Anesthesia Control: A Personal Opinion
Milella L*
General Neonatal and Pediatric Anesthesia and Intensive Care Unit-Neonatal and Pediatric Cardiac Anesthesia and Intensive Care Unit, Italy
Submission: February 01, 2018; Published: February 08, 2018
*Corresponding author: L Milella, General Neonatal and Pediatric Anesthesia and Intensive Care Unit--Neonatal and Pediatric Cardiac Anesthesia and Intensive Care Unit , Pediatric Hospital "Giovanni XXIII”-Viale Amendola 207, 70121-Bari, Italy, Tel: +39-080-559-6624; Fax: +39-080-559-6814; Email:giovaniosl@bol.com.br
How to cite this article: Gerald C, Zahra K, Farima K, Mehrian J, Seyedmahdi P, Adeel J, Arzu K. High-Density Lipoprotein-Targeted Therapy for Coronary Heart Disease: is the HDL Hypothesis Operational or Defunct?. Curr Trends Biomedical Eng & Biosci. 2018; 12(3): 555836. DOI: 10.19080/CTBEB.2018.12.555836.
Abstract
Supported by evidence from basic science, clinical, and epidemiological studies, the high-density lipoprotein (HDL) hypothesis posits that reduction in HDL levels may accelerate the development of coronary heart disease (CHD). HDL may also exert cardiovascular protection via antioxidative, anti-thrombotic, anti-inflammatory, and anti-apoptotic properties. However, controversial results from clinical trials involving HDL- raising interventions have cast doubts on the validity of HDL hypothesis. The coevolution of HDL physiology and CHD pathogenesis has prompted the revision of hypothesis, with an alternative focus on the functionality of HDL particles. Instead of raising the quantity of HDL, improving the quality of HDL by promoting reverse cholesterol transport may be a more appropriate strategy in preventing adverse cardiovascular events. Preliminary data suggest that administration of apolipoprotein A1 mimetic peptides and reconstituted HDL particles could be safe and effective in promoting plaque stabilization and regression. Future studies are warranted to clarify the effect of HDL subspecies on atherosclerosis and determine whether the effect could translate into cardiovascular benefits.
Keywords: High-density lipoprotein; Cholesterol; Apolipoprotein; Atherosclerosis; Thrombosis; Coronary heart disease
Abbreviations: ABCA1: ATP-Binding Cassette Subfamily A Member 1; ABCG1: ATP-Binding Cassette Subfamily G Member 1; ACS: Acute Coronary Syndrome; Apoa1: Apolipoprotein A1; CETP: Cholesteryl Ester Transfer Protein; CHD: Coronary Heart Disease; HDL: High-Density-Lipoprotein; LCAT: Lecithin-Cholesterol Acyltransferase; PC: Phosphatidylcholine; RCT: Reverse Cholesterol Transport; rHDL: Recombinant High-Density- Lipoprotein
Introduction
The potential beneficial role of high-density lipoprotein (HDL) in preventing coronary heart disease (CHD) was first reported in the Framingham Study [1]. The inverse relationship between HDL levels and the incidence of CHD has prompted the development of "HDL hypothesis", which posits that increasing HDL may reduce the risk of atherosclerosis. However, data from large clinical trials of HDL-raising interventions have cast doubts on the validity of HDL hypothesis. In the AIM-HIGH study (ClinicalTrials.gov: NCT00120289) [2], the addition of niacin to statin therapy showed no clinical benefit among patients with atherosclerotic disease despite a significant improvement in HDL and triglyceride levels. In the dal-OUTCOMES study (ClinicalTrials.gov: NCT00658515) [3], dalcetrapib was associated with an increase in HDL but failed to reduce the risk of recurrent cardiovascular events among patients with recent acute coronary syndrome (ACS). As a result, it is suggested that HDL concentration may be a risk factor but not an appropriate therapeutic target.
Mechanistically, HDL is a pivotal intermediary in the process of reverse cholesterol transport (RCT), in which cholesterol is removed from macrophages within atherosclerotic plaques and transferred to the liver for biliary excretion (Figure 1) [4]. Furthermore, HDL particles may possess several protective effects to the circulatory system, including anti-oxidative [5], anti-inflammatory [6], anti-apoptotic [7], and anti-thrombotic properties [8]. The HDL hypothesis has been revised to reflect the alternative focus on the functionality of HDL particles (i.e., cholesterol efflux) [9]. To examine the revised "HDL flux hypothesis", this review summarizes the findings from human studies involving the administration of apolipoprotein A1 (ApoA1) mimetic peptides and recombinant HDL (rHDL) particles (Figure 2).
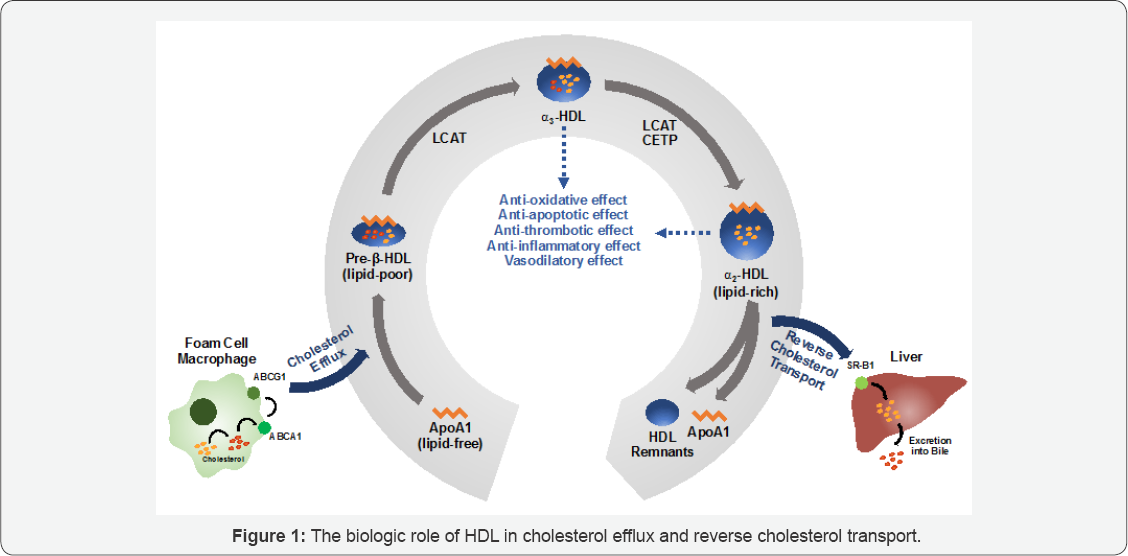
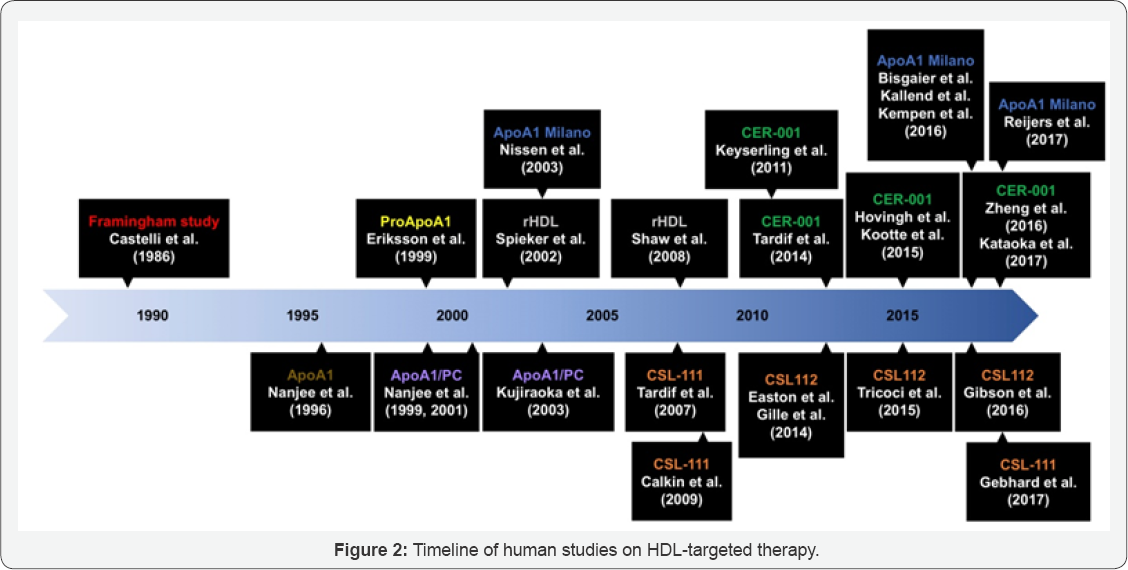
Discussion
ApoAl infusion
ApoA1, the main protein component of HDL, was postulated to initiate reverse cholesterol transport and provide a substrate for the formation of mature HDL particles via various mechanisms. Nanjee et al. first examined the effect of intravenous infusion of lipid-free ApoA1 and showed that there was no associated increase of HDL unesterified or esterified cholesterol [10]. In contrast, infusions of apolipoprotein A1/phosphatidylcholine (ApoA1/PC) over 4 h resulted in HDL increase that persisted for more than 72h [11]. It was concluded that acute ApoA1/PC infusions promote the intravascular production of nascent pre- P-HDLs. Results from a later study from Nanjee et al. support the hypothesis that these pre-p-HDLs readily cross endothelium into tissue fluid and enhance the efflux of cholesterol from tissues [12]. Furthermore, infusions of nascent HDL particles were associated with increased concentrations of key mediators of RCT including lecithin-cholesterol acyltransferase (LCAT) and cholesteryl ester transfer protein (CETP) [13]. In the study from Eriksson et al. [14], subjects with heterozygous familial hypercholesterolemia received intravenous infusion of proApoAl (the precursor of human ApoAl). Although the improvement in plasma concentrations of HDL was transient, the fecal excretion of cholesterol was increased in all subjects, indicating that ApoAl may stimulate RCT and eventually lead to enhanced cholesterol excretion.
Reconstituted HDL infusion
Spieker et al. examined the effect of reconstituted HDL (rHDL) infusion on endothelial function in hypercholesterolemic subjects [15]. Intravenous rHDL infusion was found to restore endothelium-dependent vasodilation by increasing the bioavailability of nitric oxide among patients with hypercholesterolemia. Shaw et al. demonstrated that in patients with claudication scheduled for percutaneous superficial femoral artery revascularization, there was an increase in HDL levels and cholesterol efflux capacity after infusion of rHDL. Additionally, rHDL treatment led to acute changes in plaque characteristics with a reduction in lipid content, macrophage size, and inflammatory markers [16]. Taken together, these studies support the potential therapeutic benefit of rHDL in patients at risk of atherosclerosis.
ApoAl Milano
ApoAl Milano is an ApoAl variant identified in individuals who exhibit very low levels of HDL. Nissen et al. [17] investigated the effect of intravenous infusion of ETC-216 (recombinant ApoAl Milano/phospholipid complexes) on atheroma volume in a randomized controlled trial of patients with ACS. Compared to placebo, ApoA1 Milano significantly reduced the atheroma burden as measured by intravascular ultrasound. Bisgaier et al. [18] demonstrated a dose-dependent elevation of HDL following ETC-216 administration to normal human volunteers. The formula ETC-216 was considered to contain host cell proteins that carry a risk for eliciting inflammatory reactions. MDCO- 216, the successor of ETC-216, is a complex of dimeric ApoA1 Milano and 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine. Kallend et al. [19] investigated the use of MDCO-216 in healthy volunteers as well as patients with coronary artery disease, and demonstrated that MDCO-216 was well tolerated and associated with an enhancement of ABCAl-mediated cholesterol efflux. Kempen et al. [20] evaluated single ascending doses of MDCO- 216 on HDL in relation to changes in cholesterol efflux capacity in healthy volunteers and in patients with stable angina pectoris. MDCO-216 was found to increase the formation of lipid-rich HDLs (al-HDL and a2-HDL) that may directly augment cholesterol efflux capacity. Reijers et al. further examined the safety profile of MDCO-216 with respect to adverse immunostimulation [21]. It was concluded that intravenous administration of MDCO-216 did not induce inflammatory response in healthy volunteers and subjects with stable coronary artery disease. Further clinical studies are warranted to determine whether MDCO-216 would reduce the risk of atherosclerotic disease or cardiovascular events.
CER-001
CER-001 is comprised of recombinant human ApoA1 complexed with phospholipids and has similar beneficial effects of nascent pre-^-HDLs on RCT. The safety of CER-001 was examined in a double-blind, randomized cross-over study from Keyserling et al. [22]. CER-001 infusion was reported to be safe and well tolerated. In addition, administration of CER-001 elevated total and free cholesterol in HDL fraction, indicating an increase in reverse cholesterol transport. Tardif et al. investigated the effects of various dosage of CER-001 on atheroma volume as assessed by intravascular ultrasonography and quantitative coronary angiography [23]. CER-001 infusions did not reduce coronary atherosclerosis when compared with placebo. Hovingh et al. studied the effect of serial infusions with CER-001 on carotid artery wall dimensions in patients with homozygous familial hypercholesterolemia on top of maximal low-density lipoprotein-lowering therapy [24]. Compared with placebo, there was a significant reduction in carotid mean vessel wall area among subjects who received CER-001. Kootte et al. [25] studied the effect of CER-001 infusions among patients with familial hypoalphalipoproteinemia in an open-label, uncontrolled, proof-of-concept study [25]. Similarly, it was concluded that CER-001 increased cholesterol efflux and reduced carotid artery wall area. In patients with atherosclerotic carotid artery disease, Zheng et al. [26] demonstrated that infusion of CER-001 increased plasma ApoA1 concentration and cholesterol efflux capacity [26]. CER-001 was also found to specifically target plaque regions as evidenced by contrast enhancement. Kataoka et al. compared the effect of 6 weekly infusions of CER- 001 versus placebo on coronary atherosclerosis in patients with ACS [27]. CER-001 induced atheroma regression in patients with baseline atheroma volume percentage of >30% but not in those with <30%, suggesting that the efficacy of CER-001 would increase with the severity of atherosclerosis. The ongoing phase 2, multicenter CARAT trial (ClinicalTrials.gov: NCT02484378) will further investigate the impact of serial 3mg/kg infusions of CER-001 on plaque burden in patients with recent ACS [28].
CSL-111 and CSL112
CSL-111 is synthesized from combining human plasma derived-ApoA1 with phosphatidylcholine from soybean to mimic the biochemical profile of native HDL. In the A randomized, placebo-controlled ERASE trial (ClinicalTrials.gov: NCT00225719) [29], patients with coronary atherosclerosis were randomized to receive placebo or CSL-111 at the dosage of 40mg/kg or 80 mg/kg. CSL-111 treatment was discontinued prematurely in the 80mg/kg group due to liver function abnormalities. Infusions of CSL-111 40mg/kg was relatively well tolerated, albeit with self-limiting elevations of transaminase. Although there was no significant reduction in plaque burden, CSL-111 was associated with a statistically significant improvement in the plaque characterization index and coronary score on quantitative coronary angiography. In the study from Calkin et al. [30], patients with type 2 diabetes mellitus were infused with placebo or CSL-111 20mg/kg/h for four hours. In addition to an increase in plasma HDL, CSL-111 was shown to inhibit platelet hyper-reactivity via reducing the cholesterol content of platelet membranes. The study from Gebhard et al. points to a potential cardiovascular repair effect of CSL-111 in the ACS setting [31]. Administration of CSL-111 appeared to preserve the circulating levels of CD34, possibly via its beneficial effects on increased migration and reduced apoptosis of progenitor cells. CSL112 is the reformulated version of CSL-111 and its pharmacokinetics, safety, and tolerability have been studied in healthy subjects by Easton et al. [32]. A dose-dependent sustained increase in ApoA1 concentration was observed following CSL112 infusions. Multiple infusions of CSL112 were also found to be safe and well tolerated without evidence of major organ toxicity or immunogenicity. Gille et al. reported that CSL112 administration was associated with an elevation of cholesterol efflux mediated by ATP-binding cassette transporter A1 (ABCA1) [33]. The safety, pharmacokinetics, and pharmacodynamics of CSL112 infusion among patients with stable atherosclerotic disease was studied in a phase 2a, randomized, double-blind, multicenter, dose-ranging trial by Tricoci et al. (ClinicalTrials.gov: NCT01499420) [34]. CSL112 was well tolerated and resulted in an immediate elevation of ApoAl levels as well as an increase in the cholesterol efflux capacity. The study by Didichenko et al. [35] demonstrated that CSL112 may enhance the HDL functionality by remodeling of HDL particles. Three sizes of particles were generated after spontaneous fusion of CSL112 with HDL. Lipid-poor and small HDL displayed both high capacity for ABCA1-mediated cholesterol efflux and anti-inflammatory effects, whereas the large particles possessed strong antioxidant functions. These findings offer a novel approach in devising HDL-targeted therapy. It is plausible that enrichment of a specific pool of HDL particles may confer a specific facet of cardiovascular protection, and achieving the right blend of HDL compositions may be required to optimize the benefits of rHDL infusion. In the post- myocardial infarction setting, CSL112 infusions were also well tolerated and not associated with significant alterations in liver or kidney function [36]. The efficacy of CSL112 in improving cardiovascular outcome needs to be evaluated in future studies.
Conclusion
Infusion of reconstituted HDL agents may promote reverse cholesterol transport from peripheral tissues to liver and eventually into the bile. Additional potential cardiovascular benefits of HDL-targeted therapy include stabilization of atherosclerotic plaque, suppression of inflammatory response, and inhibition of thrombogenesis or oxidative stress. Given its safety profile and preliminary evidence in reducing atheroma burden, future clinical studies are warranted to assess the therapeutic and prophylactic value of HDL-targeted infusion agents for patients with coronary heart disease.
Conflict of Interest
The article is not funded and the authors declare no conflict of interest.
References
- Castelli WP, Garrison RJ, Wilson PW, Abbott RD, Kalousdian S, et al. (1986) Incidence of coronary heart disease and lipoprotein cholesterol levels. The framingham study. JAMA 256: 2835-2838.
- AIM-HIGH Investigators, Boden WE, Probstfield JL, Anderson T, Chaitman BR, et al. (2011) Niacin in patients with low hdl cholesterol levels receiving intensive statin therapy. N Engl J Med 365: 2255-2267.
- Schwartz GG, Olsson AG, Abt M, Ballantyne CM, Barter PJ, et al. (1980) Effects of dalcetrapib in patients with a recent acute coronary syndrome. N Engl J Med 367: 2089-2099.
- Fielding CJ, Fielding PE (1995) Molecular physiology of reverse cholesterol transport. Journal of lipid research 36: 211-228
- Soran H, Schofield JD, Durrington PN (2015) Antioxidant properties of HDL. Frontiers in pharmacology 6: 222.
- Barter PJ, Nicholls S, Rye KA, Anantharamaiah GM, Navab M, et al. (2004) Antiinflammatory properties of HDL. Circulation research 95: 764-772.
- Suc I, Escargueil-Blanc I, Troly M, Salvayre R, Negre-Salvayre A (1997) HDL and ApoA prevent cell death of endothelial cells induced by oxidized ldl. Arteriosclerosis, thrombosis, and vascular biology 17: 2158-2166.
- Mineo C, Deguchi H, Griffin JH, Shaul PW (2006) Endothelial and antithrombotic actions of HDL. Circulation research 98: 1352-1364.
- Rader DJ, Tall AR (2012) The not-so-simple hdl story: Is it time to revise the hdl cholesterol hypothesis? Nature medicine 18: 1344-1346.
- Nanjee MN, Crouse JR, King JM, Hovorka R, Rees SE, et al. (1996) Effects of intravenous infusion of lipid-free apo a-i in humans. Arteriosclerosis, thrombosis, and vascular biology 16(9): 1203-1214.
- Nanjee MN, Doran JE, Lerch PG, Miller NE (1999) Acute effects of intravenous infusion of apoa1/phosphatidylcholine discs on plasma lipoproteins in humans. Arteriosclerosis, thrombosis, and vascular biology 19: 979-989.
- Nanjee MN, Cooke CJ, Garvin R, Semeria F, Lewis G, et al. (2001) Intravenous apoa-i/lecithin discs increase pre-beta-HDL concentration in tissue fluid and stimulate reverse cholesterol transport in humans. Journal of lipid research 42: 1586-1593.
- 13. Kujiraoka T, Nanjee MN, Oka T, Ito M, Nagano M, et al. (2003) Effects of intravenous apolipoprotein a-i/phosphatidylcholine discs on lcat, pltp, and cetp in plasma and peripheral lymph in humans. Arteriosclerosis thrombosis and vascular biology 23: 1653-1659.
- Eriksson M, Carlson LA, Miettinen TA, Angelin B (1999) Stimulation of fecal steroid excretion after infusion of recombinant proapolipoprotein a-i. Potential reverse cholesterol transport in humans. Circulation100: 594-598.
- Spieker LE, Sudano I, Hurlimann D, Lerch PG, Lang MG, et al. (2002) High-density lipoprotein restores endothelial function in hypercholesterolemic men. Circulation 105: 1399-1402.
- Shaw JA, Bobik A, Murphy A, Kanellakis P, Blombery P, et al. (2008) Infusion of reconstituted high-density lipoprotein leads to acute changes in human atherosclerotic plaque. Circulation research 103: 1084-1091.
- Nissen SE, Tsunoda T, Tuzcu EM, Schoenhagen P, Cooper CJ, et al. (2003) Effect of recombinant apoa-i milano on coronary atherosclerosis in patients with acute coronary syndromes: A randomized controlled trial. Jama 290: 2292-2300.
- Bisgaier CL, Ackermann R, Rea T, Rodrigueza WV, Hartman D (2016) Apoa-imilano phospholipid complex (etc-216) infusion in human volunteers. Insights into the phenotypic characteristics of apoa- imilano carriers. Pharmacological research 111: 86-99.
- Kallend DG, Reijers JA, Bellibas SE, Bobillier A, Kempen H, Burggraaf J, et al. (2016) A single infusion of mdco-216 (apoa-1 milano/popc) increases abca1-mediated cholesterol efflux and pre-beta 1 hdl in healthy volunteers and patients with stable coronary artery disease. European heart journal. Cardiovascular pharmacotherapy 2: 23-29.
- Kempen HJ, Asztalos BF, Moerland M, Jeyarajah E, Otvos J, et al. (2016) High-density lipoprotein subfractions and cholesterol efflux capacities after infusion of mdco-216 (apolipoprotein a-imilano/palmitoyl- oleoyl-phosphatidylcholine) in healthy volunteers and stable coronary artery disease patients. Arteriosclerosis, thrombosis, and vascular biology 36: 736-742.
- Reijers JAA, Kallend DG, Malone KE, Jukema JW, Wijngaard PLJ, et al. (2017) Mdco-216 does not induce adverse immunostimulation, in contrast to its predecessor etc-216. Cardiovascular drugs and therapy 31: 381-389.
- Keyserling CH, Hunt TL, Klepp HM, Scott RA, Barbaras R, et al. (2011) Cer-001, a synthetic hdl-mimetic, safely mobilizes cholesterol in healthy dyslipidemic volunteers.
- Tardif JC, Ballantyne CM, Barter P, Dasseux JL, Fayad ZA, et al. (2014) Effects of the high-density lipoprotein mimetic agent cer-001 on coronary atherosclerosis in patients with acute coronary syndromes: A randomized trial. European heart journal. 35: 3277-3286.
- Hovingh GK, Smits LP, Stefanutti C, Soran H, Kwok S, et al. (2015) The effect of an apolipoprotein a-i-containing high-density lipoproteinmimetic particle (cer-001) on carotid artery wall thickness in patients with homozygous familial hypercholesterolemia: The modifying orphan disease evaluation (mode) study. American heart journal 169: 736-742.
- Kootte RS, Smits LP, van der Valk FM, Dasseux JL, et al. (2015) Effect of open-label infusion of an apoa-i-containing particle (cer-001) on rct and artery wall thickness in patients with fha. Journal of lipid research 56: 703-712.
- Zheng KH, van der Valk FM, Smits LP, Sandberg M, Dasseux JL, et al. (2016) HDL mimetic cer-001 targets atherosclerotic plaques in patients. Atherosclerosis 251: 381-388.
- Kataoka Y, Andrews J, Duong M, Nguyen T, Schwarz N, et al. (2017) Regression of coronary atherosclerosis with infusions of the high- density lipoprotein mimetic cer-001 in patients with more extensive plaque burden. Cardiovascular diagnosis and therapy 7: 252-263.
- Andrews J, Janssan A, Nguyen T, Pisaniello AD, Scherer DJ, et al. (2017) Effect of serial infusions of reconstituted high-density lipoprotein (cer- 001) on coronary atherosclerosis: Rationale and design of the carat study. Cardiovascular diagnosis and therapy 7: 45-51.
- Tardif JC, Gregoire J, L'Allier PL, Ibrahim R, Lesperance J, et al. (2007) Effects of reconstituted high-density lipoprotein infusions on coronary atherosclerosis: A randomized controlled trial. JAMA 297: 1675-1682.
- Calkin AC, Drew BG, Ono A, Duffy SJ, Gordon MV, Schoenwaelder SM, et al. (2009) Reconstituted high-density lipoprotein attenuates platelet function in individuals with type 2 diabetes mellitus by promoting cholesterol efflux. Circulation 120: 2095-2104.
- Gebhard C, Rheaume E, Berry C, Brand G, Kernaleguen AE, et al. (2017) Beneficial effects of reconstituted high-density lipoprotein (rhdl) on circulating cd34+ cells in patients after an acute coronary syndrome. PloS one 12: e0168448.
- Easton R, Gille A, D'Andrea D, Davis R, Wright SD, Shear C (2014) A multiple ascending dose study of csl112, an infused formulation of apoa-i. Journal of clinical pharmacology 54: 301-310.
- Gille A, Easton R, D'Andrea D, Wright SD, Shear CL (2014) Csl112 enhances biomarkers of reverse cholesterol transport after single and multiple infusions in healthy subjects. Arterioscler Thromb Vasc Biol 34: 2106-2114.
- Tricoci P, D'Andrea DM, Gurbel PA, Yao Z, Cuchel M, Winston B, et al. (2015) Infusion of reconstituted high-density lipoprotein, csl112, in patients with atherosclerosis: Safety and pharmacokinetic results from a phase 2a randomized clinical trial. Journal of the American Heart Association 4: e002171.
- Didichenko SA, Navdaev AV, Cukier AM, Gille A, Schuetz P, et al. (2016) Enhanced hdl functionality in small hdl species produced upon remodeling of hdl by reconstituted hdl, csl112: Effects on cholesterol efflux, anti-inflammatory and antioxidative activity. Circulation research 119: 751-763.
- Michael Gibson C, Korjian S, Tricoci P, Daaboul Y, Yee M, et al. (2016) Safety and tolerability of csl112, a reconstituted, infusible, plasma- derived apolipoprotein a-i, after acute myocardial infarction: The aegis-i trial (apoa-i event reducing in ischemic syndromes i). Circulation 134: 1918-1930.






























