- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Novel Biocompatible Ultrananocrystalline Diamond (UNCD) Coating Technology for a New Generation of Medical Implants, Devices, and Scaffolds for Developmental Biology
Orlando Auciello*
Departments of Materials Science and Bioengineering, University of Texas at Dallas, USA
Submission: July 11, 2017; Published: August 30, 2017
*Corresponding author: O Auciello, Distinguished Endowed Chair Professor, Departments of Materials Science and Bioengineering, University of Texas at Dallas, USA, Email: orlando.auciello@utdallas.edu
How to cite this article: Orlando A. Novel Biocompatible Ultrananocrystalline Diamond (UNCD) Coating Technology for a New Generation of Medical Implants, Devices, and Scaffolds for Developmental Biology. Curr Trends Biomedical Eng & Biosci. 2017; 8(3): 555740. DOI 10.19080/CTBEB.2017.08.555740.
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Abstract
A new biomaterial paradigm for a new generation of implantable medical devices and prostheses, and for scaffold for stem cells growth and differentiation for developmental biology, is provided by a novel multifunctional/biocompatible/body fluid corrosion-resistant ultrananocrystalline diamond (UNCD) film developed and patented in recent years. The UNCD coating provides a unique combination of multi functionalities, including biocompatibility, bioinertnes to chemical attack by body fluids, and surface chemistry based on Carbon (C) atoms (element of life], to enable a new generation of implantable medical devices and prostheses, and scaffolds for tissue engineering, to impact the quality of life of people.
This article include the following topics:
Brief description of the synthesis and properties of UNCD films and comparison with other diamond film technologies.
Descriptions of key medical devices and prostheses under development, based on UNCD-coated base materials, and demonstration of UNCD as an outstanding scaffold for growing stem cells and induce differentiation for developmental biology, to treat specific medical conditions requiring effective therapies, namely:
UNCD-coated microchip (artificial retina) implantable inside the eye on the retina to restore partial vision to people blind by retinitis picmentosa and macular degeneration produced by genetically-induced degeneration of the retina photoreceptors (The Argus II device, developed by an interdisciplinary team of researchers and doctors from universities, national laboratories and Second Sight, is currently implanted commercially in the USA and Europe, and it was selected by TIME magazine as one of the top 25 inventions for 2013).
UNCD-coated intraocular device for treatment of glaucoma (intraocular pressure associated optical neuropathy that could lead to blindness].
UNCD-coated metal dental implants with order of magnitude longer life and superior performance than current implants.
UNCD-coated prostheses (hips, knees and many other metal-based implants); new generation of Li-ion batteries (LIBs) for defibrillator/ pacemakers, with ≥10 x longer life and safer than current LIBs.
Keywords: Ultrananocrystalline diamond (UNCD); Coating; Encapsulated Si chip (artificial retina); Coted dental implants; Glaucoma drainage devices; Prostheses
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Introduction
The novel ultrananocrystalline diamond (UNCD] coating technology, developed by the author of this article and many colleagues (see acknowledgements] [1], and patented (see selected patents, of seventeen, in references [2-5]], in recent years, is providing a new paradigm material for a new generation of implantable medical devices, prostheses, and new scaffolds for stem cell growth and differentiation for developmental biology. It is important to point out that the UNCD coating technology is already in the market, commercialized by Advanced Diamond Technologies (www.thindiamond.com), a company co-founded by O. Auciello and J. A. Carlisle in 2003, profitable in 2014).
Products in the market include:
Low manufacturing cost/high performing UNCD coatings for mechanical pumps seals and bearings, with order of magnitude lower coefficient of friction (0.020.04) than any other material (≥0.3) previously used, thus providing up to 20% savings in cost of energy of running the mechanical pumps, because much less torque needed.
Electrically conductive Boron-doped UNCD coating to cover metal electrodes from corrosion in a new generation of water purification systems for domestic and industrial uses, based on electrolysis, to create ozone that kills all bacteria in the water and destroy any other contaminant (www.thindiamond.com). In the field of application to medical technologies, the UNCD coating exhibits a unique combination of multi functionalities and properties, including biocompatibility, bio-inertness to chemical attack by body fluids and unique surface chemistry, enabling new generations of implantable prostheses, medical devices and scaffolds for tissue engineering.
Biomedical products under development include:
UNCD-coated microchip, implantable inside the human eye, to restore partial vision to people blind by retinitis picmentosa, produced by genetically-induced death of retina photoreceptors (1st generation device (2009 R & D and Editors Choice Award); the first generation device (Argus II) without the UNCD coating yet, is currently marketed by Second Sight (CA, USA) restoring partial vision to blind people (selected as a top invention in 2013 by TIME magazine).
UNCD-coated intraocular device for treatment of glaucoma;
UNCD-coated metal dental, hips, knees and other prostheses with ≥10x longer life and superior performance than current implants, Li-ion batteries with electrically conductive UNCD-coated electrodes and membranes and inner walls of metal LIB's cases, providing ≥10x longer life and safer, for new defibrillator/pacemakers with order of magnitudes longer life without changing batteries, s it occurs with these devices powered by current LIBs.
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
Several types of diamond thin films have been and are currently being grown on surfaces of insulators, semiconductors and metals based materials used in many devices and components oftechnological products, and the properties studied systematically. The diamond films produced until now exhibit different microstructure, surface morphology, and properties. Diamond films are grown, using mainly microwave plasma enhanced chemical vapor deposition (MPCVD] [1-6] or hot filament chemical vapor deposition (HFCVD) processes [7-9]. A conventional diamond film growth process involves "seeding" the surface of the substrate with micro or nanocrystalline diamond particles by immersing the substrate in a solution of methanol and diamond particles in a sonicator, which sounds waves, shake the particles and embed them on the surface as "seeds" to nucleate and induce the growth of diamond film. For the MPCVD method, a mixture of gases, containing CH4 molecules, as the main species, is inserted in an air evacuated chamber and microwave power coupled to the gas to produce a plasma involving ionized and neutral atoms and molecules containing C, H and other components such as Ar [1-6]. For the HFCVD process an array of hot filaments (~2200 °C) induces cracking of CH4 molecules impacting on the hot surface, producing radicals that induce growth of diamond films [7-9]. Use of a hydrogen-rich chemistry [H2 (balance)/CH4 (0.1 to 4%]] [1,6] results in microcrystalline diamond (MCD] (1-5μm grains and columnar microstructure] (for~1% CH4] and nanocrystalline diamond (NCD] (10-100nm grains) (for up to~4% CH4) films. The H2/CH4 chemistry-based growth process is driven by CHx (x=2-3] radical's interaction with the substrate surface, involving hydrogen abstraction process, ultimately resulting in carbon atoms bonding into positions corresponding to the diamond lattice. a graphitic phase that codeposits with the diamond phase is etched by chemical reaction of the carbon sp2 bound atoms with atomic hydrogen in the plasma, which also etches the diamond phase, although at ~ 50 times lower rate than for graphite, which results in formation of inter-granular voids and columnar large grains (≥1μm). The MCD films exhibit high compressive stress, poor inter-granular adhesion, and rough surfaces (Figure 1a), which are not suitable for coating prostheses, requiring smooth (Figure 1b) low friction coating surfaces, as provided by the UNCD coating (Figure 1c) [1]. Therefore, MCD films are not well-suited for application to the medical devices and implants discussed in this article (read details in Ref. [1]). Diamond films grain size can be reduced to 10-100nm, characteristic of NCD films, by increasing the CH4/ H2 ratio in the plasma, resulting in relatively smooth NCD films (Figure 1d), [1,6] but with increased non-diamond components in the grain boundaries.

Figure 1 shows clearly why UNCD coatings are far superior than MCD and NCD coatings for prostheses and other implantable devices, and even far superior than DLC films, currently used as coating for some prostheses.
Differently to MCD and NCD films, the UNCD films discussed in this article are produced by microwave MPCVD [1] and HFCVD, using a novel (patented [2-5]] argon-rich chemistry [Ar (99%]/ CH4(1%]], [1] which produces carbon dimers (C2) and CHx radicals in the plasma, from methane decomposition, via 2CH4 ⊠ C2H2 +3H2 and C2H2 ⊠ C2 + H2 chemical reactions.
Initial experimental and theoretical work indicated that C2 dimers play a critical role in the UNCD film nucleation and growth [1,10]. Calculations predicted that the C2 dimers have low activation energy (~6kcal/mol) for insertion into the substrate surface, thus establishing the nucleation characteristics of UNCD films. Recent modeling [11] suggested that while the C2 population in the plasma is high, the population near the surface may be low, and other hydrocarbon radicals (e.g., CH3, C2H2] are also substantial or the main contributors to the UNCD film growth [11]. However, this model did not fully explain the low temperature growth (≤400 °C) of UNCD films. Clearly, further experimental and theoretical work is needed. In any case, the uniqueness of UNCD films is that they exhibit a nanostructure with the smallest grain size observed today (Figure 1a) for diamond films. It is also relevant that insertion of nitrogen in the gas mixture (e.g, Ar (79sccm)/CH4 (1sccm]/N2 (20sccm] results in UNCD films, named N-UNCD, with ~10nm grain size and ~2nm wide gran boundaries [2,12], which exhibit good electrical conductivity, via nitrogen incorporation into the grain boundaries, satisfying dangling C bonds, and providing electrons for electrical conduction.
A critical outcome of the UNCD nucleation and growth processes is that it produces diamond films at the lowest temperatures (350-400 °C) demonstrated today. This unique property of UNCD coatings enables encapsulation of Silicon (Si]-based microchips for implantation in the human body, as demonstrated by the UNCD coating encapsulation of a Si microchip implantable inside the eye [13], as part of an artificial retina to restore partial vision to people blind by retinitis picmentosa. Low temperature UNCD film growth on CMOS devices may provide the bases for monolithically integrated UNCD-MEMS/NEMS/CMOS devices [14] for biosensors and MEMS-drug deliver devices implantable in the human body.
In relation to the synthesis of UNCD films, a process named bias enhanced nucleation/bias enhanced growth (BEN/BEG) was demonstrated in recent years for the MPCVD process [1], and for the HFCVD process as well [7,8]. The latter being scaled up now to cover large areas [15] for commercialization of the process. The BEN-BEG process involves biasing the substrate with a negative voltage (—150-300V) to accelerate C+ and CHx+ ions from the plasma towards the substrate's surface, where they get implanted producing a carbide layer, which nucleates and induce grow UNCD films [15]. BEN/BEG provides several advantages over conventional UNCD growth with chemical diamond seeding, as discussed above, namely:
Comparable or better seeding efficiency.
Stronger adhesion to substrates, due to the energetic ions sub-plantation in the sub-surface region and
An integrated fully dry nucleation/growth process using plasmas only. BEN/BEG processes, using H2/CH4 chemistries produced NCD (30-100nm grains) films. However, this process resulted n the formation of diamond clusters, relatively high surface roughness, high compressive stress, film delamination, and high non-diamond phase content [16]. More recently, our group developed a low pressure MPCVD BEN/BEG process, using the Ar/CH4 chemistry, which yielded UNCD films with the UNCD nanostructure see Figure 1a, low stress (~80-100MPa), smooth surfaces (rms ~4-6nm), and higher growth rates (~ 1μm/hr) [1,17,18] than for UNCD films grown without bias (~02-0.3μm/h) [1]. BEN/BEG UNCD films may provide the most appropriate process for coating of dental and other prosthesis implants, as discussed in this article.
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Properties of UNCD Films Relevant to Medical Implants and Devices
Properties of UNCD films, relevant to the development of the medical implants and devices discussed in this article include:
Hardness (98GPa) and Young's modulus (980GPa) [1] close to corresponding values for single crystal diamond (100GPa and 1200GPa, respectively).
The lowest coefficient of friction (0.02-0.04) [1] of any biocompatible coating known today.
High fracture strength (~ 5.3GPa) [1].
Relatively high electrical conductivity by incorporation of N atoms in the grain boundaries (N-UNCD] [1]. With respect to this property, Garret et al. [19] recently demonstrated excellent performance of N-UNCD coated metallic electrodes for neural stimulation. An alternative process that was developed to produce electrically conductive UNCD coatings is by inserting B atoms substituting C atoms in the diamond lattice and supplying electrons to the conduction band, to produce what is known now as electrically conductive B-UNCD films [20].
Extreme resistance to chemical attack by body fluids, as demonstrated by UNCD-coated Si microchip implanted inside rabbit eyes as main component of an artificial retina to restore partial sight to people blind by genetically-induced degeneration of photoreceptors [13].
Tailoring of UNCD surface chemistry to use UNCD films as scaffolds for efficient stem cell growth and differentiation for developmental biology [21] in this sense, the demonstration that biological cells can grow efficiently on the surface of UNCD coatings [21] may induce enhanced grafting of UNCD-coated dental and other prostheses, via bone cell growth onto the UNCD surface of the implant, currently under investigation by our group.
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
Devices for restoring vision to blind people
Background: Human vision results from photons penetrating the eye and exciting the retina photoreceptors. The photonic excitation transforms into electrical charges, which excite the bipolar cells connected to the photoreceptors. The bipolar cells operate amplifying the electrical pulses produced by the carriers. These charges are then injected into the ganglion cells, which transmit the charges through their axons, forming the optical nerve, to the brain, where the images are finally formed.
R&D performed during several recent years focused on developing implantable devices for neural stimulation and hybrid bionic systems to restore lost human motor and sensory functions. First generation of neural prostheses are being used to restore hearing and sight, via cochlear implants [22] and artificial retina [23,24] (the Argus II device, marketed by Second Sight, is the first device worldwide being implanted in people blind by retinitis picmentosa, who regain partial vision], respectively.
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
Background on technologies for vision restoration
Several vision restoration technologies have been and are being explored. A review of those technologies can be seen in a recent article by Auciello et al. [25] and will not be repeated here. The focus of this part of the article is on briefly describing the R&D performed to develop the Argus II Retinal Prosthesis [26] . This device involves a Si-base microchip that in the final rendition should be implanted inside the eye on the ganglion cell layer (see schematic in 2 left side], receive images, wireless, from a camera on glasses and inject processed electrical pulses to the ganglion cells through a large electrode array, finally transmitted to the brain via the ganglion cell axons bundle (optical nerve] [26]. The Argus II device was developed by a team of researchers from Universities, National Laboratories, and Second Sight (company currently commercializing the device, http://www.secondsight.com), during a ten years Department of Energy (USA]-funded project. The Argus II is currently the most advanced artificial retina device [26], and the only one currently implanted commercially in USA and Europe to restore partial sight to blind people (The Argus II device was named one of the top 25 inventions for 2013 by TIME magazine -November 25, 2013].
Coatings technologies for encapsulation of implantable microchips
Silicon (Si]-based microchips implanted inside the eye, should be encapsulated with a hermetic/bioinert/ biocompatible coating to inhibit chemical destructive attack of Si by the eye's humour. The hermetic coatings should have the double functionality of protecting the implantable device and the surrounding tissues, to enable long service time devices, free of electronic failure. A packaging technology (titanium based hard shell] is already used in the the commercial Argus II device located outside the eye inside the shell and connected to the retina though polymer encapsulated Pt wires, to transmit charges from the microchip to the electrodes in contact with the ganglion cells on the retina]. The best approach would be to have a coating encapsulating the Si-microchip, protecting it from chemical attach from the eye humor, to implant the chip fully inside the eye. The coatings should be hermetic and biocompatible, because the Si CMOS device performance can be affected by sodium ion incorporation from the eye's saline humour, and Si can be attached chemically leading to destruction [27].
Coating materials that have been evaluated as encapsulating coatings include SiO2 [28], SiC [29], polytetrafluoroethylene and polyimide [30], and parylene [30].
There are several problems with these coatings, namely:
a) SiO2 coatings exhibited dissolution and decay when implanted in animals up to six months [27].
b) Polyimide and other polymers are inexpensive and flexible, but absorb significant quantities of water, resulting in electrical leakage, and are not hermetic, enabling chemical attack and eventual destruction of the encapsulated chip.
UNCD coating technology for encapsulation of implantable microchips
Contrary to the drawbacks of the coatings described above, UNCD coatings exhibit a unique combination of properties described above, and, for the encapsulation of artificial retina Si microchip, the UNCD coatings are the only diamond-based coatings that have been demonstrated to be grown at ~400 °C (non-destructive thermal budget temperature) on CMOS devices (Figure 2a in [25] and Figure 2 (left) in this article), without any degradation [1], Extensive in vivo implantation of UNCD-coated Si chips in rabbit eyes for long periods of time (up to three years) (Figure 2b in [25] ), followed by scanning electron microscopy studies (Figure 2c-2d in [25]) and X-ray photoelectron spectroscopy (XPS) (Figure 2e in [25]), for surface chemical analysis, showed that the UNCD coating provides the encapsulating coating that may enable long term (years] implantation of a Si chip inside the eye without any degradation [9].
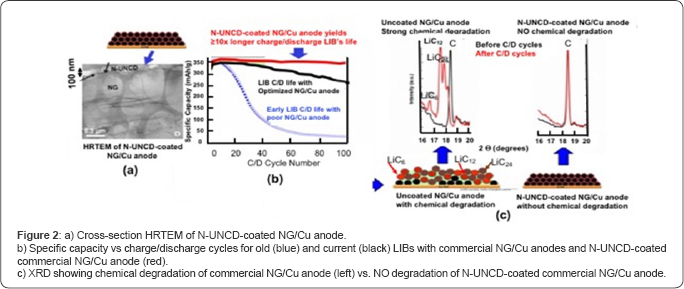
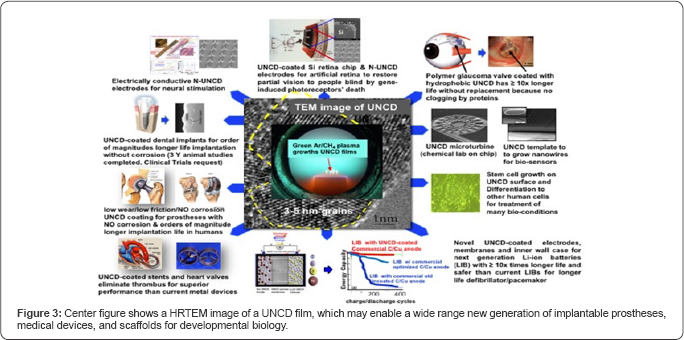
Details of the R&D performed to develop UNCD films as bioinert/biocompatible coatings for encapsulation of the artificial retina microchip can be found in Ref. [13]. On the other hand, details on the Argus II device development are found in References [26] and [31]. The Argus II device features the microchip encapsulated in a biocompatible metallic box located outside the eye and connected, via polymer encapsulated Pt wires, to Pt electrodes on a polymer support matrix implanted on the retina's ganglion cell layer. When approval from regulatory agencies is obtained to insert UNCD in the eye, the UNCD-encapsulated microchip could be implanted inside the eye, receiving images wireless from the CCD camera, thus, eliminating undesirable hard wire connection to the retina through the eye’s wall [26,31] (Figure 3).
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
Background
Glaucoma is the condition related to the clogging of the tubes in the trabecular mesh of the eye, which continuously evacuate the aqueous humour form the inner region of the eye towards the outside to keep the inner eye pressure constant to avoid increased intra-ocular pressure (IOP), which can destroy the optical nerve, leading to blindness (Figure 4 in ref. 25].
Other causes of glaucoma, although less common, include blunt/chemical injury in the eye, eye infection, clogging of blood vessels or eye inflammation, and eye surgery done to correct some other eye condition. Glaucoma generally occurs in both eyes, although each is affected differently. Glaucoma is the second leading cause of blindness after cataracts. Glaucoma affects one in 200 people aged 50 and younger, and one in 10 over the age of 80. Early detection of glaucoma arrest or slow progression, via pharmacological and surgical means. Further details about glaucoma can be found in Ref. [32].
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
Current commercial glaucoma treatment devices inserted in the eye to drain the aqueous humour are based mainly in polymers and are rather large (~1.7 x 1cm), and the implant surgery requires two incisions, one to insert the body of the valve in the eye sclera and the other to insert the polymer tube inside the eye to drain the fluid (Figure 3 & 5a in ref [25]).
A problem, not yet solved, with current glaucoma treatment devices is the eye's reaction against the implant due to protein layer adhesion to the hydrophilic surface of the polymer, resulting in inflammation in short time. Macrophages are attracted, by chemical signals, to the implant surface, where they release growth factors that stimulate growth of fibroblast cells that yield collagen production that result in a fibrous capsule development around the implant. Subsequently, complete fibrosis occurs on the surface of the valve, isolating the implant from the rest of the body. This fibrous capsule induces materials degradation effects, not only on glaucoma valves discussed here, but also on other medical implants, resulting in degraded sensor performance or transport of drugs in drug delivery devices. Several approaches have been investigated to overcome these problems, but with not proven success.
Thus, this section focuses on presenting a brief description of animals studies done in a joint collaboration between researchers at the University of Texas-Dallas and the University of Buenos Aires and Hospital Austral in Buenos Aires, Argentina, directed at investigating the effectiveness of a UNCD coating in eliminating the development of fibrotic capsules on the surface of commercial glaucoma silicone-based valves, which over time degrade the performance of the valve to drain the eye's humour. The animal studies were performed following NIH and ANMAT guidelines (Argentina FDA) for animal care. The studies involved using two rabbits, each receiving one uncoated drainage device (Figure 5b in ref. [25]) on one eye, and one coated with a low temperature (~ 400 °C) UNCD film (Figures 5c in ref [25]) on the other eye, in order to avoid ambiguities from using different animals in one experiment. The uncoated valve developed fibrosis, due to eye proteins adhesion, 24hrs. after implantation, while the UNCD-coated device remained clear even after several months of implantation (Figure 5c in [25]) and Figure 2 in this article-top right), thus demonstrating the power of UNCD films as bio-inert/biocompatible encapsulating coatings, to improve by orders of magnitude the lifetime of the implanted glaucoma valve. Approvals for clinical trials are needed and our group is talking to a company manufacturing these valves to explore interest in heling to request clinical trial approval.
The elimination of protein adhesion on the surface of the UNCD-coated drainage device is attributed to the fact that the surface of UNCD films is extremely hydrophobic (no water adhesion), due to the hydrogen termination of the surface of the as-deposited UNCD films [1].
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
Background
Pure Ti and Ti-alloys are widely used in dental and orthopedic implants, because past R&D indicated that Ti-based implants exhibit suitable mechanical properties and biocompatibility [33]. However, recent R&D on, and use of Ti-based implants in patients indicates that they exhibit failures, specifically of osseointegration and corrosion induce by chemical attack by body fluids [34].
Currently, worldwide statistic indicate that about 5-10% Ti- based dental implants fail in shorter times than desired, due to mouth fluids-induced Ti electrochemical corrosion [35], which release metallic particles (mainly TiO2, since Ti oxidizes readily when exposed to air before the prostheses is implanted inside the human body) from the Ti surface into the local tissue [36], resulting in elevated levels of metals released from implants, which induce potential long-term systemic toxicity [37]. The corrosion of Ti occurs when the natural oxide passivation layer, formed by atmospheric exposure, is removed or has been partially formed only, and metallic Ti is exposed to corrosive body fluids. The metallic implant acts as an in vivo electrode leading to electrochemical degradation of the implant’s surface [35,37], weakening the implant/bone interface, thus the attachment to the bone [37]. In addition, TiO2 particles released from the implant can migrate to distant sites producing additional biological deleterious effects [38].
UNCD-coated metallic implants exhibit extreme resistance to chemical attack by body fluids [1,12]. In addition, roughening of the Ti implant surface by micro-machining, followed by coating with UNCD films, produces a chemically resistant microroughened surface, which can enhance osseointegration, as demonstrated in R&D for roughened Ti implant surfaces [39]. Carbon based materials, including diamond, have emerged as attractive coatings for biomedical implants, due to good tissue compatibility, resistance to chemical attack by body fluids, radiation resistance (suitable for sterilization processes), hemocompatibility, low friction coefficient, negligible wear, and good adhesion to Ti. In addition, recent work demonstrated that NCD coatings exhibit increased resistance to bacterial adhesion compared to stainless steel and Ti [40].
Synthesis and characterization of uncd-coated dental implants
Ti laminar implants were used as samples for in vivo tests in the School of Dentistry, University of Buenos Aires, namely:
a) Uncoated Ti.
b) UNCD-coated Ti, and
c) UNCD/W-coated Ti, where the W interface layer was grown by magnetron sputter-deposition and the UNCD coatings were grown using the MPCVD and HFCVD metho [1].
Biological studies of dental implants with and without UNCD coating
Animal studies were performed using Male Wistar rats to investigate the peri-implant reparation process. Guidelines from NIH and the Ethics regulations of the Faculty of Dentistry, University of Buenos Aires, for surgical procedures and care of laboratory animals, were observed, as done in recent work.
The histological analysis of the samples from the control and experimental groups revealed areas of lamellar bone in close contact with the surface (osseointegration) (see Figure 7b in Ref. [25]) and areas of bone marrow in contact with the implant surface (myelointegration). These studies have shown that UNCD has good biocompatibility as a coating material for dental implants.
Among other factors, the clinical success of implants depends on osseointegration of the implant, and if coated, on the coating/ substrate reliability. Histological studies indicated that UNCD- coated dental implants achieved excellent osseointegration, without exhibiting any inflammatory reactions (Figure 7b in Ref. [25]). In addition, more recently, studies on chemical corrosion of UNCD-coated dental implants [41], as potentially induced by saliva, demonstrated that the surface of the UNCD coating is order of magnitude superior to bare Ti-based implants, and would resist corrosion by saliva [41].
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
Background
Lithium-ion batteries (LIBs) are in the market today powering electric vehicles and electronic devices (e.g., cell phones, laptop computers, and many other electronic systems). However, the application relevant to this article is LIBs powering of medical devices, and particularly defibrillator/pacemakers to protect people with weak heart conditions from life threatening events, due to heart failure. In spite of being in the market, LIBs still have shortcomings, and do not meet all requirements for an energy storage device for future high performance electric vehicles [42] or electronic or medical devices. LIBs require mayor improvements in electrode materials to minimize or eliminate the capacity energy degradation vs charge/discharge cycles, due to chemical attack by Li-ions. Various anode materials exhibit excellent reversible insertion of lithium [43], via intercalation in layered carbons (e.g., graphite), adsorption in hard carbons surface layers, and binding on hydrogen atoms in carbons containing hydrogen [44]. Mesocarbon Microbeads (MCMB) are in standard anode material for LIBs due to their high theoretical capacity (372mAh/g based on LiC6) and low potential profile (0-0.3V VS. Li/Li+). Natural graphite (NG) has emerged as a strong candidate anode material to replace (MCMB), because the NG high Coulombic efficiency and electronic conductivity (0.4-2.5x104Scm-1 on the basal plane), low volume expansion after whole lithiation (~10%), and low cost (≤US$10/Kg) [45]. However, large irreversible capacity loss and short charging/discharging cycling life limit the use of NG electrodes in LIBs. Chemical reactivity of Li-ion-inserted NG with organic electrolytes needs to be eliminated to produce efficient NG-based LIB anodes. Reactions of Li-ion-inserted NG with electrolytes form local solid-electrolyte-interphase (SEI) films of increasing thickness and electrical resistance, generating gases (e.g., hydrogen, CO, CO2, methane, ethylene, and propylene within interlayers of NG crystals, and resulting in irreversible capacity loss and damage to the NG structure (Figure 2b), reducing battery life.
Properties of the SEI layer depend on the electrolyte compositions, microstructures and surface characteristics of the NG anode. The SEI films are formed when negative electrodes are polarized to low potentials in non-aqueous electrolytes, containing Li salts, resulting in the reduction of electrolyte species to insoluble salts. The films block electron transfer between electrodes and electrolyte, but conduct Li ion, as explained by the SEI model. However, the SEI passivation films crack due to the volume change of the graphite anode and expose fresh surface of electrodes, which is repaired by further graphite anode surface-electrolyte reaction, resulting in continuous SEI films formation and Li-ions deficiency, causing the battery capacity to fade. The process described above becomes worse on electrodes where the local charging and discharging current density is higher than average. In addition to the SEI films, dendritic growth of compound structures occurs in highly reactive graphite electrode areas, resulting in electrical shorting and hazardous failure of the LIB. The LIB anode problems described above limit increase of LIB market share for electrical vehicles, requiring inexpensive LIBs with NG anodes exhibiting long-life and high-rate charging/discharging cycle performance.
Recent R&D by Auciello/Tzeng and colleagues [46,47], demonstrated that by coating the NG/Cu commercial anodes of LIBs with electrically conductive N-UNCD, the chemical degradation of the NG layer, due to Li-NG chemical reaction is practically eliminated (Figure 2c right) and there is not degradation in the specific energy vs. charge /discharge cycles (Figure 2b-red curve).
The data shown in Figure 2 indicates N-UNCD-coated anodes can enable the next generation of LIBs with >10x longer life and safer to enable the next generation of defibrillator/pacemaker with orders of magnitude longer life that may not require change in many more years than current LIBs require. Further R&D is underway to also develop N-UNCD-based coatings for cathodes and insulating UNCD coatings for membranes and inner walls of LIBs’ cases [48].
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Summary View of the Multiple Applications of the Biocompatible UNCD Coating
Technology
A summary of the multiple applications of the novel UNCD coating, to enable the next generation of implantable prostheses and other medical devices, and a new generation of scaffolds with superior surface chemistry for stem cell growth and differentiation for developmental biology, is presented in (Figure 3).
Summary
The discussion of the fundamental and applied materials science and implant and device development and testing for some key medical implant and devices based on UNCD coatings shows that this material provides a unique combination of mechanical, tribological, electrical, chemical and biological properties, which may enable a new generation of medical implants and devices with superior performance with respect to those currently based on other materials.
In addition, the work described in this article also shows that UNCD coatings provide superior scaffolds for stem cell growth and differentiation. Work is in progress to develop and introduce all products into the market.
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
Acknowledgement
Work performed at the University of Texas-Dallas was supported by the Endowed Chair funds of Prof. O. Auciello (#37139010).
Work related to the artificial retina project was funded by the DOE-Artificial Retina Project, and Auciello acknowledges the outstanding collaboration with all team members, and especially Dr. M. Humayun (Director of the artificial retina project), and P. Gurman and J.A. Carlisle for contributions to the work on UNCD coating of the Si- microchip.
Auciello acknowledges the strong contributions of Drs. M. Saravia (Hospital Austral-Argentina), P. Gurman (UTD), and A. Berra (Universidad de Buenos Aires-Argentina) on the R&D related to the development of UNCD-coated glaucoma valves.
Auciello acknowledges the outstanding contributions of Profs. M. Guglielmotti, D. Olmedo, D. Tasat. M. Bruno, M. Domingo, M. Paparella, and P. Evelson (University of Buenos Aires (UBA)-Argentina), and P. Gurman and K. Kang (UTD) for the work on UNCD-coated dental implants (work in UBA- Argentina was funded by grants: UBACyT 20020100100812 (UBA); PIP 11220090100117 (CONICET) and PDTS PO 01 2013 (UBA)-Ministry of Science, Technology and Productive Innovation, Argentina.
Auciello acknowledges the outstanding contributions of Prof. T. TZeng, Y-W. Cheng, C-K. Lin, and Y.-C. Chu (NCKU-Taiwan), A. Abouimrane, Z. Chen, Y. Ren, and C.-P.
- Mini Review
- Abstract
- Introduction
- Synthesis and Structure of Biocompatible UNCD Films and Comparison with Other Diamond Films
- Properties of UNCD Films Relevant to Medical Implants and Devices
- UNCD Coatings on Prostheses and Implantable Medical Devices: Comparison with Alternative Biocompatible Coatings
- UNCD-encapsulated Silicon Microchip: Main Component of Artificial Retina to Restore Partial Vision to Blind People
- Application of UNCD Coatings to Cover Surfaces of Drainage Valves for Treatment of Glaucoma
- Hydrophobic UNCD Films as Hermetic Bio-inert/ Biocompatible Coatings for a New Generation of Long Life Glaucoma Treatment Devices
- Application of Mouth Fluids Corrosion-Resistant UNCD Coatings to Develop a New Generation of Dental Implants with Superior Performance
- Corrosion-Resistant Electrically Conductive N-UNCD-Coated Electrodes and Insulating UNCD- Coated Membranes and Inner Walls of Cases for a New Generation of Li-Ion Batteries for Longer Life Implantable Defibrillator/Pacemaker
- Summary View of the Multiple Applications of the Biocompatible UNCD Coating Technology
- Acknowledgement
- References
References
- Auciello O, Sumant AV (2010) Status review of the science and technology of ultrananocrystalline diamond (UNCD™) films and application to multifunctional devices. Diamond & Related Materials 19(7-9): 699-718.
- Gruen DM, Krauss AR, Auciello O, Carlisle JA (2004) N-Type Doping of NCD Films with Nitrogen and Electrodes Made There from.
- Naguib N, Birrell J, Elam J, Carlisle JA, Auciello O (2006) A Method to Grow Carbon Thin Films Consisting Entirely of Diamond Grains 3-5nm in Size and High-Energy Grain Boundaries.
- Carlisle JA, Gruen DM, Auciello O, Xiao X (2009) A Method to Grow Pure Nanocrystalline Diamond Films at Low Temperatures and High Deposition Rates.
- Tzeng Y, Auciello O, Liu CP, Lin CK, Cheng YW (2015) Nanocrystalline- Diamond/ Carbon and Nanocrystalline-Diamond/Silicon Composite Electrodes for Li-Based Batteries..
- Butler JE, Sumant AV (2008) The CVD of Nanodiamond Materials. Chemical Vapor Deposition 14(7-8): 145-160.
- Janischowsky K, Ebert W, Kohn E (2003) Bias enhanced nucleation of diamond on silicon (100) in a HFCVD system Diamond and Related Materials 12(3-7): 336-339.
- Ansari SG, Anh TL, Seo HK, Sung KG, Mushtaq D, et al. (2004) Growth kinetics of diamond film with bias enhanced nucleation and H2/CH4/Ar mixture in a hot-filament chemical vapor deposition system. Journal of Crystal Growth 265(3-4): 563-570.
- Peña JJA, Montes J, Jimenez MJA, Mendoza DB, García R, et al. (2016) Low temperature hot filament chemical vapor deposition of Ultrananocrystalline Diamond films with tunable sheet resistance for electronic power devices. Diamond and Related Materials 69: 207-213.
- Sternberg M, Zapol P, Curtiss LA (2003) Carbon dimers on the diamond (100) surface: Growth and nucleation. Physical Rev B 68(20): 205330.
- May PPW, Allan NL, Ashfold MNR, Richley JC, Mankelevich YA (2009) Simplified Monte Carlo simulations of chemical vapour deposition diamond growth. Journal of Physics: Condensed Matter 21(36): 364203.
- Xiao X, Birrell J, Gerbi JE, Auciello O, Carlisle JA (2004) Low temperature growth of ultrananocrystalline diamond. Journal of Applied Physics 96(4): 2232.
- Xiao X, Wang J, Carlisle JA, Mech B, Greenberg R, et al. (2006) In vitro and in vivo evaluation of ultrananocrystalline diamond for coating of implantable retinal microchips. J Biomedical Materials 77B(2): 273281.
- Sumant V, Auciello O, Carpick RW, Srinivasan S, Butler JE (2010) Ultrananocrystalline and Nanocrystalline Diamond Thin Films for MEMS/NEMS Applications. MRS Bulletin 35(4): 281-288.
- Peña JJA, de Obaldia E, Gutierrez JM, Kang K, Jimenez MJA, et al. (2017) Diamond and Related Materials (in press, July 2017).
- Stoner BR, Ma GH, Wolter SD, Glass JT (1992) Characterization of bias- enhanced nucleation of diamond on silicon by invacuo surface analysis and transmission electron microscopy. Phys Rev B 45(19): 11067.
- Lee YC, Lin SJ, Chia CT, Cheng HF, Lin IN (2005) Effect of processing parameters on the nucleation behavior of nano-crystalline diamond film. Diamond and Related Materials 14(3-7): 296-301.
- Chen YC, Zhong XY, Konicek AR, Grierson DS, Tai NH, et al. (2008) Synthesis and characterization of smooth ultrananocrystalline diamond films via low pressure bias-enhanced nucleation and growth. Appl Phys Lett 92: 133113.
- Garrett DJ, Ganesan K, Stacey A, Fox K, Meffin H, et al. (2012) Ultra-nanocrystalline diamond electrodes: optimization towards neural stimulation applications. J Neural Eng 9(1).
- Zhanga J, Zimmer JW, Howe RT, Maboudian R (2008) Characterization of boron-doped micro- and nanocrystalline diamond films deposited by wafer-scale hot filament chemical vapor deposition for MEMS applications. Diamond and Related Materials 17(1): 23-28.
- Shi B, Jin Q, Chen L, Auciello O (2009) Fundamentals of ultrananocrystalline diamond (UNCD) thin films as biomaterials for developmental biology: Embryonic fibroblasts growth on the surface of (UNCD) films. Diamond and Related Materials 18(2-3): 596-600.
- House WF (1976) Cochlear implants. Ann Otol Rhinol Laryngol 85 suppl 27(3 Pt 2): 1-93.
- Weiland JD, Liu W, Humayun MS (2005) Retinal prosthesis. Annu Rev Biomed Eng 7: 361-401.
- http://www.eurekanetwork.org/project/-/id/5558
- Auciello O, Gurman P, Guglielmotti MB, Olmedo DG, Berra A, et al. (2014) Biocompatible ultrananocrystalline diamond coatings for implantable medical devices. MRS Bulletin 39(7): 621-629.
- Humayun M, Dorn JD, da Cruz L, Dagnelie G, Sahel JA (2012) Interim Results from the International Trial of Second Sight’s Visual Prosthesis. Ophthalmology 119(4): 779-788.
- Hammerle H, Kobuch K, Kohler K, Nisch W, Sachs H, et al. (2002) Biostability of micro-photodiode arrays for subretinal implantation. Biomaterials 23(3): 797-804.
- Meyer J (2001) Retina implant-a bioMEMS challenge. Sensor and Actuator A 97-98: 1-9.
- Cogan SF, Edell DJ, Guzelian AA, Ping Liu Y, Edell R (2003) J Biomedical Mater Res A 67(3): 856-867.
- Seo J, Kim S, Chung H, Kim H, Yu H, et al. (2004) Mater Sci Engin C 24 185.
- Zhou DD, Greenbaum E (2010) Implantable Neural Prostheses. In: Zhou DD, Greenbaum E (Eds.), Biological and Medical Physics. Biomedical Engineering, Vol 1 & 2, Springer, Germany
- Auciello O, Gurman P, Berra A, Zaravia M, Zysler R (2013) Ch 6 In: Roger Narayan (Ed.), Diamond Based Materials for Biomedical Applications. Woodhead Publishing Ltd, UK, p. 151.
- Gepreel MAH, Niinomi M (2013) Biocompatibility of Ti-alloys for longterm implantation. J Mech Behav Biomed Mater 20: 407-415.
- Palmquist A, Omar OM, Esposito M, Lausmaa J, Thomsen P (2010) Titanium oral implants: surface characteristics, interface biology and clinical outcome. J R Soc Interface 7 Suppl 5: S515-S527.
- Gittens RA, Navarrete RO, Tannenbaum R, Boyan BD, Schwartz Z(2011) Electrical implications of corrosion for osseointegration of titanium implants. J Dent Res 90(12): 1389-1397.
- Olmedo DG, Cabrini RL, Duffo G, Guglielmotti MB (2008) Local effect of titanium implant corrosion: an experimental study in rats. Int J Oral Maxillofac Surg 37(11): 1032-1038.
- (2012) Olmedo DG, Tasat D, Duffó G, Cabrini RL, Guglielmotti MB, p. 94.
- Olmedo DG, Paparella ML, Spielberg M, Brandizzi D, Guglielmotti MB, et al. (2012) Oral mucosa tissue response to titanium cover screws. J Periodontol 83(8): 973-980.
- Hacking SA, Boyraz P, Powers BM, Sen-Gupta E, Kucharski W, et al. (2012) Surface roughness enhances the osseointegration of titanium headposts in non-human primates. J Neurosci Methods 211(2): 237244.
- Amaral M, Gomes PS, Lopes MA, Santos J, Silva R (2008) Nanocrystalline Diamond as a Coating for Joint Implants: Cytotoxicity and Biocompatibility Assessment. J Nanomater p. 9.
- Patel B, Martinez ACD, Gurman P, Auciello O, Barao V, et al. (2017) Surface Innovations 5(2): 106.
- Chiang YM (2010) Materials science. Building a better battery. Science 330(6010): 1485-1486.
- Magasinski A, Dixon P, Hertzberg B, Kvit A, Ayala J, et al. (2010) High- performance lithium-ion anodes using a hierarchical bottom-up approach. Nature Materials 9: 353-358.
- Dahn JR, Zheng T, Liu Y, Xue JS (1995) Mechanisms for Lithium Insertion in Carbonaceous Materials. Science 270(5236): 590-593.
- Shim J, Striebel KA (2004) The dependence of natural graphite anode performance on electrode density. Journal of Power Sources 130(1-2): 247-253.
- Tzeng Y, Auciello O, Liu CP, Lin CK, Cheng YW (2015) Nanocrystalline- Diamond/Carbon and Nanocrystalline Diamond/Silicon Composite Electrodes for Li-Based Batteries.
- Cheng YW, Lin CK, Chu YC, Abouimrane A, Chen Z, et al. (2014) Electrically Conductive Ultrananocrystalline Diamond-Coated Natural Graphite-Copper Anode for New Long Life Lithium-Ion Battery. Advanced Materials 26(22): 3724-3729.
- www.nexeon.co.uk






