In vivo Acute Toxicity (LD50) Studies and Phytochemical Screening of Stem Bark Extracts of Detarium microcarpum Guilt and per (Caesalpinioideae) and Myosortisscorpioides L. (boraginaceae)
Abdu Zakari1*, Dimas Kubmarawa2, Sunday Osemeohon A2, Sunday Dzarma3 and Elisha Barde Baba3
1Department of Chemical Sciences, Federal University, Nigeria
2Department of Chemistry, Modibbo Adama University of Technology, Nigeria
3Department of Pharmacology, National Institute for Pharmaceutical Research and Development (NIPRD), Nigeria
Submission: November 11, 2017; Published: May 16, 2018
*Corresponding author: Abdu Zakari, Department of Chemical Sciences, Federal University, Kashere PMB 0182, Gombe, Nigeria, Email: zabdu70@gmail.com
How to cite this article: Abdu Z, Dimas K, Sunday O A, Sunday D, Elisha B B. In vivo Acute Toxicity (LD50) Studies and Phytochemical Screening of Stem Bark Extracts of Detarium microcarpumGuill and per (Caesalpinioideae) and Myosortisscorpioides L.(boraginaceae). Curr Trends Biomedical Eng & Biosci. 2018; 14(4): 555891. DOI: 10.19080/CTBEB.2018.14.555891
Abstract
The methanol extract of stem bark of Detarium microcarpum and Myosortisscorpioides were screened for in vivo safety pharmacological studies (acute toxicity LD50) using Swiss Albino Miceand for the presence of some phytochemicals. The result of the phytochemical screening indicated the presence of important secondary metabolites including alkaloids, saponins, tannins, anthraquinones, carbohydrates, flavonoids. The results showed that the plant extracts were toxic to the mice and caused death at a concentration of =2900mg/Kg and 1600mg/kg body weight of the live mice and LD50 of 3,807.89mg/Kg and =2,828.43mg/Kg respectively. The result of the study was a useful guide to a practically safe dose in an ongoing antimalarial study of extracts of these plants.
Keywords: In vivo; Acute toxicity; LD50; Phytochemical; Detarium microcarpum; Myosortisscorpioides
Introduction
Paracelsus once said, 'All substances are poisons; there is none which is not a poison. The right dose differentiates a poison and a remedy [1]. Toxicological screening is very important for the development of new drugs and for the extension of the therapeutic potential of existing molecules. Toxicity tests are mostly used to examine specific adverse effects or specific end points such as cancer, cardio toxicity, and skin/eye irritation. Toxicity testing also helps calculate the No Observed Adverse Effect Level (NOAEL) dose and is helpful for clinical trials. Necessities of toxicological studies include the benefit of calculation of risk ratio and the prediction of therapeutic index obtained as ratio of maximum tolerated dose and minimum curative dose as expressed by following equation;
Therapeutic index = (Maximum tolerated dose)/(Minimum curative dose)
The smaller the ratio, the better safety of the drug.
The present in vivo safety pharmacology study was designed to define the dose-response relationship of the adverse effect of the plant extracts and the time course of the adverse effect e.g. onset and duration of response was also investigated. In the present study extracts of the plants under investigation were administered at different dose levels, and the effects were observed for 14 days. All mortalities caused by the extractsduring the experimental period were recorded and behavioral changes in the animals were investigated and recorded D. microcarpum occurs naturally in the drier regions of West and Central Africa (Benin, Cameroon, Central African Republic, Chad, Gambia, Ghana, Guinea, Guinea Bissau, Côte d'Ivoire, Mali, Niger, Nigeria, Senegal, Sudan and Togo).
D. microcarpum belongs to the Fabaceae family (legumes). It flowers during the rainy season and bears fruit from September - January/May and in November [2]. D. microcarpum is highly appreciated by local peoples due to its variety of uses; it is said to be one of the most appreciated in the environments where it occurs naturally. The fruit can be eaten raw or cooked, but for the most part, its pulp is transformed into flour. The seed flour is a traditional emulsifying, flavouring and thickening agent used to prepare cakes, bread, couscous, baby food and local beer. Its seed kernels are added to egusi soup, or are cooked and eaten as vegetables. The leaves are used as a condiment or vegetables, as are its flowers. Medicinal properties are in the roots, stems, bark, leaves and fruits to treat ailments including tuberculosis, meningitis and diarrhea. The species showed strong inhibitory effects on HIV-1 or HIV-2 infection in methanol extracts. Leaves and roots are also used to treat farm animals [3].
The fruit pulp has been found to have high proportions of carbohydrate (40-42.0%) and protein (29.1-30.9%). The seeds yield 7.5% oil with the predominant fatty acid being linoleic acid. The hulled seed flour contains per 100g: 3.5-6.5g water, 3g crude fibre, 13-15g crude fat, 13.5-27g crude protein, 39g carbohydrate, Ca 500mg, Mg 500mg, Fe 100mg [4]. Myosotisscorpioides (syn. myosotispalustris), commonly calledwater forget-me-not or true forget-me-not, is a herbaceousperennial flowering plant in the borage family Boraginaceae. It is native to Europe and Asia, but can be found elsewhere, including Africa and much of North America, as an introduced species and sometimes a noxious weed. It grows in bogs and wet places and beside streams and rivers. Myosotisscorpioides is a rhizomatous marginal aquatic perennial that typically grows 6-10" (less frequently to 18") tall on decumbent to upright angular stems (Missouri Botanical Garden).
The review of chemistry, pharmacology and clinical properties of Detarium microcarpum indicated the presence of some phytochemicals while a co-chromatographic analysis revealed the presence of catechol i.e condensed tannins and various phytoconstituents in bark of Detarium microcarpum [5]. However not much was reported on Myosortisscorpioides. Although Plants are potential sources of modern drugs, yet only about 13% of drugs produced within developed countries are derived from plants and less than 15% of the plants were known to have been investigated pharmacologically as reported in a survey study conducted by Kubmarawa et al. 2013. The present study is an effort to further investigate the Chemistry and Pharmacology of Detarium microcarpum and Myosortisscorpioidesto justify the claim of traditional medicine practitioners in the area of study (Adamawa State - Nigeria).
Materials and Methods
Plant collection and identification
The fresh samples of the plants were collected in Girei Local Government Area of Adamawa State, Nigeria. The plant was identified by Clifford Emmanuel (research assistant) and authenticated by Botanists in the Department of Biological sciences Federal University Kashere.
Preparation of plant parts for extraction
The plant samples were air dried and ground into powder with pestle and mortal and weighed. Exactly 60g of the powder was packed in an improvised thimble of white clean cloth sewed manually. The thimble was then inserted into the Soxhlet apparatus, 500mL ethanol was transferred down the thimble into the pot. Extraction was maintained at a temperature of 75 °C and continued for 6 hours. Then the ethanol extract was collected and the cake recovered from the thimble was kept. The methanolic extract was evaporated on a rotary evaporator (R110) at 40 °C, altogether, 200g of each sample were extracted and labeled F01 [5].
Phytochemical screening
Standard method described by Abdul Rani et al. [6] and modified by Abdu et al. [7] was adopted for the screening of some important secondary metabolites.
Acute toxicity test (LD50) the lokes method
In the acute oral toxicity study, 36 Swiss albino mice were employed to observe the toxicity effects of ethanol crude extract of E. angustifolia leaves. From the result, mortalities were reported as well as adverse toxicity signs were observed on the tested mice right from the lower dose. The physical appearance such as, fur, raised tails, salivation, paw licking were observed which indicated that the crude extract did affect the animals.
Laboratory animals used in the research
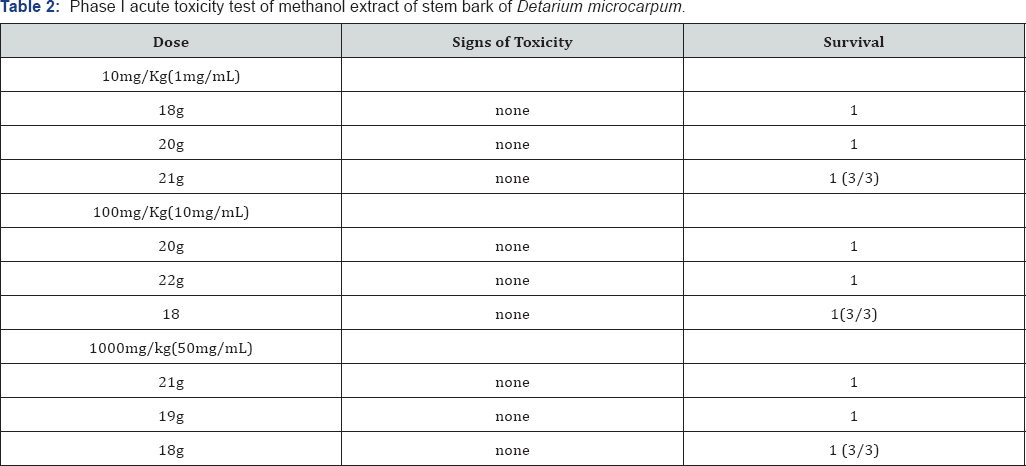
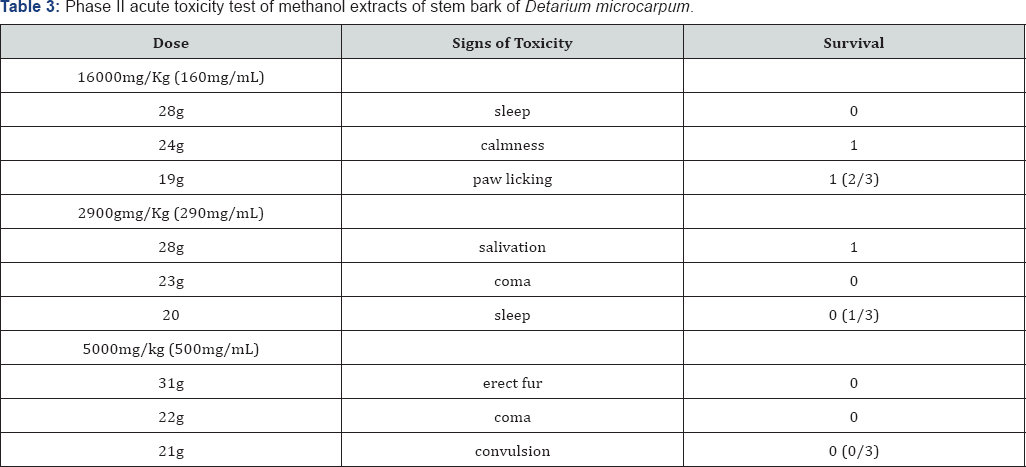
The animal models involved in this study were the Swiss albino mice acquired from the National Veterinary Research Institute (NVRI) Vom in Jos, Plateau State-Nigeria. The experimental procedures relating to the animals were authorized by the Ethical committee of National Institute for Pharmaceutical Research and Development (NIPRD), Abuja-Nigeria before starting the study and were conducted under the internationally accepted principles for laboratory animal use and care (EEC Directive of 1986; 86/609/EEC) [5]. The parameters evaluated during this assay included body weight of the mice, survival time, paw licking, salivation, stretching/writhing, erect fur, calmness, reduced movement, weakness, coma, convulsion, sleep and death. Albino mice were coded, weighed and randomized into three groups/dose with 3 mice per group/dose in two phases. In phase I, nine mice were randomized into three groups of three mice each and were given (5mL) each of 30, 300 and 500mg/kg body weight (b. wt) of the extract orally [8].
Volume of extract per live weight of mice
The volume of extract per live weight of mice was calculated using the following formula:
Volume of extract solution = ((Weight of mice x Dose))/(Stock concentration)
Where Dose refer to 'expected therapeutic level'
The Limit Dose as provided by EPA/OECD for all Acute Oral Toxicity was 5000mg/kg, which is a dose considered high enough that if no mortality or significant toxicity was seen in animals receiving this dose, no higher doses are required.
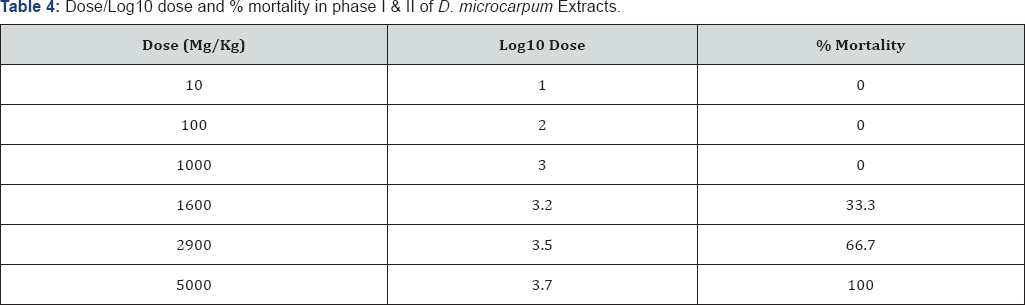
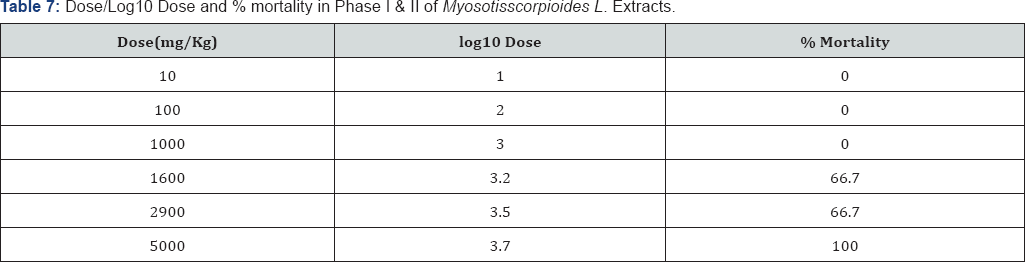
The mice were observed for paw licking, salivation, stretching of the entire body, weakness, sleep, respiratory distress, coma and death in the first four (4) hours and subsequently daily for seven (7) days. In phase II, another fresh set of nine mice were randomized into three groups of three mice each and were given 1600, 2900 and 5000mg kg-1 body weight of the extract orally on the result of the first phase. They were observed for signs of toxicity and mortality for the first four critical hours and thereafter, daily for 7 days. The LD50 was then calculated as the square root of the product of the lowest lethal dose and highest non-lethal dose i.e., the geometric mean of the consecutive doses for which 0% and 100% survival rates were recorded in the second phase, the oral median lethal dose were calculated using the formula: LD50=√(Minimum toxic dose x maximum toxic dose)
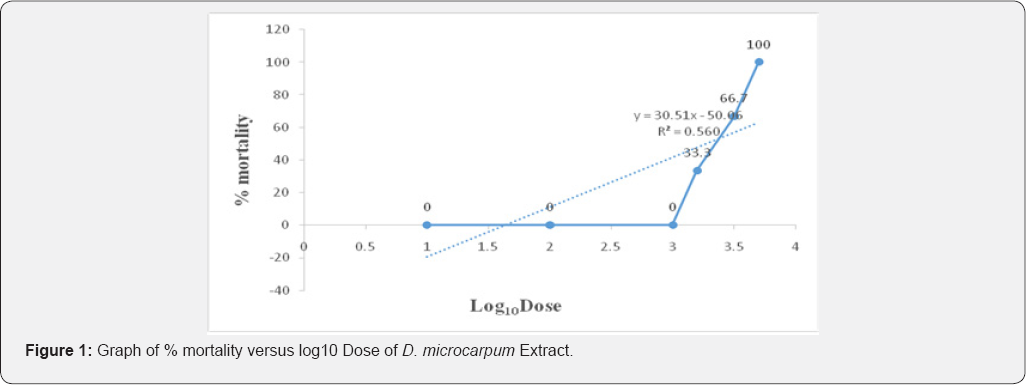
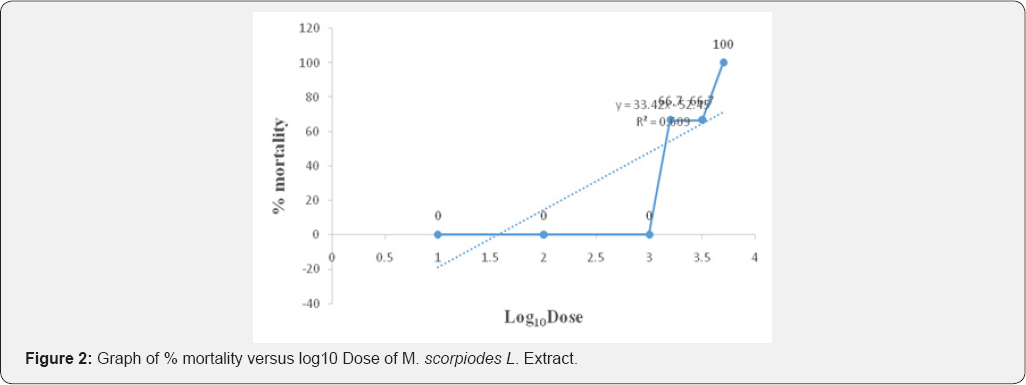
Verified by a graph of percentage mortality versus logarithmto base tenof doses administered.
The Organization for Economic Cooperation and Development (OECD) Test Guidelines for Acute Toxic Class method (OECD TG 423) were strictly adhered to.
Result
Percentage yield of the extracts was presented in Table 1. The result of the Phase I and II Acute Toxicity Test were presented in Table 2-7. The result of the phytochemical screening were presented in Table 7.



Discussion
The result of the phytochemical screening indicated the presence important secondary metabolites such as alkaloids, flavonoids, steroids, tannins which partly justified the use of these plants traditionally as remedy to ailments. The in vivo acute oral toxicity of methanol extracts of Detarium microcarpum and Myosotisscorpioides Las well as phytochemical screening were determined in the present study. The screening of the toxicity of the plant was crucial to assure the safety and effectiveness of the plant extract. Signs of toxicity such as pain, distress, allergic reactions, and physical changes in the tested animals were detected in the in vivo assays. The plant extracts have caused acute toxicity effects with an LD50 values of 3,807.89mg/Kg and 2,828.43mg/Kg for Detarium microcarpum and Myosotisscorpioides L respectively. The results suggest the possibility of the use of the extract as a potential source for the development of pharmacological agent to treat various types of ailment. Studies on the antimalarial properties of these plants is in advance stage which will be followed immediately by isolation and characterization pure compounds from the plants extract (Figure 1 & 2).
Conclusion
Findings of the study were useful guide to the expected therapeutic level of the extracts through an increasing dose phase-wise manner in the ongoing antimalarial investigation of these plants.Specific phytoconstituents will be target especially alkaloids in isolation process ahead.
Acknowledgement
The authors wish to acknowledge TET Fund - Nigeria, the managements of Federal University Kashere Gombe-Nigeria, Modibbo Adama University of Technology Yola-Nigeria and National Institute for Pharmaceutical Research and Development Abuja - Nigeria for their support.
References
- Borzelleca, Joseph F (2000) Paracelsus: herald of modern toxicology. Toxicological Sciences 53(1): 2-4.
- Kouyaté AM, Van Damme P (2006) Medicinal plants/ Plantesmédicinales: Detarium microcarpum Guill & Perr Prota 11(1).
- Kouyaté AM (2GG7) Enquêteethnobotaniquesur Detarium microcarpum Guill Et Perr au sud du Mali quoted in Kalinganire. Improving Rural Livelihoods through Domestication of Indigenous Fruit Trees in the Parklands of the Sagel, p. 189.
- Abdalbasit UN (2GG7) Detarium microcarpum Guill and Perr fruit proximate chemical analysis and sensory characteristics of concentrated juice and jam, p. 4217.
- Sani A, Agunu A, Danmalam UH, Hajara I (2G14) Pharmacognostic studies of the stem bark of detarium microcarpum - guill and perr (fabaceae). Nat Prod Chem Res Sl: GG4.
- Abdul R, Muhamad S, Soundararajan V, Sreenivasan S, Lachimanan Y, et al. (2G1G) Acute oral toxicity and brine shrimp lethality of elaeisguineensisjacq, (oil palm leaf) methanol extract. Molecules 15(11): 8111-8121.
- Abdu Z, Dimas K (2G16) In vitro cytotoxicity studies and qualitative investigation of phytochemicals of stem bark Extracts of Detarium microcarpum (caesalpinioideae), Echinaceae angustifolia (compositae) and Isoberlinia doka (fabaceae): National Journal of Multidisciplinary Research and Development 1(1): 22-26.
- https://en.wikipedia.org/wiki/Paracelsus accessed 19/1G/2G17.






























