In Vitro Anti-Inflammatory Activity of L-Asparaginase from Soil Rhizosphere Fungus Aspergillus tamarii
NS Mahabal1 and BB Kaliwal2*
1Department of Biotechnology and Microbiology, Karnatak University, India
2Department of Microbiology, Davangere University, India
Submission: March 03, 2017; Published: May 30, 2017
*Corresponding author: BB Kaliwal, Department of Microbiology, Davangere University, Shivagangotri, Davangere, Karnataka, 577 002, India, Tel: 91-08192-208444; E-mail: b_kaliwal@yahoo.com
How to cite this article: N Mahabal, B Kaliwal. In Vitro Anti-Inflammatory Activity of L-Asparaginase from Soil Rhizosphere Fungus Aspergillus tamarii. Curr Trends Biomedical Eng & Biosci. 2017; 4(5): 555647. DOI:10.19080/CTBEB.2017.04.555647
Abstract
In recent years L-asparaginase has been gaining interest of researchers as it is a potential anti-tumor drug. It has also been reported that the enzyme also possesses anti-inflammatory property. That present investigation is an insight to the in vitro anti-inflammatory property of L-asparaginase extracted from fungus Aspergillus tamarii. The in vitro assessment methods included albumin denaturation assay, proteinase inhibitory activity and membrane stabilization using Diclofenac sodium and Aspirin as standard drugs. In the present study L-asparaginase (10^g/ml to 50^g/ml) was used as the test sample for anti-inflammatory tests. The results indicated that 50 ^g/ml of L-asparaginase showed significant inhibition values. 52.38% of albumin denaturation, 46.87% of proteinase inhibition, 55.26% of haemolysis inhibition and 51.21% of inhibition of hypotonic induced haemolysis. The results indicate that L-asparaginase extracted from Aspergillus tamarii possesses antiinflammatory property apart from being an anti-tumor drug.
Keywords: L-asparaginase; Anti-inflammatory activity; Aspergillus tamarii
Introduction
Inflammation is a part of the complex biological response of vascular tissues to harmful stimuli, such as pathogens, damaged cells or irritants. It is a self defense reaction in its first phase, hence regarded as the main therapeutic target and often, the best choice to treat the disease and alleviate the symptoms. It is a complex response that protects the host against tissue injury and microbial invasion. The redness, swelling, heat, pain, and loss of functions are considered as symptoms of inflammation. Prolonged inflammation contributes to the pathogenesis of many diseases such as asthma, arthritis, multiple sclerosis and even cancer [1,2].
Inflammation can be classified as acute and chronic based on the commencement time. Acute inflammation is the primary response ofthe body to injurious stimuli and is involved in vascular changes i.e. vasodilatation, increased capillary permeability and migration of leukocytes. Chronic inflammation is prolonged inflammation characterized by progressive destruction and retrieving of injured tissue from the inflammatory process [3,4]. The process of inflammatory response is mediated by a variety of signaling molecules produced by mast cells, macrophages, granulocytes, platelets, lymphocytes and complement activation factors.
The activation of these immune cells can be obtained by the release of chemical mediators from injured tissue and migrating cells. It has been suggested that [5] macrophage cells play an important role in inflammation through the release of inflammatory mediators such as biologically active amines (histamine, serotonin), proteins and peptides (hydrolytic enzymes, cytokines, growth factors, and antibodies), Reactive Oxygen Species (ROS) like superoxide anion, hydroperoxide, hydroxyl radicals, Reactive Nitrogen Species (RNS) and lipids (platelet activating factors and prostaglandins) involved in the immune response. Delayed macrophage enactment is to a great extent in charge of the pathology of intense and unending incendiary conditions. Likely, Interleukin (IL-1) and Tumor Necrosis Factor (TNF) are master incendiary cytokines involved in fever, irritation, and tissue pulverization [6].
The generation and their natural impacts are firmly controlled through different mechanisms, including mitigating cytokines, solvent cytokine receptors or receptor enemy proteins to avert undesirable irritation and tissue damage [7]. The discovery of each chemically vary large group, so called nonsteroidal anti-inflammatory drugs (NSAIDs) such as diclofenac and indomethacin, have emerged as the most commonly used anti-inflammatory agents, including the therapy of rheumatoid arthritis which creates a shield against COX-1 and COX-2 enzymes (which responsible for inflammation) but the side effect including gastrointestinal ulceration, bleeding, renal damage and platelet dysfunction [8,9].
Several sources of anti-inflammatory compounds have been studied, which include medicinal plants, bacteria and fungi. Potent component from halo-tolerant fungus Alternaria alternate has also been studied. Based on this background of a variety of anti-inflammatory compounds the present investigation focuses on the assessment of anti-inflammatory property of L-asparaginase by in vitro methods.
Materials and Methods
Extraction of L-asparaginase
Production of L-asparaginase was done through Solid State Fermentation. The identified fungal strain Aspergillus tamarii BBKNM3 was grown on 10g of the substrate is moistened with water and inoculated with 1ml of spore suspension in a 250ml conical flask. The flasks were incubated at 37±2°C for 72hrs. After incubation the fungal extract was obtained and centrifuged at 10,000rpm for 10mins at 4°C. This clear supernatant served as crude enzyme extract and was used in determining the enzyme activity. The enzyme was purified to 105.21 fold purity and further it was assessed for its properties. Further the enzyme activity of the extract was measured using direct Nesslerization test [10].
Assessment of in vitro anti-inflammatory activity
Inhibition of albumin denaturation: The anti-inflammatory activity of L-asparaginase was studied using the inhibition of albumin denaturation [11]. The method included the test extract and 1% aqueous solution of bovine serum. pH of the reaction mixture was adjusted using 1NHCl. The samples were incubated at 37 °C for 20mins and then heated at 50 °C for 20mins. The mixture was allowed to cool and the absorbance was measured at 660nm. The experiment was performed in triplicates.
The Percentage inhibition of protein denaturation was calculated as follows:
Percentage inhibition=(Abs Control-Abs Sample) X 100/ Abs control.
Anti-proteinase action: The purified L-asparaginase was studied for its anti-inflammatory activity by anti-proteinase action [12,13]. Briefly, 2ml of the reaction mixture along with 0.06mg trypsin, 1ml of 20mM Tris HCl buffer (pH 7.4) and 1ml of enzyme of varying concentrations (100-500|ig/ml). All test tubes were incubated at 37 °C for 5mins and 1ml of 0.8% (w/v) casein was added. After 20mins of incubation, the reaction was stopped by adding 70% perchloric acid. The formed cloudy suspension was centrifuged and supernatant was collected. The absorbance of the supernatant was read at 210nm against buffer blank. The experiment was carried out in triplicates.
The percentage inhibition of protein was calculated using the formula:
Percentage inhibition = (Abs Control-Abs Sample) X 100/ Abs control.
Preparation of Human Red Blood Cell suspension (HRBC) [14]: Blood was collected from healthy volunteer who has not received any Non-steroidal Anti Inflammatory Drugs (NSAID's) two weeks prior to the experiment. The blood sample was centrifuged at 3000rpm for 10mins and washed with normal saline. The step was repeated thrice and the volume of blood was reconstituted with normal saline to 10% (v/v) suspension.
Heat induced hemolysis: The hemolysis test was performed according to the method of Shinde et al. [15] with a slight modification. Reaction mixture was prepared by adding 1ml purified L-asparaginase, 1ml of 10% RBC suspension. Control was prepared by adding saline and RBC suspension. The tubes were incubated at 55°C for 30mins in water bath. The tubes were cooled and centrifuged at 2500rpm for 5mins and the supernatant was collected. Absorbance was measured at 560nm. The experiment was conducted in triplicates.
The Percentage inhibition of Haemolysis was calculated as follows:
Percentage inhibition = (Abs control-Abs sample) X 100/ Abs control.
Hypotonicity-induced haemolysis [16]: Haemolysis of HRBC suspension was tested by preparing test sample containing 1ml L-asparaginase+1ml phosphate buffer+2 ml hyposaline and 0.5ml HRBC suspension. Diclofenac sodium was used as standard (50|ig/ml). The reaction mixture was incubated at 37 °C for 30mins and centrifuged at 3000rpm for 10mins. Supernatant was collected and estimated spectrophoto metrically at 560nm.
The Percentage inhibition of Haemolysis was calculated as follows:
Percentage inhibition = (Abs control-Abs sample) X 100/ Abs control.
Statistical analysis
All the experimental data were carried in triplicates and expressed as means±standard errors. The statistical analyses of the data were performed using one way ANNOVA variance SPSS version 20.0 software with advanced models (SPSS Japan, Tokyo, Japan). Differences between means were located using Tukey's test (P<0.05).
Results and Discussion
In the present investigation of in vitro anti-inflammatory property of L-asparaginase the results indicate that as the concentration of L-asparaginase increase there is an increase in the activity of L-asparaginase. This directly indicates that the enzyme is dose dependent drug. The enzyme shows maximum inhibition of albumin denaturation at 50μg/ml concentration (Table 1). Similarly 50μg/ml concentration is also effective for proteinase inhibition (Table 2), haemolysis inhibition (Table 3 & 4). Inflammation is a complex biological response of vascular tissue to harmful stimuli, pathogens, irritants characterized by redness, warmth, swelling and pain [17]. Prolonged inflammation leads to the rheumatoid arthritis, atherosclerosis, hay fever, ischemic heart diseases [18-20].
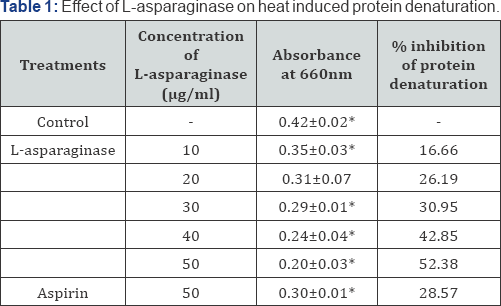
Each value represents the mean±SD. Significance p<0.05.
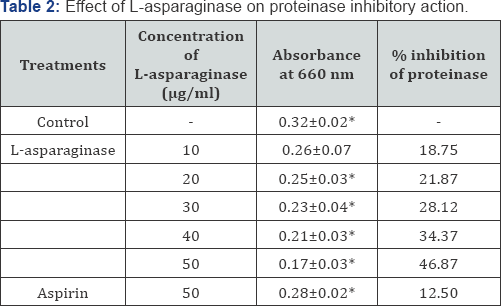
Each value represents the mean±SD. Significance p<0.05.

Each value represents the mean±SD. Significance p<0.05
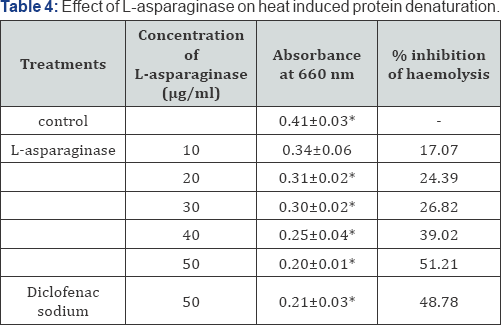
Each value represents the mean±SD. Significanc p<0.05
Inflammationisacommonmanifestationofinfectiousdiseases like leprosy, tuberculosis, syphilis, asthma, inflammatory bowel syndrome, nephritis, vasculitis, celiac diseases, auto-immune diseases etc [21]. Denaturation of proteins is a well-documented cause of inflammation. Proteinases have been embroiled in joint responses. Neutrophils are known to be a wellspring of proteinase which conveys in their lysosomal granules numerous serine proteinases. It was beforehand detailed that leukocytes proteinase assume critical part in the improvement of tissue harm amid in fiery responses and noteworthy level of insurance was given by proteinase inhibitors [22] Some researchers report that denaturation of protein is one of the reason for rheumatoid joint pain because of the generation of auto-antigens in certain rheumatic sicknesses. It might be cause to in vitro denaturation of proteins [11,23].
Mechanism of denaturation is involved in alteration of electrostatic force, hydrogen, hydrophobic and disulphide bonds. Several authors have shown anti-inflammatory drugs to show dose dependent ability to inhibit the thermally induced protein denaturation [24]. The extracts of anti-inflammatory sources may potentially hinder the arrival of lysosomal substance of neutrophils at the site of irritation. These neutrophils lysosomal constituents incorporate bactericidal chemicals and proteinases, which upon extracellular discharge bring on additional tissue irritation and harm [25]. This report includes the use of extracellular L-asparaginase extract from fungal source. Fungal L-asparaginase is well reported to have lesser immunological allergies as compared to bacterial L-asparaginase [26].
Karunakaran et al. [27] reported the fungus A. oryzae which exhibited anti-inflammatory activity in dose dependent manner. Zhu et al. [28] reported new cadinane-type sesquiterpenes and hypocreaterpenes A and B along with five known compounds from marine-derived fungus Hypocreales sp. Showed moderate in vitro anti-inflammatory activity with average maximum inhibition (Emax) values of 10.22% and 26.46% at 1|iM, respectively. High anti-inflammatory activity of the compound asperlin from marine-derived fungus Aspergillus sp was reported by Lee et al. [29]. Belofsky et al. [30] reported that Oxepinamides isolated from tunicate fungi Acremonium sp. exhibited comparable higher activity than standard drugs. Remarkable in vitro anti-inflammatory activity was reported by Bhimba et al. [31] in comparision with M. gulliermondii and marine fungal extracts from N. crassa. Nijveldt et al. [32] and reported that some flavonoids and steroids compounds from Dipthera canthus prostatusare known to show significant antiinflammatory activity by inhibiting the COX and LOX systems.
Conclusion
The present investigation concludes that L-asparaginase which is a potent anti-tumor drug, also possesses antiinflammatory property. L-asparaginase from many bacterial sources has been reported and is widely used as potential antitumor drugs. Further, these drugs also have properties of antiinflammation and also anti-bacterial. Bacterial originated drugs have allergic reaction on some users on long term usage. Added to this literature, the present study reveals that the enzyme extracted from the source Aspergillus tamarii can cause lesser allergies and hence can be a potent drug.
Acknowledgement
The authors are thankful to the Department of Biotechnology (DBT), Ministry of Science and Technology, Government of India, New Delhi, for funding the Bioinformatics Infrastructure Facility Project (BT/BI/25/001/2006 VOL II date 05-03-2012), The Interdisciplinary Program for Life Science Project (BT/PR/4555/ INF/22/126/2010 dated 30-09-2010), UGC-UPE fellowship and P.G Department of Biotechnology and Microbiology, Karnatak University, Dharwad, for providing the necessary facilities for the research.
Conflict of Interest
The authors hereby declare that they have no conflict of interest
References
- Mahesh G, Ramkanth S, Mohamed Saleem TS (2011) Anti-inflammatory drugs from medicinal plants-comprehensive a review. Int J Rev Life Sci 1: 1-10.
- Karin M, Greten FR (2005) NF-kB: Linking inflammation and immunity to cancer development and progression. Nat Rev Immunol 5: 749-759.
- Nathan C (2002) Points of control in inflammation. Nature 26: 846852.
- Sharon B, Elizabeth AA (2003) Wound care essentials: practice principles. Lippincott Williams and Wilkins, pp. 91-93.
- Dinarello CA (2009) Immunological and inflammatory functions of the interleukin-1 family. Annu Rev Immunol 27: 519-550.
- Standiford TJ (2000) Anti-inflammatory cytokines and cytokine antagonists. Curr Pharm Des 6(6): 633-649
- Bennett PN, Brown MJ (2005) New Delhi: Churchill Livingstone; Clinical Pharmacology (9th edn).
- Tripathi KD (2008) New Delhi: Jaypee Brothers Medical Publishers (P) Ltd; Essentials of Medical Pharmacology (6th edn).
- Imada A, Igarasi S, Nakahama K, Isono M (1973) Journal of General Microbiology 76: 85-99.
- Mizushima Y, Kobayashi M (1968) Interaction of anti-inflammatory drugs with serum proteins, especially with some biologically active proteins. J of PharmaPharmacol 20: 169-173.
- Oyedepo OO, Femurewa AJ (1995) Anti-protease and membrane stabilizing activities of extracts of Fagrazanthoxiloides, Olaxsubscorpioides and Tetrapleuratetraptera. Int J of Pharmacong 33: 65-69.
- Sakat S, Juvekar AR, Gambhire MN (2010) in vitro antioxidant and antiinflammatory activity of methanol extract of Oxalis corniculata Linn. International Journal of Pharma and Pharmacological Sciences 2(1): 146-155.
- Leelaprakash G, Mohan Dass S (2011) in vitro anti-inflammatory activity of methanol extract of Enicostemmaaxillare. Int J Drug Dev and Res 3(3): 189-196.
- Shinde UA, Kulkarni KR, Phadke AS, Nair AM, Dikshit Mungantiwar VJ, et al. (1999) Mast cell stabilizing and lipoxygenase inhibitory activity of Cedrusdeodara(Roxb) Loud Wood Oil. Indian J Exp Biol 37(3): 258261.
- Azeem AK, Dilip C, Prasanth SS, Junise V, HananShahima (2010) Anti-inflammatory activity of the glandular extracts of Thunnuasalalunga. Asia pac J for Med pp. 412-420.
- Sherwood ER, Toliver Kinsky T (2004) Mechanisms of the inflammatory response. Best Practice and Research Clinical Anaesthesiology 18(3): 385-405.
- Libby P (2008) Role of inflammation in atherosclerosis associated with rheumatoid arthritis. The American journal of medicine 121(10): S21-S31.
- Stevens RJ, Douglas KM, Saratzis AN, Kitas GD (2005) Inflammation and atherosclerosis in rheumatoid arthritis. Expert reviews in molecular medicine 7(07): 24.
- Black PH, Garbutt LD (2002) Stress, inflammation and cardiovascular disease. Journal of psychosomatic research 52(1): 1-23.
- Robbins SL, Cotran RS (1979) Pathologic basis of disease, WB Saunders Company. Philadelphia, London and Toronto. 1333.
- Das SN, Chatterjee S (1995) Long term toxicity study of ART-400. Indian Indigenous Med 16(2): 117-123.
- Vane JR, Botting RM (1995) New insights into the mode of action of anti-inflammatory drugs. Inflammation Research 44(1): 1-10.
- Grant NH, Alburn HE, Kryzanaukas C (1970) Stabilization of serum albumin by anti-inflammatory drugs. Biochem Pharmacol 19: 715-722.
- Chou CT (1997) The antinflammatory effect of Triptergiumwilfordii Hook F on adjuvant- induced paw edema in rats and inflammatory mediators release. Phyto Res 11: 152-154.
- Sarquis MI, Oliveira EM, Santos AS, Costa GL (2004) Production of L-asparaginase by filamentous fungi. Epub 99(5): 489-492.
- Karunakaran K, Raja P, Diraviyaraj K, Murugan A (2013) Antiinflammatory, analgesic, anti-pyretic, anti-ulcer and CNS stimulant activities of marine fungi Aspergillus oryzae, pharmacologia, pp. 359364.
- Zhu X, Zeng K, Qiu Y, Yan F, Lin C (2013) Therapeutic effect of emodin on collagen-induced arthritis in mice. Inflammation 36: 1253-1259.
- Lee HB, Kim EK, Park SJ, Kim TG, Chung DW (2011) Isolation and anti-inflammatory effect of astragalin synthesized by enzymatic hydrolysis of tea seed extract. J Sci Food Agric 91(13): 2315-2321.
- Belofsky GN, Anguera M, Jensen PR, Fenical W, Kock M (2000) Oxepinamides A-C and Fumiquinazolines H-I: Bioactive Metabolites from a Marine Isolate of a Fungus of the Genus Acremonium. 6: 8.
- valentine B, Angelin C, Pushpam T, Shilpa G, Geena M, et al. (2011) Anti-inflammatory effect of marine fungi Neurosporacrassa and Meyerozymaguilliermondii. Int J Univ Phar and Life sciences, pp. 22496793.
- Nijveldt RJ, Van Nood E, Van Hoorn DE, Boelens PG, Van Norren K, et al. (2001) Flavonoids: a review of probable mechanisms of action and potential applications. Am J Clin Nutr 74: 418-425.






























