Modulation of Radio Response by an Isoquinoline Alkaloid Berberine in Cultured Hela Cells Exposed to Different Doses of γ -Radiation Radiosensitizing Activity of Berberine in Hela Cells
Ganesh Chandra Jagetia1* and SK Rao2
1Department of Zoology, Mizoram University, India
2Department of Pharmacognosy, C. U. Shah College of Pharmacy & Research, India
Submission: June 14, 2017; Published: August 28, 2017
*Corresponding author: Ganesh Chandra Jagetia, Department of Zoology, Mizoram University, Aizwal-796004, India, Tel:913892330724/2330227; Email: gc.jagetia@gmail.com
How to cite this article: Ganesh C J, SK R. Modulation of Radio Response by an Isoquinoline Alkaloid Berberine in Cultured Hela Cells Exposed to Different Doses of γ -Radiation Radiosensitizing Activity of Berberine in Hela Cells. Curr Trends Biomedical Eng & Biosci. 2017; 8(1): 555726. DOI: 10.19080/CTBEB.2017.08.555726.
Abstract
The radiomodulatory effect of berberine chloride (BCL) was studied in cultured HeLa cells exposed to γ-rays, where optimum treatment duration of BCL for its radiosensitizing activity was determined by treating HeLa cells with BCL for different times before exposure to 2Gy γ-rays. Exposure of HeLa cells to BCL for 4h before exposure to 2Gy, caused a significant decrease in the cell viability (50%) and surviving fraction (0.42) after 4h treatmetn with 4μg/ml BCL. The radiosensitizing activity was studied by treating HeLa cells for 4h with diffrent cocentrations of BCL before expsoure to 0, 0.25, 0.5, 1, 2, 3, or 4Gy γ-rays. Exposure of HeLa cells to different doses of γ-rays resulted in a dose dependent decline in the viability of HeLa cells, while treatment of HeLa cells with various concentrations of BCL further decreased the cell viability and clonogneic potenial of cells depending not only on the irradiation dose but also BCL concentration dependent manner. The lowest cell viability and clonogenicity was observed for 4μg/ml BCL for all exposure doses. The analysis of lactate dehydrogenase release in HeLa cells before exposure to 3Gy irradiation at various post-irradiation times, showed a time dependent elevation in its release up to 12h post-irradiation. Our study demonstrates that BCl treatment increased the effect of γ-rays and this may be due to membrane damage as indicated by lactate dehydrogenase release.
Keywords: Berberine; HeLa cells; Survival; Radiation; Lactate dehydrogenase
Introduction
Ionizing radiations are a used regulary to treat various tumors since the discovery of X-rays by Wilhelm Conrad Röntgen in the year 1895 and approximately 40% of patients are cured by radiotherapy and 50% of the patients receive radiotherapy during their disease [1,2]. It is used to treat tumors or as an adjuvant or neoadjuvant therapy and in several instances it is the only modality to treat tumors [3]. It is also used as a palliative therapy if the treatment is impossible [4,5]. Despite the success of radiotherapy its full potential remains underutilized due to the presence of hypoxic fraction of cells in the tumor and also due to the fact that with the increase in the radiation dose the normal tissue radiotoxicity increases [6]. The normal tissue toxicity can be alleviated and the tumor response to radiotherapy may be increased by combining certain drugs with radiotherapy.
There has been an enthusiasm initially to combine hypoxic cell sensitizers with radiotherapy due to its success in preclinical models however; the clinical success has been limited. This has led to development of alternative approach, where chemotherapeutic drugs were combined with radiotherapy. This approach met with a remarkable success in clinical situation to treat different types of malignant tumors [7,8]. The modern chemoptherapeutic drugs such as 5-fluorouracil, cis-dichlorodiammine-platinum (II), crytophycins, mitomycin C, docetaxel, paclitaxel, camptothecin, irinotecan, topotecan, combretastatin A-4, and gemcitabine have been used successfully combined with radiotherapy to treat several neoplasia [8-16]. The chemoradiotherapy has been beneficial in the treatment of solid tumors in many randomized clinical trials, however, the main drawback has been increased high toxicity and development of second malignancies [17,18]. This indicates the need for newer paradigms to reduce the toxic side effects of combination treatment regimens, and extract optimum therapeutic benefits to the patients with good quality of life.
Berberine containing plants are used medicinally in virtually all traditional medical systems, and have a history of long usage in Ayurvedic and Chinese systems of medicine, dating back at least 3,000 years. Berberine is an isoquinoline alkaloid present in a number of clinically important medicinal plants, including Hydrastis canadensis (goldenseal), Coptis chinensis (coptis or golden thread), Berberis aquifolium (Oregon grape), Berberis aristata (tree turmeric) and Tinospora cordifolia (giloy). Many useful activities of berberine including antibacterial, antifungal, anticancer antidiarrheal, antioxidant, anti-inflammatory, antiarrhythmic, antihypertensive, chemosensitizing and hepatoprotective have been reported [19-21]. Berberine has been found to have conducive effect on memory impairment in rats and also block inflammatory colitis in mice [22,23]. Berberine induced apoptosis by activation of Bax and caspase 3 and inhibited the growth of prostate cancer cells in vitro in an earlier study [24]. Berberine afforded protection against Alzheimer's disease, diabetes, diarrhea, hyperlipidemia, fatty liver disease, gastroenteritis, coronary artery diseases, hypertension, obesity, metabolic syndrome, and polycystic ovary [23,25]. Clinically it has been found to improve cardiac performance in patients with heart failure and exert a direct depressive action on myocardial vasculature and smooth musculature [26]. The administration of 1.2 to 2g of berberine daily in the patients suffering from chronic congestive heart failure has been reported to improve left ventricular ejection fraction and ventricular premature complexes [27]. Clinically berberine treatment has been reported to be effective in type 2 diabetes in humans [28-30]. Berberine has been reported to be clinically effective in the treatment of Behcet's disease, dementia dyslipidemia, non-fatty liver disease, hyperlipidemia, and ocular [31-36]. Berberine has been reported to trigger molecular damage into the DNA of cultured HeLa cells and also augment the effect of radiation [37,38]. Therefore, the present study was conceptualized to obtain an insight into the radiomodulatory activity of berberine chloride in cultured HeLa cells.
Materials and Methods
Preparation of drug
Berberine chloride (BCL) was dissolved in sterile double distilled water (MEM) at a concentration of 5mg/ml and diluted in MEM in such a way so as to obtain the required concentrations. All drug solutions were prepared afresh immediately before use.
Cell line and culture
HeLa S3 cells (doubling time of 20±2h) were supplied by National Centre for Cell Science, Pune, India. The cells were usually grown in 25cm2 culture flasks (Techno Plastic Products, Trasadingën, Switzerland) in Eagle’s minimum essential medium (MEM) supplemented with 10% fetal calf serum, 1% L-glutamine and 50μg/ml gentamicin sulfate at 37 °C in an atmosphere of 5% CO2 in 95% humidified air in a CO2 incubator (NuAir, Plymouth, USA) with loosened caps.
Experimental design
A fixed number (5X105) of exponentially growing cells were seeded into several culture flasks (Techno Plastic Products, Trasadingën, Switzerland) and divided into the following groups according to the treatment they received:
MEM+irradiation: The cells of this group were cultured in minimum essential medium and exposed to different doses of γ-rays.
BCL+irradiation: This group of cell cultures was treated with different concentrations of BCL before exposure to various doses of γ-rays.
Irradiation
The 25cm2 flask containing cells were exposed to 0.0, 0.25, 0. 5, 1, 2, 3 or 4Gy of Cobalt-60 γ-rays from a Telecobalt therapy source (Theratron Atomic Energy Agency, Ontario, Canada). The cell culture flasks were irradiated at a dose of lGy/minute at a SSD of 91cm (midpoint of the flask) at room temperature.
Determination of optimum time
The optimum time of BCL treatment or radiosensitization of HeLa cells was determined by performing the following experiments, where grouping and other conditions were essentially similar except that the cells were exposed to 2Gy γ-rays after treating the cells with different concentration of BCL.
Pratt and Willis assay
The optimum duration for drug exposure before irradiation was evaluated by Pratt and Willis assay [39] in HeLa cells. Briefly, 1X105 HeLa cells were inoculated into 25cm2 culture flasks (Cellstar, Greiner, Germany) and grown for 24h. Thereafter, 1, 2 or 4μg/ml BCL was added and cells were incubated for 0, 1, 2, 4 or 6h at 37 °C and were exposed to 2Gy γ-radiation. The drug- containing medium was replaced with a fresh drug-free medium and transferred to CO2 incubator. After 72h of drug inoculation, the cells were harvested, stained with trypan blue and the cells were counted using a hemocytometer (Labovert microscope, Ernst Leitz, Wetzlar GmbH, Germany). The cells incorporating the dye were considered as dead cells whereas those excluding the trypan blue were considered viable. The results obtained were further confirmed by clonogenic assay.
Clonogenic assay
The grouping and other conditions were similar to that described above except that the clonogenicity of cells was estimated by colony forming assay [40], where 200 cells were plated on to several individual culture dishes (Cellstar, Greiner, Germany) containing 5ml drug free medium in triplicate for each drug dose for each group. The cells were exposed to 1, 2 or 4μg/ ml BCL for 0, 1, 2, 4 or 6h, respectively before exposure to 2Gy γ-rays. Immediately after irradiation the cells were transferred back to the CO2 incubator and were allowed to grow for 11 days to form colonies. The resultant colonies were stained with 1% crystal violet in methanol and clusters containing 50 or more cells were scored as a colony. The plating efficiency of cells was determined and the surviving fraction calculated.
Determination of radiosensitizing activity
A separate experiment was carried out to determine the radiosensitizing activity of BCL, where the cells were treated with 0, 1, 2 or 4μg/ml BCL for 4 h before exposure to 0, 0.25, 0.5,1,2, 3 or 4Gy of γ-rays as described above within five minutes of irradiation, the medium containing the drug was removed from each culture flask of each group and the cells were washed twice with sterile PBS. The cells from each group of flasks were dislodged by trypsin EDTA treatment and divided into two parts.
Pratt and Willis assay
One part of the cells i.e. 1x105 were inoculated into several culture flasks in triplicate and the cytotoxicity of various treatments was determined by Pratt and Wills test [39] as described above.
Clonogenic assay
The results obtained from Pratt and Willis assay were confirmed by clonogenic assay as described above, where the cells left after Pratt and Wills assay were inoculated into several petridishes in triplicate for each doses of radiation for each group and left undisturbed for 11 days for colony formation. The colony thus formed were counted. The plating efficiency of cells was determined and the surviving fraction was fitted on to a linear quadratic model SF=exp (αD+βD2).
Biochemical analysis
In a separate experiment, effect of 0, 1, 2, or 4μg/ml BCL was studied on the lactate dehydrogenase (LDH) enzyme release in the medium. The grouping and other conditions were similar to that described in the experimental section. The cells were treated with different concentrations of BCL for 4h and exposed to 3Gy. The LDH was estimated at 0, 0.5, 1, 2, 4, 8 and 12h postirradiation.
Estimation of lactate dehydrogenase activity
The estimation of LDH release in the culture medium of above groups was carried out by the earlier described method with minor modifications [41]. The whole medium from each cell culture of each group was removed and collected separately immediately after irradiation (within 5min after irradiation) and was considered 0h after treatment. The cells were fed with a fresh 5ml medium and the above procedure (removal of media) was successively repeated at each assay period (i.e., 0.5, 1, 2, 4, 8 and 12h) until the termination of the experiment. Briefly, the tubes containing media were centrifuged and 50μl of the medium was transferred into the individual tubes containing Tris-EDTA-NADH buffer followed by 10 min incubation at 37 °C and the addition of pyruvate solution. The absorbance was read at 339nm in a UV-Vis spectrophotometer (UV-260, Shimadzu Corp, Tokyo, Japan) and the data have been expressed as units/ litre (U/L).
Statistical analysis
The statistical analyses were performed using GraphPad Prism 5 statistical software (GraphPad Software, San Diego, CA, USA). The significance among all groups was determined by one-way ANOVA and Bonferroni's post-hoc test was applied for multiple comparisons. The experiments were repeated for confirmation of results. The results are the average of five individual experiments. The test of homogeneity was applied to find out variation among each experiment. The data of each experiment did not differ significantly from one another and hence, all the values have been combined and means calculated. A p value of <0.05 was considered statistically significant.
Results
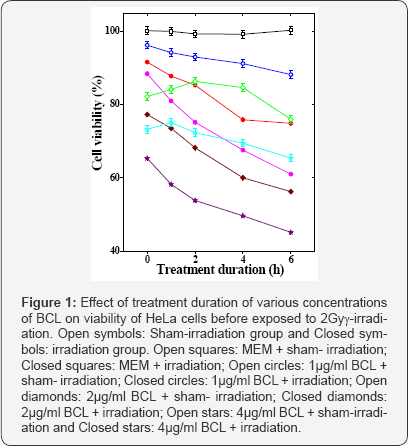
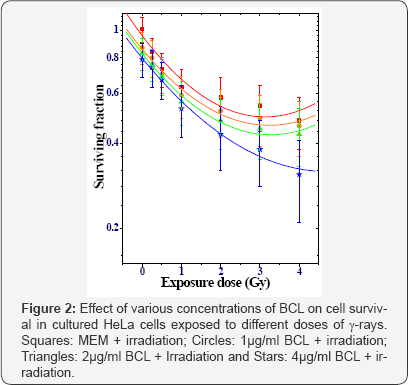
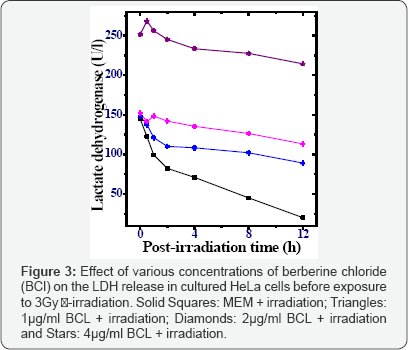
The results are expressed as % viability for Pratt and Willis assay and surviving fraction (SF) for clonogenic assay in (Figure 1 & 2). The results of biochemical analysis are expressed as LDH (units/L) (Table 1) and (Figure 3).
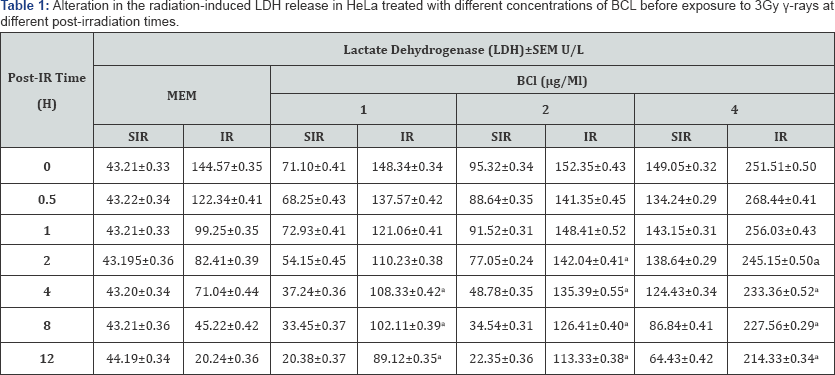
a = p<0.001 (Comparison of BCl concentrations with DDW + IR).
SIR: Sham Irradiation (0Gy); MEM: Minimum Essential Medium; BCL:Berberine Chloride.
Determination of optimum time
Pratt and Willis assay: MEM did not alter the spontaneous viability of HeLa cells with estimation time. However, treatment of HeLa cells with 1, 2 or 4μg/ml reduced the cell viability in a concentration and time dependent manner (Figure 1). Irradiation of HeLa cells with 2Gy γ-rays depleted the cell viability and the cytotoxic effect was almost similar at different MEM treatment duration. Treatment of HeLa cells with 1, 2 or 4μg/ml BCL for different times reduced the cell viability in a concentration and time dependent manner and highest cytotoxic effect was discernible for 4μg/ml BCL for 6h treatment duration (Figure 1). Treatment of HeLa cells with BCL for 4h caused a significant decrease in the cell viability (approximately 50%) and a gradual decline thereafter however difference between 4 and 6h was not statistically significant (Figure 1).
Clonogenic assay: The reproductive integrity of HeLa cells remained unaffected in MEM+irradiation group with time, as evidenced by non-significant changes in the survival of HeLa cells (Figure 4). Treatment of HeLa cells with 1, 2 or 4μg/ml BCL for various time periods before exposure to 2Gy exhibited a time and BCL concentration dependent decrease in the SF. The SF of HeLa cells treated with 2μg/ml BCL for 4h before irradiation reduced to 0.54 and, thereafter, clonogenicity of HeLa cells declined negligibly with exposure time up to 6h post- treatment, the last BCL treatment time evaluated (Figure 4). Therefore further studies were carried out using 4h treatment time for BCL.
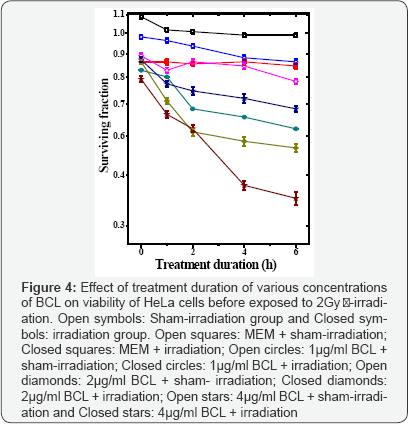
Determination of radiosensitizing activity
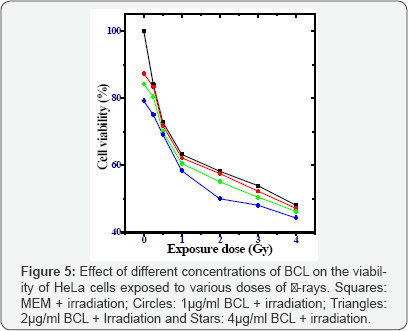
Pratt and Willis assay: MEM treatment did not alter the spontaneous viability of HeLa cells significantly (Figure 5). When HeLa cells were treated with different concentrations of BCL for 4h, the cell viability declined in a concentration dependent manner and a lowest viability was observed for 4μg/ml, the highest concentration of BCL evaluated (Figure 5). Exposure of HeLa cells to different doses of γ-rays resulted in a dose dependent decline in the cell viability and lowest viable cells were recorded for 4Gy γ-irradiation. Treatment of HeLa cells with various concentrations of BCL before exposure to different doses of γ-rays led to a further decrease in the cell viability depending not only on the irradiation dose but also on the concentration of BCL (Figure 5). The lowest dose of 1μg/ ml BCL increased cytotoxic effect of y-rays very significantly when compared with the non-drug treated irradiated control. Exposure of HeLa cells to 2 μg/ml BCL further decreased the cell viability at all exposure doses in comparison with MEM + irradiation group where an approximate 2 fold decline in the cell viability was observed for 2 and 3Gy, respectively. The increase in irradiation dose up to 4Gy resulted in a 3 folds rise in cell death in the cultures receiving 2μg/ml BCL. A subsequent increase in BCL concentration up to 4μg/ml, before exposure to different doses of γ-rays further increased the radiation-induced cytotoxicity of HeLa cells, which was maximum among all three BCL doses studied. The effect of BCL treatment was always greater than that of non-drug treated group (Figure 5).
Clonogenic assay: Sham irradiation did not alter cell survival after MEM treatment, whereas treatment of HeLa cells with different concentration of BCL resulted in a concentration dependent attrition in cell survival (Figure 2). Exposure of HeLa cells to 0 to 4Gy γ-rays resulted in a radiation-dose dependent depletion in the cell survival and the greatest reduction in cell survival was recorded for 4Gy exposure (Figure 2). A similar pattern was observed in HeLa cells treated with different concentrations of BCL, except that the clonogenecity of cells declined significantly with BCL concentration when compared with MEM + irradiation group. The lowest SF was observed for 4μg/ml BCL for all irradiation doses. This reduction in the clonogenic potential of HeLa cells was always greater than MEM pretreatment (Figure 2).
Lactate dehydrogenase
Irradiation of HeLa cells with 3Gy γ-rays caused an elevation in LDH release in the medium when compared to sham- irradiated controls (Table 1). Treatment of HeLa cells with various concentrations of BCL before irradiation elevated LDH significantly when compared to 3Gy γ-ray exposure. The LDH activity was highest immediately after irradiation (0h) in all the groups. However, this elevation was 2 folds greater at other postirradiation assay times in BCL + IR group (Table 1 and Figure 3). The LDH release declined with assay time (since the whole media was removed at each time, the values in tables and graphs are lower) reaching a nadir at 8h post-irradiation (Figure 3), however, the LDH contents were significantly higher (p<0.001) than the sham-irradiated control (MEM + 0Gy) as well as MEM + 3Gy irradiation group for all BCL concentrations (Table 1).
Discussion
Radiotherapy and chemotherapy have multiple negative effects on the immune system, as do surgery, anesthetics, antibiotics, blood transfusions and all drug and chemical exposures that may form part of cancer treatment. The inclusion of a drug in radiotherapy regimens will improve the therapeutic index by killing neoplastic cells and reducing radiation toxicity to normal tissues [42]. Our earlier study has shown that 10mg/ kg b.wt. berberine chloride (BCL) killed neoplastic cells in vivo [21] and reports regarding the systematic evaluation of radiosensitizing activity of BCL in vitro are lacking. Therefore the present study was undertaken to study the radiosensitizing activity of berberine in cultured HeLa cells exposed to different dosed of γ-rays.
Treatment of HeLa cells with different concentrations of BCL before irradiation caused radiation dose-dependent decline in the cell survival. Berberine has been reported to increase the radiosenstivity of prostate cancer cells and xenografted tumor earlier [43]. Our earlier study has shown that berberine treatment enhanced the effect of radiation by triggering molecular DNA damage and cell death [38]. Similarly, the other drugs like vinblastine, chlorpromazine, bleomycin, vincristine, teniposide, taxol, adriamycin, vindesine and echitamine have been reported to increase the effect of radiation in vitro and in vivo [44-48]. Treatment of HeLa cells with various concentrations of BCL caused a significant decline in cell viability after exposure to 1 to 4Gy γ-radiation. The highest concentration of 4μg/ml BCL increased cytotoxic effect of γ-radiation significantly when compared with the non-drug treated control. A similar effect has been observed for V79 cells treated with taxol, vindesine and teniposide before irradiation [49-51]. Acyclovir and azidothymidine have also been found to enhance the effect of radiation in HeLa cells [52,53].
BCL treatment increased the radiation-induced LDH release significantly at all post-irradiaiton times. The measurement of LDH release is useful in assessing the cytotoxicity of cells in cases of advanced cell membrane damage [54]. An increase in LDH contents after paclitaxel and VM-26 treatment has been reported earlier [45]. The increased LDH activity is closely related to the reduced surviving fraction in the present study. A direct correlation between increase in LDH release and a consequent decline in cell survival has been reported earlier [45,51,55,56].
The mechanisms involved in the increased radio response by BCL in the present investigation may not be ascribed to a single mechanism, but multiple mechanisms may be acting in concert with other to increase the radiosensitivity of Hela cells. The ionizing radiations interact with cellular aqueous milieu by triggering formation of free radicals [57,58]. The combination of berberine with radiation would have enhanced the free radical formation as berberine has also been reported to produce free radicals [59]. This free radical production would have damaged DNA causing increased cell death. Our earlier studies have shown that berberine induces molecular DNA damage and it also augmented radiation-induced DNA damage [37,38]. The inhibition of DNA topoisomerase-II by berberine may have increased the cell killing effect of radiation as it has been actually found to repress the activity of topoisomerase II in vitro [60]. Acceleration of radiation-induced apoptosis by berberine may have also contributed to increased cell killing effect in BCL + irradiation group. Berberine has been reported to increase the apoptosis in MCF-7 cell by increasing the activity of cytochrome C, caspase 9, PARP cleavage and reducing the activity of Bcl2 [61]. The HeLa cells constitutively express NF-kB and suppression of NF-kB by BCL may have played an important role in increasing the radiosensitivity. BCL has been found to downregulate NF- kB, COX-II, p53, activator protein 1, and cyclins that may have played a very significant role in enhancing the radiosenstivity of HeLa cells in the present study. Inhibition of HIF-1a and VEGF expression by berberine may have also been responsible for increased radiosensitivity [43]. Apart from the mechanisms described above, increased LDH activity- may have played an important role in increasing the cytotoxicity of radiation.
Conclusion
Treatment of HeLa cells with BCL before exposure to different doses of γ-rays increased the cytotoxic effect of radiation. The upsurge in the cytotoxic effect of radiation by BCL may be due to inhibition of topoisomerase II activity, NF- kB, COX-II, p53, activator protein 1, HIF-1α and VEGF and cyclins and increase in LDH release by changing the fluidity of the cell membrane. Apart from these, elevation in radiation-induced DNA damage and apoptosis by BCL may have also contributed to increased cell death after irradiation. The present study in preclinical model shows that BCL is capable of enhancing the radiosensitivity of HeLa and deserve attention as radiosensitizer in clinical situation, where it may bring out therapeutic gains in the cancer patients.
Acknowledgment
The authors wish to thank Prof. M.S. Vidyasagar and Dr. J. G. R. Solomon, Department of Radiotherapy and Oncology, Kasturba Medical College, Manipal, India, for providing the irradiation facilities and for help in radiation dosimetry, respectively. The financial Assistance from Indian Council of Medical Research and Council of Scientific and Industrial Research, Government of India, New Delhi to carry out the above study is gratefully acknowledged.
References
- Ahmad SS, Duke S, Jena R, Williams MV, Burnet NG (2012) Advances in radiotherapy. BMJ 345: e7765.
- Hellevik T, Zubiaurre IM (2014) Radiotherapy and the tumor stroma: the importance of dose and fractionation. Front Oncol 4: 1.
- Liauw SL, Connell PP, Weichselbaum RR (2013) New paradigms and future challenges in radiation oncology: an update of biological targets and technology. Sci Transl Med 5(173): 173sr2.
- Horvat A, Kovac V, Strojan P (2009) Radiotherapy in palliative treatment of painful bone metastases. Radiology and Oncology 43(4): 213-224.
- Rodin D, Grover S, Elmore SN, Knaul FM, Atun R, et al. (2016) The power of integration: radiotherapy and global palliative care. Ann Palliat Med 5(3): 209-217.
- Barnett GC, West CM, Dunning AM, Elliott RM, Coles CE, et al. (2009) Normal tissue reactions to radiotherapy: towards tailoring treatment dose by genotype. Nat Rev Cancer 9(2): 134-142.
- Gregoire V, Hittelman WN, Rosier JF, Milas L (1999) Chemo- radiotherapy: Radiosensitzing nucleoside analogues (Review). Oncol Rep 6(5): 949-957.
- Zhang L, Jiang N, Shi Y, Li S, Wang P, et al. (2015) Induction chemotherapy with concurrent chemoradiotherapy versus concurrent chemora- diotherapy for locally advanced squamous cell carcinoma of head and neck: a meta-analysis. Scientific Reports.
- Siemann DW, Chaplin DJ, Horsman MR (2004) Vascular-targeting therapies for treatment of malignant disease. Cancer 100(12): 2491-2499.
- Pauwels B, Korst AE, Lardon F, Vermorken JB (2005) Combined modality therapy of gemcitabine and radiation. Oncologist 10(1): 34
- Rapidis A, Sarlis N, Lefebvre JL, Kies M (2008) Docetaxel in the treatment of squamous cell carcinoma of the head and neck. Ther Clin Risk Manag 4(5): 865-886.
- Kuwahara A, Yamamori M, Nishiguchi K, Okuno T, Chayahara N, et al. (2010) Effect of dose-escalation of 5-fluorouracil on circadian variability of its pharmacokinetics in Japanese patients with Stage III/IVa esophageal squamous cell carcinoma. Int J Med Sci 7(1): 48-54.
- Sun W, Wang T, Shi F, Wang J, Wang J, et al. (2015) Randomized phase III trial of radiotherapy or chemoradiotherapy with topotecan and cis- platin in intermediate-risk cervical cancer patients after radical hysterectomy. BMC Cancer 15: 353.
- Lammers PE, Lu B, Horn L, Shier Y, Keedy V (2015) nab-Paclitaxel in Combination With Weekly Carboplatin With Concurrent Radiotherapy in Stage III Non-Small Cell Lung Cancer. Oncologist 20(5): 491-492.
- Larsen FO, Markussen A, Jensen BV, Fromm AL, Vistisen KK, et al. (2016) Capecitabine and Oxaliplatin before, during, and after radiotherapy for high-risk rectal cancer. Clin Colorectal Cancer 16(2): e7- e14.
- DeMasi J, Edwards JM, Shahidain S, Desimone P, Tzeng CW, et al. (2016) Superior outcomes with gemcitabine-based systemic and concurrent administration with radiation therapy in inoperable or unresect- able cholangiocarcinomas. Int J Radiat Oncol Biol Phys 96(2 Suppl): E207-E208.
- Oeffinger KC, Baxi SS, Friedman DN, Moskowitz C (2013) Solid tumor second primary neoplasms: who is at risk, what can we do? Semin Oncol 40(6): 676-689.
- Morton LM, Swerdlow AJ, Schaapveld M, Ramadan S, Hodgson DC, et al. (2014) Current knowledge and future research directions in treatment-related second primary malignancies. EJC Suppl 12(1): 5-17.
- Singh A, Duggal S, Kaur N, Singh J (2010) Berberine: Alkaloid with wide spectrum of pharmacological activities. J Nat Prod 3: 64-75.
- Letasiová S, Jantová S, Miko M, Ovádeková R, Horváthová M (2006) Effect of berberine on proliferation, biosynthesis of macromolecules, cell cycle and induction of intercalation with DNA, dsDNA damage and apoptosis in Ehrlich ascites carcinoma cells. J Pharm Pharmacol 58(2): 263-270.
- Jagetia GC, Baliga MS (2004) Effect of Alstonia scholaris in enhancing the anticancer activity of berberine in the Ehrlich ascites carcinoma bearing mice. J Med Food 7(2): 235-244.
- Li J, Li O, Kan M, Zhang M, Shao D, et al. (2015) Berberine induces apop- tosis by suppressing the arachidonic acid metabolic pathway in hepatocellular carcinoma. Mol Med Rep 12(3): 4572-4577.
- de Oliveira JS, Abdalla FH, Dornelles GL, Adefegha SA, Palma TV, et al. (2016) Berberine protects against memory impairment and anxiogen- ic-like behavior in rats submitted to sporadic Alzheimer's-like dementia: Involvement of acetylcholinesterase and cell death. Neurotoxicology 57: 241-250.
- Choi MS, Oh JH, Kim SM, Jung HY, Yoo HS, et al. (2009) Berberine inhibits p53-dependent cell growth through induction of apoptosis of prostate cancer cells. Int J Oncol 34(5): 1221-1230.
- Ortiz LM, Lombardi P, Tillhon M, Scovassi AI (2014) Berberine, an epiphany against cancer. Molecules 19(8): 12349-12367.
- Neto FR (1993) Electropharmacological effects of berberine on canine cardiac Purkinje fibres and ventricular muscle and atrial muscle of the rabbit. Br J Pharmacol 108(2): 534-537.
- Zeng XH, Zeng XJ, Li YY (2003) Efficacy and safety of berberine for congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol 92(2): 173-176.
- Yin J, Xing H, Ye J (2008) Efficacy of berberine in patients with type 2 diabetes mellitus. Metabolism 57(5): 712-717.
- Zhang H, Wei J, Xue R, Wu JD, Zhao W, et al. (2010) Berberine lowers blood glucose in type 2 diabetes mellitus patients through increasing insulin receptor expression. Metabolism 59(2): 285-292.
- Lan J, Zhao Y, Dong F, Yan Z, Zheng W, et al. (2015) Meta-analysis of the effect and safety of berberine in the treatment of type 2 diabetes mellitus, hyperlipemia and hypertension. J Ethnopharmacol 161: 69-81.
- Hu Y, Ehli EA, Kittelsrud J, Ronan PJ, Munger K, et al. (2012) Lipid-lowering effect of berberine in human subjects and rats. Phytomedicine 19(10): 861-867.
- Chang X, Wang Z, Zhang J, Yan H, Bian H, et al. (2016) Lipid profiling of the therapeutic effects of berberine in patients with nonalcoholic fatty liver disease. J Transl Med 14: 266.
- Di Pierro F, Putignano P, Ferrara T, Raiola C, Rapacioli G, et al. (2016) Retrospective analysis of the effects of a highly standardized mixture of Berberis aristata, Silybum marianum, and monacolins K and KA in patients with dyslipidemia. Clin Pharmacol 9: 1-7.
- Huang M, Chen S, Liang Y, Guo Y (2016) The role of berberine in the multi-target treatment of senile dementia. Curr Top Med Chem 16(8): 867-873.
- Koppen LM, Whitaker A, Rosene A, Beckett RD (2017) Efficacy of berberine alone and in combination for the treatment of hyperlipidemia: A systematic review. J Evid Compl Altern Med 1-13.
- Yang Y, Wang Q, Xie M, Liu P, Qi X, et al. (2017) Berberine exerts an anti-inflammatory role in ocular Behcet's disease. Mol Med Rep 15(1): 97-102.
- Jagetia GC, Rao SK (2015) Isoquinoline alkaloid berberine exerts its antineoplastic activity by inducing molecular DNA damage in HeLa cells: A comet assay study. Biology and Medicine 7(1): 223.
- Jagetia GC, Rao SK (2016) Berberine an isoquinoline alkaloid augments the effect of radiation by enhancing the molecular DNA damage in HeLa cells exposed to different doses of y-radiation: correlation between DNA damage and clonogenicity. J Mol Genet Med 10: 235.
- Pratt RM, Willis WD (1985) In vitro screening assay for teratogens using growth inhibition of human embryonic cells. Proc Natl Acad Sci USA 82(17): 5791-5794.
- Puck TT, Marcus I (1956) Action of X-rays on mammalian cells. J Exp Med 103(5): 653-666.
- Decker T, Matthes MLL (1988) A quick and simple method for the quantitation of lactate dehydrogenase release in measurements of cellular cytotoxicity and tumor necrosis factor TNF activity. J Immunol Methods 115(1): 61-69.
- Tannock IF (1996) Treatment of cancer with radiation and drugs. J Clin Oncol 14(12): 3156-3174.
- Zhang Q, Zhang C, Yang X, Yang B, Wang J, et al. (2014) Berberine inhibits the expression of hypoxia induction factor-1 alpha and increases the radiosensitivity of prostate cancer. Diagn Pathol 9: 98.
- Jagetia GC (1992) Effect of chlorpromazine hydrochloride on the formation of micronuclei in the bone marrow of mice exposed to gamma radiation. Hereditas 115(3): 195-199.
- Jagetia GC, Adiga SK (1995) Influence of various concentrations of taxol on cell survival, micronuclei induction, and LDH activity in cultured V79 cells. Cancer Lett 96(2): 195-200.
- Jagetia GC, Adiga SK (1998) Influence of vindesine exposure on the micronucleus formation and cell survival in V79 cells. Mutat Res 421(1): 55-63.
- Jagetia GC, Nayak V (2000) Effect of doxorubicin on cell survival and micronuclei formation in HeLa cells exposed to different doses of γ-radiation. Strahlenther Onkol 176(9): 422-428.
- Jagetia GC, Baliga MS (2002) Vincristine increases the genomic instability in irradiated cultured human peripheral blood lymphocytes. Toxicology Lett 126(3): 179-186.
- Jagetia GC, Adiga SK (1997) Correlation between micronuclei- induction and cell survival in V79 cells exposed to paclitaxel (taxol) in conjunction with radiation. Mutat Res 377(1): 105-113.
- Jagetia GC, Adiga SK (2000) Correlation between cell survival and micronuclei formation in V79 cells treated with vindesine before exposure to different doses of gamma radiation. Mutat Res 448(1): 5768.?
- Adiga SK, Jagetia GC (1999) Correlation between cell survival, micronuclei-induction, and LDH activity in V79 cells treated with teniposide (VM-26) before exposure to different doses of gamma radiation. Toxicology Lett 109(1-2): 31-41.
- Jagetia GC, Aruna R, Shivananda B (2000) Alteration in the radiation- induced LD release in HeLa cells by acyclovir. Clinica Chimica Acta 294(1-2): 129-138.
- Jagetia GC, Aruna R, Shivananda B (2000) Effect of azidothymidine on the radiation-induced LDH release in HeLa cells. Pharmazie 55(11): 837-840.
- Adiga SK, Jagetia GC (1999) Effect of teniposide (VM-26) on the cell survival, micronuclei-induction and lactate dehydrogenase activity on V79 cells. Toxicology 138(1): 29-41.
- Bunel V, Ouedraogo M, Nguyen AT, Stevigny C, Duez P (2014) Methods applied to the in vitro primary toxicology testing of natural products: state of the art, strengths, and limits. Planta Med 80(14): 1210-1226.
- Chenoufi N, Raoul JL, Lescoat G, Brissot P, Bourguet P (1998) In vitro demonstration of synergy between radionuclide and chemotherapy. J Nucl Med 39(5): 900-903.
- Le Caёr S (2011) Water radiolysis: influence of oxide surfaces on H2 production under ionizing radiation. Water 3(1): 235-253.
- Fidalgo AB, Dahlgren B, Brinck T, Jonsson M (2016) Surface Reactions of HO, H, and O2 in Aqueous Systems Containing ZrO2. J Phys Chem C l20[3): 16 09-1614.
- Chen TC, Lai KC, Yang JS, Liao CL, Hsia TC, et al. (2009) Involvement of reactive oxygen species and caspase-dependent pathway in berberine- induced cell cycle arrest and apoptosis in C6 rat glioma cells. Int J Oncol 34(6): 1681-1690.
- Makhey D, Gatto B, Yu C, Liu A, Liu LF, et al. (1995) Protoberberine alkaloids and related compounds as dual inhibitors of mammalian topoisomerase I and II. Med Chem Res 5: 1-12.
- Patil JB, Kim J, Jayaprakasha GK (2010) Berberine induces apoptosis in breast cancer cells (MCF-7) through mitochondrial-dependent pathway. Eur J Pharmacol 645(1-3): 70-78.






























