Decellularized Human Liver Tissue Fragments to Create Cell - and Tissue-Engineered Liver Constructs
Nemets EA, Kirsanova LA, Basok Ju B, Lymareva MV, Schagidulin M Ju and Sevastianov*
Shumakov Federal Research Center of Transplantology and Artificial Organs of the Ministry of Healthcare of the Russian Federation, Russia
Submission: August 10, 2017; Published: August 31, 2017
*Corresponding author: Viktor I Sevastianov, Head and Professor, Department of Biomedical Technology and Tissue Engineering, Shumakov Research Center of Transplantology and Artificial Organs, Moscow, Russia, Email: viksev@yandex.ru
How to cite this article: Nemets EA, Kirsanova LA, Basok J B, Lymareva MV, Schagidulin M J, Sevastianov. Decellularized Human Liver Tissue Fragments to Create Cell - and Tissue-Engineered Liver Constructs. Curr Trends Biomedical Eng & Biosci. 2017; 8(5): 555747. DOI:10.19080/CTBEB.2017.08.555747
Abstract
One of the problems of creating a bioengineered liver as an alternative to the donor liver transplantation for the treatment of end-stage liver failure, is finding the matrix, capable of performing temporarily the functions of a natural extracellular matrix (ECM) and providing the necessary conditions to maintain the viability and functionality of liver cells. The main disadvantage of resorbable biopolymer matrices is the absence of tissue-specific properties and the impossibility of reproducing the unique structure of the liver ECM. The aim of this work was the development of technology for decellularization of liver tissue fragments while preserving the structural properties of the native liver ECM.
Materials and Methods: The decellularization of mechanically grinded human liver fragments was carried out in three changes of buffer solution (pH=7.4) containing 0.1% sodium dodecyl sulfate and increasing concentrations of Triton X100 (1%, 2% and 3%, respectively). While developing the technology, we investigated the effects of duration, rinsing conditions (static, dynamic, rotary system, magnetic stirrer), and methods of liver tissue grinding on the complete removal of cellular elements and detritus and preservation of the liver ECM structure. Slices of decellularized liver tissue samples were stained with hematoxylin and eosin, as well as by Masson's method for the detection of connective-tissue elements.
Results and Discussion: Methods of histological analysis showed that the most effective for decellularization and structural preservation of the native human liver ECM is a mode of rinsing the liver fragments for three days at room temperature in static conditions, accompanied by stirring with a magnetic stirrer 2-3 times a day for one hour. A longer time or a greater repetition of stirring is accompanied by increased risk of liver tissue damage. On the basis of the test results the algorithm was obtained for a preliminary study of donor human liver, designed to optimize the process of obtaining decellularized liver tissue fragments.
Conclusion: The algorithm of evaluation of donor human liver for decellularization is suggested and optimal conditions of obtaining decellularized liver tissue fragments are found while preserving the liver ECM structure and removing completely cellular elements and detritus.
Keywords: Liver; Decellularization; Resorbable biopolymer matrix; Tissue specific properties; Tissue-engineered construct
Introduction
Due to the shortage of donor organs, which, according to forecasts, will only increase in the coming years, the search for alternatives to transplantation in the treatment of end-stage liver failure remains extremely vital. Lately, the main emphasis in resolving this issue is made on the use of technologies of tissue engineering and regenerative medicine, directed at creation of bioengineered liver equivalents [1]. One of the key tasks is the creation of scaffolds (matrices) with the necessary physicochemical and biological properties. When developing cell- and tissue-engineered liver constructs, the matrices from resorbable biopolymer materials are used the most widely [1,2]. However, despite all their advantages, the main shortcoming of biopolymer matrices is the absence of tissue-specific properties and the impossibility of reproducing the unique structure of the liver ECM.
The methods are known for making a tissue-specific 3D matrix while preserving the tissue architecture and microvasculature by decellularization of liver, kidneys, heart, and other organs [3]. Literature describes successful experiments on decellularization of a whole liver or its lobes of rats [4], rabbits [5], pigs [6], and humans [7]. However, the issues still remain of high percentage of incomplete decellularization of cadaveric donor grafts, of complete cell colonization of the entire volume of decellularized matrices, and maintenance of their viability, especially in their depth, etc. One of the solutions to the existing problems when using whole decellularized liver as a matrix for the creation of bioengineered liver constructs is decellularization of liver fragments. The aim of this work was the development of technology for obtainment of decellularized liver fragments while preserving the structural properties of its ECM.
Material and Methods
The donor human liver
The experiments were performed on human post-mortem donor liver that could not be used for transplantation. The samples were transported in sterile saline and frozen at -20°C.
Decellularization process
Human liver fragments (HLF) were obtained by grinding with a mechanical homogenizer MPW-302 (Poland) for 15 to 30 seconds or by hand using scissors and a scalpel (the size of fragments did not exceed 1 x 1 x 3 mm). The decellularization was performed in three changes of phosphate buffer solution (PBS) (138 mm NaCl, 2.67 mm KCl, 1.47 mm KH2PO4, 8.1 mm Na2HPO4, pH=7.4) containing sodium dodecyl sulfate (SDS) and increasing concentrations of Triton X100:
A. 400 ml. of PBS containing 1% Triton X-100 and 0.1% SDS.
B. 400 ml. of PBS containing 2% Triton X-100 and 0.1% SDS.
C. 400 ml. of PBS containing 3% Triton X-100 and 0.1% SDS.
Sample was treated by each solution during one day. At the end of the decellularization process samples were rinsed on the filter (400 ml FBS).
Histological examination of decellularized HLF samples
Samples of decellularized HLF were fixed in 10% formalin solution in PBS, rinsed for 15 minutes in running water and dehydrated in alcohols of increasing concentration ( 70%, 80 %, 96% for 5 minutes in each change of alcohol solution), and kept for 5-7 minutes in ethanol and chloroform mixture, then in pure chloroform, and were embedded in paraffin. 4-5 micron thick sections obtained by using the microtome Leica RM3255 were deparaffinized, rehydrated and stained with hematoxylin and eosin dye, as well as by Masson's method for the detection of connective-tissue elements. The analysis and photographing of the preparations obtained were performed using a Nikon eclipse microscope equipped with a digital camera.
Results and Discussion

Figure 1 presents the results of a comparative analysis of the influence of a preliminary mechanical grinding method and the stirring mode on the efficiency of decellularization of HLF and rinsing out cellular elements and detritus from HLF. The experiment was carried out on the liver fragments of the same donor.
As seen in Figure 1, after mechanical grinding of liver fragments with the homogenizer and their rinsing on the rotating system, the decellularized sample had a fibrous- granular structure with a predominance of fibrous component. A significant number of pyknotic nuclei and intact liver cells was found in the cells formed by the fibers of the stroma. After mechanical grinding by hand and subsequent decellularization with the use of rotation system, poorly rinsed areas of liver tissue were observed with high content of cell detritus and severe swelling of the stroma components.
As seen in Figure 1, the preservation of fibrous structure with a minimal amount of fragments of cell detritus was obtained by crushing the liver tissue by hand and rinsing using a magnetic stirrer in a cyclic mode (3 times per day for one hour). However, when staining for connective tissue (Masson's) the decellularized preparation revealed some ruptures of the collagen fibers of the stroma. In order to test the stirring algorithm during the decellularization process, the effect of stirring mode and process length on the efficiency of decellularization was studied.
Three modes were studied: (a) stirring once a day on a magnetic stirrer for 1 hour, the rest of the time (23 hours)-static rinse; (b) stirring twice a day on a magnetic stirrer for 1 hour, the rest of the time (22 hours)-static rinse; (c) stirring three times a day on a magnetic stirrer for 1 hour, the rest of the time (21 hours)-static rinse. The total length of treatment for each mode is 3 days. Figure 2 summarizes the results of the first experiment.
As seen in Figure 2, the longer HLF was rinsed on a magnetic stirrer, the more efficient was the decellularization process. On the first day of processing, the HLF samples, rinsed under conditions of single- or double stirring, had mostly a dense structure with a large number of surviving cells and a high content of cell detritus. In case of triple stirring, already after the first day of rinsing the fibrous component of the stroma in the sample became more pronounced. However, a denser area remained in the thickness of HLF with few whole cells with hyperchromatic nuclei and small fragments of cellular detritus. On the second day of rinsing all samples had predominantly loose, fibrous structure, however, the sample treated with a magnetic stirrer once a day revealed small denser (less rinsed) areas with high content of cellular detritus fragments. Best rinsed was the sample after three stirrings a day where only single cells in a state of degradation were detected (Figure 2). After the third day of decellularization it became apparent that a single stirring on a magnetic stirrer is clearly not sufficient for an efficient rinsing of HLF: in the samples, along with loose structures, the inclusions of dense, poorly rinsed areas were detected. Samples obtained with the use of double and triple treatment with a magnetic stirrer showed a fibrous-cellular structure of the stroma with a minimal content of cell detritus. The rinsing efficiency in case of triple stirring was slightly higher, however, numerous ruptures of collagen fibers were detected, which indicates a partial destruction of the unique matrix structure.
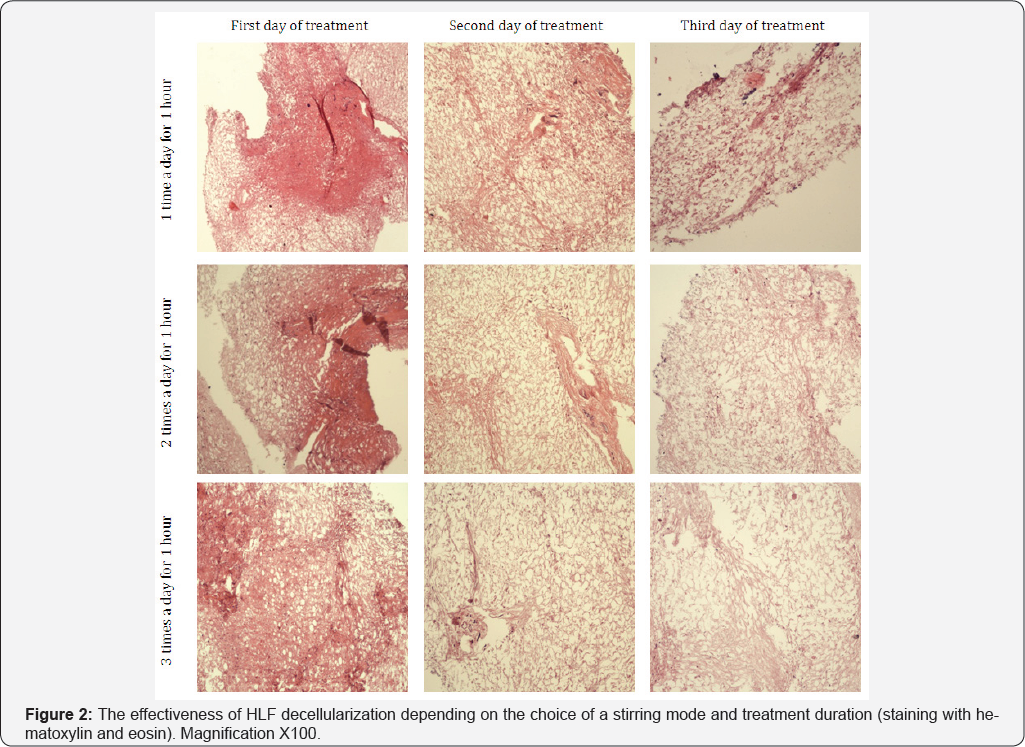
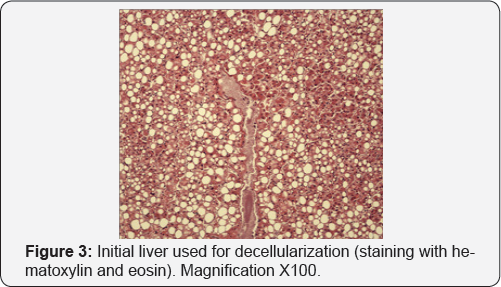
Therefore, the twice a day for 1 hour stirring mode by a magnetic stirrer is the best from the point of view of decellularization efficiency and preservation of the unique structure of the extacellular matrix. In the subsequent experiments it was shown that the optimal mode of rinsing depends on the condition of the donor liver, but the duration of decellularization should not exceed 3 days. However, it should be noted that in all HLF samples studied, decellularized per the chosen rinsing mode (three cycles with total duration of 1 day, in a solution of 0.1% SDS and Triton X-100 with increasing concentrations) the presence of pyknotic cell nuclei was observed. The histological picture of the initial liver source (Figure 3) shows that pyknotic nuclei were already present in the tissue and after all the processing modes remained intact and were not destroyed. Thus, the samples of donor liver with signs of karyopyknosis cannot be used as a source of nonimmunogenic tissue-specific liver matrix. Consequently, for decellularization it is necessary to use only the donor liver not containing pyknotic nuclei or find a method of their removal.
Based on the experimental data obtained, the process of HLF decellularization was optimized and the algorithm of a preliminary study of donor human liver was developed for the purpose of obtaining tissue-specific liver matrix, suitable for use as a carrier in tissue engineering.
The algorithm is as follows:
a. donor liver is examined for the presence of pyknotic hyperchromatic nuclei of hepatocytes and in case of their absence one proceeds to trial decellularization;
b. pilot fragments of liver tissue obtained by mechanical grinding are subjected to decellularization in solutions of 0.1% sodium salt of sodium dodecyl sulfate and Triton X-100 with increasing concentrations under the two modes of stirring on the magnetic stirrer: two or three times a day;
c. per the results of histological examination of the pilot decellularized samples obtained, the optimal stirring mode for this donor liver is selected.
Conclusion
The algorithm of evaluation of suitability of donor human liver for decellularization is suggested and the optimal conditions for the obtainment of decellularized liver fragments with preserved liver ECM structural properties and practically complete removal of cellular elements and detritus are found. However, there remains an issue of the removal of pyknotic nuclei during the process of decellularization of human liver fragments.
References
- Sevastianov VI (2014) Technologies of tissue engineering and regenerative medicine. Russian J of Transplantology and Artificial Organs 16(3): 93-108.
- Shagidulin M, Onishchenko N, Krasheninnikov M (2012) Treatment of chronic liver failure by transplantation of liver cells and bone marrow stem cells: 1 year experience. Proceeding of the European Society for Surgical Research, ESSR 2012, 47th Annual Congress, France, pp. 77-81.
- Morris AH, Stamer DK, Kyriakides TR (2017) The host response to naturally-derived extracellular matrix biomaterials. Semin Immunol pii: S1044-5323(16)30110-5.
- Sabetkish S, Kajbafzadeh AM, Sabetkish N, Khorramirouz R, Akbarzadeh A, et al. (2015) Whole-organ tissue engineering: decellularization and recellularization of three-dimensional matrix liver scaffolds. J Biomed Mater Res A 103(4): 1498-1508.
- Nari GA, Cid M, Comín R, Reyna L, Juri G, et al. (2013) Preparation of a three-dimensional extracellular matrix by decellularization of rabbit livers. Rev Esp Enferm Dig 105(3): 138-143.
- Bühler NE, Schulze Osthoff K, Königsrainer A, Schenk M (2015) Controlled processing of a full-sized porcine liver to a decellularized matrix in 24h. J Biosci Bioeng 119(5): 609-613.
- Zheng X, Xiang J, Wu W, Liu X, Liu W, et al. (2015) Preparation of a decellularized scaffold derived from human liver tissue. Nan Fang Yi Ke Da Xue Xue Bao 35(7): 1028-1033.






























