Evaluation of Aegle marmelos Extracts for Antibacterial, Antifungal, Antioxidant and Cytotoxic Properties
Preeti Choudhary*, Madhulika Bhagat, Sahil Gupta and Monica Sangral
University of Jammu, India
Submission: August 10, 2017; Published: September 27, 2017
*Corresponding author: Preeti Choudhary, School of Biotechnology, University of Jammu, India, Email: preetichoudhary172@gmail.com
How to cite this article: Preeti Choudhary, Madhulika Bhagat, Sahil Gupta, Monica Sangral.Evaluation of Aegle marmelos Extracts for Antibacterial, Antifungal, Antioxidant and Cytotoxic Properties. Curr Trends Biomedical Eng & Biosci. 2017; 9(4): 555766. DOI: 10.19080/CTBEB.2017.09.555766
Abstract
Aegle marmelos has emerged out to be one of the most important medicinal properties bearing plant these days. In the present research an attempt is made to evaluate the phytochemical properties of the fruit extract of A. marmelos which demonstrated its significant antibacterial, antifungal and antioxidant properties and has potential in finding its application in medicine and herbal formulations. Leaf methanol extract exhibited in vitro cytotoxic potential against breast T47D cell line for which the IC50 values were 34.52μg/mL and 45.57μg/mL respectively. Leaf methanol extract exhibit high antibacterial and antifungal activity in comparison to other extracts.
Keywords: Herbal; Phytochemical; Cytotoxic; Formulations
Introduction
Bael (Aegle marmelos) is an Indian medicinal plant which has enormous traditional values against various diseases and many bioactive compounds have been isolated from this plant. Bael (Linn), family Rutaceae, is also known as Bale fruit tree, is a moderate sized, slender, aromatic tree, 6.0-7.5m in height, and 90 to 120cm in girth, with a somewhat fluted bole of 3.0-4.5 meter growing wild throughout the deciduous forests of India, ascending to an altitude of 1200 meter in the western Himalayas and also occurring in Andaman island. This is generally considered as sacred tree by the Hindus, as its leaves are offered to Lord Shiva during worship. According to Hindu mythology, the tree is another form of Lord Kailashnath. Leaves, fruit, stem and roots of this tree at all stages of maturity are used as ethno medicine against various human ailments. The different parts of Bael are used for various therapeutic purposes, such as for treatment of Asthma, Anaemia, Fractures, Healing of Wounds, Swollen Joints, High Blood Pressure, Jaundice, Diarrhoea Healthy Mind and Brain Typhoid Troubles during Pregnancy. Aegle marmelos has been used as a herbal medicine for the management of diabetes mellitus in Ayurvedic, Unani and Siddha systems of medicine in India (Table 1-3).


The plant has been employed for long time in folk therapy. Poultice made of leaves are used for ophthalmia and ulcers. The leaves are use to lowering the blood glucose levels [1]. Other actions like antifungal [2], antibacterial, antiprotozoal [3], antispermatogenic [4] are also reported. The plant has been found to contain number of phytoconstituents like aegeline, agelinine, rutin, sterol, -sitosterol, -D-glucoside, marmesinine [5], lupeol, tannins, phlobatannins, flavonoids, umbelliferone, quercetin and volatile oils (Eugenol and methyl eugenol) [6]. It has been reported that leaves possess cardiotonic effect, antifungal, analgesic and antioxidant activities [7]. No scientific evaluation of antioxidant activity of A. marmelos has been reported so far. Therefore, it was thought worthwhile to evaluate antioxidant activity of A. marmelos to confirm its folk medicine claim. Many naturally occurring products have been reported to contain large amount of antioxidant other then vitamin C, E and carotenoid [8]. These antioxidant play a vital role in delaying, intercepting or preventing oxidative reactions, catalyse by free radical [9]. This antioxidant activity might be due to the presence of phenolic compounds such as 002 Afr. J. Plant Sci. flavonoids [10], phenolic acids and phenolic diterpine [11]. Antioxidants may guard against reactive oxygen species (ROS) toxicities by the prevention of ROS construction, by the disruption of ROS attack, by scavenging reactive metabolites and converting them to less reactive molecules or by enhancing the resistance of sensitive biological target to ROS attack.
Materials and Methods
Collection of plant material
The plant was identified by morphological features and collected from the local areas of Jammu. Plant material was authenticated by Department of Botany, University of Jammu, J&K, India.
Preparation of crude extracts
Sequential extraction: The plant material leaves were shade dried and then ground into fine powder. 100gms of fine powder was taken and put into the flask and then 250mL of chloroform was added to each flask. With occasional shaking and stirring the sample was extracted into chloroform. After that it was filtered and the filtrate was kept in a new flask and covered. Again 250ml of chloroform was added to the previous flask for extraction. Then the flask was kept overnight & filtered and the filtrate was added to previously collected filtrate. The extract was poured into petri dishes till chloroform was evaporated completely. The process was repeated until the entire chloroform was evaporated. The extract left in petriplates was transferred into autoclaved vials. The vials were kept in -20C for further use. Then it was further fractionated by successive solvent extraction with ethyl acetate (EtOAc fraction), acetone, methanol and then finally with methanol. After organic solvent extraction, the remaining aqueous fraction was also used for activity testing and the marc was discarded. The solvents were removed under reduced pressure and the extracts were concentrated under vacuum at 40-60 °C and weight of the dried mass was recorded.
Extraction of leaf essential oil
Hydro-distillation apparatus and procedure: One hundred and fifty grams leaves were hydro-distillated with a Clevenger-type apparatus and extracted with 2L of water for 4h until no more essential oil was obtained. The essential oil was collected, dried and kept dry in vials and stored at 4 0C before chemical and bioassay analyses. The amount of oil obtained was measured and the oil was calculated based on the fresh weight (mL/100g fresh weight) was based on comparison of their mass spectra with those if NISTO5 (version 2.0) library.
Antioxidant Studies
Free radical scavenging potential
DPPH (1, 1-diphenyl-2-picrylhydrazyl) radical scavenging assay: In this assay, free radical scavenging activity was determined according to the method of Blois with modifications. A total of 1ml from a 0.5mM methanol solution of the DPPH radical was mixed to 300|il sample (1mg/ml) and to this 0.1M sodium acetate buffer (pH 5.5) was added. The mixtures were well shaken and kept at room temperature in the dark for 30 min. The absorbance was measured at 517nm using a double beam UV-VIS spectrophotometer.
Radical scavenging activity (RSA) was expressed in percentage and calculated using the formula:
% RSA = (A control- A sample)/A control x 100.
Result was presented as IC50, the concentration of extract required to scavenge 50% of the DPPH radical.
Ferrous ion chelating (FIC)
The chelating effect on ferrous ions of the prepared extracts was estimated by the method of Singh and Rajini. Briefly, 500|il of each extract (1mg/ml) and 2500|il of methanol were added to 60|il of 2mM FeCl2. The reaction was initiated by the addition of 120μl of 5mM ferrozine into the mixture, which was then left at room temperature for 10min before determining the absorbance of the mixture at 562nm.
The ratio of inhibition of ferrozine-Fe2+complex formation was calculated using the equation:
% inhibition = ([abs. of control- abs. of test sample] / abs. of control) x 100
Antibacterial activity
The antibacterial activity was performed using agar well diffusion technique as described by NCCLS, 1999. Briefly, 100|il each of the bacterial suspensions (108CFU/ml) i.e. Bacillus subtilis, Bacillus cereus, Staphylococcus aureus, Escherichia coli, Enterococcus fecalis, Alcaligens denitrificans, Camphylobacter coli, Pseudomonas alcaligens, Alcaligens denitrificans, Micrococcus luteus and Klebsiella pneumonia were spread on nutrient agar plate. The wells were created in the pre-seeded plates using borer. Extract was added into each well. Finally the plates were kept for incubation at 37 0C for 24h. Chloramphenicol (108g/ml) was used as positive reference. Microbial growth was determined by measuring the zone of inhibition.
Antifungal activity
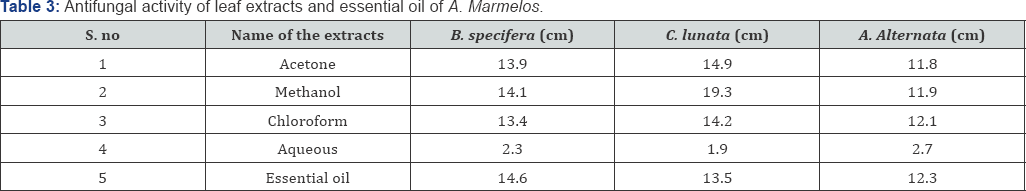
Antifungal activity of the extracts was determined by poisoned food technique against three different plant pathogenic fungi viz. Alternaria alternata, Curvularia lunata, and Bipolaris specifera. The test component was added to the sterilized potato dextrose agar in 9cm petri plates. After the preparation of plates containing different concentrations of extracts, test fungus (5mm) was inoculated in the centre of the agar plate (upside down). Plates were then incubated in dark at 26 0C. The extension diameter of hyphal growth was measured at 24 hour interval, till growth of fungus in the plate without test component (control) reached the edge of the plate. The experiment was conducted in three replicates and results were expressed as average of the three. Fungal growth diameter in each plate containing different concentrations of test component was determined to calculate per cent growth inhibition.
The antifungal indices were determined as:
Antifungal index (%) = (1-Da/Db) x 100.
Results
In the present study, leaves of A. marmelos (Bael) were collected from bhaderwah area of Jammu. The different types of solvents viz. Acetone, methanol, chloroform and aqueous were used for the extraction. Essential oil was extracted using hydro distillation method. These extracts and essential oil were further used to check antimicrobial, antifungal, antioxidant and cytotoxic potential.
Antibacterial and antifungal activity
The antibacterial and antifungal properties of the extract and essential oil was assessed by using agar well diffusion technique against 7 pathogenic bacterial and fungal strains, and the results were compared with the activity of the positive control, chloramphenicol. The results of antifungal assay are obtained by Food Poisoned Technique. It is an assay method by which we can find the growth of the fungi. The extract showed considerable antibacterial activities against most of the test organisms. The most prominent anti-bacterial profile was exhibited against S .aureus with the zones of inhibition of 9mm followed by A. denitrificans 8mm, E. coli 7mm and B subtilis 5.5mm respectively in case of chloroform extract. The most noteworthy activity was observed against all three fungal pathogens.
Cytotoxic activity
In vitro cytotoxic potential of the leaf methanol extract and essential oil of A. marmelos against breast T47D cell line was assessed by the MTT assay for which the IC50 value were 34.52|ig/mL and 45.57|ig/mL respectively. Earlier reports showed the cytotoxic activity of the leaf extracts of A. marmelos against cell lines [12].
Tetrazolium-based colorimetric assay (MTT) which measures the reduction of the tetrazolium salt MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide; Roche) into a blueformazan product, mainly by the activity of the mitochondrial enzymes, cytochrome oxidase and succinatedehydrogenase [13]. Typically, 100|il of cells suspension were plated at a density of approximate 2 *104 cells per well in a 96-well plate, and were subsequently incubated at 37 °C in a 5% CO2 humid incubator for 24 hours. Then essential oil and methanol extract with different concentrations were added to each group (triplicate wells) and the incubation was continued for 24h, followed by adding l0|il (5mg/ml) of MTT dye solution to each well for 4 hours at 37 °C. After removal of the MTT dye solution, cells were treated with 100|il DMSO and the absorbance at 490nm was quantified using ELISA reader. The cytotoxicity was calculated after comparing with the control (treated with 0.1% DMSO). Cytotoxicity is expressed as the concentration of drug inhibiting cell growth by 50% (IC50) that came to all tests and analysis were run in triplicate and mean values recorded [14-16].
Conclusion
It is quite evident from the research that Aegle marmelos contains a number of phytoconstituents which reveals its uses for various therapeutic purposes. The Plant or its individual parts can be used for the treatment of various disorders in human being such as fungal infection, microbial infection, inflammation etc. Still so much work is required with the Aegle marmelos to investigate the mechanism of actions with other therapeutic activities.
References
- Ayurvedic Pharmacopoeia of India (1988) The Ayurvedic Pharmacopoeia of India (1st edn), Part- I, Govt of India, Ministry of health and family welfare, Deptt of AYUSH, New Delhi, India, 4: 1-179.
- Renu (1983) Fungi toxicity of leaf extracts of some higher plants aginst Rhizoctonia solani Kuehn. Natl Acad Sci Lett 6: 245-246.
- Banerjee A (1980) Antimicrobial and antihelminthic screening of the fixed oil and unsaponefiable matter of Aegle marmelos. Conf Proc, Indian Science Congress. Chemistry Section abstr, India, p. 247.
- Sur TK, Pandit S, Pramanik T (1999) Antispermatogenic activity of leaves of Aegle marmelos, corr In albino rats: a preliminary report. Biomed 19(3): 199-202.
- Sharma BR, Rattan RK, Sharma P (1980) Constituent of leaves and fruits of Aegle marmelos. Indian J Chem, p. 162.
- Banerjee A, Nigam SS (1979) Studies on the fixed oil from the seeds of Aegle marmelos Corr. J Am Oil Chem Soc 56(6): 647.
- Rai MK (1996) In vitro evaluation of medicinal plant extract against Pestalotiopsis mangiferae. Hindustan Antibiot Bull 38(1-4): 53-56.
- Javanmardi J, Stushnoff C, Locke E, Vivaco JM (2003) Antioxident activity and total phenolic content of Iranian Ocimum accessions. Food Chem 83(4): 547-550.
- Vilioglu YS, Mazza G, Gao L, Oomah BD (1998) Antioxidant activity and total phenolics in selected fruits, vegetables and grain products. J Agric Food Chem 46(10): 4113-4117.
- Pietta P, Simonetti P, Mauri P (1998) Antioxidant activity of selected medicinal plants. J Agric Food Chem 46(11): 4487-4490.
- Shahidi F, Wanasundara PK (1992) Phenolic antioxidants. Crit Rev Food Sci Nutr 32(1): 67-103.
- Rahman S, Parvin R (2014) Therapeutic potential of Aegle marmelos (L.)-An overview. APJTD 4(1): 71-77.
- Kalaivani T, Premkumar N, Meignanam E, Vijayakumar V, Rajasekaran C (2008) Conventional and recent uses of Aegle marmelos (L.) corr. A vital tree of India, International Conference on Biotechnology, India.
- Banerji N, Kumar R (1980) Studies on the seed oil of A. Marmelos and its effect on some bacterial species. J Inst Chem Calcutta, India, 52: 59.
- Charakbraty B, Malik C, Bhatthacharya S (1960) Studies on the effect of green leaves of Aegle marmelos and Piper nigrum on the glucose and cholesterol levels of blood in diabetes mellitus. Indaian Med Forum 9: 285-286.
- Kessler M, Ubeaud G, Jung L (2003) Anti-and pro-oxidant activity of rutin and quercetin derivatives. J Pharm Pharmacol 55(1): 131-142.






























