Development Model of Micro vascular Network in Porous Silk Fibroin Film
Lun Bai1,2*, Kuihua Zhan13, Qi Hu1 and Jianmei Xu1
1School of Textile and clothing Engineering, Soochow University, China
2Faculty of Textile Science and Technology, Shinshu University, Japan
3School of Mechanical and Electronic Engineering, Soochow University, China
Submission: February 21, 2017; Published: March 16, 2017
*Corresponding author: Lun Bai, School of Textile and Clothing Engineering, Soochow University, China, Email: bailun@suda.edu.cn
How to cite this article: Lun Bai, Kuihua Z, Qi Hu, Jianmei X. Development Model of Micro vascular Network in Porous Silk Fibroin Film. Curr Trends Biomedical Eng & Biosci. 2017; 2(2): 555583. DOI: 10.19080/CTBEB.2017.02.555583
Abstract
Aims: This study was designed to explore the construction process and the characteristics of micro vascular network in biomaterials by implanting porous silk fibroin film (PSFF) into muscle tissue of rat and follow-up observing the morphology of angiogenesis and micro vascular network formation.
Methods: After the PSFFs are implanted into the leg muscle tissue of SD rats, the micro vascular network development was histologically monitored and the micro vessel densities (MVD) changed with time were investigated in order to understand the forming process of micro vessels at various levels and vascular network.
Results: In the early stages of implant PSFFs it was observed that a large number of inflammatory cells migrate into the interior of the materials. Then during the period of the rapid growth of capillaries the capillaries grew progressively inters from the outer edge to the interior of PSFF, and there were the phenomenon of vascular growth in the pairs in PSFFs. The micro vessel density reached the maximum about 10 days after implantation. About three weeks later the micro vascular distribution approached the steady state and arterioles had been observed, which prompt that the micro vascular network had been basically formed.
Conclusion: Based on the experimental results and analysis, three mechanisms for micro vascular network construction in PSFF were proposed:
i. Endothelial cells migrate toward the source of VEGF and its movement is constrained by the temporary matrix.
ii. The metabolic demand of the tissue cells regulates MVD.
iii. To automatically regulate the coordinate growth of various levels of microvasculars. Based on these mechanisms the new capillaries will develop into a healthy micro vascular network.
Keywords: Angiogenesis; Micro vascular network; Temporary matrix; EC migration; Porous silk fibroin film
Abbreviations: PSFF: Porous Silk Fibroin Film; SPF: Specific Pathogen Free; SD: Sprague-Dawley; MVD: Micro Vessel Density; EC: Endothelial Cell
Introduction
Within 10 days after implantation of Porous silk fibroin film (PSFF) into hypodermal tissue of rat, it was observed that the micro vascular network system had been formed in materials [1] and the arterioles were found in materials, which shows that in the process of capillary formation, the arterioles development is also underway simultaneously [2,3]. There are a lot of reports about the angiogenesis so far, in which the main focus is on the angiogenesis mechanism [1], the proliferation and growth behavior of endothelial cells and the stimulation mechanism [2] and the angiogenesis mode [3] etc. However, the relevant reports about the construction of micro vascular networks in implant materials were seldom seen. We do not know what mechanism makes the newborn capillaries develop into sound micro vascular networks within a short period of time, which have a suitable micro vessel density and a coordinated structure of vascular network to meet the metabolic demands of tissues. In the process of damaged tissue repair, this is an important issue related to whether the survival of the tissues in implant material. This study focuses on this issue, through the experiments implanting PSFFs into muscle tissue of rat, the process of micro vascular network building was observed and analyzed, the construction mode and its mechanism were discussed. The results are considered to be meaningful for tissue engineering and regenerative medicine.
Materials and Methods
Preparation of PSFFs
With the same manner to the previous report [4] the PSFFs were prepared. The size of films were 2cm×2cm, where the pore size 50-100|im, porosity 70% -90% and thickness of about 0.5mm (Figure 1) show the macrostructure of PSFFs by SEM.

Animals and material collection
A total of 16 SPF (Specific Pathogen Free) SD (Sprague- Dawley) healthy male rats weighing 250±50g, were used for study. Before surgery rats were anesthetized with sodium pentobarbital. The surgical areas in hind legs was shaved after disinfection, cut full-thickness skin along the long axis of thigh about 2cm. The thigh muscle was dissected bluntly and implanted the SPFFs with size 1cm × 2cm, then stitched and bandaged to resume feeding the rat. After surgery at 3d, 5d, 7d, 10d, 13d, 16d, 19 and 22d, making two rats death to produce the samples for TEM and SEM and (Paraffin slide with H E) and taking consecutive slices for immunohistochemical staining and observation of optical microscope. The experiments were conducted in accordance with guidelines for the Care and Use of Laboratory Animals and the Management Committee of Experimental Animal Center (Soochow University, Medical Faculty, Suzhou, China).
Common pathologic histology section-staining
The specimens were washed with PBS buffer (0.1m PBS, pH 7.2), fixed by osmic acid, dehydrated conventionally, embedded, sliced into ultrathin section of about 50~70nm thickness, and then stained with uranylacetate and lead nitrate. The locations of angiogenesis appearance in the specimens were searched and observed with Hitachi H-600 TEM.
CD34, VEGF immunohistochemistry and data processing
With immunohistochemical SABC staining method and CD34 marker, ECs and the expression level of VEGF in implanted materials were observed. The micro vessels were counted by referencing to the Weidner's method [5], and the micro vessel density (MVD) was calculated in accordance with the number of blood vessels in the vision of 0.475 mm2. The expression levels of VEGF were judged with the improved Wang's scoring criteria [6]. As statistical values, the micro vessel density (MVD) and the expression quantity of VEGF were checked by t-test with SPSS 11.5 statistical software.
Results
Distribution of VEGF in implanted PSFFs
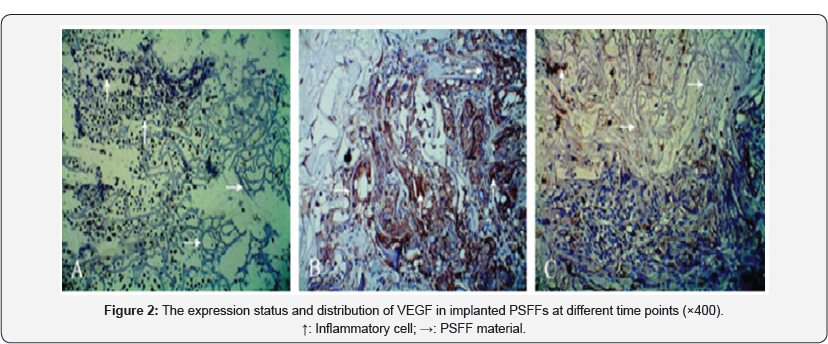
Based on immunohistochemical SABC staining method (Figure 2), shows the expression status and distribution of VEGF in implanted PSFFs at different time points. At 3 days after the surgery ( Figure 2A), there are a lot of inflammatory cells in the junction areas (on the left of figure) between of materials and tissues. It can be seen that VEGF (brown) is abundantly expressed in dense regions of inflammatory cells, and the amount of VEGF expression becomes smaller along with access to the interior materials, which means that the angiogenesis will start from the junction areas between of materials and tissues, then gradually enter into 3d 7d 19d the material. At 7 days after the surgery ( Figure 2B), VEGF expression was the most active inside the materials, and not only by inflammatory cells, but also by the endothelial cell (EC)s entered into materials, which was showed by EC marker (CD34) . At this stage the capillaries quickly generated. It is noted that there is little VEGF expression and EC enter in the zone not communicating in the right of the image. At 19 days after the surgery ( Figure 2C), VEGF expression could barely see inside the materials, representing the capillaries have tended to stop generating. From these figures it can be seen that VEGF expression showed a complex distribution patterns in implanted PSFFs. In such a complex environment, however, a sound capillary network is how to be formed?
Angiogenesis extension layer by layer
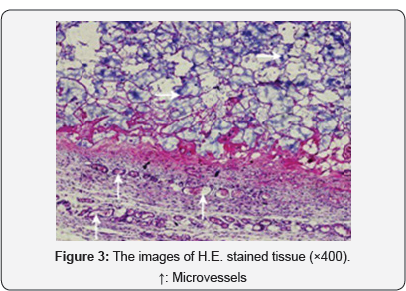
Figure 3 shows an immunohistochemistry image of angiogenesis in the implanted material edge, in which there are the materials in upper part and the tissues in lower part. It is shown that the capillaries grow from the material contact areas and then gradually enter into the interior of the materials layer by layer. There are two growing capillary layer in lower part of the image. In the capillary layer near the tissues, a large number of micro-vessels with larger diameter can be observed, which means a mature vascular layer has been formed. In the capillary layer near the materials, the number of micro-vessels are smaller, and the diameter slightly smaller than the former. But it is noted that the micro-vessels are filled by RBCs, and some larger venules have been formed, which suggests that the layer of blood vessels play an active role in the hypoxic environment and it will be needed to continue the development of micro vascular networks. This result shows a basic characteristic of angiogenesis in implanted materials, that is the angiogenesis extended from existing blood vessels to the interior of materials layer by layer, which directly affect the formation of micro vascular networks.
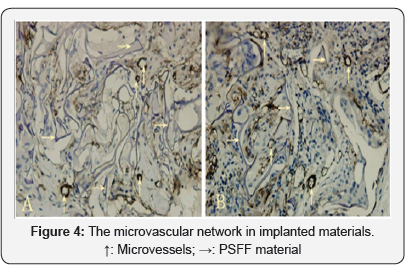
Angiogenesis must be accompanied by the growth of arterioles. Figure 4 is the immunohistochemical (CD34) images of the micro vascular network in implanted materials and illustrates the results of angiogenesis within the materials on days 19 and days 21. In two images the patterns of micro vascular distribution are 19d (×400), 22d (×400). Similar, in which the micro-vessels are evenly distributed and some of arterioles and venules are observed, showing a more complete vascular network has been formed during this period. The issues on the arteriolar formation in materials were reported by Bai [7]. In this article we mainly focus on the process of micro vascular network formation accompanied by the growth of arterioles which will be discussed later.
MVD in implanted materials
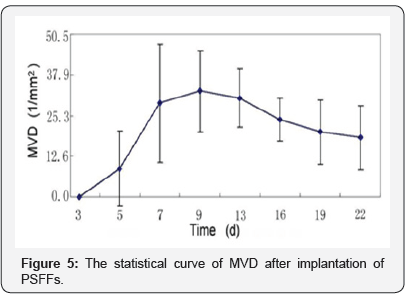
Based on expression of the EC marker CD34, the MVDs of various images were calculated. Figure 5 is a statistical curve of MVD at different time-points after implantation of PSFFs into leg muscle of rats. It is shown that new blood vessels beginning to appear in the materials was from third day after surgery, and the MVD reached the maximum about 10 day after surgery, then decreased slightly, and tended to a stable state finally. It is an interesting phenomenon that the MVD appears a tendency downward after increase, which will be discussion below.
The pairs of neo vascular
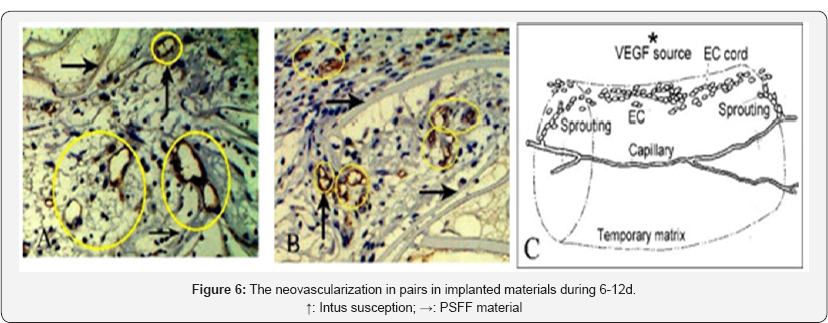
From the immunohistochemistry image of angiogenesis with marker CD34, it is noted that there were many neo vascularization in pairs in implanted materials during 6-12d. Figure 6 shows two enlarged images in this period when the new capillaries rapidly grow (Figure 6A & 6B). These pairs of capillaries generally are in one larger and another smaller, some have been separated, and some are still together. In contrast, there are no such pairs of capillaries in Figure 4 . From many of these images it is speculated that in the early stage after surgery, when a large number of inflammatory cells and other cells entered into the pores of the material and abundantly expressed VEGF, the ECs of capillaries in the connected areas between tissues and material bound VEGFs and occurred the strong response [1], leading to rapid proliferation of ECs and then newborn ECs left the mother vessels and migrated toward the source of VEGF. These newborn ECs clustered on the edge of the temporary matrix and formed the EC cords (Figure 6C). Under the conditions of strong hypoxic inside materials in the early stages, there are a lot of newborn ECs on the edge of temporary matrix around original capillaries, which may come together to form one or two endothelial cell cords. The end portion of EC cord will is connected to the parent capillaries by sprouting angiogenesis. We feel that it may be an effective mechanism to generate pairs of capillaries in order to respond the strong hypoxia, which make adequate new capillaries form during a short time. The new capillary cord which first connected with the parent capillary will be thicker than the other. On the other hand, in the areas of lower VEGF concentration, the capillaries will occur in tussusception to branch and expand to two new capillaries T arrows in Figure 6A & 6B, which is slower than the above way for the angiogenesis.
Discussion
The above results show, at about 20 days after the surgery of implanting PSFFs in to muscle tissue of rat, the tissue cells have entered the pores of material inside, and a functional micro vascular network has been constructed. In this section, the issues related on the micro vascular network construction model in materials and its mechanism will be discussed.
The role of temporary matrix
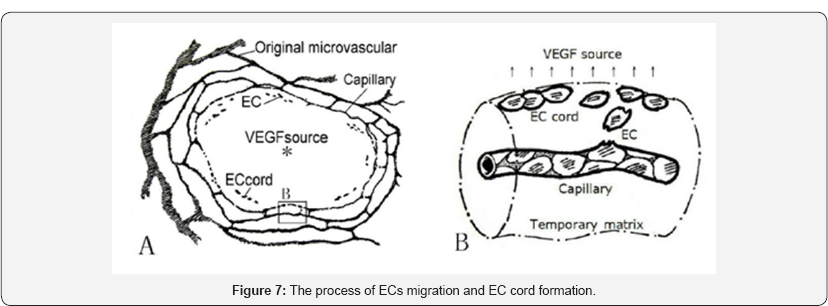
After implantation of PSFFs into muscle tissues, along with inflammatory cells and other tissue cells enter in to the pores of material inside, a large number of VEGF expression led to the strong response of capillaries in material edge and induced angiogenesis within the material. From the experimental results it was seen that the angiogenesis within the materials are the beginning from the edge and advancing toward the interior of the material. However only by such a guide for ECs it is still impossible to form a healthy micro vascular network, because leaving the mother vessels the ECs migrate toward hypoxia direction and will form a random distribution inside materials, rather than the orderly EC cords which meet the metabolic needs of tissue cells. There is also need to have some other factors that influence the migration of cells. We note that the role of the temporary matrix around mother capillary. It already was known that VEGF induce capillary permeability to increase, and extravasation of plasma proteins to form a temporary matrix, and also induce EC proliferation and increase of blood metalloproteinases and plasminogen activator to degrade the extracellular matrix that impede EC migration [8-10]. Accordingly, the process of EC cord formation is considered as shown in Figure 7A & 7B, where it is shown the ECs migrate toward direction of VEGF source, and stop when reach the edge of the temporary matrix. The ECs which was isolated from different parts of mother vessel connected into an EC cord along the edge of the temporary matrix. On the other hand, in the areas of the lower VEGF concentration, the original capillaries are induced to occur the sprouting angiogenesis that the capillary buds extend toward VEGF source and, in last, are connected together with the EC cord. Although, there are a lot of factors which affect ECs' behavior, and the mother capillaries may have complex vascular morphology, which make the generation of new blood capillaries appear to be random, the angiogenesis process should comply with the following rules according to the above [1]. The EC migration is toward the higher expression areas of VEGF, so the newborn capillaries occur between mother vessel and VEGF source [2]. The ECs leaving the mother capillary cluster in the temporary matrix edge parallel with the mother capillary [3]. The new capillary network gradually advances toward VEGF source in layer by layer. These rules promote orderly growth of capillaries. Figure 5C of literature [11] by Matthias WL has provided an image of micro vascular network formation in synthetic bone materials, which is considered as an experimental result for illustrating this model. Where a micro vascular network in edge developed toward to the implant center of the materials in layer by layer as Figure 7A. Here, the temporary matrix plays a key role for regulating new capillaries to ordered growth. Here, the temporary matrix plays a key role for regulating new capillaries to ordered growth.
Regulation of MVD
In this section we try to analyze the mechanism of the MVD curve of downward trend after reaching the maximum. There was the presumption formula of oxygen concentration in of radius r around capillary in the following [12].
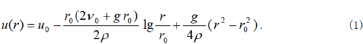
Where, p is diffusion coefficient; u0the oxygen concentration in the capillary; v0 the oxygen diffusing intensity in the capillary wall and g the oxygen consuming intensity. If the critical oxygen concentration of cell survival is ud(rd)=ß0u0, where Pβ0 is called critical oxygen concentration coefficient and rd critical radius of cell survival, we can derive φ, the maximum density of the capillaries, that is MVD as the following. Here the together effect of the oxygen supply from the adjacent capillaries had been considered.
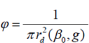
Which is related with g, the oxygen consuming intensity and β0 (related with type of cells) and increases with the increase in metabolic rate. For the specific cells, therefore, if the MVD changes in cell tissues, the reason should be the changing of the cell metabolic rate.
According to experimental results it is known that early in implantation of PSFFs, a large number of cells, including inflammatory cells enter into the interior of the material, and the areas within the material quickly fall into severe hypoxia state. These hypoxic cells abundantly express VEGFs which quickly spread and induce angiogenesis. After 10 days of implant surgery the newborn capillaries already meet metabolic demands of cells in materials, and VEGFs are no longer expressed in various cells. The inflammatory cells which enter into material in early stage and abundantly express VEGFs play an active role for the rapid formation of new blood vessels. However after the formation of new tissue, inflammatory cells gradually subside, and tissue oxygen metabolic demand reduce. Then some surplus of capillaries induced by inflammatory cells appear in the tissues, which will gradually degrade due to their prolonged off time. As the results MVD in materials will decrease. Thus it is understood that MVD decline on the late angiogenesis process in PSFFs is due to the reduction of metabolic rate in tissues, which result excess and degradation of newborn capillaries. The parts that are first stimulated by the increase of blood flow are the sphincter capillaries and arterioles.
Micro vascular formation in different levels
It has been reported that micro-arteries are formed through the arterialization of capillaries, which relies mainly on weeks cells or mesenchymal cells which are adhered to the surface of capillary wall, to transformed to smooth muscle cells [13]. After implantation of material into tissues, once the blood flow through the newborn capillaries in materials, the perfusion of micro vascular network will increase. The pre capillary sphincter and arterioles are first stimulated by the increase of blood flow, which lead transformation of some pre capillaries to arterioles. The transformation will promote upgrade of the existing arterioles, that is its surface smooth muscle will proliferate, blood vessel wall thicken and blood vessel diameter increase. This process is started from the formation of capillary network in the most downstream [7]. Corresponding to this, the venules which collect blood from capillary bed are stimulated also to become large. According to the experimental results it was seen that with the gradual deepening of the new capillaries into material inside, the arterioles gradually develop to the inner layer of materials from the joint areas between materials and tissues. Therefore, the micro vascular network forming in the implantation material is an automatic adjustment process, in which the key mechanism is the effect of EC proliferation and transformation of pericytes to smooth muscle cells under conditions that the ECs of capillary continue to be in filling state by blood perfusion. As the results of this adjustment process, the micro vessels at all levels are generated and the micro vascular network is established in a coordinate constitute.
Conclusion
In summary, we showed in the present study for construction of a sound micro vascular network in the implantation material, the following three mechanisms are necessary, that is
i. The newborn ECs migrate toward the orientation of VEGF source and EC migration is restricted by the scope of temporary matrix, which makes new capillaries orderly generate.
ii. The metabolic demands of cell tissues determine the MVD, which makes the number of capillaries just to meet the metabolic demands.
iii. The micro vessels growth are adjusted to meet the perfusion needs at all levels, which makes neo vascular network get adequate blood supply. Based on these mechanisms, the newborn capillaries in the inner materials will be able to develop into a healthy micro vascular network.
Acknowledgment
We are grateful to Yongzhen Chen and Quan Bai for their excellent assistance.
References
- Bai L, Wu D, Xu J, Liu H, Xie M, et al. (2011) On Modes of angiogenesis and the Mechanism in Porous Silk Fibroin Films. J Mater Sci: Mater Med 22(4): 927-933.
- Bai L, Baoqi Z, Guoping G, Lixing D, Yongzhen C, et al. (2008) On the Growth Morphous of the Capillaries and Tissue in the Porous Silk Fibroin Films, Front. Mater Sci 2(3): 266-270.
- Bai L, Xu J, Sun Q, Di C, Wu Z (2007) Research on the Growth Density of the Capillaries and the Configuration of the Porous Silk Fibroin Film. Key Engineering Materials Vol. 342-343, pp. 229-232.
- Lun B, K Z, Qi H, Jianmei X (2014) Endothelial tubes form from intracellular vacuoles in implanted biomaterial in vivo of rat. J Mater Sci: Mater Med 25(5): 1275-1282.
- Weidner N, Folkman J, Pozza F, Bevilacqua P, Allred E, et al. (1992) Tumor angiogenesis: a new significant and independent prognostic indicator in early-stage breast carcinoma. J Natl Cancer Inst 84(24): 1875-1887.
- Wang C, Zhang X, Yang F, et al. (1995) 23nm gene product/NDPK expression and its clinical significance in human colorectal carcinoma. Chinese J Pathology 24(6): 356-358.
- Lun B, Guangqian W, Xiaoyan T, Jianmei X (2012) Exploring the mechanism of micro arteriogenesis in porous silk fibroin film. International Journal of Biomaterials 2012(2012): 6.
- Dvorak HF (1986) Tumors: Wounds that do not heal. Similarities between tumor stroma generation and wound healing. N Engl J Med 315(26): 1650-1659.
- Elias PM, Arbiser J, Brown BE, Rossiter H, Man MQ, et al. (2008) Epidermal vascular endothelial growth factor production is required for permeability barrier homeostasis, dermal angiogenesis, and the development of epidermal hyperplasia: implications for the pathogenesis of psoriasis. Am J Pathol 173(3): 689 -699.
- Bergers G, Brekken R, McMahon G, Vu TH, Itoh T, et al. (2000) Matrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesis. Nature Cell Biol 2(10): 737-744.
- Matthias W Laschke, Kristina Witt, Tim Pohlemann, Michael D Menger (2007) Injectable Nano crystalline Hydroxyapatite Paste for Bone Substitution: In-Vivo Analysis of Biocompatibility and Vascularization. J Biomed Mater Res B Appl Biomater 82(2): 494-505.
- Bai Lun, Xu Jianmei, Sun Qilong, Di Chuanxia, Song Jiangchao, et al. (2007) On The Growth Model of the Capillaries in the Porous Silk Fibroin Films. Materials Science: Materials in Medicine 18(10): 19171921.
- Fay hansen-Smith, Stuart Egginton, Ai-ling Zhou, Hudlicka O (2001) Growth of Arteioles Precedes That of Capillaries in Stretch-Induced Angiogenesis in Skeletal Muscle. Microvascular Research 62(1): 1-14.






























