In Silico Identification of the Antimastitis I Components in Allium cepa
Sruthy B1, Latha MS2* and Sherly P Anand3
1Department of Chemistry, Sree Narayana College, India
2Department of Chemistry, Sree Narayana College, Ala, India
3Department of Zoology, TKMM College of Arts and Science, India
Submission: January 19, 2018; Published: March 26, 2018
*Corresponding author: Latha MS, Department of Chemistry, Sree Narayana College, Ala, Chengannur, Kerala, India, Tel: 9400648068; Email: lathams2014@gmail.com
How to cite this article: Sruthy B, Latha MS, Sherly P A. In Silico Identification of the Antimastitis Components in Allium cepa. Curr Trends Biomedical Eng & Biosci. 2018; 13(2): 555860. DOI: 10.19080/CTBEB.2018.13.555860
Abstract
Bovine mastitis is a pathogenic inflammation to the mammary gland of cattle results in huge reduction of milk production. Antibiotics are used for the treatment of mastitis. Limitted cfficacy of the antibiotic treatment is a threat to the diary industry. Farmers of South Kerala use the crude extract of Allium cepa for the treatment of mastitis and is found to be highly effective. Present study focus on the identification of the antimastitis components in Allium cepa which can perform efficient docking interaction with the target protein (PDB ID: 1JIL) of Staphalococus aureus bacteria using Schrodinger suit v 9.2. The phytochemicals, Myricetin, Quercetin-4'-glucoside and Quercetin-3-glucoside in Allium cepa are identified as the antimastitis components. The results of the study are helpful in developing novel drugs for the treatment of mastitis.
Keywords: Bovine mastitis; Docking; Staphalococus aureus; Target protein
Introduction
Bovine mastitis is associated with the presence of infectious agents as bacteria, Bovine Herpes virus, non bacterial pathogens like mycoplasmas, fungi, yeast and chalmydia [1,2]. Among these, Staphalococus aureus bacterial infections are most common. Transfer complex protein (PDB ID:1JIL) [3] is an excellent drug target in Staphalococus auerus. Inhibition of which results in bacterial cell lysis. Allium cepa is a biennial plant, a good source of nutrients. Various phytochemicals present in the plant are responsible for its therapeutic potential [4]. 20 phytochemicals in Allium cepa were selected for the docking study based on ADMET properties predicted by Qik prop option.
Methodology
Bioinformatics analysis
Preparation of Protein: Crystal structure of the target proteins of Staphalococus aureus transfer complex protein was obtained from RCSB Protein Data Bank (PDB ID: 31JIL). Automatically imported PDB files from the RCSB PDB website to the Maestro working interface [5-7]. Subsequently the protein was optimized and minimized.
Ligand preparation
Twenty phytochemicals present in Allium cepa were selected to find out the inhibitory activity towards the target protein. The structure of the phytochemicals and commonly used commercially available antibiotic Pirlimycin hydrochloride were downloaded from pub chem in the (.sdf) format. These ligands were subjected to ligand preparation using the ligand preparation wizard (ligprep) of Schrodinger software in the Maestro interface 12 [6,7].
Docking Studies
The compounds were screened by Schrodinger docking software to study inhibitors of the target protein. Grid generation was done using the centroid of workspace ligand R0 48-8071. The rigid receptor docking using the Glide program was carried out against the target protein with the set of ligands. The mode of docking was selected as XP (Extra precision) for a high docking accuracy. The glide docking was carried out for the minimised protein [6-8]
Docking Studies
The bioactive compounds from Allium Cepa were checked for their ADMET properties using Qik prop module [8,9]. QikProp helped in analyzing the pharmacokinetics and pharmacodynamics of the ligand by accessing the drug likeness.
Results and Discussion
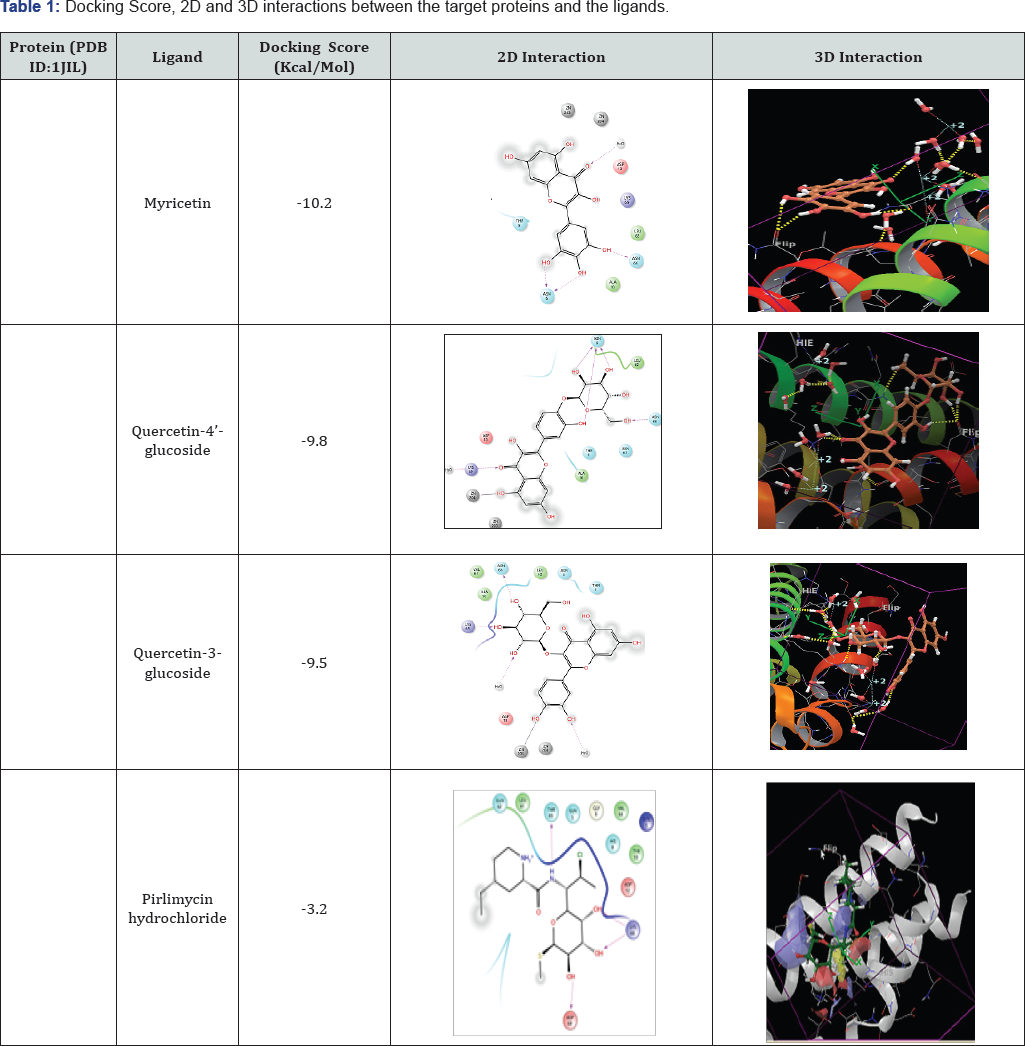
(Range of properties - Molecular weigh less than 500, Hydrogen bond donor 0-6, Hydrogen bond acceptor 2-20 and QP logP(o/w) -2 - 6.5), From the results obtained it has been understood that the phytochemicals Myricetin, Quercetin- 4'glucoside and Quercetin-3-glucoside are responsible for the antimastitis activity of the plant Allium cepa. These phytochemicals posess excellent docking interactions to the target protein than the commonly used commercially available drug, Pirlimycin hydrochloride. Three hydroxyl groups in Myricetin forms three side-to-side hydrogen bonds with 66 ASN and 6ASN amino acid residues of the target protein. Ketonic oxygen in the compound forms a hydrogen bond with water molecule in the protein. Four hydroxyl groups in Quercetin-4'- glucoside forms four hydrogen bonds with 6ASN and 66ASN amino acid residues. One hydroxyl group in the compound forms a metal coordination with Zn 203. Ketonic oxygen forms a hydrogen bond with water molecule in the protein. Four hydroxyl groups in Quercetin-3-glucoside forms four hydrogen bonds with 66ASN, 69LYS and two water molecules in the target protein. Also it forms a metal coordination with Zn 203 in the target protein. Pirlimycin hydrochloride forms four hydrogen bonds with 60ASP, 66 LYS and 65THR amino acid residues of the target protein. All the three phytochemicals having better docking score than the commonly used antibiotic (Table 1) Qik prop option helped in analyzing the ADMET properties. The antimastitis components in Allium cepa satisfied the ADMET properties for drug likeness (Table 2).

Conclusion
The phytochemicals Myricetin, Quercetin-4'-glucoside and Quercetin-3-glucoside have potent inhibitory activity towards the staphalococus aureus transfer complex protein compared to the commercial drug Pirlimycin Hydrochloride. These phytochemicals are responsible for the antimastitis activity of Allium cepa. Results of this study are helpful for the development of novel drugs for mastitis.
References
- Reena J, Prashant D, Chaitanya G , Anju P (2015) Identification of purative drug targets in mastitis causing Staphalococus aureus by insilico approach. International journal of Pharmacy and Pharmaceutical sciences 7(9): 496-502.
- Wellenberg GJ, van der Poel WH, Van Oirschot JT (2002) Viral infections and Bovine mastitis-a review. JT Veterinary Microbiology 88(1): 27-45.
- Qiu X, Janson CA, Smith WW, Green SM, McDevitt P, et al. (2001) Crystal structure of Staphylococcus aureus tyrosyl-tRNA synthetase in complex with a class of potent and specific inhibitors. Protein Sci 10 (4): 2008-2016.
- Chitra S, Patil MB, Ravi K, Swati P (2009) Preliminary phytochemical investigation and wound healing activity of Allium cepa Linn(LILIACEAE). International journalof pharmacy and pharmaceutical sciences 2(2): 56-62.
- http://www.rcsb.org/pdb
- Schrodingersuit (2009) Protein Preparation Wizard; Epik version 2.0; Impact version 5.5. Schrodinger, LLC, New York, USA.
- Friesner RA, Banks JL, Murphy RB, Halgren TA, Klicic JJ, et al. (2004) Glide: a new approach for rapid, accurate docking and scoring.l.Method and assessment of docking aacuracy. J Med Chem 47(7): 1739-1749.
- Friesner RA, Murphy RB, Repasky MP, Frye LL, Greenwood JR, et al. (2006) Extra precesion glide: docking and scoring incorporating a model of hydrophobic enclosure for protein-ligand complexes. J Med Chem 49(21): 6177-6196.
- Schrodinger S (2010) Protein Preparation Guide, Site Map 2.4; Glide Version 5.6, Lig Prep: 2.4, Qik Propn 3.3, Schrodinger, LLC, New York, USA.






























