Histopathological Effect of Fasciolasis Infection in Goats Slaughtered in Three Abattoirs in Port Harcourt Metropolis, Rivers State, Nigeria
GN Wokem1, AE Abah2* and II Sounyo2
1Department of Medical Laboratory Sciences, Rivers State University of Science and Technology, Nigeria
2Department of Animal and Environmental Biology, University of Port Harcourt, Nigeria
Submission: August 03, 2017; Published: August 30, 2017
*Corresponding author: AE Abah, Department of Animal and Environmental Biology, Faculty of Science, University of Port Harcourt, PMB 5323 Port Harcourt 50001, Rivers State, Nigeria; Email: austin.abah@uniport.edu.ng
How to cite this article: GN Wokem, AE Abah, II Sounyo. Histopathological Effect of Fasciolasis Infection in Goats Slaughtered in Three Abattoirs in Port Harcourt Metropolis, Rivers State, Nigeria. Curr Trends 005 rends Biomedical Eng & Biosci. 2017; 8(4): 555742. DOI: 10.19080/CTBEB.2017.08.555742
Abstract
There is only scanty information about the histopathological effects of fasciolasis in goats in Nigeria and especially in the southern region. This study was aimed at establishing the status of fasciolasis infection and the histopathological effects in goats slaughtered in Port Harcourt metropolis. Liver samples were collected from randomly selected goats from three different Abattoirs namely; Trans-Amadi, Mile 3 and Rumuokwurusi and processed following standard method after ethical clearance. Out of 323 Livers Examined, 8 (2.48%) were found to be positive for fasciolasis. eight livers were found to be infected with Fasciolagigantica from random sampling during meat inspection. Trans-Amadi had 5 (slaughters average of 1500/daily), while 3 was from Mile 3 (slaughters average of 150/daily). Also one liver was found to be infected with tuberculosis from Trans-Amadi abattoir. Average liver weight was between 0.5-1.8 kg. Adult Fasciolagigantica were also recovered from the bile duct of the infected liver. Also in the gall bladder of infected liver were Fasciola ova recovered in cluster. Average measurement of adult Fasciola species ranges between 5.0 x 1.5cm and 3.3 x 0.9cm; length/width. The histological examination of livers infected with fasciolopsis (F gigantica) revealed histopathological effect such as necrosis of hepatocytes and occurrence of fibrosis were common observation in most of the Fasciola infected livers as against the normal architectural arrangement of the liver cells. Proliferation of bile ductules and expansion of the portal tracts by marked infilteration from chronic inflammatory cells. There were cases of dilation of the bile ducts filled with massive necrotic debris. The fluke infestation on liver lobes also showed diffuse vacuolations. These have adverse effect on the quality and quantity of what is available for eating and also on the nutritional status of the populace in the affected environment. There is also the danger of human infection since fasciolasis is a zoonotic infection.
Keywords: Fasciolasis; Prevalence; Histopathological effects; Abattoirs; Goats; Nigeria .
Introduction
Fascioliasis, a parasitic infection caused by the trematodes worms, Fasciola species, is distributed globally especially in ruminants-rearing region of the world [1]. Infection is endemic in Central and South America (especially Bolivia and Peru), Europe (especially Portugal, France, Spain, and Turkey), Asia (especially China, Vietnam, Taiwan, Korea, and Thailand), Africa, and the Middle East [2]. Scattered occurrences have also been reported in Australia and the United States of America [3,4]. One hundred per cent (100%) of animals living in endemic area can become harmfully or even fatally infected, especially sheep [5]. An estimated 2.4-17 million people are infected in about 51 countries, and 91 million globally are at risk of infection worldwide [6,7].
Majority of clinical knowledge about fascioliasis were established on the infections caused by F. hepatica though similar clinical features and principles also apply for F. gigantic [8]. High eosinophilia, above 68% has been reported in severe fascioliasis. Anaemia and leukocytosis may also be present [5]. Light infections due to F. hepatica may be asymptomatic though they may present with symptoms such as emesis, coughing, headache, perspiration, fever, diarrhoea and anaemia. Fasciola species have been reported to elicit pharyngitis in people who eat raw infected livers from goats and sheep as observed in Lebanon, Armenia, and the Middle East [5,8].
Howlader and Huq [9], in their study on the histologic alteration of the liver of goats infected with Fasciolagigantica reported that all the studied samples suffered from chronic infection, with the presence of flukes surrounded by capsules in the liver. The proliferation of the fibrous tissues led to the thickening of blood vessels around them. Infilteration of lymphocytes in the lobules, occurrence of eosinophils and neutrophils in patches were found in the liver tissue. Also observed were that the bile ductules in the hepatic trinity clogged by the flukes proliferated. Chiezey et al. [10] in their study on Yankasa ewe reported that at post-morterm hepatic fibrosis of the liver with intra-hepatic haemorrhage was observed. Also liver from F gigantica infected Yankasa ewes at 12 weeks had distended gall bladder and enlarged hepatic lymph node.
Arjmand et al. [11] in their study onprevalence, macroscopic and microscopic lesions of Bovine fasciolosis at Ahvaz abattoir, Khozestan Province, Iran reported histopathological changes in infected livers to be chronic portal hepatitis (14.91%), eosinophilic hepatitis (21.1%), bile duct hyperplasia (14.91%), and mild hepatitis (9.64%) respectively. There is only scanty information about the histopathological effects of fasciolasis in goat in Nigeria and especially in the southern region. Abraham and Jude [12] in their study on fascioliasis in cattle and goat slaughtered in Calabar abattoir, Nigeria reported that 2-7% of the infected goat liver (out of a total of 350 goats) studied were rejected and discarded. This was as a result of damaged parenchyma leading to severe haemorrhage, thickening and grosses fibrosis of bile duct. They also observed dislodged hepatic cells that became wandering cells among macrophages within the sinusoid. This study was undertaken to increase the baseline data in the south-south and was aimed at establishing the histopathological effects of fasciolasis in goats slaughtered in Port Harcourt metropolis.
Material and Methods
Study area
There are many abattoirs in Port Harcourt City, Rivers State, but most of them are not officially recognized by Rivers State Government. However, Trans-Amadi abattoir which was chosen as one of the study areas is among the officially recognized ones in the state. Slaughtering of goat is done in the early morning of each day. Trans-Amadi abattoir is the main abattoir in the city of Port Harcourt; it is located in Trans-Amadi Industrial Area. Trans- Amadi lies at 4°48'53" N latitude and 7°2'14" E longitude (Figure 1), is a thousand-hectare industrial area situated in an area that supports astrong manufacturing sector and is considered to be a major industrial zone in Obio-Akpor local Government Area, Rivers State [13]. Trans-Amadi abattoir is the largest abattoir in the Niger Delta. The animals are killed in thousands on daily basis and sold to inhabitants of Rivers State and the rest of the neighbouring states. Rumuokwurusi lies at 4°51’6”N, 7°2'653"E, latitude and longitude. It is located on the outskirt of Port Harcourt, close to Port Harcourt-Aba expressway and Rivers State College of Art and Science. Mile 3 lies at 4°47'24”N latitude and longitude 6°59'36”E. It is one of the three extensions of Diobu. It is a densely populated neighbourhood of Port Harcourt, Rivers State located within Port Harcourt metropolis. This neighbourhood is rank among the most commercially lively places in the city. Rumuokwurusi and Mile 3 abattoirs are not as large as the Trans-Amadi abattoir. All the locations were motorable and easily accessible by goat consumers and traders who buy and sell goat products (Source URL: http://www. worldofmatter.net/trans-amadi-slaughterhouse accessed 15th Sept 2016).
Study population
The study population were male and female goats from the Trans-Amadi, Rumuokwurusi and Mile 3 abattoirs all in ObioAkpor Local Government Area, Rivers State situated in the southern part of Nigeria. The goats were brought mainly from the northern part of Nigeria (Kano and Nassarawa states), as well as from Chad Republic. The samples were selected based on random sampling, a total of 323 goats were sampled.
Sample size determination and design
The sample size for the study was determined by taking the estimated prevalence of 30% fasciolosis using the formula given by Cochran in 1963 [14]. They were 323 goats investigated in this present study.
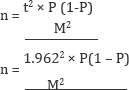
Where n = required sample size
t = confidence level of 95% (standard value of 1.96)
P = estimated prevalence = 30%
M= Margin of error @ 5% (standard value of 0.05)
Hence, M2 = 0.05 and p = 0.3 (30%). 323 animals were sampled.
Ethical permit
Ethical clearance was obtained from the Research Ethics Committee of the University of Port Harcourt. Consent was also sought from the Veterinary Section, Ministry of Agriculture, Rivers State, before samples were collected.
Sample collection
The abattoirs were visited between 7am to 9am thrice weekly (Mondays, Wednesdays and Fridays). Samples of faeces, gall bladder and liver were collected from randomly selected goats in each of the abattoirs. Fresh faecal materials were collected directly from the rectum of the slaughtered animals using a pair of sterile hand gloves. The samples were placed in clean labelled bottles containing 10% formalin and transferred to the parasitology laboratory of Animal and Environmental Biology, University of Port Harcourt for examination for eggs of F. gigantica and F.hepatica. The abattoirs were visited in the early hours of the day when the animals were always slaughtered. The sex and sources of each randomly selected goat were noted and recorded. Adult flukes seen were removed, measured and recorded. The content of the gall bladder were emptied into a clear sterile bottle containing 10% formol saline, and later examined for various stages and species of Fasciola..
Identification of fasciola eggs
The Fasciolaova were identified with their characterized large, oval and yellowish colour with a distinct flat operculum were seen as described by [15-17]. Also, species identification of the recovered Fasciola from post mortem specimen of the livers collected was based on the morphological features of the parasites [15-17].
Laboratory examination of samples
Formol-ether concentration technique for faecal samples [18]: All the faecal samples were concentrated using Formol-Ether concentration technique, four milliliter of a well- mixed stool sample was put in a tube containing 4 ml of 10% formalin. The suspension was sieved using a coffee strainer into a centrifuge tube. The filtrate was centrifuged at 2,000 rpm for 2 mins. The supernatant was discarded and the sediment resuspended in 10 ml of physiological saline. The sediment was suspended in 7 ml of formol saline, after which 3 ml of ether was added. The tube was closed with a stopper and shaken vigorously. The stopper was removed and the tube centrifuged at 2,000 rpm for 2 mins. Four layers became visible: the top layer of ether, a second layer of plugs of debris, a third layer of formalin and a fourth layer of sediment. The plug of debris was detached from the side of the tube with the aid of a glass rod and the liquid was discarded leaving a small amount of formal saline for re- suspending the sediment. A little was transferred to a clean glass slide at a time, covered with a cover slip and examined under the microscope at x10 and x40 objectives respectively, to view for eggs and this was repeated until the whole sediment was examined.
Histological examination of the liver
Post-mortem examinations of the liver and gall bladder samples were carried out in the Parasitological Laboratory of University of Port Harcourt for adult flukes. The infected livers were weighed, and then placed on a clear board and dissected in-situ there after a reasonable portion of each liver sample was cut and send to the Histology laboratories of the University of Port Harcourt and Teaching Hospital to determine the histopathological effects of fascioliases in goats. The liver tissue cut off was processed and stained with Haematoxylin and Eosin technique (H & E) [19] as follows.
Histological Steps
Involves Processing-dehydration, clearing, and infiltration; Embedding, Microtomy or Sectioning, Staining and Microscopy were carried out.
Haematoxylin and Eosin staining technique (H and E)
Haematoxylin stained the nucleus violet or blue and the 1% eosin stained the cytoplasm pink. Before the sections were stained, the paraffin wax had to be removed to make the stain permeable to the tissue. Thus the process of staining a paraffin section with H& E was as follows:
The slides were deparaffinised in xylene for 5 minutes after which they were immersed in absolute alcohol for 1 minute. They were also immersed in 90% and 70% alcohol for 30 seconds respectively. They were washed and rinsed in water. After which the sections were stained. Slides were stained with Haematoxylin for 15 minutes. The slides were rinsed under tap running water for 5 minutes till colour of the section became blue (blueing). Slides were differentiated in 1% acid alcohol for 5 seconds and washed with squashed water. Slides were stained with 1% aqueous eosin for 1 minute. The slides were rinsed in water for few seconds. The slides were dehydrated using 70%, 90% and absolute alcohol and then cleared in xylene for 1 minute. The slides were cleaned, blotted and mounted in Distyrene Plasticizer Xylene (DPX). The slides were viewed under the microscope using x40 objective and the observations noted.
Data analysis
Data obtained were statistically analysed using SPSS version 2010 and imported into the statistical package for social sciences (SPSS) ver. 21 for data analysis. Categorical data was presented in the form of frequencies and percentages (%). Chi-square test was used to test for association or differences between categorical variables. A p-value of ≤ 0.05 was considered statistically significant. The descriptive statistics was generated in simple proportions and some of the results presented in tables.
Percentage based prevalence was calculated using the formula:
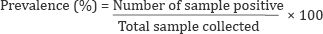
Results
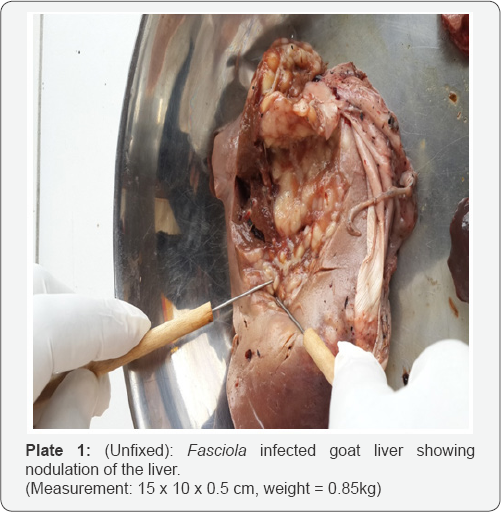
Eight livers were found to be infected with Fasciolagigantica from random sampling during meat inspection (Plate 1). Trans- Amadi had 5 (slaughters average of 1500/daily), while 3 was from Mile 3 (slaughters average of 150/daily). Also one liver was found to be infected with tuberculosis from Trans-Amadi abattoir. Average liver weight was between 0.5-1.8 kg. Adult Fasciolagigantica were also recovered from the bile duct of the infected liver. Also in the gall bladder of infected liver were Fasciola ova recovered in cluster. Average measurement of adult Fasciola species ranges between 5.0 x 1.5cm and 3.3 x 0.9cm; length/width (Plate 2).

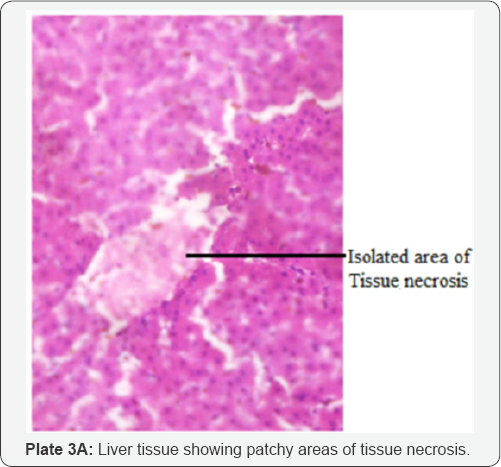
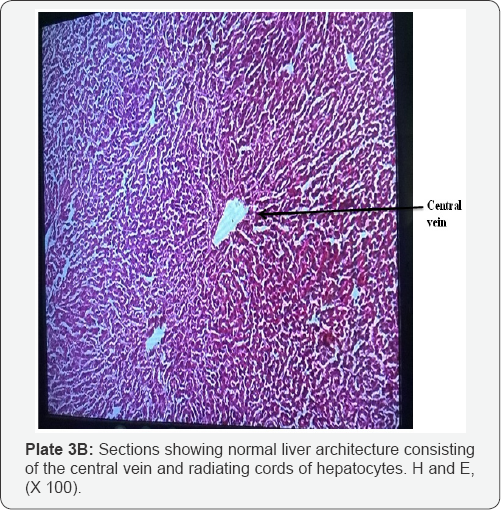
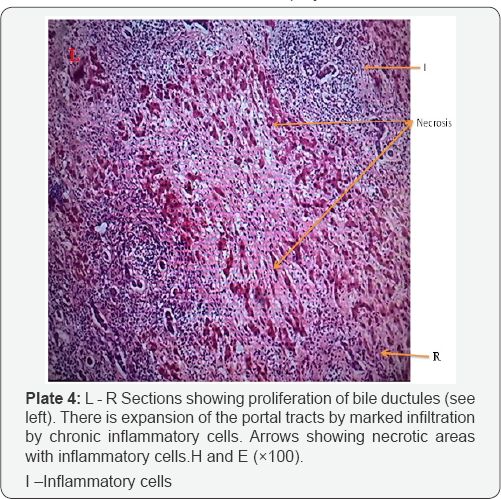
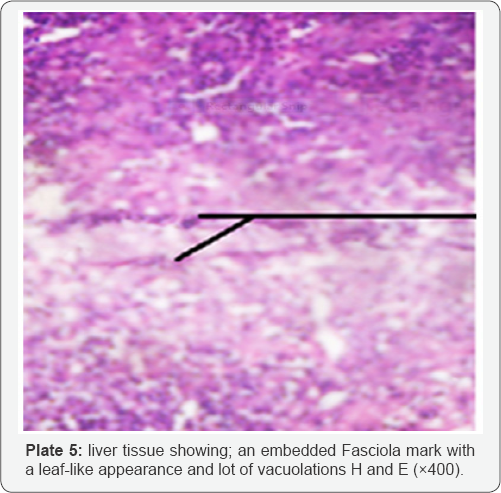
The histological examination of livers infected with fasciolopsis (F. gigantica) revealed histopathological effect such as necrosis of hepatocytes and occurrence of fibrosis were common observation in most of the Fasciola infected livers (Plate 3A) as against the normal architectural arrangement of the liver cells shown in (plates 3B). Proliferation of bile ductules and expansion of the portal tracts by marked infilteration from chronic inflammatory cells (Plate 4). There were cases of dilation of the bile ducts filled with massive necrotic debris. The fluke infestation on liver lobes also showed diffuse vacuolations (plate 5)
Discussion
The observed histological effects of necrosis of hepatocytes and occurrence of fibrosis, proliferation of bile ductules as well as marked infilteration from chronic inflammatory cells seen in most of the Fasciola infected livers were in line with similar studies. Chiezey et al. [10] in their study on Yankasa ewe reported on post-mortem hepatic fibrosis of the liver. These observations agreed with the works of Arjmand et al. [11] who also reported chronic portal hepatitis in infected livers in bovine fasciolosis at Ahvaz abattoir in Iran, and Howlader & Huq [9] who reported on proliferation of fibrous tissue and infilteration of cells. The histological effect is also similar to that of Abraham & Jude [12] in their study on fascioliasis in cattle and goat slaughtered in Calabar abattoir, Nigeria who reported damaged parenchyma leading to severe haemorrhage, thickening and grosses fibrosis of bile duct [12]. These observations buttress the fact that necrotic hepatitis or black diseases are essentially intoxication as a result of exotoxins and injury to the liver due to presence of flukes. More so, it has been established that proliferation of bile ductules is a hepatic cellular reaction observed in most form of liver diseases associated with liver injury.
These histopathological effects on the infested livers may result in liver dysfunction, anaemia, hepatic haemorrhage, abortion and sometimes death if the animals are not treated [8,10,11]. Liver tissue is known to be a rich source of nutrients including protein, iron and some important vitamins such as A, D, K, and E and minerals. This is often recommended for pregnant women, lactating mothers, patients who are recovering from various illnesses both young and adult, as well as for prevention and treatment of anaemia. In situations where a large quantity of liver tissues are condemned as a result of fascioliosis leading to reduction of available liver, it will consequently result in increase in price and lead to competition [20]. These will adversely affect the quality and quantity of what is available for eating hence the nutritional status of the populace in the affected environment; particularly in Nigeria where most people hardily meet the minimal protein nutritional requirement. There is also the danger of human infection since fasciolopsis is a zoonotic infection and one of the neglected tropical diseases.
References
- Leder K, Weilder PF, Baron EL (2015) Liver flukes: Fascioliasis solutions. Wolters Kluwer Health Clinical Solutions.
- Mahanty S, Maclean JD, Cross J (2011) Liver, Lung, and Intestinal Fluke Infections. Guerrant RL, Walker DH, Weller PF (Eds.), In: Tropical Infectious Diseases: Principles, Pathogens and Practice, (3ri edn), Philadelphia, USA, p. 854.
- Sivagnanam S, Van der Poorten D, Douglas MW (2014) Hepatic lesions and eosinophilia in an urban dweller. Liver International 34(4): 643.
- Weisenberg SA, Perlada DE (2013) Domestically acquired in fascioliasis in northern californa. Am J Trop Med Hyg 89(3): 588-591.
- Junquera P (2015) Liver fluke, Fasciola hepatica, parasite of cattle, sheep, Goats, Horses, Dogs, and Cats. Biology, prevention and control. Parasitipedia.net.
- Mas Coma S, Valero MA, Bargues MD (2009) Fasciola, lymnaeids and human fascioliasis, with a global overview on disease transmission, epidemiology, evolutionary genetics, molecular epidemiology and control. Adv Parasitol 69: 41-146.
- Keiser J, Utzinger J (2009) Food-borne trematodisses. Clin Microbiol Rev 22(3): 466-483.
- CDC (2014) Fasciola-Resources for Health Professionals. Global Health-Division of Parasitic Diseases and Malaria. pp. 30329-34027.
- Howlader MMR, Huq MM (1997) Histological alterations in the bengal goats infected with Fasciolagigantica. Asian-Australasian Journal of Animal Sciences 10(1): 114-117.
- Chiezey NP, Adama JY, Ajanus J, Lawal I (2013) Disruption of estrus and conception in the acute phase of Fasciolagigantica infections in Yankasa ewes. Journal of Veterinary Medicine and Animal Health 5(8): 206-214.
- Arjmand J, Hajipour N, Khojasteh SMB, Baran AI, Mirshekar F, et al. (2015) Prevalence, Macroscopic and Microscopic lesions of bovine. Fasciolosis at Ahvaz Abattoir, Khozestan Province, Iran. Biological Forum-An International Journal, 7(1): 539-545.
- Abraham JT, Jude IB (2014) Fascioliasis in cattle and goat slaughtered in Calabarabattoir. Journal of Biology, Agriculture and Healthcare 4(18): 2224-3204.
- Encyclopedia Britannica (2014) Port Harcourt (Nigeria). Retrieved.
- Israel GD (1992) Sampling the evidence of extension program impact. Program Evaluation and Organizational Development, IFAS, University of Florida, PEOD. p. 5.
- WHO (1995) Control of foodborne trematode infections: report of a WHO Study Group. World Health Organ Tech Rep Ser 849: 1-157.
- Cheesbrough M (2005) Parasitological testing, In, District Laboratory Practice in Tropical countries, Part 1 2nd edition. Cambridge University press, England, USA, pp. 196-198.
- Ngele KK, Ibe E (2014) Prevalence of fasciolopsis in cattle slaughtered at Eke market abattoir, Afikpo, Ebonyi State, Nigeria. Animal Research International 11(2):1958-1963.
- Arora DR, Brij BA (2010) Medical Parasitology, (3rd edn), CBS Publishers and Distributors Pvt Ltd., USA, pp. 158-159.
- Singh SP (2010) Manual of Practical Histology. (Second Reprint) AAU Vital Book Co. Ariaria, Aba, Nigeria, pp. 3-7.
- Okoli IC, Opara MN, Iheukwumere FC, Herbert U (2006) Analysis of abattoir records for Imo State, Nigeria from 1995-1999 IV: Diseases of incidences in cattle, sheep and goats. Journal of Agriculture and Social Research (JASR) 6: 2.






























