Citrus limon as an Abundant Source of Phytochemicals their Evaluation and Antioxidant Analysis with Bacterial Endophytes
Rathod Zalak R, Shah Dhrumi, Shukla Pooja, Meghani Sonali, Patel Charmi, and Saraf Meenu S*
Department of Microbiology and Biotechnology, University School of Science, Gujarat University, India
Submission:April 01, 2022;Published:April 28, 2022
*Corresponding author:Saraf Meenu S, Department of Microbiology and Biotechnology, University School of Science, Gujarat University, Ahmedabad, Gujarat, India 380009
How to cite this article: article: Rathod Z R, Shah D, Shukla P, Meghani S, Patel C. Citrus Limon as an Abundant Source of Phytochemicals Their Evaluation and Antioxidant Analysis with Bacterial Endophytes. Curr Trends Biomedical Eng & Biosci. 2022; 20(4): 556045. DOI:10.19080/CTBEB.2022.20.556045
Abstract
Citrus limon (Lemon) belongs to Family Rutaceae, is commonly known as Kagdi Limbu or Lime. The peel is a leathery exocarp, or skin of the fruit containing numerous oil glands. Lime peel tea was traditionally used as an appetite stimulant, for cold and cough, for sore throat and as a digestive stimulant. It can also be used as an astringent for skin. Lime peel essential oil is use as an aroma and flavour enhancer. The goal of this study was to determine the phytochemical analyses of endophytes isolated from several parts of the Citrus limon plant, including the leaf, steam, root, seed, and peel. The presence of various phytochemical compounds was determined through qualitative analysis. This study will provide referential information for the correct identification of the bioactive compounds and suitable solvent system for separation of those compounds from the isolates of Citrus limon. The quantitative analysis of the phytochemical compounds allows, by using standard analytical technique, the determination of chemical groups, or compounds in aqueous extracts from different plants. For the determination of the antioxidant activity of the isolate is detected by DPPH, and FRAP method. DPPH gave the idea about % scavenging activity of the isolate.
Keywords: Citrus limon; Folin–Ciocalteu reagent; DPPH; FRAP
Introduction
The rhizosphere and phyllosphere bacteria of Citrus limon, a common evergreen fruit crop, have been examined. Citrus is one of the most important fruit crops on the planet, with significant economic, social, and cultural implications in our community. By employing plants as a natural source, it would be possible to synthesize bioactive compounds that cannot be produced through chemical reactions. Interactions between plants and endophytic bacteria must be explored in order to comprehend the ecological and environmental implications of this symbiotic relationship. The antioxidant area is rapidly expanding, owing to the fact that oxidative stress is linked to the onset of a wide range of diseases, including diabetes, cancer, and numerous aging-related and central nervous system ailments. Various free radicals are generated during rigorous oxidative reactions, which can be significant precursors of systemic cell and tissue damage. Antioxidants operate as oxidation inhibitors, eliminating free radical intermediates such as Reactive Oxygen Species (ROS) and Reactive Nitrogen Species (RNS) and oxidizing themselves to protect the organism from free radicals and hazardous chain reactions. Despite their low concentration, they can perform a variety of physiological tasks in the body to prevent the dangerous oxidation reaction. Antioxidant is not a new phrase; herbal plants have been considered good antioxidants since ancient times. Tocopherol, ascorbic acid, and flavonoids are the most well investigated antioxidants in medicine and pharmacology due to their anticancer, antimutagenic, and antitumor properties. UV filtration, symbiotic nitrogen fixation, and flower coloring are all aided by flavonoids in higher plants. Chemical messengers, physiological regulators, and cell cycle inhibitors are all possible functions of these molecules. Saponins can protect plants from bacteria and fungus by acting as anti-feedants. Tannins defend plants from predators, and they may also act as insecticides and regulate plant growth. Alkaloids have a wide range of pharmacological activities including antimalarial (e.g. quinine), antiasthma (e.g. ephedrine), anticancer (e.g. homoharringtonine), cholinomimetic (e.g. galantamine), vasodilatory (e.g. vincamine), antiarrhythmic (e.g. quinidine), analgesic (e.g. morphine), antibacterial (e.g. chelerythrine) and antihyperglycemic activities (e.g. piperine).
Materials and Methods
Isolation and characterization of endophytic bacteria
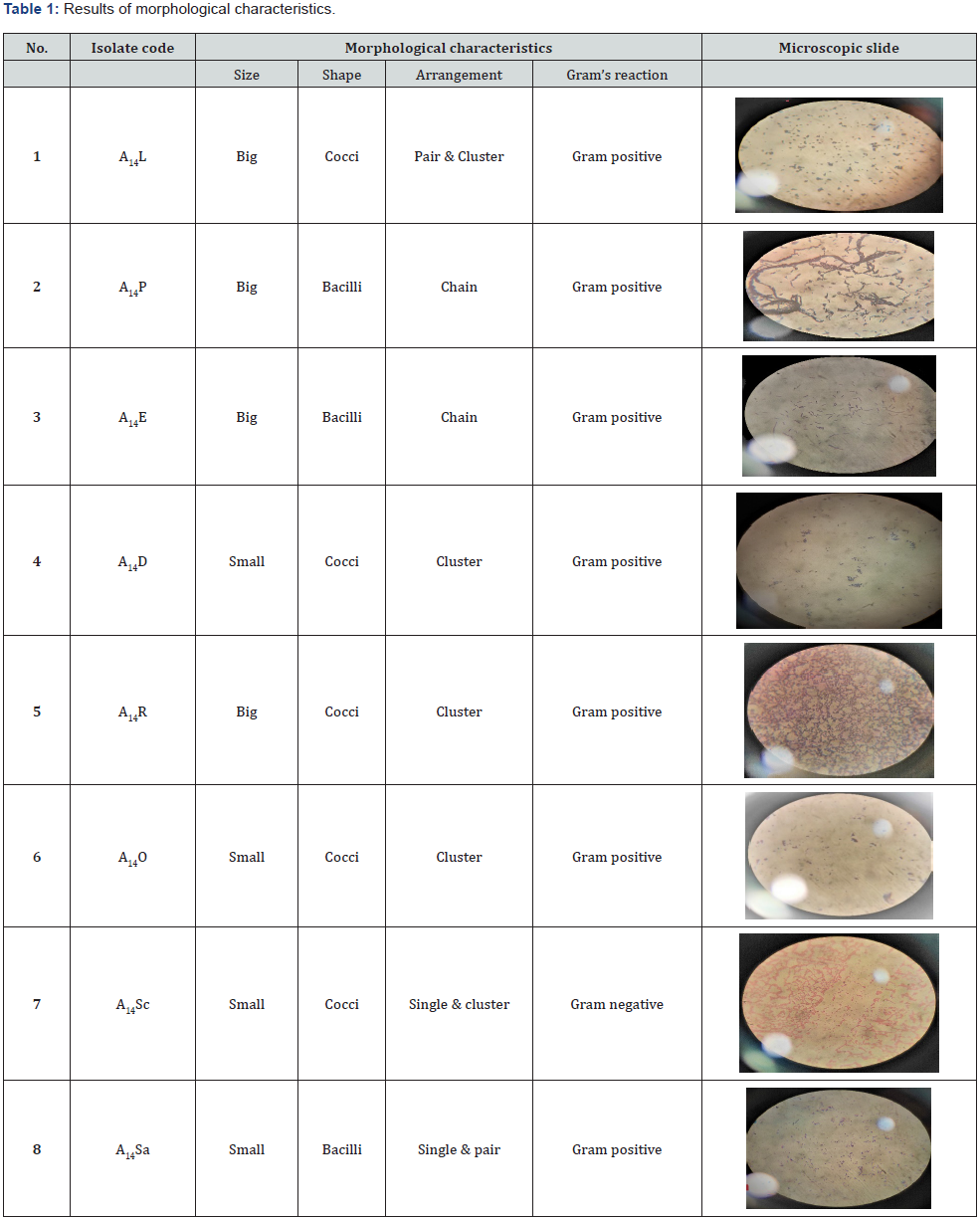
Endophytic bacteria were recovered from the leaves, stems, roots, and seeds of Citrus limon. Eight isolates were described using morphological and cultural characteristics. Gram staining offered morphological information during a microscopic examination. The visual analysis of colonies grown on Nutrient agar media revealed cultural characteristics.
Extraction
After incubation, 100ml of fermentation broth of each culture was taken in centrifuge tube and then broth was centrifuged at 4℃ for 5 minutes at 10000 rpm by using cooling centrifuge. Supernatant was discarded and cell pellets were resuspended in 20ml prechilled methanol (solvent: broth ratio is 1:4). By using vortex mixture, cell pellets were vortexed for 3 min. The cell pellets were preserved in deep freezer at -20℃ until the antioxidant test and amino acids estimation tests were performed. All the standards for the test like phenols, Flavonoids, Alkaloids, Saponins and Tannins were taken in the powder form belonging to the sigma company HPLC brand
Quantitative evaluation by TLC- Thin layer Chromatography
Due to natural variability, chemical analysis of plant material is a great challenge and requires special approaches. TLC was performed with two different chromatographic systems. Thin layers were prepared from microcrystalline cellulose and silica gel G. standard solution and samples were spotted on precoated plates. Silica plates act as stationary phase. The standard and unknown solution moves along with the solvent system. Here eight isolates were taken as an unknown sample, which is isolated from the citrus limon plant. The illumination with UV light causes fluorescence and makes the spots visible. The Rf value is counted for both standard and unknown solution. In addition, the value of unknown sample was compared with the standard solution.
Phenolic compounds
The solvent system used for phenolic compounds is Ethyl acetate: Toluene: Formic acid in ratio of 2.2: 1.1: 1.1. The detection reagent use for the detection of phenolic compounds is Ferric chloride (FeCl3). After using the detection reagent, the spots were examined under the UV light because some of the components can’t be seen with visible light and need to be projected by ultraviolet light.
Flavonoids
The solvent system used for the Flavonoids is Toluene: acetone: ethyl acetate in the ratio of 4.5: 4.5: 1. The detection of sheets were carried out under UV light. Rf value is counted for both standard and unknown eight samples. And the value of unknown samples was compared with the standard solution.
Chromatography for Alkaloids
The solvent system used for the Alkaloids is Chloroform: ethanol in the ratio of 9: 1. The detection of sheets were carried out under UV light. Rf value is counted for both standard and unknown eight samples. In addition, the value of unknown samples was compared with the standard solution.
Saponins
The solvent system used for the Saponins is Chloroform: methanol: water in the ratio of 65: 35: 10. The detection of sheets were carried out by using 10% H2SO4. Rf value is counted for both standard and unknown eight samples. And the value of unknown samples was compared with the standard solution.
Tannins
The solvent system used for the Tannins is Toluene: Acetone: Formic acid in the ratio of 60: 60: 10. The detection of sheets were carried out under UV light. Rf value is counted for both standard and unknown eight samples. In addition, the value of unknown samples was compared with the standard solution.
Qualitative Evaluation
The presence of specific bioactive components in plants confers therapeutic efficacy. Both qualitative and quantitative analyses are used in phytochemical analysis. While qualitative analysis is concerned with the presence or lack of a phytochemical. Different qualitative phytochemical investigations are known that allow the determination of chemical groups or compounds in aqueous extracts from various plants using standard analytical techniques.
Phenols: To determine the presence of phenol phytochemicals, 1 mL of extract was mixed with 2 mL of distilled water in a test tube, followed by a few drops of 10% ferric chloride (FeCl3). The presence of phenols is indicated by the presence of a blue or green colour. The extract (50mg) is mixed in distilled water, then 3ml of a 10% lead acetate solution is added to the mixture. The presence of phenolic chemicals is indicated by a bulky white precipitate (Rathod Zalak R. et al., 2021).
Flavonoids: 1 ml of extract and a few drops of dilute NaOH were added. The intense yellow colour produced in the plant extract which becomes colourless on addition of few drops of dilute mineral acid indicated the presence of flavonoids (Rathod Zalak R. et al., 2021).
Alkaloids: A few drops of Mayer’s reagent are added to solvent-free extracts to detect the presence of alkaloids. In the presence of Mayer’s reagent, alkaloids solution generates a creamcolored precipitate. 50mg solvent free extract is mixed with a few ml of dilute HCl and filtered, then 1 or 2 ml of Hager’s reagent is added to the filtrate. The presence of alkaloids is indicated by a bright yellow precipitate (Rathod Zalak R. et al., 2021).
Saponins: To determine the presence of saponin phytochemicals in various extracts, the extract was diluted with 20 mL distilled water and agitated for 15 minutes in a graduated cylinder. The presence of saponin is indicated by the creation of a 1cm layer of foam (Rathod Zalak R. et al., 2021).
Tannins: 1 ml of 5 percent ferric chloride added to solvent free extract in a test tube to check for tannin phytochemical content. The production of a bluish black or greenish black precipitate indicates the presence of tannin (Rathod Zalak R. et al., 2021).
Determination of Total phenolic content
Using the Folin-Ciocalteu method (Ahmed, Howladar, Mohamed, & Al-Robai, 2016; McDonald, Prenzler, Antolovich, & Robards, 2001), the total polyphenol content in the extracts was calculated. 5 ml Folin–Ciocalteu reagent, 4 ml sodium carbonate (75 g/l), and 4.9 ml distilled water were added to a 1 ml sample. After that, the mixture was incubated at room temperature for 90 minutes. Total polyphenol content was measured at 765 nm. Gallic acid was used as standard (Van Tan, et al., 2018).
Determine total Flavonoid content
The method was used to determine flavonoid content. Flavonoid will be a yellow complex formulation with AlCl3 solution in the samples. The amount of flavonoid content is directly related to the color intensity. The solution’s absorbance was measured at 510 nm wavelength, allowing the calibration curve and regression equation between quercetin content and absorbance to be established (Shoib A. Baba et al., 2014).
Determination of Total Alkaloid content
Measured out aliquots of the caffeine standard solution and place them in separate separatory funnels. Then, add 5 ml pH 4.7 phosphate buffers and 5 ml BCG solution to a combination of 1, 2, 3, and 4 ml chloroform and shake well. The extracts were collected in a 10-ml volumetric flask and diluted to achieve the desired concentration using chloroform as a solvent The complex’s absorbance in chloroform was measured at 470 nm against a blank made in the same way but without caffeine (Manjunath Ajanal et al., 2012).
Determination of Total Saponin content
In separate test tubes, ginsenoside Rb1 (2.0 g) was dissolved in 10 mL ethanol and serial dilutions of 0.2, 0.6, 1.0, 1.4, 1.8, 2.2, and 2.6 were made. These concentrations were evaporated to dryness and 5% w/v of 2.0 mL vanillin-acetic acid and 0.8 mL of perchloric acid were added to each test tube. Each test tube’s mixture was heated to 70°C for 15 minutes and then cooled for 20 seconds in an ice bath. After that, each test tube received 5 mL of glacial acetic acid. A UV spectrophotometer was used to measure the absorbance of each dilution at a wavelength of 550 nm. The absorbance data was utilised to create an absorbance vs. concentration graph (Theresa Edewor et al., 2016).
Determination of Total Tannins content
Folin-Ciocalteu was used to determine the tannins. A volumetric flask (10 ml) was filled with 0.1 ml of the sample extract, 7.5 ml of distilled water, 0.5 ml of Folin Ciocalteu phenol reagent, 1 ml of 35% sodium carbonate solution, and 10 ml of distilled water. The solution was thoroughly mixed before being stored at room temperature for 30 minutes. The same procedure was used to generate a set of tannic acid standard solutions (20, 40, 60, 80, 100 g/ ml). An UV/ Visible spectrophotometer was used to measure the absorbance of test and standard solutions against a blank at 700 nm (Kavitha Chandran et al.,2016)
Antioxidant activity by DPPH and FRAP method
DPPH method was developed by Blois (1958) by taking it in mind to determine the antioxidant activity by using a stable free radical α, α-diphenyl-β-picrylhydrazyl (DPPH). DPPH is stable free radical that display maximum absorbance at 517nm. When DPPH radicals come across proton (hydrogen) donating substrate such as antioxidant, free radical should be scavenged and absorbance would be reduced. The decreased in the absorbance is taken as measure for “Radical Scavenging action”. After 30 minutes of incubation, free radical scavenging property with sample is compared with Ascorbic acid as control. 95% scavenging activity of 0.02mg DPPH was achieved by 1mg/ml Ascorbic acid. Generally, 50μl of sample extract in methanol is mixed with 100μl of DPPH solution and 850μl methanol, by making final volume 1ml. Methanol is used as blank and distilled water + DPPH solution is used as control (Ktima SIRIVIBULKOVIT et al., 2018). Percentage DPPH radical scavenging activity (% DRSA) was calculated by the following equation,(%DRSA) = {(Ao–A1)/Ao} × 100 Ferric reducing Ability of Plasma (FRAP) assay is based on the principle of reduction of ferric tripyridyl triazine (Fe3+- TPTZ) complex to ferrous tripyridyl triazine (Fe2+- TPTZ) by the antioxidants of the sample at low pH. At absorption maximum 593 nm end product, Fe2+-TPTZ gives blue color and the change in absorbance is related to the antioxidant capacity of the plasma (Benzie IF et al., 1996).
Result and Discussion
Isolation and characterization
The endophytes isolated from various sections of the citrus limon plant, including the leaf, steam, root, seed, and peel, were analysed for phytochemicals. Eight isolates of citrus limon were identified and morphologically described using microscopic inspection and the gram staining technique which can be observed in Table 1.
Colony characterization of isolates
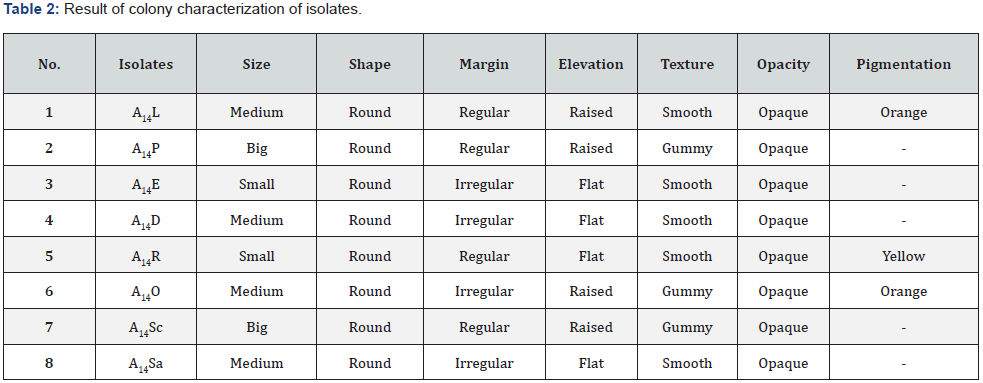
Phytochemicals were extracted from endophytes isolated from several parts of the citrus limon plant, including the leaf,steam, root, seed, and peel. Eight isolates of citrus limon were described by colony characteristics on Nutrient agar plate which can be observed in Table 2.
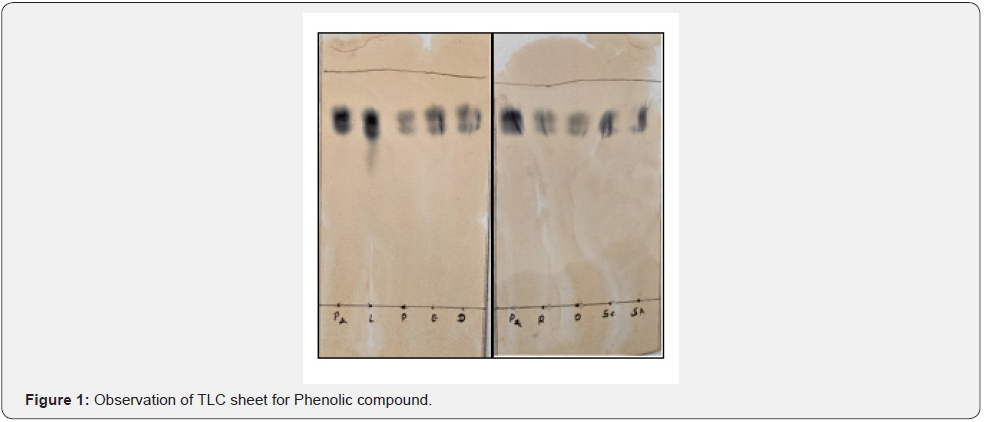
TLC (Thin Layer Chromatography)
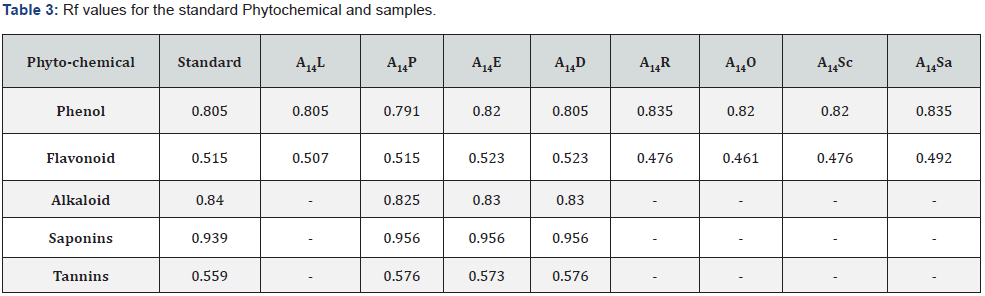
In the chromatogram shown in Figures 1-5, eight isolated and extracted sample fractions were detected in the TLC sheet, and the eluted species were labelled A14L, A14P, A14E, A14D, A14R, A14O, A14Sc, and A14Sa. For each standard and sample, the Rf value was calculated (Table 3). A14L, A14R, A14O, A14Sc and A14Sa did not give any spot near to standard spot for the Alkaloid, Saponins and Tannin. The Rf value of the sample was then compared to standard Phenol, Flavonoid, Alkaloid, Saponins and Tannin which can be observed in Table 3 & Figure 1-5.


Rf value is calculated as, Rf value = Distance travelled by solute/Distance travelled by solvent
Qualitative analysis of phytochemical
The qualitative tests were performed for all the endophytic bacterial isolates. All of the endophytic bacterial isolates were subjected to qualitative analysis of Phytochemical. All of the isolates gave positive results for the test of Phenol, Flavonoids where A14P, A14E and A14D gave positive results for the test of Alkaloid, Saponins and Tannins. A14P, A14E and A1414D gave better growth with positive results for the test of Phenol, Tannins compared to other isolates. A14R, A14O, A14Sc and A14Sa gave negative result for the test of Alkaloid, Saponins and Tannins which can be observed in Table 4.
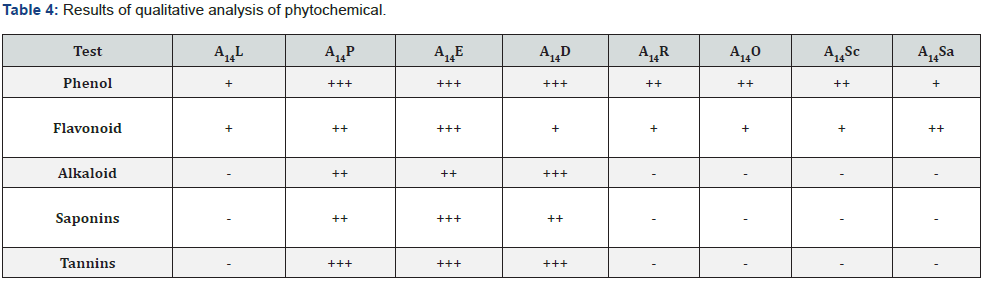
Key: +++= Excellent growth, ++= Better growth, +=Good growth, -= Negative growth
Quantitative analysis of phytochemical
Quantitative analysis of phytochemical performed for the eight isolates. A14E gave the best results for the production of all Phytochemical, like it produces 0.82mg/g phenol in highest amount. On the contrary, A14L and A14Sa gave the least result for tannins, A14R gave the least result of Flavonoid in the concentration of 0.11mg/g, All the isolates gave the good results for the phenol. Among the all isolates A14P, A14E, A14D gave the better result for saponin other gave it in very minute concentration. The graph for the all phytochemicals with their concentration(mg/g) are mentioned, in the Graph 1 Alkaloid shows 102 times higher concentration where, Saponins shows 103 times higher concentration.
Antioxidant activity by DPPH
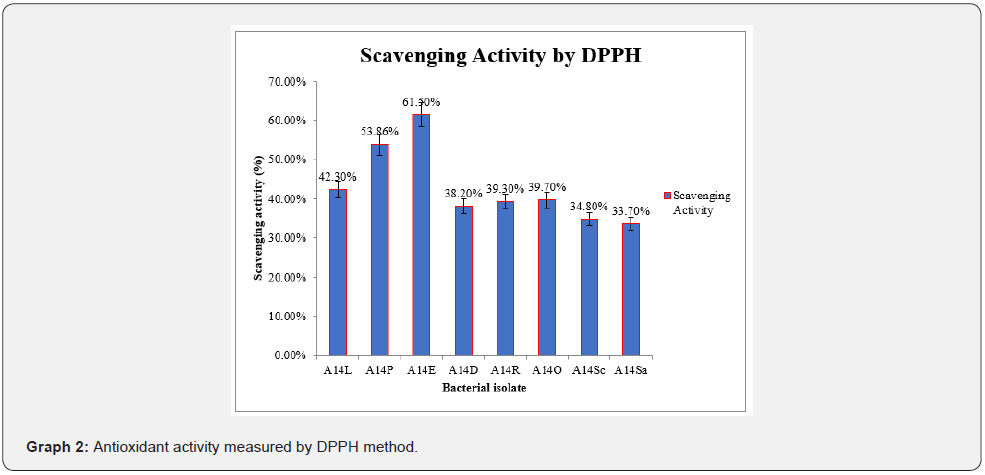
For antioxidant activity, DPPH and FRAP tests were performed. The eight isolates were tested for antioxidant activity by DPPH, and FRAP method. Intensity of colour was measured spectrophotometrically for DPPH at 517 nm. Ascorbic acid used as standard for DPPH (Figures 6) (Graph 2). Antioxidant activity by DPPH for A14E gave the highest scavenging activity 61.50% whereas for A14Sa gave the least scavenging activity 33.70% which can be observed in (Figure 6) (Graph 2).

Antioxidant activity by FRAP
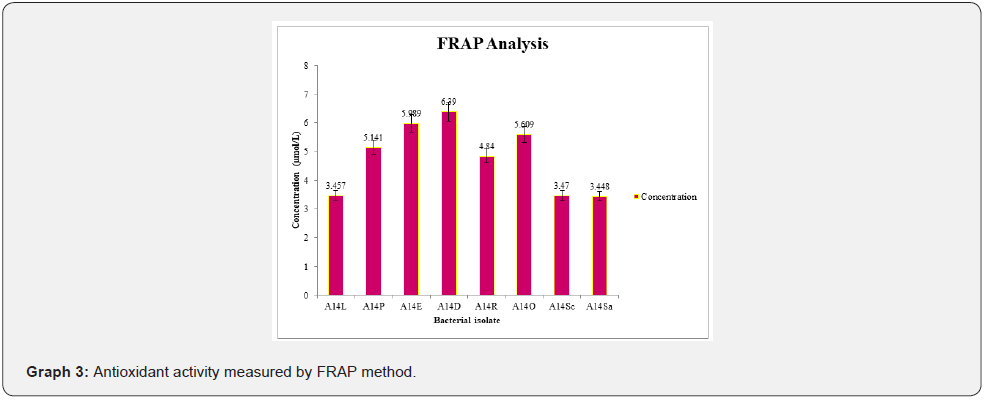
Intensity of color was measured spectrophotometrically for FRAP method at 593 nm. Ferrous sulphate used as standard for FRAP (Figure 7) (Graph 3). Increased absorbance of the reaction mixture indicated increase reducing capacity. Antioxidant activity by FRAP for A14D gave the highest concentration 6.39 μmol/L among the al isolates whereas for A14Sa gave the least concentration 3.448 μmol/L which can be observed in Figure 7 and Graph 3.

Conclusion
Citrus limon isolates from its leaf, root, seed and stem were analysed for phytochemical contents appeared to have potential as a source of beneficial medications as well as to improve the health status of its users due to the presence of numerous phytochemical substances that are essential for good health. Macroscopy, microscopy, physio-chemical parameters, and TLC fingerprinting provide the majority of information on the identity, purity, and quality of the isolates (Rathod Zalak R. et al., 2021). The qualitative analysis gave the positive results for the presence of Phenols and Flavonoid in all the 8 isolates. Also, some of the isolates also gave the results for the Alkaloid, saponin and Tannins. By the quantification study the concentration of phytochemicals present in the isolates is obtained. Phytochemical screening aids in the discovery of plant isolates constituents and the one that predominates over the others, as well as the search for bioactive compounds that can be employed in the development of therapeutic medications. So it can be concluded the further scale up of phytochemicals with endophytes the processes for secondary metabolites may be a promising source of medicines. As a result, isolates from these plants could be considered a promising source of therapeutics. The traditional medicine practice is recommended strongly for these plant as well as it is suggested that further work should be carried out to purify, and characterize the active constituent responsible for the activity of these plants isolates. Also, additional work is encouraged to elucidate the possible mechanism of action of these isolates (RNS Yadav et al., 2011).
Acknowledgement
DST-FIST sponsored Department of Microbiology & Biotechnology, University School of sciences, Gujarat university, Ahmedabad, Gujarat, India.
References
- Ajanal M, Gundkalle MB, Nayak SU (2012) Estimation of total alkaloid in Chitrakadivati by UV-Spectrophotometer. Anc Sci Life 31(4): 198-201.
- Baba SA, Malik SA (2015) Determination of total phenolic and flavonoid content, antimicrobial and antioxidant activity of a root extract of Arisaema jacquemontii J Taibah Univ Sci 9(4): 449-454.
- Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": the FRAP assay. Anal Biochem 239(1): 70-76.
- Benzie IF, Choi SW (2014) Antioxidants in food: content, measurement, significance, action, cautions, caveats, and research needs. Adv Food Nutr Res 71: 1-53.
- Bourhia M, Laasri FE, Moussa SI, Ullah R, Bari A, et al. (2019) Phytochemistry, antioxidant activity, antiproliferative effect, and acute toxicity testing of two Moroccan aristolochia Evidence-Based Complementary and Alternative Medicine.
- Boyer J, Liu RH (2004) Apple phytochemicals and their health benefits. Nutr J 3(1): 5.
- de Souza LB, Gindri AL, de Andrade Fortes T, Kubiça TF, Enderle J, et al. (2020) Phytochemical analysis, antioxidant activity, antimicrobial activity, and cytotoxicity of Chaptalia nutans Adv Pharmacol Pharm Sci 2020: 3260745.
- Chaves N, Santiago A, Alias JC (2020) Quantification of the antioxidant activity of plant extracts: analysis of sensitivity and hierarchization based on the method used. Antioxidants (Basel) 9(1): 76.
- Kavitha Chandran CI, Indira G (2016) Quantitative estimation of total phenolic, flavonoids, tannin and chlorophyll content of leaves of Strobilanthes kunthiana (Neelakurinji). J Med Plants Stud 4(4): 282-286.
- Deshmukh MA, Theng MA (2018) Phytochemical screening, quantitative analysis of primary and secondary metabolites of Acacia arabica bark. Int J Curr Pharm Res 10(2): 35-37.
- Dontha S (2016) A review on antioxidant methods. Asian J Pharm Clin Res 9(2): 14-32.
- Edge R, Truscott TG (2018) Singlet oxygen and free radical reactions of retinoids and carotenoids-A review. Antioxidants (Basel) 7(1): 5.
- Edewor TI, Owa SO, Ologan AO, Akinfemi F (2016) Quantitative determination of the saponin content and GC-MS study of the medicinal plant Cassytha filiformis (linn.) leaves. J Coastal Life Med 4(2): 154-156.
- Ezeonu CS, Ejikeme CM (2016) Qualitative and quantitative determination of phytochemical contents of indigenous Nigerian softwoods. New Journal of Science.
- Foti MC (2007) Antioxidant properties of phenols. J Pharm Pharmacol 59(12): 1673-1685.
- Palma IG, Escalona-Buendia HB, Ponce-Alquicira E, Tellez-Tellez M, Gupta VK, et al. (2016) Evaluation of the antioxidant activity of aqueous and methanol extracts of Pleurotus ostreatus in different growth stages. Front Microbiol 7: 1099.
- Gwatidzo L, Dzomba P, Mangena M (2018) TLC separation and antioxidant activity of flavonoids from Carissa bispinosa, Ficus sycomorus, and Grewia bicolar Nutrire 43(1): 1-7.
- Haile M, Kang WH (2019) Antioxidant activity, total polyphenol, flavonoid and tannin contents of fermented green coffee beans with selected yeasts. Fermentation 5(1): 29.
- Hossain MA, AL-Raqmi KA, Al-Mijizy ZH, Weli AM, Al-Riyami Q (2013) Study of total phenol, flavonoids contents and phytochemical screening of various leaves crude extracts of locally grown Thymus vulgaris. Asian Pac J Trop Biomed 3(9): 705-710.
- Jelena J, Yustiantara PS (2021) Antioxidant activity of fermented coffee beans. Pharm Rep 1(2): 25.
- John BIJU, Sulaiman CT, George S, Reddy VR (2014) Spectrophotometric estimation of total alkaloids in selected Justicia species. Int J Pharm Pharmaceutic Sci 6(5): 647-648.
- Kitima S, Souksanh N, Yupaporn S (2018) Paperbased DPPH assay for antioxidant activity. Anal Sci 34(7): 795-800.
- Sonali M (2021) Classification, mechanism and assay of antioxidant compounds. In: PLANTA Research Book Series. 3: 816-829.
- Monica S (2016) Antioxidant and its applications. J Pharmacol Toxicol 4(4): 28-31.
- Nateghpour B, Kavoosi G, Mirakhorli N (2021) Amino acid profile of the peel of three citrus species and its effect on the combination of amino acids and fatty acids Chlorella vulgaris. J Food Comp Analysis 98: 103808.
- Oleszek W, Kapusta I, Stochmal A (2008) 20TLC of Triterpenes (including saponins) thin layer chromatography in phytochemistry. 1672.
- Puri B, Hall A (1998) Phytochemical dictionary: a handbook of bioactive compounds from plants. CRC press. pp. 976.
- Zalak R, Meenu SS (2021) Spectrophotometric and HPLC estimation of total alkaloids of Citrus limon L. Burm. f. (Lemon) and its endophytes. Acta Scientific Biotechnol 2(3): 21-26.
- Zalak R, Jain R, Sharma S, Rupal KS, Lucie KM, et al. (2020) To determine the minimum inhibitory concentrations and minimum bactericidal concentrations of the effective plants extract and endophytes of Citrus limon. Biosc Biotech Res Comm. Special Issue 13(1): 210-215.
- Zalak R, Sarita S, Meenu SS (2021) Endophytic bacterial effects on seed as enhancement in germination with mobilization of reserves and microbial disinfection in Citrus limon (L) by biopriming under salinity stress. Curr Trends Biomedical Eng & Biosci 20(2): 01-04.
- Zalak R, Meenu SS (2021) Plant as an inexhaustible medicinal source of secondary metabolites, Vidya A J Gujarat Univ 1(1): 64-78.
- Zalak R, Venisha VC, Sonal KM, Meenu SS (2021) Preliminary phytochemical investigation of extraction from Citrus limon. Int J Sci Engg Tech 9:1
- Zalak R, Meet AD, Amita YM, Ritika J, Baldev VP, et al. (2020) Compatibility studies on different endophytes of Citrus limon antagonistic to bacterial with plant pathogen. Adv Biores 11(3): 126-129.
- Zalak R, Meet AD, Amita YM, Baldev VP, Meenu SS (2020) A review on Phytochemical analysis of Secondary metabolites and extraction techniques. Int Engg J Res Develop 5(5): 1-10.
- Zalak R, Meet AD, Amita YM, Baldev VP, Meenu SS (2020) A review article: Citrus as salt tolerance under plant tissue culture. Int J Engg Res Develop 7(8): 34-44.
- Zalak R, Meet AD, Amita YM, Baldev VP, Meenu SS (2020) A review on extraction, tools and techniques of phytochemicals. In: PLANTA Research Book Series 1: 129-140.
- Zalak R, Meet AD, Meenu SS, Baldev VP (2021) Developing Plant Probiotic bacteria based Bioformulation having PGPR properties. In: PLANTA Research Book Series. 2: 324 – 342.
- Zalak R, Meet AD, Amita YM, Baldev VP, Meenu SS (2020) A review on phytochemical screening of secondary metabolites. Vidya A J Gujarat Univ 15(2): 175-190.
- Zalak R, Sonal KM, Venisha VC, Meenu SS (2021) Pharmacognostic and phytochemical evaluation of Citrus limon (Lemon) fruit and leaf. Int J Inn Res Sci Engg Technol 10(2): 1015-1019.
- Santos-Sanchez NF, Salas-Coronado R, Villanueva-Cañongo C, Hernandez-Carlos B (2019) Antioxidant compounds and their antioxidant mechanism. In: Shalaby E (Ed.), Antioxidants, Intech Open Ltd, London, UK. pp. 1-28.
- Senguttuvan J, Paulsamy S, Karthika K (2014) Phytochemical analysis and evaluation of leaf and root parts of the medicinal herb, Hypochaeris radicata for in vitro antioxidant activities. Asian Pac J Trop Biomed 4(Suppl 1): 359-367.
- Sogandi S, Triandriani W, Saputri D, Suhendar U (2020) Antioxidant activity of endophytic bacterial extract isolated from clove leaf (Syzygium aromaticum L.). J Agri Applied Biol 1(1): 9-17.
- Sulistiyani S, Ardyati T, Winarsih S (2017) Antimicrobial and antioxidant activity of endophyte bacteria associated with Curcuma longa J Exp Life Sci 6(1): 45-51.
- Supritha P, Radha KV (2018) Estimation of phenolic compounds present in the plant extracts using high pressure liquid chromatography, antioxidant properties and its antibacterial activity. Ind J Pharm Edu Res 52(2): 321-326.
- Urquiaga IN, Leighton F (2000) Plant polyphenol antioxidants and oxidative stress. Biol Res 33(2): 55-64.
- Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, et al. (2007) Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol 39(1): 44-84.
- Van Tan P (2018) The determination of total alkaloid, polyphenol, flavonoid and saponin contents of Pogang gan (Curcuma sp.). Int J Biol 10(4): 42-47.
- Xiao F, Xu T, Lu B, Liu R (2020) Guidelines for antioxidant assays for food components. Food Frontiers 1(1): 60-69.
- Yadav RN, Agarwala M (2011) Phytochemical analysis of some medicinal plants. J Phytology 3(12).
- Yahia EM, Maldonado Celis ME, Svendsen M (2017) The contribution of fruit and vegetable consumption to human health. In: Yahia EM (Ed.), Fruit and vegetable phytochemicals (2nd Edn). Hoboken: John Wiley & Sons 3-52.
- Zalak R, Saraf M, Patel B (2018) Developing plant probiotic bacteria from rohu fish on bioformulation by PGPR activity. National conference on synthetic biology approaches in drug discovery and microbial biotechnology, Department of Microbiology and Biotechnology, Gujarat University, Ahmedabad, Vidya.






























