Effect of Arsenic (As2o3) on Haemetological Parameters of Freshwater Air Breathing Fish, Channa punctatus (Bloch)
A Amsath1*, J Sugumaran1 and S Vanitha2
1Department of Zoology, Khadir Mohideen College, India
2Department of Zoology, ADM College for Women, India
Submission: February 03, 2017; Published: August 14, 2017
*Corresponding author: A Amsath, Associate Professor and Research Advisor, P.G. and Research Department of Zoology, Khadir Mohideen College, Adirampattinam- 614 701, Tamil Nadu, India, Tel: +91 9524582977; Email: aamsath@gmail.com
How to cite this article: A Amsath, J Sugumaran, S Vanitha. Effect of Arsenic (As2o3) on Haemetological Parameters of Freshwater Air Breathing Fish, Channa punctatus (Bloch). Curr Trends Biomedical Eng & Biosci. 2017; 7(1): 555702. DOI: 005 10.19080/CTBEB.2017.07.555702.
Abstract
The study was conducted to investigate the effect of sub lethal concentration of heavy metal Arsenic (NaAsO2) exposure on hematological parameters such as red blood cells (RBC), Hemoglobin (Hb), white blood cells (WBC) and clotting time (CT) in the blood of fresh water fish, Channa punctatus The present study shows the level of RBC and Hb were significantly decreased simultaneously the WBC significantly increased due to arsenic exposure. While the clotting time also increased with exposure. Thus the present study concludes that the hematological parameters of fish of Channa punctatus affected by exposure to arsenic. Further the duration of exposure determines the rate the impact on blood parameters.
Keywords: Channa punctatus; Arsenic; Hemoglobin; RBC; WBC
Introduction
Arsenic is a naturally occurring element found widely in the environment. However, on recent days the level of arsenic in the environment has increased several folds due to its use as pesticide, defoliant, electronics, thermal power plants, wood preservatives and metal industry. Arsenic has been reported as one of the most alarming chemical in the environment [1], present in different forms and the toxicity depends upon its chemical form and oxidation states [2]. The toxicity of arsenical in animals depends on species, sex, age, exposure dose, duration of exposure, organic or inorganic form, valence state, etc. [3]. In natural water, arsenic is mostly exist in inorganic and organic form [4] and the inorganic form has been found to be more toxic. Among the various arsenical compounds, arsenic trioxide (As2O3) is mostly used in synthesis of various inorganic and organic compounds and in agricultural chemicals. It is also used as a chemotherapeutic agent for the treatment of hematological malignancies [5]. Arsenic trioxide (As2O3) was a dominant species in most of the arsenic contaminated areas in India and which is almost 50% of the total area arsenic level [6].
Among the aquatic fauna fish appear to be particularly susceptible to arsenic toxicity as they are continually exposed to it through gills and intake of arsenic contaminated food [7]. Sensitivity of fish to arsenic is variable in terms of 96hr of LC50 with range of 10.8 to 105mg/L [8]. Channa punctatus is one of the most important freshwater fish species of India [9]. It is a common freshwater fish which is abundantly found in ponds, lakes and canals of India. In fisheries ground water is widely used in various stages as in hatchery operation and in aquaculture. Furthermore surface water reserves are also getting polluted due to release of treated and untreated industrial effluents and also in urban waste water. In the present study, arsenic toxicity on hematology of C. punctatus has been under taken on the fishes in controlled laboratory condition. Freshwater fish C. punctatus were exposed to different concentrations of Arsenic trioxide for varied span of time in controlled laboratory condition to measure hematological parameters as indicators of the health of the larger population and community.
Behavioral studies on C. punctatus are known from various natural and laboratory investigations. It is not clear what extend C. punctatus differ from other wild fish species to the response of toxicant environment. Few studies are available on C. punctatus in toxicant contaminated environment. In particular, the effect of arsenic in the form of Sodium arsenate has been reported. There is no information is available on the effect of Arsenic trioxide (As2O3) on C. punctatus. The present study aims to assess the survival response in terms of acute toxicity test (LC50) and hematological changes of C. punctatus exposed to Arsenic trioxide (As2O3) in laboratory condition.
Materials and Methods
Selection of animal
A common freshwater air breathing teleost fish, C. punctatus, was selected as the test species owing to its availability throughout the year, hardy nature to survive under laboratory conditions and high sensitivity to small environmental changes. The medium sized freshwater fish, C. punctatus, weighing 14±1.8g and measuring 11±2.1cm, were collected with the help of local fisherman from water bodies of Thanjavur district, Tamilnadu. The fishes were properly washed in tap water and treated with 0.02% KMnO4 and 0.005% formalin solution to remove external infection. Prior to the experimentation the normal uninfected healthy fish were selected for experiment.
Experimental conditions
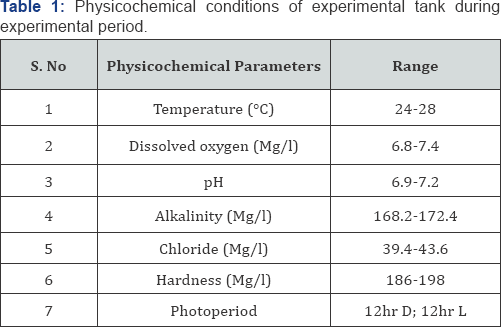
Healthy C. punctatus were maintained at 26±2 °C in 50L aquarium tank filled with one week prevaricated and de chlorinated tap water. The physicochemical conditions of experimental tank are given in Table 1. The parameters were measured daily according to standard experimental procedures [10]. The laboratory photoperiod was 12hr D; 12hr L. Fishes were fed with fish feed twice per day. The aquarium water was renewed at the 1/3 with filtered tap water. Feeding was suspended 24 hr before the start of experiment on mortality test for the fish.
Arsenic exposure
Arsenic was chosen in the present study because today large quantities of arsenic waste are disposed into aquatic system through agriculture and industrial activities. Chemicals used were of analytical grade, purchased from Merck (Mumbai, India). A stock solution of Arsenic trioxide (As2O3) was prepared by dissolving 1g in de ionized water and then diluted with tap water to obtain the desired concentration.
Determination of LC50
Based on the progressive bisection of intervals, log concentrations were fixed after conducting the range finding test. The fish were starved for 24 hours prior to the experiment. Prior to treatment, LC50 value of Arsenic trioxide for C. punctatus was calculated following Finney [11]. The fishes were divided into ten test groups and one control, each group consists of 15 fishes each. Each group was transferred separately to aquaria of 50 liter volume. The control was maintained without any treatment, the groups I to X were exposed to various concentrations of arsenic trioxide for four days to determine the median lethal concentration (LC50) for selection of sub lethal dose.
Sub lethal studies
Based on the 96hrs acute toxicity test, sub lethal concentrations (10% of LC50) were derived for Arsenic. Ten fish were exposed to 10% concentration of arsenic trioxide for a period of 10, 20 and 30 days. A control batch also maintained simultaneously.
Hematological studies
Blood samples were collected from caudal region after piercing the caudal peduncle. A thin Blood smear was prepared and stained with Giemsa stain for total counts. Blood parameters like RBC, WBC, Hb and clotting time (CT) were calculated following the methods of Dacie and Lewis [12].
Results
Acute toxicity test
The values of LC50, upper and lower confidence limits results of Arsenic (as arsenic trioxide) on C. punctatus at 96 hrs of time period are 24.19mg/L, 21.03mg/L, 27.33mg/L, respectively. At 96hrs of exposure 100% mortality was recorded at 50mg/L.
Hematological changes
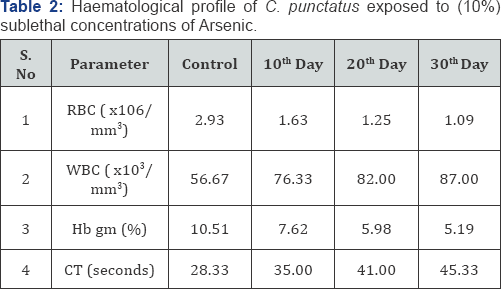
Exposure of fish to 10% sub lethal concentration (i.e., 2.42) of Arsenic for 10, 20 and 30 days caused significant alterations in hematological parameters, C. punctatus along with development of lesion in epidermis. The exposure of C. punctatus to 10% sub lethal concentrations of arsenic trioxide for 10, 20 and 30 days showed significant decrease in RBC count and Hb%. While WBC count increased significantly following arsenic exposure (Table 2). While, the clotting time (CT) increased with duration of exposure.
Discussion
Aquatic environments are flooded with contaminants released by man. These contaminants affect the aquatic life especially the fishes. Since fishes are one of the important food sources of man, the manmade pollution will return to him through the food they eat. In many aquatic systems, metal concentrations are elevated over natural background levels due to a continuous release of metals from industrial and agricultural sources [13]. Arsenic is one such metal found in fish, which thus may give rise to human exposure. Arsenic levels are higher in the aquatic environment than in most areas of land as it is fairly water soluble and may be washed out of arsenic bearing rocks [14]. Recently, treatment of agricultural land with arsenical pesticides, treating of wood using chromate copper arsenate, burning of coal in thermal power stations and the operations of gold-mining have increased the environmental pervasiveness of arsenic and its rate of discharge into freshwater habitat [15].

The results of present acute and chronic studies will give new insights in the field of aquatic toxicology. It is important to determine the lethal concentration of the substance before releasing them into the aquatic environment. There is relatively little information about the acute toxicity of arsenic to fish, with only a few species having been tested. Median lethal concentration (LC50) value of arsenic trioxide in C. punctatus was determined to be 24.19mg/L for 96hrs of exposure. The safe levels, estimated by different concentrations at 96 hr exposure are listed in Figure 1. Determination of LC50 values of arsenic trioxide of C. punctatus in different time span was necessary to formulate the sub lethal dose of this toxin for the entire experiment. The LC50 value of the present study is similar to Mukherjee et al. [16]. Sensitivity of fish to arsenic is variable in terms of 96 hr of LC50 with range of 10.8 to 105mg/L. Further, Pimphales promelas (LC50=25.6mg/L) was similar to our study while, mosquito fish Gambusia affinis (LC50=49.0 mg/L) had a higher value. Squawfish Ptychocheilus lucius was the most tolerant fish to arsenic exposure, with 96 hr LC50 of 150 mg/L arsenate at their larval stage (Hamilton and Buhl, 1997). Indian major carp Labeo rohita is the most sensitive species to arsenic exposure, and the LC50 2.73mg/L [17]. Therefore, the arsenic toxicity to animals depends on species and the test condition. The results indicate the tested fish C. punctatus moderate in facing arsenic toxicity.
The previous literature indicates that the acute toxicity of arsenic is varying from one species to another species. In general, toxicity of arsenic depends on species, sex, age, dose, exposure period, their valence, nature, concentration and organic and inorganic form. Gupta & Sastry [18] have reported that difference in acute toxicity may be due to changes in water quality and test species. Among the different forms, inorganic arsenicals are more toxic than the organic compounds.
Based the findings of 96 hrs LC50 value, the 10% sub lethal concentration of arsenic was given to C. punctatus to study the effect for prolonged period of 10, 20 and 30 days on its hematology. Measurement of hematological parameters helps in diagnosing the structural and functional status of animals exposed to toxicant because blood parameters are highly sensitive to environmental or physiological changes and health condition [19]. In the present study significant decrease in RBC count and Hb% was observed. While WBC count and clotting time (CT) increased significantly following arsenic exposure.
The reduced RBC content may be due to inhibition of erythropoietin or by the destruction [20]. Reported that reduced hemoglobin content in toxicant exposed fish may be due to disruption of haemopoietic processes and accelerated disintegration of erythrocyte cell membrane. In this study, the significant decrease in RBC count and hemoglobin content was observed in C. punctatus treated with Arsenic trioxide might have resulted from destructed of RBC's due to erythroblast sis leading to anemia [21]. The anemic condition in fish results from an unusually low number of red blood cells or too little hemoglobin in the red blood cells. Similar results with significant reduction of RBC and Hb% content in fishes exposed to different heavy metals have been reported previously by Goel & Sharma [22].
White blood cells are involved in the regulation of immunological functions and their numbers increase as a protective response in fish to stress. High white blood cell counts indicate damage due to infection of body tissues, severe physical stress, and as well as leukemia. In most cases, abnormal red cell morphology is noted. White blood cell counts were found increased following arsenic exposure. Similar findings were also documented significantly higher in fish exposed heavy metals [23,24].
When a wound is made in any blood vessel a clot is formed as the end product of blood coagulation. C. punctatus under sub lethal exposure of arsenic trioxide exhibits prolonged clotting time. The blood clotting substance in fish blood is prothrombin which is present in high percentage. A substance released by the platelet (thrombosthenin) is responsible for clot retraction [25]. Comparable results have also been reported in Labeo rohita exposed to copper sulphate [26], and Catla catla exposed to cadmium [27].
The changes in the hematological parameters of fish are a helpful biomarker for evaluating their health status. The arsenic induced impairment in the blood parameters are recorded in the present study indicates alterations in various haematological parameters may be due to haemolysis or hemorrhage under the action of arsenic induced toxins to the fish [28].
Conclusion
The environmental monitoring programmes were evolved to measure impact of stress inducers on aquatic fauna [29]. Aquatic ecosystem is too complex and hence indicators are an efficient means to obtain useful and representative information about the condition of fauna and flora. Acute toxicity studies are very first step in determining the water quality requirement of fish. Hematological parameters of fish can be helpful to identify the target organ toxic effects and also the general health condition of harmful changes in stressed organisms. The findings of the present study reflect that arsenic exposure of C. punctatus affect its hematological profile. The parameter studied in the present study indicates that arsenic affects the hematological profile of C. punctatus. Since, C. punctatus is known as a hardy fish, can tolerate pollutants to a greater extend. If arsenic affects this fish, the extent of damage it may cause to the sensitive fishes and other lower components of the aquatic food web such as phytoplankton and zooplankton will me more serious. Hence the use of arsenic should be avoided or reduced to an extent that our future generations should be protected from the deleterious effects of arsenic [30].
Acknowledgement
The authors are thankful to the Principal and Head of Department of Zoology, Khadir Mohideen College to for the facilities provided to carry out this work.
References
- ATSDR (2002) Toxicological profile for Arsenic. Agency for Toxic substances and Disease registry. SUDHHS PHS Washington D C.
- Agusa T, Takagi K, Kubota R, Anan Y, Iwata H, et al. (2008) Specific accumulation of arsenic compounds in green turtles (Chelonia mydas) and hawksbill turtles (Eremochelys imbricata) from Ishigaki Island Japan. Environ Pollut 153(1): 127-136.
- Allen T, Singhal R, Rana, SV (2004) Resistance to oxidative stress in a freshwater fish Channa punctatus after exposure to inorganic arsenic. Biol Trace Elem Res 98(1): 63-72.
- Luh MD, Baker RA, Henley DE (1973) Arsenic analysis and toxicity-a review. Sci Total Environ 2(1): 1-12.
- Liao CM, Tsai JW, Ling MP, Lain HM, Ghou Y, et al. (2004) Organ-specific toxicokinetics and dose-reponse of arsenic in tilapia Oreochromis mossambicus. Arch Environ Contam Toxicol 47(4): 502-510.
- List FA (2002) New approaches to the treatment of myelodysplasia. Oncologist 7: 39-49.
- Chatterjee A, Das D, Chakraborti D (1993) A study of ground water contamination by arsenic in the residential area of behala Calcutta due to industrial pollution. Environ Pollut 80(1): 57-65.
- Ahmed K, Anwaral A, Hasan M, Islam M, Hasan A (2008) Toxicity of arsenic (sodium arsenite) to Fresh water spotted snake head Channa punctatus (Bloch) on cellular death and DNA content. American Eurasian J Agric & Environ Sci 4(1): 18-22.
- USEPA (U S Environmental Protection Agency) (1985) Guidelines for Deriving Numerical National Water Quality Criteria for the Protection of Aquatic Organisms and Their Uses EPAPB 85-227049.
- Mishra A, Niyogi PA (2011) Hematological changes in the Indian Murrel (Channa punctatus, Bloch) in response to phenolic industrial wastes of the Bhilai steel plant (Chhaittisgarh, India). Int J Res Chem Environ 1(2): 83-91.
- APHA (2005) Standard methods for the examination of water and wastewater. (21st edn), Washington DC, USA.
- Finney DJ (1971) Probit Analysis. Cambridge University Press London, USA, p. 333.
- Dacie JV, Lewis SM (1977) Practical Hematology. Elsevier 653.
- Hameid NH (2009) A protective effect of calcium carbonate against arsenic toxicity of the Nile Cat fish Clarias gariepinus Turk J Fish Aquat Sci 9: 191-200.
- Duker AA, Carranza EJM, Hale M (2005) Arsenic geochemistry and health. Environ Int 31(5): 631-641.
- Mukherjee S, Ray D Adhikari D, Ghosh T (2015) Impairment of hematological profile of Channa punctatus exposed to Sodium arsenite. Int J Appl Biol Pharm Tech 6(2): 223-229.
- Vutukuru S, Prabhath S, Arun N, Raghavender M (2007) Effect of Arsenic and Chromium on the Serum Amino-Transferases Activity in Indian Major Carp, Labeo rohita. Internat J Environ Res Public Health 4 (3): 224-227.
- Gupta PK, Sastry KV (1981) Alterations in the activities of 3 dehydrogenises in the digestive system of 2 teleost fishes exposed to mercuric chloride. Environ Res 24(1): 15-23.
- Saravan M, Ramesh M, Malarvizhi A, Petkam R (2011) Toxicity of neem leaf extracts (Azadirachta indica A. Juss) on some hematological, ion regulatory, biochemical and enzymological parameters of Indian major carp. Cirrhinus mrigala J Trop for Environ 1(1):14-26.
- Mazon AF, Monteiro EA, Pinheiro GH, Fernandes MN (2002 ) Hematological and physiological changes induced by short-term exposure to copper in the freshwater fish, Prochilodus scrofa. Braz J Biol 62: 621-631.
- Nath R, Banerjee V (1995) Effects of various concentrations of lead nitrate on haematological parameters of an air breathing fish. Clarias batrachus J Freshwater Biol 7: 267-268.
- Pandey K, Shukla J P (2005) A textbook of Fish and Fisheries. Rastogi Publications, Meerut, India.
- Sinha A K, Sinha M K, Adhikari S (2000) Effect of the copper toxicity on hematological profile of Indian major corp, Lobeo rohita. Hand book Industry Environment and Pollution, p. 172.
- Vincent S, Ambrose T, Cyril L, Kumar A, Selvanayagan M (1996) Heavy metal cadmium influenced anaemia in the riverine major crop Catla catla (Ham). J Environ Biol 171: 81-84.
- Singh M (1995) Hematological responses in a fresh water teleost, Channa punctatus to experimental copper and Cr poisoning. J Environ Biol 16: 339-341.
- Cairns J, McCormick PV, Niederlehner BR (1993) A proposed framework for developing indicators of ecosystem health. Hydrobiologia 263(1): 1-44.
- Chatterjee A, Das D, Chakraborti D (1993) A study of ground water contamination by arsenic in the residential area of behala Calcutta due to industrial pollution. Environ Pollut 80(1): 57-65.
- Hamilton SJ, Buhl KJ (1997) Hazard assessment of inorganics, individually and mixtures to two endangered fish in the San Juan River, New Mexico. Environ Toxicol Water Qual 12: 195-209.
- Kumar P, Singh A (2010) Cadmium toxicity in fish: An over view. GERE Bulletin Biosciences 1(1): 41-47.
- Singh AK, Banerjee TK (2008) Toxic effects of sodium arsenate (Na2HAsO4 7H2O) on the skin epidermis of air-breathing catfish Clarias batrachus (L). Veterinar ski Arhiv 78(1): 73-88.






























