological adaptation in the Glycine max L. plant under stress conditions (Figure 7). According to Trivedi et al. their endophytes boost proline content in Glycine max L. under stress circumstances as compared to a non-inoculated control [42].
Conclusion
The current study is an important step in determining beneficial rhizobacteria and investigating their potential to improve plant growth under abiotic stress conditions. Plant development and growth are mostly dependent on rhizobacteria, which encourage plant growth. This study focuses on the production of cytokinin by isolated rhizobacteria, a phytohormone that promotes plant survival under abiotic stress such as salt by assisting in plant cell proliferation and differentiation. Isolation of such varied rhizobacterial isolates in the current study might be a beneficial step toward better agricultural yield and production under salt stress. The future use of these isolates with increased cytokinin production will maximum values the development of bio-based products it can be used as bio-enhancers.
Acknowledgement
I would like to express my sincere gratitude to my guide Prof. (Dr.) Meenu Saraf for support and guidance and DIST-FIST sponsored Department of microbiology and biotechnology, University school of sciences, Gujarat university, Gujarat, India for providing required facility.
Conflict of Interest
The author declares no conflict of interest in preparing this article.
References
- Shrivastava P, Kumar R (2015) Soil salinity: A serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J Biol Sci 22(2): 123-131.
- Gupta B, Huang B (2014) Mechanism of salinity tolerance in plants: physiological biochemical and molecular characterization. Int J Genomics 2014:701596.
- Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59: 651-681.
- Shaikh NB, Shaikh N, Rochlani A, Dalwani A, Sharma S, et al. (2022) Rhizobacteria that promote plant growth and their impact on root system architecture root development and function. Acta Sci Microbiol 5(4): 53-62.
- Ansari FA, Ahmad I (2018) Biofilm development plant growth promoting traits and rhizosphere colonization by Pseudomonas entomophila FAP1: a promising PGPR. Adv Microbiol 8(3): 235.
- Rochlani A, Dalwani A, Shaikh NB, Shaikh N, Sharma S, et al. (2022) Plant growth promoting Rhizobacteria as Biofertilizers: application in agricultural sustainability. Acta Scientific Microbiol 5(4): 12-21.
- Saraf MS, Sharma S, Thakkar A (2017) Production and optimization of siderophore from plant growth promoting Rhizobacteria. Scholar Press 1-85.
- Yasin NA, Akram W, Khan WU, Ahmad SR, Ahmad A, et al, (2018) Halotolerant plant-growth promoting rhizobacteria modulate gene expression and osmolyte production to improve salinity tolerance and growth in Capsicum annum L. Environ Sci Pollut Res Int 25(23): 23236-23250.
- Khan N, Zandi P, Ali S, Mehmood A, Adnan Shahid M (2018) Impact of salicylic acid and PGPR on the drought tolerance and phytoremediation potential of Helianthus annus. Front Microbiol 9: 2507.
- Skoog F, Strong FM, Miller CO (1965) Cytokinins. Science 148(3669): 532-533.
- Letham R (1994) The work of Christ. Scottish Bulletin of Evangelical Theology 12: 53-76.
- Conrad K, Bettin B, Neumann S (1992) The cytokinin production of Azospirillum and Klebsiella possible ecological effects, In Physiology and biochemistry of cytokinins in plants. Symposium Liblice República de Checoslovaquia pp. 401-405.
- Tirichine L, Sandal N, Madsen LH, Radutoiu S, Albrektsen A, et al. (2007) A gain-of-function mutation in a cytokinin receptor triggers spontaneous root nodule organogenesis. Science 315(5808): 104-107.
- Liu F, Xing S, Ma H, Du Z, Ma B (2013) Cytokinin-producing plant growth-promoting rhizobacteria that confer resistance to drought stress in Platycladus orientalis container seedlings. Appl Microbiol Biotechnol 97(20): 9155-9164,
- Werner T, Motyka V, Laucou V, Smets R, Van Onckelen H, et al. (2003) Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. The Plant Cell 15(11): 2532-2550.
- Timmusk S, Nicander B, Granhall U, Tillberg E (1999) Cytokinin production by Paenibacillus polymyxa. Soil Biol Biochem 31(13): 1847-1852.
- Shah RK, Jain R, Sharma S, Lucie KM, Raval VH, et al. (2020) Salinity Alleviation in Trigonella foenum-graecum by Employing Cytokinin from Rhizobium sp. Biosci Biotechnol Res Comm 13(1): 27-34.
- Sharma S, Shah RK, Rathod ZR, Jain R, Lucie KM, et al. (2020) Isolation of heavy metal tolerant Rhizobacteria from Zawar Mines area Udaipur Rajasthan India. Biosci Biotechnol Res Comm 13(1): 233-238.
- Sharma S, Rathod ZR, Saraf MS (2021) Elucidate the influence of heavy metal on bacterial growth isolated from a mining location and a waste dump: using their inducible mechanism. Curr Trends Biomedical Eng & Biosci 20(2): 556034.
- Sharma S, Rathod ZR, Saraf MS (2022) Exploring the biotic stress tolerance potential of heavy metal tolerate rhizobacteria isolated from mines area and landfill site. Acta Scientific Microbiol 5(2): 31-37.
- Sapre S, Gontia-Mishra I, Tiwari S (2018) Klebsiella sp, confers enhanced tolerance to salinity and plant growth promotion in oat seedlings (Avena sativa). Microbiol Res 206: 25-32,
- Kothari VV, Kothari RK, Kothari CR, Bhatt VD, Nathani NM, et al. (2013) Genome sequence of salt-tolerant Bacillus safensis strain VK isolated from saline desert area of Gujarat, India. Genome Announc 1(5): e00671-13.
- Akiyoshi DE, Regier DA, Gordon MP (1987) Cytokinin production by Agrobacterium and Pseudomonas spp. J Bacteriol 169(9): 4242-4248,
- Patel T, Saraf M (2017) Biosynthesis of phytohormones from novel rhizobacterial isolates and their in vitro plant growth-promoting efficacy. J Plant Inter 12(1): 480-487.
- Kamnev AA, Shchelochkov AG, Perfiliev YD, Tarantilis PA, Polissiou MG (2001) Spectroscopic investigation of indole-3-acetic acid interaction with iron (III). J Mol Structur 563: 565-572.
- Sagar A, Riyazuddin R, Shukla PK, Ramteke PW, Sayyed RZ (2020) Heavy metal stress tolerance in Enterobacter sp, PR14 is mediated by plasmid. Indian J Exp Biol 58: 115-121.
- Yadav AN, Kour D, Sharma S, Sachan SG, Singh B, et al. (2019) Psychrotrophic microbes: biodiversity mechanisms of adaptation and biotechnological implications in alleviation of cold stress in plants. In: Plant growth promoting rhizobacteria for sustainable stress management. pp. 219-253.
- Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies, Plant and Soil 39(1): 205-207.
- Karadeniz AS, Topcuoglu SF, Inan SU (2006) Auxin gibberellin cytokinin and abscisic acid production in some bacteria. World J Microbiol Biotechnol 22(10): 1061-1064.
- Tranova L, Bucek J, Zatloukal M, Cankar P, Styskala J (2019) Synthesis of [15N4] purine labeled cytokinin glycosides derived from zeatins and topolins with 9‐β‐d 7‐β‐d‐glucopyranosyl or 9‐β‐d‐ribofuranosyl group. J Labelled Comp Radiopharm 62(3): 118-125.
- Khatri P, Rana JS, Sindhu A, Jamdagni P (2019) Effect of additives on enhanced in-vitro shoot multiplication and their functional group identification of Chlorophytum borivilianum Sant, Et Fernand. SN Appl Sci 1(9): 1-10.
- Bharti N, Pandey SS, Barnawal D, Patel VK, Kalra A (2016) Plant growth promoting rhizobacteria Dietzia natronolimnaea modulates the expression of stress responsive genes providing protection of wheat from salinity stress, Scientific Rep 6(1): 1-16.
- Kusale SP, Attar YC, Sayyed RZ, Malek RA, Ilyas N, et al. (2021) Production of plant beneficial and antioxidants metabolites by Klebsiella variicola under salinity stress. Molecules 26(7): 1894.
- Rajput L, Imran A, Mubeen F, Hafeez FY (2018) Wheat (Triticum aestivum L.) growth promotion by halo-tolerant PGPR-consortium. Soil Environ 37(2): 178-189.
- Albdaiwi RN, Khyami-Horani H, Ayad JY, Alananbeh KM, Al-Sayaydeh R (2019) Isolation and characterization of halotolerant plant growth promoting rhizobacteria from durum wheat (Triticum turgidum subsp, durum): cultivated in saline areas of the dead sea region, Front Microbiol 10: 1639.
- Gontia‐Mishra I, Sapre S, Sharma A, Tiwari S (2016) Amelioration of drought tolerance in wheat by the interaction of plant growth‐promoting rhizobacteria. Plant Biol 18(6): 992-1000.
- Gontia-Mishra I, Sapre S, Kachare S, Tiwari S (2017) Molecular diversity of 1-aminocyclopropane-1-carboxylate (ACC): deaminase producing PGPR from wheat (Triticum aestivum L,): rhizosphere. Plant and Soil 414(1): 213-227.
- Marraiki N (2021) Inoculation of Klebsiella variicola alleviated salt stress and improved growth and nutrients in wheat and maize. Agronomy 11(5): 927.
- Patten CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl Environ Microbiol 68(8): 3795-3801.
- Jha B, Gontia I, Hartmann A (2012) The roots of the halophyte Salicornia brachiata are a source of new halotolerant diazotrophic bacteria with plant growth-promoting potential. Plant and Soil 356(1): 265-277.
- Ain-Lhout F, Zunzunegui M, Barradas D, Tirado R, Clavijo A, et al. (2001) Comparison of proline accumulation in two Mediterranean shrubs subjected to natural and experimental water deficit. Plant and Soil 230(2): 175-183.
- Trivedi G, Patel P, Saraf M (2020) Synergistic effect of endophytic selenobacteria on biofortification and growth of Glycine max under drought stress. South Afr J Botany 134: 27-35.






























