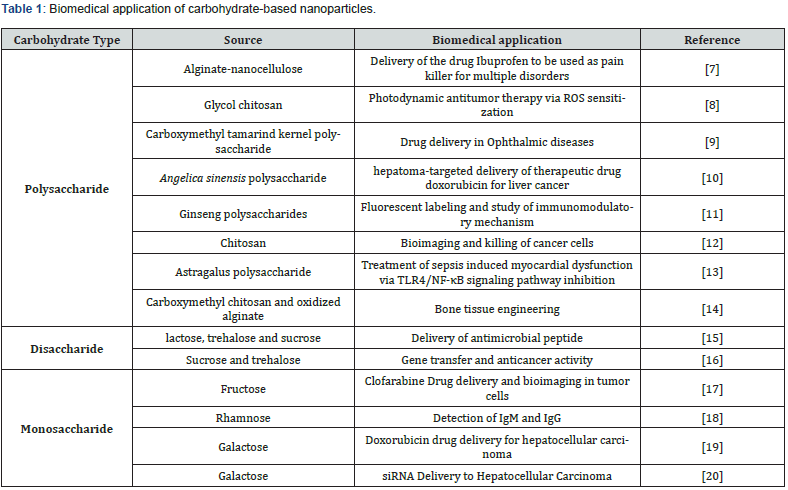
In Vitro Evaluation of Protein and Antioxidant Activity of Different Probiotic and Endophytic Bacteria
Rathod Zalak R, Vora Axita P, Mistry Parth R, Patel Prachi K, Gohel Payal K and Saraf Meenu S*
Department of Microbiology and Biotechnology, University School of Sciences, Gujarat University, India
Submission:March 24, 2022;Published:April 15, 2022
*Corresponding author:Department of Microbiology and Biotechnology, University School of Sciences, Gujarat University, Ahmedabad, Gujarat, India 380009
How to cite this article: Rathod Zalak R, Vora Axita P, Mistry Parth R, Patel Prachi K, Gohel Payal K, et al. In Vitro Evaluation of Protein and Antioxidant Activity of Different Probiotic and Endophytic Bacteria. Curr Trends Biomedical Eng & Biosci. 2022; 20(4): 556042. DOI:10.19080/CTBEB.2022.20.556042
Abstract
Antimicrobial resistance and complicated enzymatic pathways have become increasingly common in recent years. Antibiotic resistance is becoming a serious problem. The goal of an interpretative antibiogram reading is to look at the total susceptibility pattern, not just the result for a single antibiotic, in order to forecast the underlying resistance mechanisms. The goal of this study is to assist physicians in reading and comprehending antibiograms by attempting to interpret resistance mechanisms phenotypically. Protein is a fundamental component of the bacterial cell under any growth conditions. The Recommended Dietary Allowance of protein for a healthy adult with minimal physical activity is now 0.8 g protein per kg Body Weight (BW) per day, based on short-term nitrogen balance studies. Lowrey’s method is widely used method for the measurement proteins due to its simplicity, sensitivity or precision. This research article discusses the use of fish gut microbiota as potential sources for various antioxidant compounds as well as their potential for use in a range of disorders and as a functional dietary supplement due to their scavenging activities.
Keywords: Antioxidant; DPPH; Endophytes; FRAP; Probiotic
Introduction
Protein serves a variety of functions in the human body. Proteins are big, complex molecules that play a number of important tasks in the human body. They are essential for the structure, function, and control of the body’s tissues and organs, and they accomplish most of their work in cells. It aids in the repair and development of your body’s tissues, as well as allowing metabolic activities and coordinating biological operations. Proteins not only provide a structural basis for your body, but they also keep the pH and fluid balance in check. These proteins give cells shape and support. They also allow the body to move on a bigger scale. Acid hydrolysis, followed by amino acid analysis, is possibly the most accurate way of estimating protein content. Most alternative methods are sensitive to the protein’s amino acid makeup, therefore absolute amounts are impossible to acquire [1]. Lowry et al. procedure is no exception, but its sensitivity is moderately consistent from protein to protein, and it has been widely used to the point where Lowry protein estimations are a perfectly acceptable alternative to a stringent absolute determination in almost all situations involving protein mixtures or crude extracts [2-14].
The Biuret reaction, in which the peptide bonds of proteins react with copper under alkaline conditions to produce Cu+,which reacts with the Folin reagent, and the Folin-Ciocalteau reaction, in which phosphomolybdo-tungstate is reduced to heteropolymolybdenum blue by the copper-catalyzed oxidation of aromatic amino acids, are both used in the method. The reactions produce a bright blue colour, which is influenced by the amount of tyrosine and tryptophan present. The approach is sensitive down to roughly 0.01 mg of protein/mL, and it works best with protein concentrations between 0.01 to 1.0 mg/mL. An antioxidant is a component that prevents the oxidation of other molecules. A chemical reaction in which electrons are lost or the oxidation state is raised is referred to as oxidation. During oxidation reactions, free radicals can be produced [15]. Plants and animals contain complex systems that include antioxidants like glutathione, ascorbic acid, retinoic acid, and tocopherols, as well as enzymes like catalase, superoxide dismutase, and various peroxidases. Lack of antioxidants or suppression of antioxidant enzymes produce oxidative stress, which can injure or kill cells [4].
An antioxidant is a stable molecule that can supply an electron to a rogue free radical, neutralising it and restricting the free radical’s capacity to cause harm. These antioxidants can delay or prevent cellular damage due to their ability to scavenge free radicals. These low-molecular-weight antioxidants can safely interact with free radicals, stopping the chain reaction from harming vital components. The body’s natural metabolism produces some antioxidants, including glutathione, ubiquinol, and uric acid [14]. It is also possible to locate dietary sources of lighter antioxidants. Vitamin E (tocopherol), vitamin C (ascorbic acid), and B-carotene are the most important micronutrient (vitamin) antioxidants, despite the fact that the body has multiple enzyme systems that scavenge free radicals [6]. Because the human body is unable to manufacture some substances, they must be received through the diet.
When bacteria and fungi are unable to grow in the presence of one or more antimicrobial agents, this is referred to as susceptibility. This indicates that the antibiotic is capable of killing the bacterium. After bacteria or fungi causing an individual’s infection have been recovered in a culture of the samples, susceptibility testing is done.
Antibiotic Susceptibility Testing (AST) determines effective antibiotic dosage and creates an actual therapy profile for the optimal management of a patient’s health against life-threatening diseases. As a result, fast diagnosis is essential in the treatment of bacterial infections. Prior to the standardized disc diffusion approach, other diffusion-based experiments were conducted. Fleming was a pioneering contributor to AST in the 1920s. The gutter method, developed by Fleming, was the first method of antibiotic analysis, in which antibiotics were dispensed into a gutter formed of solid agar and allowed to diffuse through it. In 1941, Abraham et al. produced a modification to this design known as the “Oxford cup method,” in which the gutter is ensured and the size of the inhibition zone corresponds to the antibiotic concentration [1,11].
Using the multiple disc diffusion approach, Gould and Bowie (1952) and Stokes (1955) were able to distinguish between susceptible and resistant bacteria in the 1950s [3,16]. Because of variations in results received from different labs, all of the proposed procedures were imprecise, inappropriate, and unreliable for routine testing [8]. As a result, numerous organizations (most notably the World Health Organization) launched several attempts in 1961 to address the need for a uniform approach for antibiotic susceptibility testing. Bauer and Kirby’s method was later certified as a standard method for susceptibility testing in 1966. This technology has the potential to be used in clinical laboratories for routine susceptibility testing. Furthermore, the approach is well-liked because it provides a straightforward, lowcost protocol for detecting many targets [17,18].
Materials and Methods
Sample collection
Five Rohu fish gut isolates and three plant endophytic isolates, designated A, B, C, R, and D, E, P, respectively, were obtained from Gujarat University’s School of Sciences, Department of Microbiology and Biotechnology.
Characterization of isolates
Isolates were characterised using conventional methods based on their cultural and morphological characteristics.
Cultural and growth characteristics: The appearance and properties of bacterial colonies on solid media were used to assess growth characteristics. Eye visualisation of colonies grown on nutrient agar media revealed cultural characteristics.
Morphological characteristics: Microscopic inspection was used to determine morphological characteristics. Gram’s reactivity was determined via Gram staining. Microscopic examination using Gram staining reagents such as Crystal violet, Gram’s iodine, Decolourizer (Methanol), and Safranin was used to observe morphology [10]. Determine which microorganisms are gram - positive and gram-negative. Shape, size, and arrangement are used to further define them.
Antibiogram study of bacteria
The antibiotics from the discs diffuse into the medium and limit the development of susceptible bacteria, forming an inhibition zone around the disc. The molten tubes of nutrient agar media were sterilised (autoclaved) and cooled to 50-55°C. Thoroughly mix 20 mL of medium with 1 mL of active culture. Pour the media into a sterilised Petri plate after it has been thoroughly mixed. After solidifying the media, use sterile forceps to implant the antibiotic disc, which contains various types of antibiotics at different concentrations. For bacterial growth, incubate the plate at room temperature. The zone of inhibition must be measured. Subtract the diameter of the disc from the diameter of the zone.
Extraction: To begin, isolated bacteria were inoculated in 100 mL nutrient broth containing 250 mL volumetric flasks and incubated for 3 days in an oxygenated system under shaking conditions. After 3 days of incubation, 10 mL of fermented broth was transferred to a centrifuge tube with a capacity of 50 mL, and solvent (10 mL methanol) was added for extraction. Then it was centrifuged for 10 minutes at 10000 rpm and 4°C to get a concentrated quantity of cell mass. The supernatant and pellets were separated after centrifugation, and the pellet was vortexed with methanol for 5 to 8 minutes to disrupt the cells. Whattman filter paper number 1 was used to filter out this extract, which was then employed to check an antioxidant activity. The supernatant was used for further protein analysis.
Antioxidant scavenging activity
DPPH (1, 1 Diphenyl 2- Picryl hydrazyl) assay
Reagent Preparation: 0.1 mM or 0.004% DPPH (39.4 mg DPPH in 1000 ml methanol/ethanol); Ascorbic acid- standard solution (1000mg/L).
Analytical method: The radical scavenging activity of different extracts was determined by using DPPH assay [13].1, 1 Diphenyl 2- Picryl hydrazyl is a stable (in powder form) free radical with purple colour in solution which turns yellow when scavenged. This property is used in the DPPH experiment to demonstrate free radical scavenging activity. Antioxidants reduce DPPH to DPPH-H, lowering the absorbance. The amount of discolouration represents the extract’s scavenging potential in terms of hydrogen donating ability. Various aliquots of ascorbic acid as an antioxidant were used for standard graph creation, ranging from 0.1 to 1.0 ml. Fill each tube with distilled water to make the final volume 1 ml. The tubes were filled with 2 mL of 0.1 mM DPPH. After the addition of free radicals allow 30 minutes to incubate in the dark. In order to determine the antioxidant activity of samples from each of the extract, 1ml aliquots were collected. Stick to the same procedure as standard. After 30 min, the absorbance of the mixture was read at 517 nm. Calculate the Radical Scavenging Activity based on the absorbance of the samples
% DPPH radical scavenging activity = [(Ac – As)/Ac] × 100
Where, Ac is the absorbance of the control, and As is the absorbance of the extracts/standard [9].
FRAP (ferric reducing ability of plasma) assay
Reagent preparation: 300 mM acetate buffer(dissolve 0.16 g of sodium acetate in 100 mL of 0.28 M acetic acid; the pH should be 3.6), 10 mM TPTZ in 40 mM HCl (To make a 10 mM 2,4,6-tri(2- pyridyl)-1,3,5-triazine (TPTZ) solution; add 0.31 g to 100 mL HCl (40 mM)), 20 mM FeCl3.6H2O (Weigh 0.0541 g of ferric chloride Iron(III) chloride hexahydrate; Transfer the Iron(III) chloride hexahydrate to a beaker of appropriate size; Add 5 mL of ultrapure water to the beaker to dissolve the salt; Transfer your solution to a 10 mL volumetric flask; Using ultrapure water, complete the solution’s volume to reach 10 mL; Prepare this solution on the day of the experiment), Standard solution FeSO4.7H2O 2000 μM (0.0556 g heptahydrate iron(II) sulphate; Transfer the iron (II) sulphate heptahydrate to a suitable beaker. To dissolve the salt, fill the beaker with 90 mL of ultrapure water; Fill a 100 mL volumetric flask halfway with the solution; Using ultrapure water, complete the solution’s volume to reach 100 mL; Prepare this solution on the day of the experiment).
Analytical method: The method relies on electron-donating antioxidants, reduce Fe3+ -TPTZ complex (a colourless complex) to Fe2+ -tripyridyl triazine (a blue-coloured complex) at low pH. The change in absorbance at 593 nm is used to monitor this process. The FRAP reagent was made by combining 300 mM acetate buffer, 10 mM TPTZ in 40 mM HCl, and 20 mM FeCl3.6H2Oin a 10:1:1 ratio. A freshly made working FRAP reagent was aliquoted with a pipette (4 ml) and thoroughly mixed with 0.2 ml of the sample.
When the ferric tripyridyl triazine (Fe3+ -TPTZ) complex was reduced to its ferrous (Fe2+) form, a strong blue colour complex was generated, and the absorbance at 593 nm was measured against a reagent blank (4 ml FRAP reagent+0.2 ml distilled water) after 30 minutes at 37°C. The calibration curve was created by plotting the absorbance at 593 nm against various FeSO4 concentrations [2].
Protein estimation by Folin Lowry method
Reagent Preparation: Alkaline copper reagent (The reagent is prepared by mixing solution 1 and 2 in 50:1 proportion; Solution-1: 2% Na2Co3 in 0.1 N NaOH; Solution-2: 0.5% CuSO4•5H2O in 1%Na-K tartarate.), Folin’s Ciocalteau reagent (Dilute the commercial reagent with an equal amount of water at the time of use), Standard Protein sample (Bovine serum albuminwater soluble protein: 200 μg/ml).
Analytical method: The Biuret reaction, in which the peptide bonds of proteins react with copper under alkaline conditions to produce Cu+, which reacts with the Folin reagent, and the Folin- Ciocalteau reaction, in which phosphor-molybdo-tungstate is reduced to heteropolymolybdenum blue by the copper-catalyzed oxidation of aromatic amino acids, are both used in the method. The reactions produce a bright blue colour, which is influenced by the amount of tyrosine and tryptophan present. Various aliquots of standard protein solution were collected, ranging from 0.1 to 1.0 ml. Distilled water is used to get the final volume up to 1 ml. The tubes were filled with 5 mL of alkaline copper reagent. Allow 15 minutes to incubate at room temperature. After 15 minutes of incubation, 0.5 mL of Folin-Ciocalteau reagent was added. A 30-minute incubation period is required once again. Colorimetrically measure the intensity of the blue colour at 750 nm after incubation. Take 1 mL of each sample and follow the same process. Calculate the protein concentration in the sample by plotting the standard graph of absorbance at 750 nm vs. BSA (protein) concentration [17].
Results and Discussion
Morphological characterization
Microscopic observation can be done by electron microscopy under oil emergent lens (100x) and their gram’s reactions also visualised (Table 1).
Colony characterization
The methodical evaluation of the colony’s appearance, focusing on features such as size, shape, colour, opacity, and consistency, provides clues to the organism’s identity, allowing relevant tests to be selected to produce a positive identification also observed under light microscope using 10x and 40x (Table 2, Figures 1 and 2).


Antibiotic susceptibility
Twelve antibiotics were used to investigate the susceptibility and resistance of the bacterial isolates. The effects of antibiotics were investigated using the following antibiotics at various concentrations and their ZOI was measured in mm (Pathoteq Biological Laboratory- ‘bio-disc- 12’) (Figure 3).



All isolates show resistance towards Ampicillin and Piperacillin antibiotics. Rohu fish gut flora shows maximum inhibition zone with antibiotic Co-trimoxazole > Ceftizoxime > Ciprofloxacin > Cefotaxime = Gentamicin = Amikacin > Chloramphenicol = Tetracycline = Ofloxacin = Gatifloxacin; where Citrus limon plant endophyte shows maximum inhibition zone with antibiotic Ofloxacin Gatifloxacin > Ceftizoxime = Ciprofloxacin > Chloramphenicol = Gentamicin = Amikacin > Gatifloxacin. And in the case of Citrus limon endophyte shows resistant towards Ampicillin, Co-trimoxazole, Ceftizoxime, Piperacillin, and Tetracycline antibiotics (Table 3, Figures 4 and 5).

Antioxidant assays
A single assay method is not sufficient for the assessment of the antioxidant potential of a bacterial sample. Furthermore, different antioxidant assays vary in terms of assay principles and experimental conditions. The ability of antioxidants to donate hydrogen is assumed to be the reason for their action on DPPH. The DPPH free radical scavenging method is widely used to assess the antioxidant potential of bacterial extracts. In the DPPH assay, the extract reduces a violet-coloured DPPH solution to a yellowcoloured product, diphenylpicryl hydrazine, in a concentration dependent manner. A different approach is- The FRAP assay, a simple, automated test for determining an extract’s ferric reducing ability, is offered as a new approach for determining “antioxidant power.” At low pH, a colourful ferrous-tripyridyltriazine complex forms when ferric to ferrous ion reduction occurs. The antioxidant activity of each and every bacterial sample was different, as shown in Graph 1. When compared to other bacterial samples, bacterial sample A, which was isolated from Rohu fish gut, had higher antioxidant activity, as measured spectrophotometrically at 593 nm and 517 nm using the FRAP and DPPH methods, respectively (Figures 5 and 6) (Graph 1).




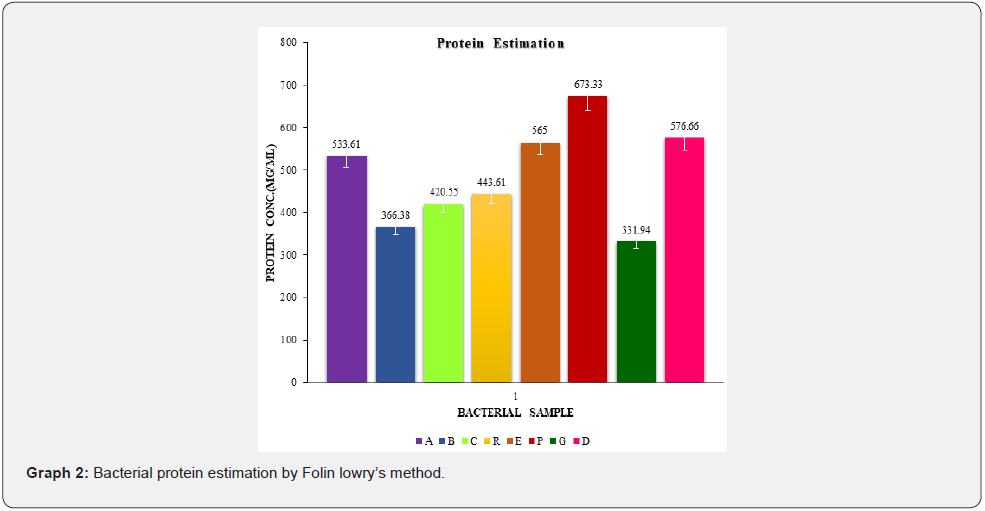
Protein estimation
The Folin Lowry method is the most often utilised method because of its sensitivity and simplicity. The alkaline copper tartrate and Folin’s reagent react with the protein in the sample to produce a deep blue colour. The colour intensity is directly proportional to protein concentration and is measured at 750 nm. Using the linear regression equation (y = mx + c), the protein content of the bacterial extract was calculated. The protein content in micrograms per millilitre changed between each and every bacterial sample, as shown in graph. Bacterial sample P, which was isolated from a citrus plant, had a higher protein content than the other bacterial samples, as evaluated spectrophotometrically at 750 nm using the Folin-lowry method.
Conclusion
Our understanding of the positive effects of antioxidants has exploded in recent years. The maximum free radical scavenging activity was reported in a methanolic extract of plant endophytic bacteria along with fish gut microbiota, which may contain antioxidant components such as vitamins. These in vitro tests show that fish gut isolates and plant isolates are a good source of natural antioxidants, which may help to prevent the advancement of diseases caused by free radicals, such as cancer. However, it is currently unknown which components of bacterial extracts are responsible for the antioxidative activity. More research is required in order to isolate and characterise the antioxidant compounds found in the bacterial extract [19-30].
Antioxidants are widely used as additives in fats and oils, as well as in the food processing industry to prevent food spoilage. Proteins are other necessary nutrients for human health. Folin Lowry’s approach was used to estimate them. It was found that bacterial isolates from fish gut probiotics and Citrus limon endophytes create a substantial amount of protein.
Acknowledgement
DST-FIST Department of Microbiology & Biotechnology, University School of sciences, Gujarat university, Ahmedabad, Gujarat, India.
References
- Bauer AW, Kirby WM, Sherris JC, Turch M (1966) Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 45(4): 493-496.
- Benzie IF, Strain JJ (1996) The Ferric Reducing Ability of Plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239(1): 70-76.
- Gould JC (1952) The determination of bacterial sensitivity to antibiotics. Edinb Med J 59(4): 178-179.
- Halliwell B, Gutteridge JM (1995) The definition and measurement of antioxidants in biological systems. Free Radic Biol Med 18(1): 125-126.
- Khan ZA, Siddiqui MF, Park S (2019) Current and emerging methods of antibiotic susceptibility testing. Diagnostics (Basel) 9(2): 49.
- Levine M, Rumsey SC, Daruwala R, Park JB, Wang Y (1999) Criteria and recommendations for vitamin C intake. JAMA 281(15): 1415-1423.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265-275.
- Mayrhofer S, Domig KJ, Mair C, Zitz U, Huys G, et al. (2008) Comparison of broth microdilution, Etest, and agar disk diffusion methods for antimicrobial susceptibility testing of Lactobacillus acidophilus group members. Appl Environ Microbiol 74(12): 3745-3748.
- Rahman MM, Islam MB, Biswas M, Alam AK (2015) In vitro antioxidant and free radical scavenging activity of different parts of Tabebuia pallida growing in Bangladesh. BMC Res Notes 8(1): 1-9.
- Zalak R, Meenu SS (2021) Effect of Endophytic Consortium on Pisum sativum and Vigna angularis plant analyst with FTIR Spectroscopy. Acta Scientific Microbiol 4(12): 79-84.
- Jorgensen JH, Ferraro MJ (2009) Antimicrobial susceptibility testing: a review of general principles and contemporary practices. Clin Infect Dis 49(11): 1749-1755.
- Sapan CV, Lundblad RL, Price NC (1999) Colorimetric protein assay techniques. Biotechnol Applied Biochem 29(2): 99-108.
- Sharma OP, Bhat TK (2009) DPPH antioxidant assay revisited. Food Chem 113(4): 1202-1205.
- Shi H, Noguchi N, Niki E (1999) Comparative study on dynamics of antioxidative action of α-tocopheryl hydroquinone, ubiquinol, and alpha-tocopherol against lipid peroxidation. Free Radic Biol Med 27(3-4): 334-346.
- Sies H (1997) Oxidative stress: oxidants and antioxidants. Exp Physiol 82(2): 291-295.
- Stokes EJ (1971) Antibiotic sensitivity testing. Br Med J 2(5763): 707.
- Waterborg JH (2009) The Lowry method for protein quantitation. In: Walker JM (Ed.), The protein protocols handbook. Humana Press, Totowa, NJ, USA. pp: 7-10.
- World Health Organization (1961) Standardization of methods for conducting microbic sensitivity tests: second report of the Expert Committee on Antibiotics [meeting held in Geneva from 11 to 16 July 1960]. pp. 24.
- Zalak R, Sarita S, Meenu SS (2021) Endophytic Bacterial Effects on Seed as Enhancement in Germination with Mobilization of Reserves and Microbial Disinfection in Citrus Limon (L) by Biopriming Under Salinity Stress. Curr Trends Biomedical Eng & Biosci 20(2): 1-4.
- Zalak R, Jain R, Sharma S, Rupal KS, Lucie KM, et al. (2020) To determine the minimum inhibitory concentrations and minimum bactericidal concentrations of the effective plants extract and endophytes of Citrus limon. Biosc Biotech Res Comm 13((1): 210-215.
- Zalak R (2015) Isolation of Probiotic Bacteria from Shoal fish and Mud fish. M.sc Dissertation thesis, Department of microbiology and biotechnology Gujarat university, Ahmedabad, Gujarat, India. pp. 28-39.
- Zalak R (2015) Developing Plant Probiotic bacteria based Bioformulation having PGPR properties. M.Phil. thesis (2016) Department of microbiology and biotechnology Gujarat University, Ahmedabad, Gujarat, India. pp. 48-65.
- Zalak R, Meet AD, Meenu SS, Baldev VP (2021) Developing plant probiotic bacteria based bioformulation having PGPR properties. In: PLANTA Research Book Series. 2: 324-342.
- Zalak R, Meenu SS, Baldev VP (2018) Developing plant probiotic bacteria from rohu fish on bioformulation by PGPR activity. Vidhya A Journal of Gujarat University Special Special issue: proceeding of national conference on synthetic biology approached in drug discovery and microbial biotechnology. pp. 147-160.
- Zalak R, Meenu SS (2021) Effect of endophytes as phosphate solubilizing bacteria on Vigna radiata plant. Vidhya A Journal of Gujarat University. 16(2): 38-50.
- Zalak R, Meet AD, Baldev VP, Meenu SS (2020) A review article: Citrus as salt tolerance under plant tissue culture. Int J Engg Res Develop 7(8): 34-44.
- Zalak R, Meet AD, Venisha VC, Meenu SS (2020) Antimicrobial activity and bacterial potency of Sphingomonas Paucimobilis as endophytes isolated from leaf of citrus limon. Int Res J Engg Technol 7(8): 4740-4744.
- Zalak R, Venisha VC, Meenu SS (2020) Pharmacognostic and phytochemical evaluation of Citrus limon (Lemon) fruit and leaf. Int J Innovat Res Sci Engg Technol 10(2): 1015-1019.
- Zalak R, Meet AD, Sarita S, Baldev VP, Meenu SS (2020) A review on phytochemical screening of secondary metabolites. Vidhya A Journal of Gujarat University. 15(2): 175-190.
- Zalak R, Meet AD, Amita YM, Ritika J, Baldev VP, et al. (2020) Compatibility studies on different endophytes of Citrus limon antagonistic to bacterial with plant pathogen. Adv Biores 11(3): 126-129.






























