Investigation of Entomopathogenic Fungi from Insects at University of Guyana Turkeyen Campus
Ferial Pestano1, Elford Liverpool2, Subramanian Gomathinayagam1 and Abdullah Ansari2*
1University of Guyana Berbice Campus, Berbice, Guyana
2Department of Biology, University of Guyana, Guyana
Submission: May 03, 2017; Published: June 06, 2017
*Corresponding author: Abdullah Ansari, Department of Biology, University of Guyana, Guyana, Email: abdullah.ansari@uog.edu.gy
How to cite this article: Ferial P, Elford L, Subramanian G, Abdullah A. Investigation of Entomopathogenic Fungi from Insects at University of Guyana Turkeyen Campus. Curr Trends Biomedical Eng & Biosci. 2017; 5(2): 555656. DOI: 10.19080/CTBEB.2017.05.555656
Abstract
An investigation to find out if entomopathogenic fungi are found on insects at the University of Guyana Turkeyen Campus, Guyana, was carried out. A total of 42 insects were collected from the Campus. These forty two insects belonged to 8 different orders. They are Orthoptera, Hemiptera, Lepidoptera, Diptera, Hymenoptera, Blattaria, Coleoptera and Odonata. Among these, Order Hymenoptera was found to be the most diverse. However, from these eight orders 11 different fungal species were obtained. They are Cladosporium, Aspergillus niger, Aspergillus flavus, Penicillum, Curvularia, Rhizopus, Pyricularia, Phytophthora, Mucor, Bipolaris, and Alternaría. None of these fungal species were entomopathogenic. Among them two (2) were opportunistic fungi and the other 9 was classified as other fungi. Aspergillus flavuswas the most dominant fungal isolate obtained with an isolation rate of 29.5%. Mucor, Alternaria. Bipolarisand Pyricularia was found to be the least.
Introduction
Insects have proven to be the most successful class of Arthropods today and the number of insect species out numbers that of any other group of invertebrates. The insect group makes up 85 percent of the animal kingdom and there are more than 920,000 identified species [1]. Over the years, insects have developed many methods of obtaining and digesting food, some have chewing mouthpart and feed by tearing or pinching off plant parts, others have sucking mouthpart that are used to pierce the stems of plants or the skin of animals [2]. Insects are important to the ecosystem by providing services to plants such as pollination, and seed dispersal, within the context of mutualistic interactions. Plants use domatia or attract insects with volatile emissions, and use them as a defense from herbivores [3]. Apart from being beneficial to plants, they are beneficial to us by removing various nuisances and deformities from the face of nature, destroy other harmful insects and supply food to other animals such as fish and birds [4].
Despite their importance, insects can act as vectors and cause disease in human or animals. They are classified as major pest to rice crops and in so doing have created large economic crisis for rice farmers in Guyana, since they have a negative effect on the quantity of rice being produced. For example the leaf miner (Hydrellia sp.) larvae tunnel and feed on cell sap and caterpillar (Spodoptera frugiperda) cut leaf tips. Apart from rice, insects damage many other crops in Guyana such as pineapple, cabbage and pepper [5].
Many chemicals are used in Guyana to control pest populations. Even though, these chemicals are effective in controlling insects, it defeats the purpose of the green revolution since it affects the health of other organisms when applied. Thus, there is need for a control mechanism such as biological methods, which are effective and much safer to use. Entomopathogenic fungi are a biological agent that is present in environments worldwide and is safer to use in the control of insect pest. However, much research is not done on these fungi and their use or presence in Guyana. As such, this project will investigate if entomopathogenic fungi are found on insects at the University of Guyana Turkeyen Campus, Guyana.
Materials and Methods
Sampling method
Stratified random sampling was used to collect insects that were both dead and alive. Using this method the campus was divided into four non-overlapping subpopulations or strata. A random sample was extracted from each stratum once a week for a period of seven weeks (From the 20th of April to the 1st of June) [6]. However, a sampling effort of one hour was given to each stratum.
Collection and Storage of insects
The dead insects were collected and placed in paper bags. The paper bags were left open so that the cadavers were air dried for 3-4 days. However, to capture live insects tools such as the areal net and sweeping net was used. Foliage sampling was also done by shaking the plants. The insects were kept in sterile vile containing fragments of host plant material, until streaking was ready.
Identification of insects
During storage the insects were identified by order using previously described keys by Dunn in 1996.
Isolation of Entomopathogenic fungi from insects
Potato dextrose agar (PDA) was used for streaking.
Method of Preparation[7]
i. 250g of sliced potato, 20g of dextrose and 15g of agar was weighed.
ii. The sliced potato was boiled in half the quantity of distilled water for 20 minutes, until the potatoes soften.
iii. After boiling, the mixture was filtered through cotton to remove most of the particulate matter.
iv. The weighed amount of dextrose was then dissolved in about 200ml of distilled water and mixed with the potato infusion. The remaining quantity of distilled water needed to make up the volume of 1L was added.
v. The agar was then added to the mixture and dissolved on a hot plate.
vi. The medium was autoclaved at 121 degree for 25 minutes in order to be sterilized.
In the laboratory, the insects were surface sterilized. They were washed in 0.5% sodium hypochlorite for 60 seconds, rinsed three times in sterile distilled water and dried with sterile absorbent paper. Sterilized cotton swabs were used for streaking onto the potato dextrose agar (PDA) plates. First, the insects were swabbed externally and streaked onto the agar. Using a sterile razor, each insect was cut longitudinally from mouth to anus. The inside of the insect was swabbed and streaked also onto another plate. After streaking the fungi was allowed to grow. Every fungal species that emerged was streaked onto potato dextrose agar (PDA) in order to obtain pure cultures.
Identification of Entomopathogenic Fungi
Using the pure culture, the spores from each fungal species and guidance from Dr. Subramanian Gomathinayagam, was used to identify it.
Method of Preparation of Slides[8]
i. Two to three drops of distilled water was placed onto a clean microscope slide using an inoculating loop.
ii. Spores from a pure culture were placed into the distilled water using an inoculating loop.
iii. The spores and distilled water was covered with a cover slip and viewed under the microscope.
iv. Pictures of the spores were taken at magnification (10*10*100).
Results and Discussion
The aim of this project was to investigate if entomopathogenic fungi [9-13] are found on insects at the University of Guyana Turkeyen Campus, Guyana. However, a total of 43 insects were collected from Campus. These insects belonged to eight different Orders. They are Orthoptera, Hemiptera, Lepidoptera, Diptera, Hymenoptera, Blattaria, Coleoptera and Odonata.
A .Objective 1: To investigate the diversity of entomopathogenic fungi on Campus
From the table it can be seen that eleven different fungi were identified from the insects. Among these are Cladosporium, Aspergillus niger, Aspergillus flavus, Penicillum, Curvularia, Rhizopus, Pyricularia, Phytophthora, Mucor, Bipolaris, and Alternaria. Some were identified up to the species level while others up to genus.
Also, from the table it can be seen that none of the fungal isolates were entomopathogenic. However, two were opportunistic fungi [14] were present and nine was neither entomopathogenic or opportunistic. None of the isolated fungal species were entomopathogenic. Reasons being that, entomopathogenic fungi [15,16] are distributed in a wide range of habitats from aquatic forest, agricultural, pasture, desert and urban habitats. Campus will not be a most suitable area to find entomopathogenic fungi. Moreover, soil is considered to a very excellent shelter for these fungi since it protects them from UV radiation and other adverse abiotic and biotic influences [9].
From the graph it can be seen that Aspergillus flavus had the greatest isolation percentage and then Aspergillus niger, Curvularia, Cladosporium, Penicillum, Rhizopus, Phytophthora, followed by Pyricularia, Mucor, Bipolaris and Alternaria which had the same and least percentage.
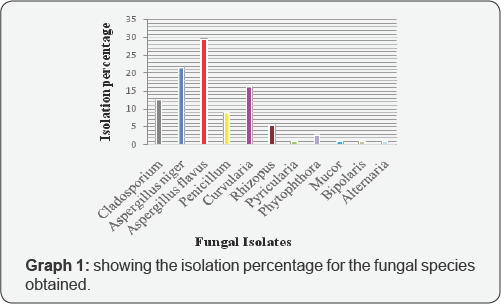

Aspergillus flavus was the most abundant isolate because it is widely distributed in the environment. It can be found in the soil, on plants and decaying vegetables, in dust, on stored food, fruits, vegetable, feed products and in the air [10] (Table 1 & Graph 1)
B .Objective 2: To examine the diversity of insect that possesses entomopathogenic fungi
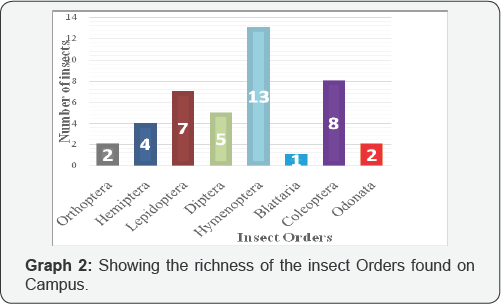
From the graph above, it can be seen that out of these eight insect orders, Hymenoptera was the richest followed by Coleoptera, Lepidoptera, Diptera, Hemiptera, Orthoptera, Odonata and finally Blattaria was the least. Hymenoptera is the richest order because it had the greatest number of species overall (Graph 2).

It can be seen that Order Coleoptera had the greatest diversity of fungal species and Odonata had the least. Even though Order Hymenoptera was the richest Coleoptera was more diverse in the number of fungal isolates. Beetles are found in dry, moist, hot, cold but also deep in the ground, in trees, inside fruits, inside seeds, inside leaves, inside dead wood, inside live plants and in carcasses. The habitat of beetles is very diverse and this can account for the great diversity of fungal isolates (Graph 3).
C . Objective 3: To determine the type of habitats associated with entomopathogenic fungi
Strata 1 were mostly a field area with little vegetation most of it was grass. It had a few building and a lot of sunlight. Strata 2 were the most vegetated area with a pond most of the strata was covered by trees. Strata 3 was the least exposed to sunlight because it was the strata occupied mostly by buildings. Finally, Strata 4 had some amount of sunlight, anthropogenic activities and a few buildings (Table 2).

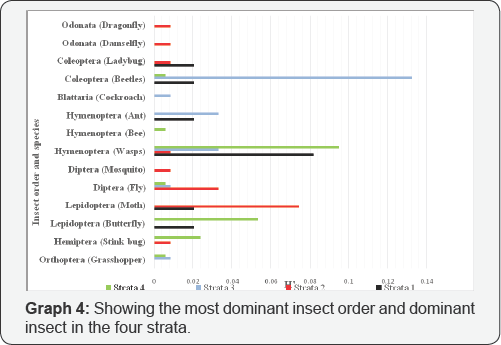
The Simpson index is a dominance index. It is denoted by the letter �D� and it gives more weight to common or dominant species [11]. D was calculated for the eight different insect orders as well as the species. It can be seen from the graph above that, in Strata 1 and 4 Order Hymenoptera was the most dominant insect order and Hymenoptera Wasp is the most dominant species. In Strata 2, Order Lepidoptera was the most abundant insect order and Lepidoptera (moth) is the dominant species. And finally in Strata 3, Order Coleoptera was the most dominant and Coleoptera (beetles) was the most abundant species. Moreover, it can be seen also that strata 2 is the most diverse, since it have more insect species (Graph 3).
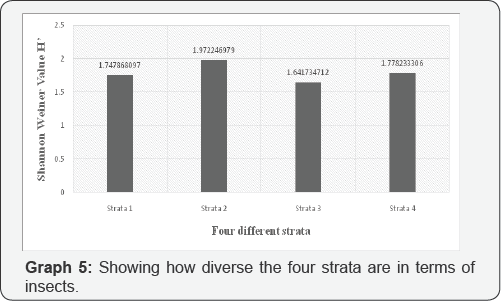
According to Pielou [11], Shannon Wiener Diversity Index is denoted by H' and calculates the diversity of the community and combines richness with how many individuals are in each taxa. According to the calculations done in excel, Shannon Wiener index is equal to 1.75 for Strata 1, 1.97 for strata 2, 1.64 for strata 3 and 1.77 for strata 4 (See appendix C for formula used). This information can be seen in the graph above. Strata 2 were the most diverse and a stratum 3 is the least. Which means that from the data collected strata 2 had the most insect species, followed by strata 1 and 4 and finally strata3 had the least.
Strata 2 had the most insects order because of its location. There was a lot of vegetation present in these strata. Strata 1 and 4 had the same amount being that they had some amount of vegetation but not as much as strata 2. However, strata 3 had the least probably because it is a more populated area with students and there is less vegetation [17-20] (Graph 4).
ANOVA was done to see how significantly different these strata were from each other. From the table, it can be seen that the p-value is greater than 5% or 0.05% this means that the four strata were not significantly different from each other; Even though strata 2 was found to be the most diverse (Table 3).
From the graph, it can be seen that Aspergillus niger, Aspergillus flavus, Penicillin and Curvularia was found in all four strata. Cladosporium and Rhizopus was found in three of the Strata, Phytophthorain two and Pyricularia, Mucor, Bipolaris and Alternariain one. It can also be seen that, Strata 3 and 2 had the greatest diversity, while Strata 1 and 4 had the least [20-27] (Graph 5 & 6).
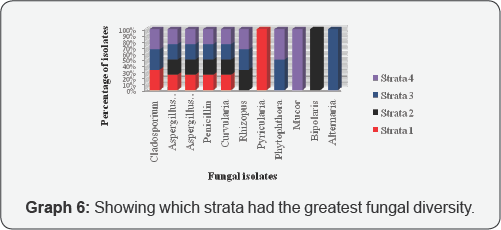
Alternaria was found only in Strata 3 this genus is very ubiquitous in the environment and is a normal agent of decay and decomposition. It was isolated from a beetle in Strata 3 since beetles have a vast majority of habitats. Genus Bipolaris and Pyricularia are pathogenic to grasses; they were in Strata 2 and Strata 3 respectively. They were found in the two strata that had a lot of grass species. Mucor are found in decaying vegeimages, foods, and animal excreta. Strata 4 had a lot of decaying vegetation and Mucor was isolated from a grasshopper found on the vegetation (U.S Micro-Solutions).
Conclusion
It can be concluded that among the eight different insect orders obtained, Order Hymenoptera was found to be the most diverse. Moreover, out of the four different strata that were investigated, Strata 2 had the greatest diversity of insects. Eleven different fungal isolates were obtained from these eight orders. Among the fungal isolates Aspergillus flavus was the most dominant fungal species obtained. However, A. flavus, A. niger, Pencillum and Curvularia were present in all four strata investigated. There was no entomopathogenic fungi isolated from the investigated insect population however, opportunistic fungi and other insect fungi were isolated. As such, the null hypothesis which states that “Entomopathogenic fungi are not present on insects found at the University of Guyana Turkeyen Campus” is accepted.
References
- Dunn G (1996) Insects of the Great Lakes Region: Insect Classification and Identification. Great Lakes environment, P. 41.
- Hadlington P, Johnston J (1998) an Introduction to Australian Insects: Feeding. pp. 11-12.
- Weisser W, Siemann E (2013) Insects and Ecosystem Function; Plant- Insect Mutualisms; Volume 173 of Ecological Studies; Springer Science & Business Media, p. 14.
- William K, William S (1816) an Introduction to Entomology, Or, Elements of the Natural History of Insects: with Plates, Longman, Hurst, Rees, Orme and Brown; p. 253.
- Homenauth O, Dwarka B (2011) Ministry of Agriculture: Pest and disease of pineapples in Guyana; p. 2-5.
- Black K (2011) Business Statistics: For Contemporary Decision Making, John Wiley & Sons, p. 228.
- Gaud SR, Gupta DG, Gokhale BS (2008) Practical Biotechnology: Culture media, Nirali Prakashan, p. 58.
- Aneja KR (1993) Experiments in Microbiology, Plant pathology, Tissue culture and Mushroom cultivation second edition, New Delhi, India, p. 140.
- Sergio R, Sanchez P, Jorge L, Raul M (2011) Occurrence of Entomopathogenic Fungi from Agricultural and Natural Ecosystems in Saltillo, México, and their Virulence Towards Thrips and Whiteflies. J Insect Sci 11(1): 2.
- U.S Micro-Solutions (2015) Fungal Library: Aspergillus species.
- Pielou C (1975) Ecological Diversity; Wiley-Intescience Publication; New York, USA, p. 165.
- Araújo J Hughes D (2014) Diversity of entomopathogens Fungi: Which groups conquered the insect body Bio Rxiv beta, pp.1-57.
- Asensio L, Carbonell T, Jimenez J, Llorca L (2003) Entomopathogenic fungi in soils from Alicante province. Spanish Journal of Agricultural Research 1(3): 37-45.
- AssafL, Haleema. R, Abdullah S (2011) Association ofEntomopathogenic and Other Opportunistic Fungi with Insects in Dormant Locations. Jordan Journal of Biological Sciences 4(2): 87-92.
- Barrag'an A, Anaya A, Alatorre R and Toriello C (2004) Entomopathogenic fungi from 'El Eden' Ecological Reserve, Quintana Roo, Mexico. Mycopathologia 158: 61-71.
- Kram A, Kram J (2012) Entomopathogenic Fungi as an Important Natural Regulator of Insect Outbreaks in Forests (Review). Forest Ecosystems - More than Just Trees, Juan AB (Ed.) 12: 265-282.
- Moore. J, Overhill R (2001) An Introduction to the Invertebrates: Why are insects such successful land animals. Cambridge University Press, USA, pp. 223-227.
- Shahid A, Rao A, Bakhsh A, Husnain T (2012) Entomopathogenic fungi as biological controllers: New insights into their Virulence and pathogenicity. Arch Biol Sci Belgrade 64(1): 21-42.
- Tuininga AR, Miller JL, Morath SU, Daniels TJ, Falco RC, et al. (2010) Isolation of Entomopathogenic Fungi from Soils and Ixodesscapularis (Acari: Ixodidae) Ticks: Prevalence and Methods. J Med Entomol 46 (3): SS7-S6S.
- Guyana Rice Development Board (2013) Pest of Rice in Guyana.
- Arnett HR (2000) American Insects: A Handbook of the Insects of America North of Mexico, Second Edition: Insect Ecology, Behavior and distribution, p. 29.
- http://www.iaszoology.com/2B.
- Da Sun B, Zhong Liu X (2008) Occurrence and diversity of insect- associated fungi in natural soils in China. Applied soil ecology 39: 100108.
- Jervis M (2007) Insects as Natural Enemies: A Practical Perspective- Genetics of Reproductive mode. Springer Science & Business Media, p. 18S.
- Lawrence AL (2012) Manual of Techniques in Invertebrate Pathology: detection and characterization. Academic Press, pp. 190-192.
- Pluke R, Permaul D, Leibee G (1999) Integrated Pest Management and the Use of Botanicals in Guyana.
- Pingali P, Roger P (199S) Impact of Pesticides on Farmer Health and the Rice Environment; Volume 7 of Natural Resource Management and Policy. Int Rice Res Inst p. 3-4.






























