Isolation, Identification and Characterization of Lactic Acid Bacteria Found in “Juju Dhau” from Bhaktapur, Nepal
Dikshya Shilpakar1*, Pawan Khanal1,2 and Rakesh Kusma1
1Pokhara Bigyan Tatha Prabidhi Campus, Tribhuvan University, Nepal
2Sujal Dairy P. LTD., Pokhara, Nepal
Submission:April 10, 2022;Published:April 25, 2022
*Corresponding author:Dikshya Shilpakar, Pokhara Bigyan Tatha Prabidhi Campus, Tribhuvan University, Nepal
How to cite this article: Dikshya S, Pawan K, Rakesh K. Isolation, Identification and Characterization of Lactic Acid Bacteria Found in “Juju Dhau” from Bhaktapur, Nepal. Curr Trends Biomedical Eng & Biosci. 2022; 20(4): 556044. DOI:10.19080/CTBEB.2022.20.556044
Abstract
The present study was undertaken to isolate, identify and characterize LAB found in juju dhau from Bhaktapur, Nepal. A total of 15 samples were collected, pooled together and finally examined microbiologically. Based on morphological, cultural, biochemical and physiological characteristics, three types (A, B and C) of Lactobacilli, two (A and B) Leuconostoc, two (A and B) Bifidobacterium, one Streptococcus thermophilus and three (A, B and C) Lactococci were isolated and identified. Moreover, it was found that all the isolated LAB were gram positive, catalase-oxidase negative, non-motile bacteria, growing optimally at 30 ᵒC (except Streptococcus thermophilus), pH 7 and 0% NaCl. All Lactobacilli grew well at 45 ᵒC and pH 4 but only type C was able to grow at 10 ᵒC and pH 2. All isolates of Leuconostoc and Lactococci were found to be mesophilic, growing well at 30 ᵒC and 10 ᵒC but not at 45ᵒC. Also, Leuconostocs grew well at pH 4 but not at pH 2 while only type C Lactococci was able to resist high acidic pH 2. Similarly, both types of Bifidobacterium grew well at 45 ᵒC and pH 2 but only type A was able to grow at 10 ᵒC. Among 11 organisms, only type C of Lactococci did not grew at 6.5% NaCl. This signifies that juju dhau comprises variety of LAB. This study can serve to document and thereby preserve as well as commercialize traditional fermented foods of Nepal.
Keywords: Juju dhau; Isolation; Identification; Characterization; Bhaktapur; Nepal; LAB
Abbreviations: LAB: Lactic Acid Bacteria; MRS: de Man, Rogosa and Sharpe; ATR: Acid Tolerance Response; HCL: Hydrochloric Acid; OD: Optical Density
Introduction
Yoghurt is one of the most popular fermented dairy products, which provides digested lactose, and viable bacterial strains of Streptococcus thermophilus and Lactobacillus bulgaricus [1]. It is prepared indigenously in different regions of world. Out of them, dahi or curd is one of the famous fermented products from South Asia.
Traditionally, it is prepared from boiled milk, fermented by natural microflora [2]. In context of Nepal itself, varieties of dahi is prepared. However, juju dhau is the most loved dahi, which is traditionally made in Bhaktapur and Kathmandu Valley of Nepal. Because of its unique texture, taste and flavor than other types of dahi, it is mostly preferred [3].
Literally, juju dhau means “king of yoghurt” in Newari language. It has been considered as a delicacy since the Malla era. Traditionally, juju dhau is prepared in earthen clay pots or kataaros by using juju dhau from previous batch as inoculum followed by insulation of these pots with rice husks in a non-ventilated room [4]. It is basically a sweetened custard-like yoghurt and an inevitable component in all feasts and celebration. It is prepared by using buffalo milk, resulting in richer taste and texture [5]. These pots have porous surface which absorbs the moisture from product especially when dahi tends to expel some whey. This provides a “muddy” flavor and thicker as well as creamy texture [6].
Microbiologically, yoghurt is a symbiotic culture of Streptococcus thermophilus and Lactobacillus delbrueckii subsp. bulgaricus [7]. In a study conducted in indigenous dahi from Nepal Himalayas, Lactobacillus fermentum, Lactobacillus delbrueckii subsp. bulgaricus, Streptococcus thermophilus, Lactococcus lactis subspecies cremoris, Lactococcus lactis subsp. lactis biovar diacetylactis and Leuconostoc mesenteroids subsp. mesenteroids. S. thermophilus were found as the dominant lactic acid bacteria [2]. Proper microbial characterization of fermented products leads to better understanding and management of food fermentation process. It also aids in improving the quality of fermented food product. There are several methods for isolation and identification of the microorganisms involved in food fermentation [8]. Moreover, scientific study of indigenous fermented food helps to authenticate its origin, preserve its culture and also improve as well as standardize its technology. This also assists in commercializing the product on global range [9].
Proper scientific documentation of indigenous fermented foods and the technology used in their preparation is lacking. This is important not only to preserve them but also to enhance their production and consumption on a larger scale. In relation to Nepal, many indigenous products are yet to be investigated due to which they are on the verge of losing their originality and identity. Many reports and articles have been published on the microbial characterization and identification of various Nepali fermented foods. However, the documentation of one of the most popular dairy products of Nepal; juju dhau is still overshadowed. On that account, this study is aimed at scientific documentation ofjuju dhau; isolation, identification and characterization of lactic acid bacteria found in “juju dhau” from Bhaktapur, Nepal.
Materials and Methods
Sample Collection
A total of 15 juju dhau samples were bought during the morning from various places of Bhaktapur, Nepal and pooled together by assembling 10 g of each samples to entirely yield 150 g of final sample. These samples were stored in a sterile plastic container and stored under ice pack system and transported to the laboratory of Pokhara, Nepal via airplane within 5 h of sample collection where it was immediately refrigerated for further use.
Microbiological Media and Chemical Reagent Collection
Lactobacilli was isolated on De Man, Rogosa and Sharpe (MRS) agar media which was purchased from Hi Media, Mumbai, India and imported to Raxual, Nepal by Flipkart, India and finally transported to Pokhara via truck. In order to improve specificity of Lactobacilli, 0.25% of L-cysteine was supplemented to isolate Bifidobacterium which was also purchased from HiMedia, Mumbai, India. Similarly, Vancomycin (20mg/L) was added to specifically isolate Leuconostoc only, which was bought from local pharmacy in Pokhara. Similarly, M17 agar was also procured from Hi Media, Mumbai, India and imported to Raxual, Nepal by Flipkart, India and finally transported to Pokhara via truck for isolating Streptococcus thermophilus. Finally, in order to isolate Lactocooci, differentiating agar media was composed in the laboratory itself wherein the chemicals and reagents required were obtained from Science House P. Ltd., Pokhara, Nepal and Kiran Scientific House, Pokhara, Nepal.
Isolation of Microbes
Initially, juju dhau samples (25 g) were taken from the pooled sample which was then homogenized (using an
electric stationary blender consisting of blender jar with rotating metal blades) with 90 mL of 0.1 % (w/v) sterile peptone water to make the initial suspension and serial decimal dilution techniques were performed using either sterile distilled water for bacterial isolation. Finally, aliquot (0.1 mL) from various dilutions was spread plated on various agar plates to isolate the microbe. Firstly, lactobacilli was isolated on MRS agar plate after complete incubation in anaerobic chamber (using gas pack chamber) at 37 ᵒC for 24-48 h. Leuconostoc was isolated on MRS-vancomycin (Vancomycin 20 mg/L) after incubating for 24 h at 30 ᵒC [2]. Similarly, after anaerobically incubating the MRS agar containing 0.25% L-cysteine at 37 ᵒC for 48 h Bifidobacterium was isolated [10, 11] In order to isolate Streptococcus thermophilus, aliquot of 0.1mL was spread plated on M17 agar plate which was then incubated at 42 ᵒC for 48 h. In regards to lactococci, Streptocoocus lactis differential agar plate (SL) was incubated anaerobically for 48 h to differentiate citrate utilizing and non-utilizing Lactococci. While Differential agar medium (D) plate was used for differentiating Streptococcus lactis and Streptococcus cremoris based on their ability to hydrolase arginine, which was incubated at 32 ᵒC for 48 h [12].
Colonies were randomly selected from the agar plates but if the plate contained less than 4 colonies, all the colonies were isolated. The isolated colonies were streaked and sub-cultured on the fresh agar plates followed by microbial examination to obtain pure culture.
Identification of isolated colonies
All the preserved isolated colonies were initially characterized on the basis of their morphological characteristics. Afterwards, biochemical tests were performed to identify them. Gram staining, catalase-oxidase test and motility test were performed as primary basis of biochemical characterization. Moreover, heat resistance at 60 ᵒC for 30 mins (Sherman test) was also performed [2]. Arginine hydrolysis test was performed by inoculating loopful of bacterial culture in arginine broth with few drops of Nesseler reagent. A brown coloration of the medium indicated positive result [13]. With regards to citrate utilization test, Simmon’s citrate agar slant was used. A small amount of bacterial colony was pricked on the center and streaked on the slant which was then incubated aerobically at 35-27 ᵒC for 4 to 5 days. Finally, blue coloration of the slant was designated as positive response [14]. Moreover, color of the bacterial colonies observed on SL and D agars was employed to interpret arginine hydrolysis and citrate utilization as well as distinguish bacteria [12,15].
Sugar fermentation test
In order to study the sugar fermentation characteristics of isolated microbes, membrane (0.45 μm) filtered 1% (w/v) solutions of different sugars (glucose, fructose, lactose, galactose, sucrose, maltose, and mannitol) were prepared. Nutrient broth (0.8%) with 1mL of phenol red indicator was prepared and transferred equally in various test tubes with Durham tube. The test tubes were then autoclaved at 121 ᵒC for 30 mins to sterilize them properly. Subsequently, 100 μL of different sugar solution was mixed to the sterilized broth. Finally, freshly cultured colonies were inoculated into the broth enriched with the mentioned sugar solutions and incubated at 37 ᵒC for 48 h. A color change of broth from red to yellow in the test tubes indicated positive result for sugar fermentation while gas production was noted in the Durham tube [16].
Characterization of Identified Microbes
Physiological tests were used to characterize the identified bacteria. Growth at different temperature (10 ᵒC, 30 ᵒC and 45 ᵒC) for 5 days, growth at different pH (2, 4 and 7) and growth in the presence of 4% and 6.5% (w/v) NaCl was used to determine whether the isolated microbe can resist high temperature, acidic and halophilic conditions respectively and to detect their optimal growth condition [17]. Growth was evaluated by sub culturing 1 mL of actively growing bacterial culture to10 mL of the respective broth for testing growth at different temperatures or to the broth whose pH was adjusted to 2 and 4 by using 1 M Hydrochloric acid (HCL) or to the broth supplemented with 4.0% and 6.50% (w/v) NaCl followed by incubation as required. Growth index was interpreted by comparing the change in optical density (OD) of the test sample by using spectrophotometer (Sky Technology, India) at 620nm with that of control sample. Control sample was prepared by using the ideal media composition and was subjected to optimum conditions (30 0C temperature, pH 7 and 0% NaCl) [18,19].

Results and Discussion
Isolation and Identification of Lactobacilli
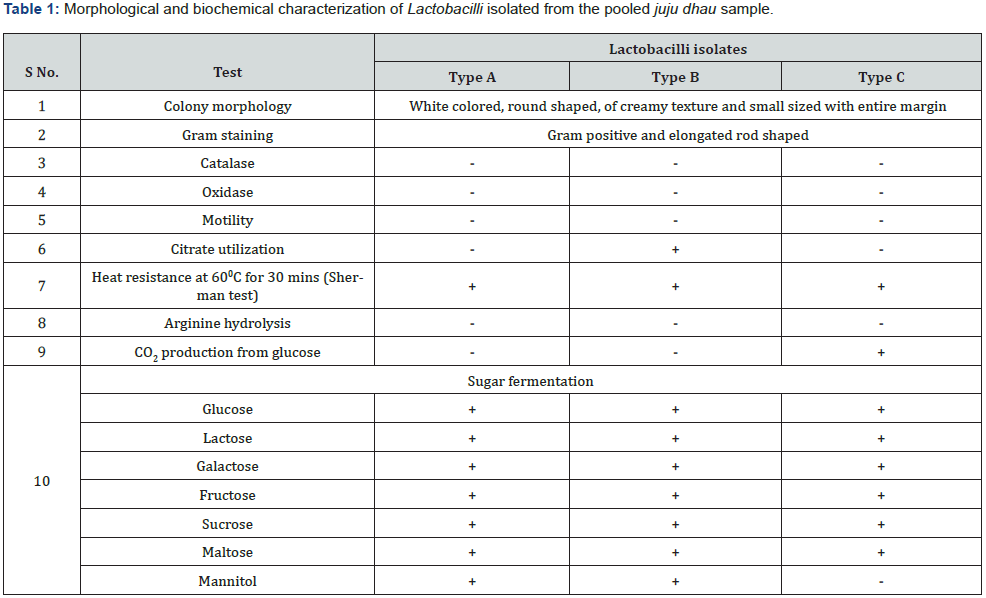
Out of numerous Lactobacilli obtained from the pooled juju dhau sample, only three types were isolated. They were designated as type A, type B and type C. The isolated Lactobacilli were further subjected to various morphological and biochemical tests for identification purpose (Table 1). Morphological observation of Lactobacilli on MRS plate is shown in Figure 1 while their microscopic observation is shown in Figure 2. The presence of different species of Lactobacilli in the samples may be attributed to the diversity of microbial community in the specific environment as well as the manufacturing processes for indigenous dahi [2]. Moreover, varieties of Lactobacilli are found in dahi produced at different altitudes. Greater varieties is found in dahi from Lalitpur and Bhaktapur district than from Gorkha district [6].

Characterization of Lactobacilli
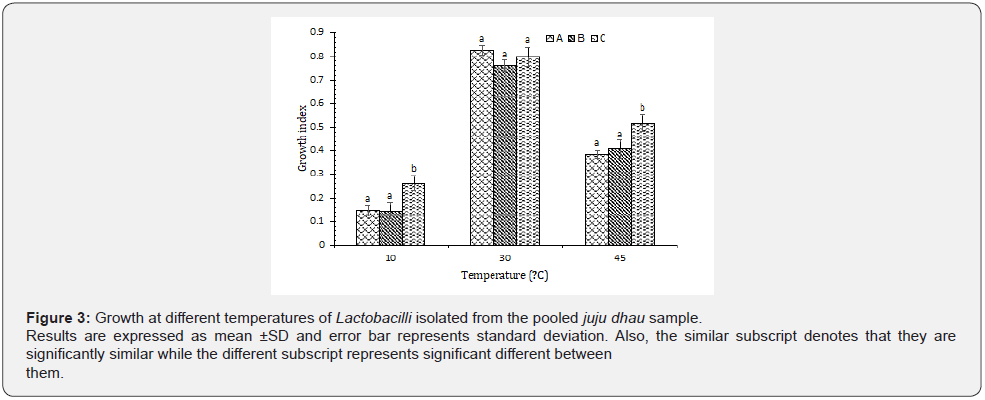
Growth at different temperature: Based on the results observed, it was found that all the isolated Lactobacilli showed best growth at 30 ᵒC. However, they were also able to grow at 45 ᵒC but not at 10 ᵒC (Figure 3). Lactobacilli are mostly found to be resistant to higher temperature (45 ᵒC) but could not grow well at lower temperature (10 ᵒC) [7]. Furthermore, their optimum growth temperature was recorded as 37-45 ᵒC during characterization of LAB from locally and commercially produced milk and yoghurts of Bangladesh, which also suggested that Lactobacilli strain cannot tolerate low temperatures (4-10 ᵒC) [20].
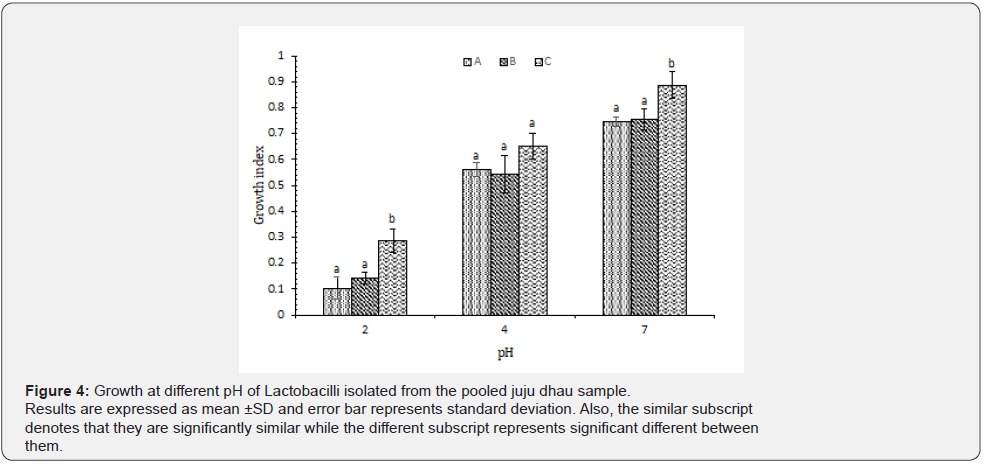
Growth at different pH: According to our results, it was found that all the isolated Lactobacilli showed best growth at pH 7. However, they were able to grow at pH 4 as well but only type C was found to be resistant to acidic pH 2 (Figure 4). In a study carried out in commercial probiotic culture, it was found that isolated Lactobacilli did not survive at the pH 2 for an incubation period of 2 hours while it survived well at pH 3 [18]. However, in a previous study, it was presumed that Lactobacilli can survive the extreme acidic condition of pH 2.5 also [17]. Although Lactobacilli are acidophilic, the high concentration of free acids (H+) may cause growth inhibition [18].
Growth at different NaCl concentration: In proportion to our results, it was found that all the isolated Lactobacilli were able to tolerate high osmotic concentration of NaCl. However, they demonstrated maximum growth at 0% NaCl (Figure 5). Similar results were recorded in a previous research which implied that Lactobacilli were able to survive extreme NaCl concentration (1-9% w/v) [17]. Furthermore, in a study carried out in Bangladesh, almost all the isolated species of Lactobacilli were able to survive well in the presence of NaCl concentration of up to 6% (w/v). However, only few of them were able to tolerate up to 7% (w/v) of NaCl concentration [20].
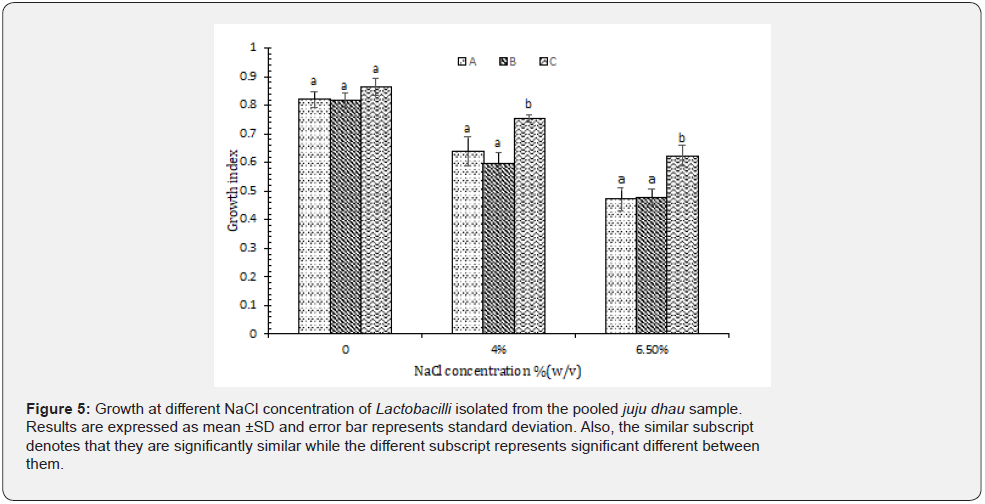
Isolation and identification of Leuconostoc
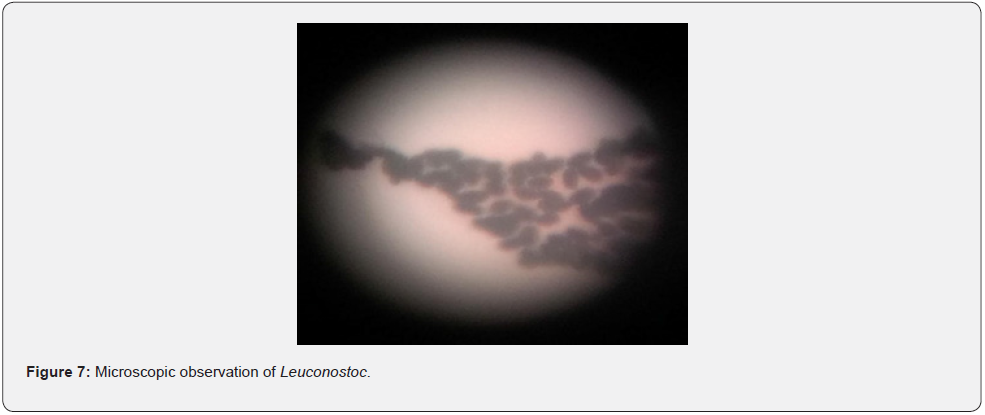
Two types of Leuconostoc were isolated from the pooled juju dhausample. These types were designated as type A and type B. The isolated Leuconostoc were further subjected to various morphological and biochemical tests for identification purpose (Table 2). Morphological observation of Leuconostoc on MRS-vancomycin plate is shown in Figure 6 and its microscopic observation is shown in Figure 7. Earlier similar observations were made in Kenya where Leuconostoc was identified as one of the dominant lactic acid bacteria in the product [21]. They were also isolated from indigenous dahi of Nepal Himalayas. It also suggested their lower number in the indigenous dahi may be due to their complex nutritional requirements and lower adaption capacity in milk [2]. Likewise, about 13 species of Leuconostoc were isolated in Bangladesh with similar properties as mentioned in our findings [22]. This signifies that Leuconostoc sp. is generally present in indigenous fermented milk product of all regions.

Characterization of Leuconostoc
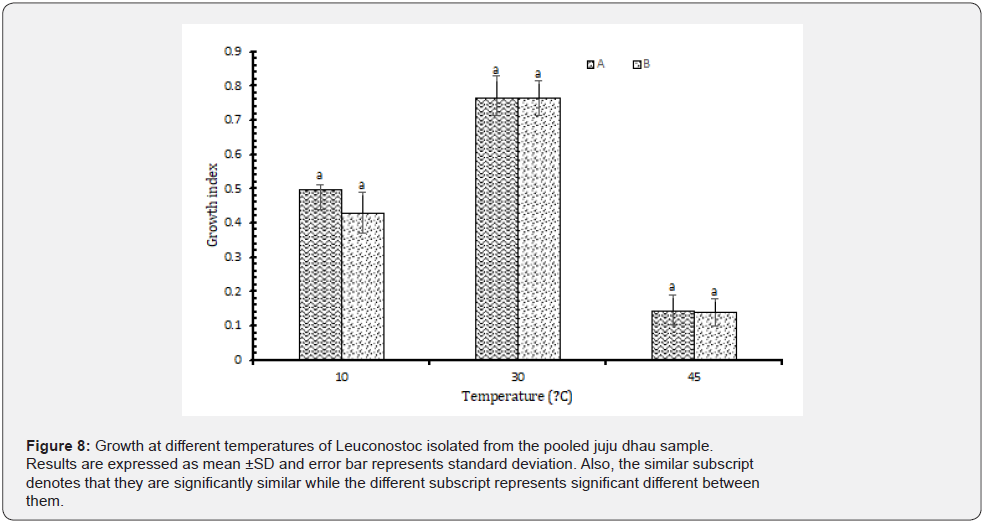
Growth at different temperatures: From the observations, it was found that all the isolated Leuconostoc showed best growth at 30 ᵒC. However, they were able to grow at 10 ᵒC as well but not at 45 ᵒC (Figure 8). Hence, they were not found to be resistant to higher temperature. In a study carried out in Bangladesh Leuconostoc isolated from dahi were able to grow well at 10 ᵒC but not at 45 ᵒC [22]. However, some strains of Leuconostoc are able to survive even at higher temperature. They demonstrated thermoduric characteristics [23]. Moreover, the heat tolerance of Leuconostoc can be improved by prior exposure to temperatures between 30-45ᵒC which led to improved viability (63-55%) upon heat shock at 60 ᵒC for 3 minutes [24].
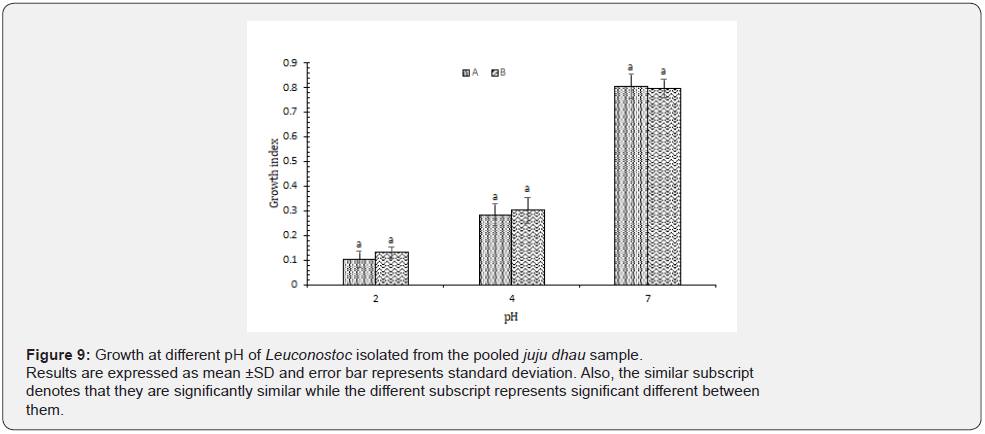
Growth at different pH: As stated by our observations, it was found that all the isolated Leuconostoc showed best growth at pH 7. However, they were able to grow at pH 4 as well but not at pH 2 (Figure 9). Leuconostoc isolated from dahi were found to be very sensitive to acidic pH [2]. Moreover, the low resistance to acidic conditions of this organism tested may be related to the low acidification ability of this genus [23].
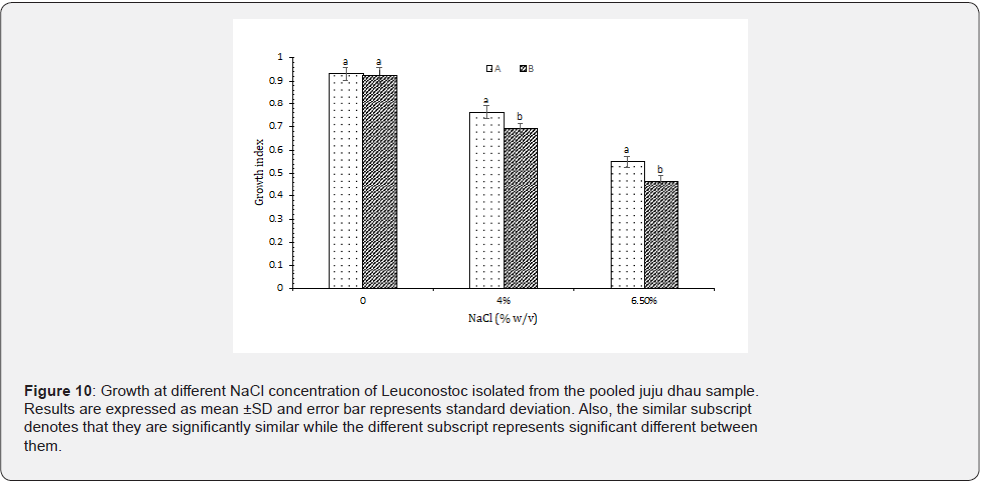
Growth at different NaCl concentration: Based on the results observed, it was found that all the isolated Leuconostoc were halophilic. However, all the isolates demonstrated maximum growth at 0% NaCl as well as reduced growth at higher NaCl concentration (Figure 10). In a study carried out in Brazilian buffalo mozzarella cheese and kimchi, Leuconostoc were found to be tolerant to high NaCl concentration. However, results suggested that the growth of the strains isolated from mozzarella cheese showed decreased growth at higher NaCl concentration than from Kimchi. This may be due to the original source of isolated organism being salty in nature [25,26].
Isolation and identification of Bifidobacterium
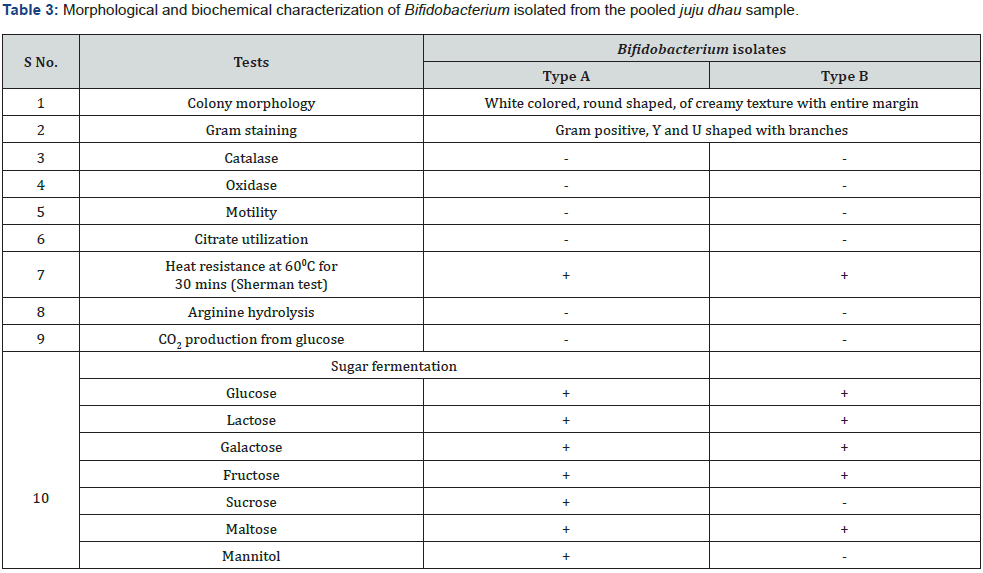
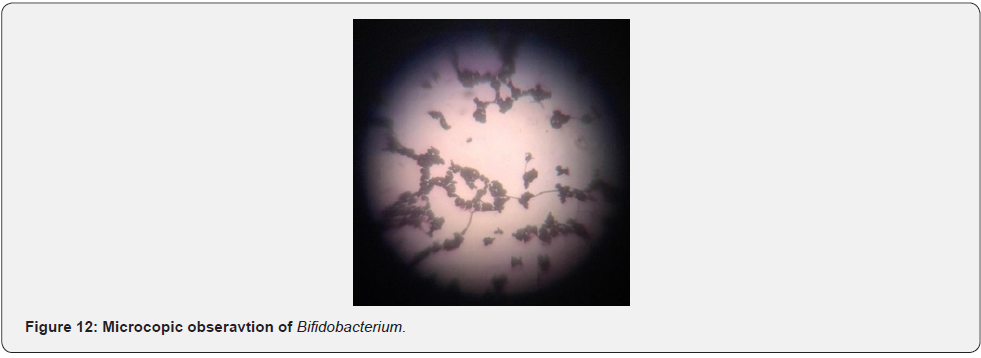
Two types of Bifidobacterium were isolated from the pooled juju dhau sample which were designated as type A and type B. The isolated Bifidobacterium were further subjected to various morphological and biochemical tests for identification purpose (Table 3). Morphological observation of Bifidobacterium on MRS-cysteine plate is shown in Figure 11 and its microscopic observation is shown in Figure 12.
Similar characteristics of Bifidobaterium were reported in study carried out in human colostrum and various fermented milk products [27, 10]. Moreover, similar carbohydrate metabolism of isolated strains was also recorded [11].

Characterization of Bifidobacterium
Growth at different temperatures: From the results, it was found that all the isolated Bifidobacterium were able to grow at various temperatures. However, only strain B was not able to grow at 10 ᵒC. Both the types showed best growth at 30 ᵒC (Figure 13). The strains of Bifidobacterium isolated from human colostrum were able to grow at 10 to 42 ᵒC [27]. Moreover, the optimum growth temperature for human bifidobacterial strains is about 36-38 ᵒC and that of animal origin is slightly higher. Also, only few species of Bifidobacterium are able to grow at low temperature. Report also suggested that Bifidobacterium can invoke advanced molecular mechanism to cope up with heat stress [28].
Growth at different pH: Based on our results, it was found that all the isolated Bifidobacterium showed best growth at pH 7. However, they were also able to grow at both pH 4 and 2 as well (Figure 14). This signifies that the isolated Bifidobacterium were resistant to acidic pH. The improved viability of Bifidobacterium in adverse acidic condition may be explained due to the provision of fermentable substrate in the medium thereby improving the organism ability to resist the detrimental effects of acidic pH [29]. Bifidobacterium have been shown to possess a wide molecular machinery allowing the degradation of a wide variety of nondigestible sugars which in turn supports the survival of this bacterial group in presence of different stress condition [30]. Moreover, it also suggested that acid tolerance response (ATR) enhances the bacterial survivability to more pH stress [11].
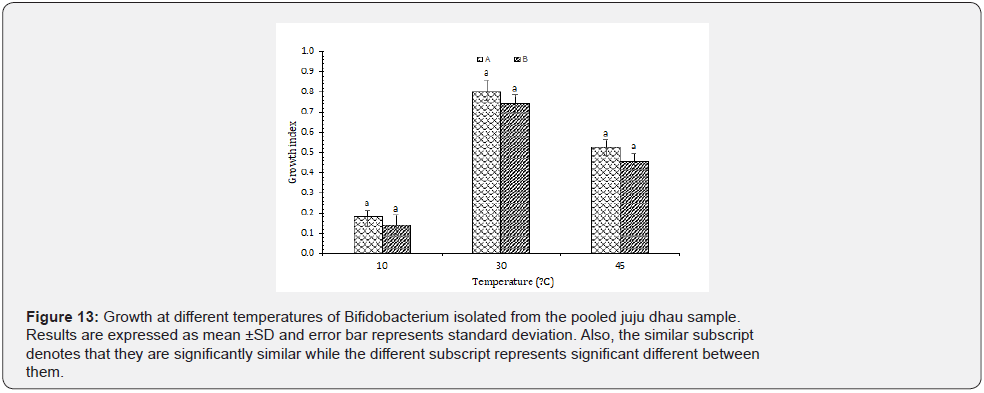
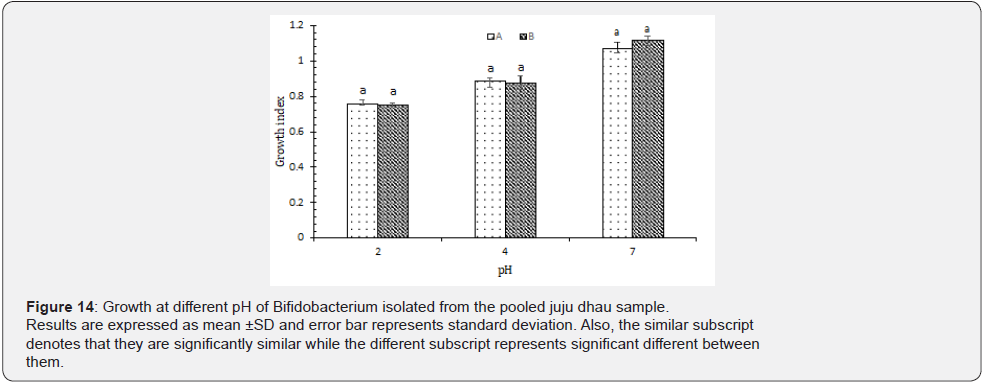
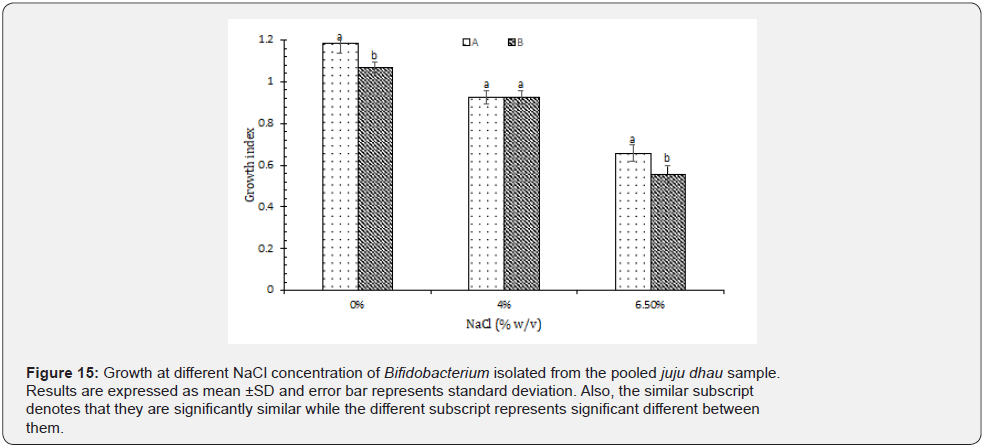
Isolation, identification and characterization of Streptococcus thermophilus
Only one type of Streptococcus thermophilus was isolated from the pooled juju dhau sample. The isolate was further subjected to various morphological, biochemical and physiological tests for identification purpose. From gram staining and microscopic observation, they were gram positive, cocci in pairs or forming chains (Figure 16). It showed negative result for catalase-oxidase, motility and citrate utilization test. It neither hydrolyzed arginine nor produce CO2 gas from glucose. In terms of sugar fermentation,it showed positive result for glucose, lactose, fructose and sucrose but negative result for the rest. Furthermore, the isolated Streptococcus thermophilus was not able to grow at 10 ᵒC, but it grew well at 30 ᵒC and demonstrated maximum growth at 45 ᵒC. Also, it was found to be resistant to heat at 60 ᵒC for 30 minutes (Sherman test). It was not able to grow at 4% and 6.5% NaCl. Finally, the isolated strain demonstrated maximum growth at pH 7 followed by at pH 4 and at pH 2. Similar characteristics of Streptococcus thermophilus was recorded in earlier studies too [2, 7, 31].
Isolation and identification of Lactococci
Three types of Lactococci were isolated from the pooled juju dhau sample. These types were designated as type A, type B and type C. The isolated Lactococci were further subjected to various morphological and biochemical tests for identification purpose (Table 4). Microscopic observation of Lactoccoci is shown in Figure 17. Various strains of Lactococci of similar properties were also isolated from various milk products [2,32]. Moreover, lactococci are also used for the preparation of yoghurt where multi-strains starter culture is used to produce the desired acidity [33].
Characterization of Lactococci
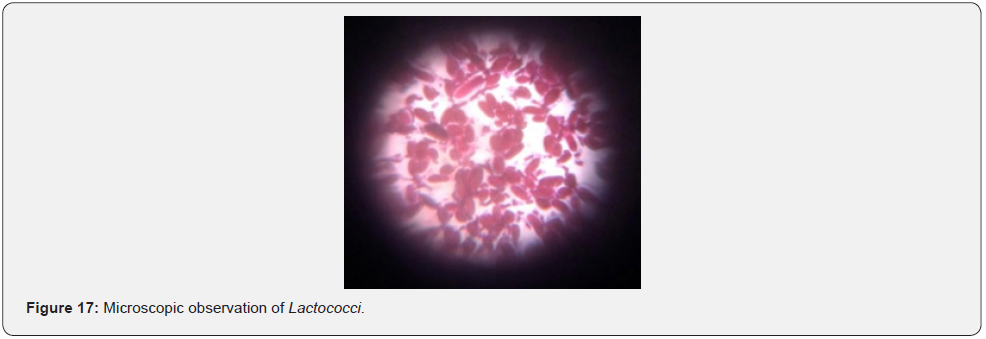
Growth at different temperatures: On the basis of our results, it was found that all the isolated Lactococci showed best growth at 30 ᵒC. However, they were able to grow at 10 ᵒC as well but not at 45 ᵒC (Figure 18). In the study carried out in indigenous dahi samples and traditionally produced cheese, the isolated Lactococci strains were able to grow at 10 ᵒC but not at 45 ᵒC. Thus, Lactococci are characterized as mesophilic bacteria [33,34].
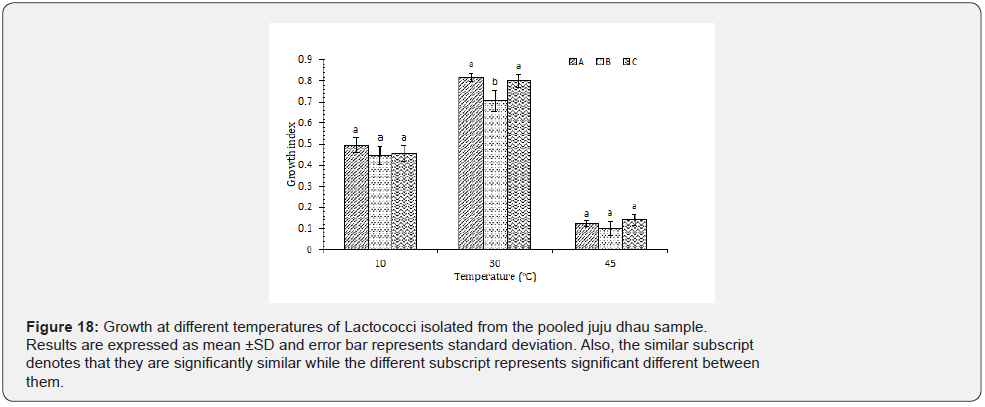
Growth at different pH: Based on the results observed, it was found that all the isolated latcococci showed best growth at pH 7. However, they were also able to grow well at pH 4 but only type A was able to resist high acidic pH 2 (Figure 19). Lactococci are quite sensitive to low pH [35]. Furthermore, growth rate of Lactococci decreases at lower pH due to decreased utilization of glucose as well as accumulation of organic acid in the cell resulting in the reduction of internal pH [36].
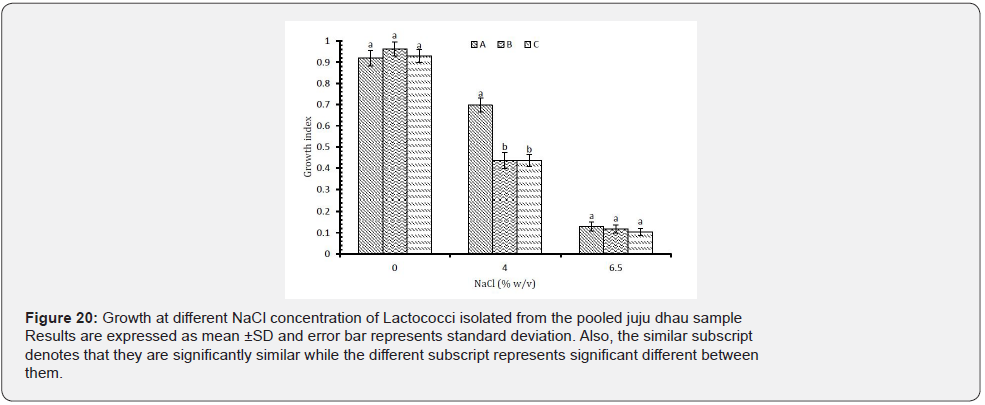
Growth at different NaCl concentration: On the basis of results, it was found that all the isolated Lactococci showed optimum growth at 0% (w/v) NaCl. However, they were able to grow at 4% (w/v) NaCl but not at 6.50% (w/v) NaCl (Figure 20). In previous researches also, Lactococci strains were not able to survive adverse stress of NaCl [33-35].
Conclusion
Varieties of LAB were isolated and identified from juju dhau of Bhaktapur, Nepal. Based on morphological, biochemical and physiological characterization, three types of Lactobacilli, two types of Leuconostoc, two types of Bifidobacterium, one type of streptococci and three types of Lactococci were identified. The results from this study can serve as a useful resource for the development of starter culture for juju dhau thereby preserving as well as commercializing them.
Acknowledgements
We would like to thank Pokhara Bigyan Tatha Prabidhi Campus, Nepal for providing us laboratory, required equipment and chemicals to conduct our research smoothly. Also, we would like to show our immense gratitude to Mr. Safal Nakarmi for helping us with sample collection.
References
- Fisberg M, Machado R (2015) History of yogurt and current patterns of consumption. Nutr Rev 73(Suppl 1): 4-7.
- Bhattarai RR, Gautam N, Nawaz MA, Lal Das SK (2016) Isolation and identification of dominant lactic acid bacteria from dahi: an indigenous dairy product of Nepal Himalayas. J Microbiol Biotech Food Sci 5(4): 358-363.
- Dahal NR, Karki TB, Swamylingappa B, Li Q, Gu G (2005) Traditional foods and beverages of Nepal-A Review. Food Rev Int 21(1): 1-25.
- Malla Y (2010) Juju Dhau: King Yoghurt.
- Pathak J (2012) Juju Dhau (The king Yoghurt from Bhaktapur Nepal).
- Koirala R, Ricci G, Taverniti V, Ferrario C, Malla R, et al (2014) Isolation and molecular characterization of lactobacilli from traditional fermented Dahi produced at different altitudes in Nepal. Dairy Sci Technol 94(4): 397-408.
- Erkus O (2007) Isolation phenotypic and genotypic characterization of yoghurt starter bacteria. Izmir Institute of Technology, Graduate School of Natural and Applied Sciences. p. 104.
- Tamang JP, Thapa N, Tamang B, Rai A, Chettri R (2015) Microorganisms in fermented foods and beverages. In: Tamang JP (Ed.), Health benefits of fermented foods and beverages (1st edn), CRC Press, USA. pp. 1-110.
- Bhattarai RR, Das SK (2013) Scientific study on indigenous technology of Dahi making of Eastern Nepal. J Food Process Tech 4(8): 253-300.
- Mishra M, Paliwal JS, Singh SK, Abharam J (2012) Isolation and characterization of Bifidobacterium from fermented milk products and their antimicrobial studies.
- Zinedine A, Faid M (2007) Isolation and characterization of strains of Bifidobacteria with probiotic proprieties In vitro. World J Dairy Food Sci 2(1): 28-34.
- Reddy MS, Vedamuthu ER, Washam CJ, Reinbold GW (1969) Differential agar medium for separating Streptococcus lactis and Streptococcus cremoris. Appl Microbiol 18(5): 755-759.
- Mehmood T, Masud T, Abbass SA, Maqsud S (2009) Isolation and identification of wild strains of lactic acid bacteria for yoghurt preparation from indigenous Dahi. Pak J Nutri 8: 866-871.
- Aryal S (2019) Citrate utilization test- principle media procedure and result.
- Kempler GM, McKay LL (1980) Improved medium for detection of citrate-fermenting Stretococcus lactic diacetylactis. Appl Environ Microbiol 39(4): 926-927.
- Mahato S, Shahani AK (2019) Identifying the diversity of dominant LABs from fermented dairy products Dahi and Yoghurt in Eastern Region of Nepal. J Food Sci Technol Nepal 11: 60-64.
- Khanal A, Koirala N (2019) Screening and analysis of probiotic properties of Lactobacillus Spp from famous commercial brand of yogurt found in Kathmandu Valley. Curr Trends Biomedical Eng Biosci 18(4): 75-82.
- Menconi A, Kallapura G, Latorre JD, Morgan MJ, Pumford NR, et al. (2014) Identification and characterization of lactic acid bacteria in a commercial probiotic culture. Biosci Microbiota Food Health 33(1): 25-30.
- Sharma A, Lavania M, Singh R, Lal B (2021) Identification and probiotic potential of lactic acid bacteria from camel milk. Saudi J Bio Sci 28(3): 1622-1632.
- Khalil I, Anwar N (2016) Isolation identification and characterization of lactic acid bacteria from milk and yoghurts. J Food Dairy Technol 4(3): 17-26.
- Mathara JM, Schillinger U, Kutima PM, Mbugua SK, Holzapfel WH (2004) Isolation identification and characterisation of the dominant microorganisms of kule naoto: the Maasai traditional fermented milk in Kenya. Int J Food Microbiol 94(3): 269-278.
- ur-Rashid H, Togo K, Ueda M, Miyamoto T (2006) Identification and characterization of dominant lactic acid bacteria isolated from traditional fermented milk Dahi in Bangladesh. World J Microbiol Biotechnol 23(1): 125-133.
- Cardamone L, Quiberoni A, Mercanti DJ, Fornasari ME, Reinheimer JA, et al. (2011) Adventitious dairy Leuconostoc strains with interesting technological and biological properties useful for adjunct starters. Dairy Sci Technol 91(4): 457-470.
- Rodriguez IC (2017) Heat acid and osmotic stress tolerance of Leuconostoc mesenteroides as influenced by prior exposure to various mild stress conditions. Master of Science in the School of Animal Science Louisiana State University.
- de Paula AT, Jeronymo-Ceneviva AB, Silva LF, Todorov SD, Franco BD, et al. (2014) Leuconostoc mesenteroides SJRP55: a potential probiotic strain isolated from Brazilian water buffalo mozzarella cheese. Annals Microbiol 65(2): 899-910.
- Kwon O, Son M (2016) NaCl Effect on dextran producing Leuconostoc mesenteroides CS-5 that isolated from chonggak kimchi. Quant Biosci 35(2): 53-60.
- Liu W, Chen M, Duo L, Wang J, Guo S, et al. (2020) Characterization of potentially probiotic lactic acid bacteria and Bifidobacteria isolated from human colostrum. J Dairy Sci 103(5): 4013-4025.
- Cronin M, Ventura M, Fitzgerald GF, van Sinderen D (2011) Progress in genomics metabolism and biotechnology of Bifidobacteria. Intl J Food Microbiol 149(1): 4-18.
- Waddington L, Cyr T, Hefford M, Hansen LT, Kalmokoff M (2010) Understanding the acid tolerance response of Bifidobacteria. J App Microbiol 108(4): 1408-1420.
- Ruiz L, Ruas-Madiedo P, Gueimonde M, de Los Reyes-Gavilan CG, Margolles A, et al. (2011) How do Bifidobacteria counteract environmental challenges? mechanisms involved and physiological consequences. Genes Nutr 6(3): 307-318.
- Dan T, Jin R, Ren W, Li T, Chen H, et al. (2018) Characteristics of milk fermented by Streptococcus thermophilus MGA45-4 and the profiles of associated volatile compounds during fermentation and storage. Molecules 23(4): 878-892.
- Guessas B, Kihal M (2004) Characterization of lactic acid bacteria isolated from algerian arid zone raw goats' milk. Afri J Biotechnol 3(6): 339-342.
- Shabana M, Fariha H, Tariq M (2013) Characterization of lactic acid bacteria isolated from indigenous dahi samples for potential source of starter culture. Afr J Biotechnol 12(33): 5226-5231.
- Buyukyoruk S, Cibik R, Cetinkaya F, Soyutemiz GE, Goksoy EO, et al. (2010) Isolation phenotypic and molecular identification of Lactococcus lactis isolates from traditionally produced village cheeses. J Animal Veterinary Adv 9(16): 2154-2158.
- Akbar A, Sadiq MB, Ali I, Anwar M, Muhammad N, et al. (2019) Lactococcus lactis subsp lactis isolated from fermented milk products and its antimicrobial potential. CyTA- J Food 17(1): 214-220.
- Mercade M, Lindley ND, Loubiere P (2000) Metabolism of Lactococcus lactis subsp cremoris MG 1363 in acid stress conditions. Intl J Food Microbiol 55(1-3): 161-165.






























