Stem Cells: Cultivation and Routes of Administration
Anusha Sudulaguntla1*, Basavarajk Nanjwade2 and Vineeth Chandy3
1Department of Pharmacy Practice, T John College, India
2Department of Pharmaceutics, Rani Chennamma College of Pharmacy, India
3Department of Pharmaceutical Chemistry, T John College, India
Submission: February 08, 2017; Published: March 08, 2017
*Corresponding author: Anusha Sudulaguntla, Assistant Professor, Department of Pharmac Practice, T John College of Pharmacy, No. 88/1, Bannerghatta Main Road, Gottigere, Bengaluru-560083, Karnataka, India, Tel: Email: anushasudulaguntla@gmail.com
How to cite this article: Anusha S, Basavarajk N, Vineeth C. Stem Cells: Cultivation and Routes of Administration. Curr Trends Biomedical Eng & Biosci. 2017; 2(1): 555579. DOI: 10.19080/CTBEB.2017.02.555579
Abstract
Objective: To personalize the treatment with stem cells, which is based on individual's symptoms and genetic condition, looks more promising, accurate and possible with the present available technology. Different routes of administration of stem cells are still being studied to improve patient's treatment and outcome as a part of individualized treatment.
Background: Stem cells have caught interest of many for their abundant use in regenerative medicine. It has always questioned one's self regarding personalized medicine and ways to improve health care system around the globe. Researchers are still trying to improve delivery system of the cure.
Conclusion: Different routes of administration of stem cells will improve in delivery care system and using of modern medicine i.e. nanotechnology along with regenerative medicine can provide even more accurate treatment along with personalized treatment.
Keywords: Stem cells; Cultivation of stem cells; Routes of administration; Nanotechnology of stem cells; Future of stem cells
Introduction
Stem cells are the future of personalized medicine. They hold the key for regeneration and reformation. Along with regenerative properties being studied at present, there are multiple factors which will affect the process of regeneration and cultivation of stem cells. Acceptance in the society and community using stem cells for their treatment has improved since last decade. Using of cells from own body to treat their own illness has created a wide range of opportunity in medicine to provide better patient care. There are many other factors we did not yet discover and apply in modern medicine regarding our own cells. Researchers today are still exploring the possibilities of using our own bodily substances to find cure and treat many diseases. This type of treatment improves patient to a larger extent. Many ADR'S and unwanted effects can be avoided with personal medicine. Using of stem cells can improve awareness around the world regarding the better out comes in present day's treatment. Legal requirements and community acceptance is needed to apply this technology for better use and useful outcomes.
Cultivation of Stem Cells
Stem cells are cultivated in different ways according to their anatomical origin and site of collection. Stem cells and other tissues which are sensitive to outer environment and can be cultivated in appropriate requirements are usually preserved by cryopreservation, this helps in monitoring the temperature in which the cells can survive for further experiments. Cryopreservation is one of the most used methods to cultivate and store cells and tissues which are either collected or stored for further use. There are chemicals which are used for the cultivation process to obtain the best results and obtain the best out of the preserved cells, substances like anti oxidants, antibiotic, different type of markers, and different compositions of mediums. Here are lists of few cultivation techniques which will help in better selection of cultivation techniques.
Use of growth factors promote to differentiation, expansion or survival of specific cell types is key in controlling the differentiation towards specific cell lineages, A variety of bioreactors used for cell cultivation and collection are used now a days and can be readily applied for stem cell cultivation and differentiation. They provide a well controlled environment suitable for studying the process of stem cell propagation and differentiation. Their use will facilitate the development of processes for stem cell application [1]. The addition of the antioxidant N-acetyl cytokine (NAC) to the differentiation medium helps in partially preventing the nuclear accumulation of APE1, increasing the neuronal differentiation of hat-MASC and helps in cultivating cells in a healthier way and better way to obtain even more accurate values [2].
In recent research there is a newer medium called serum- free defined medium (SPM) which supports the cultivation of human pluripotent stem cells (hPSCs) on recombinant human vitronectin-N coated dishes after seeding with either cell clumps or single cells. This culture system is said to support and maintain controlled cell. It is also seen that non patterned cultures produced variant, unlimited cell proliferation with non-uniform cell growth and uneven densities in which it was observed down regulated expression of some self-renewal-related markers. Patterned cultures of hPSCs allowed ordered visual inspection of every hPSCs colony, giving a number and address in patterned culture dishes. Several spots could be sampled for quality control tests of production batches, thereby permitting the monitoring of hPSCs in a single culture dish [3]. The human embryonic cells were found to be easily obtained and expanded in a serum free medium. It is said that establishing embryonic stem cells is seen to be more effective in serum free medium compared to that of the one comparing serum [4]. Cells from the forebrain of E14-15 Blab/c fetuses are grown in DMEM/F12 N2 medium supplemented with EGF and FGF-2 to form multi cellular neuro spheres. After 3 to 8 passages, neurospheres were placed on surfaces coated with poly-L-lysine, polyornithine and or laminin in dishes containing the same medium where cytokines were said to be replaced with serum. It was seen that under these conditions, neurosphere cells have been spread over the surface forming a cellular layer consisting of beta-III tubulin+neuronal, GFAP+astroglial and O4+oligodendroglial cells. When these cells were cultivated for prolonged periods, they showed formation of islands of epitheloid cells. Following 2 to 3 weeks in vitro, ependymal cells with beating cilia appeared among these cells [5]. It also strongly depends on the supply of an appropriate support with tropic factors during organogenesis. The lack of important factors, such as glial cell line-derived neurotrophic factor, leads to severe disturbances in the ENS and, thus, to movements disorders in younger kids. The isolation of neuronal precursor cells as well as their transplantation after expansion in vitro is therefore a hopeful new approach concerning all forms of dysganglionosis in children [6]. Ciliated ependymal cells observed in small clusters or as single cells scattered in certain areas. Confocal microscopy confirmed the presence of alpha-tubulin-immunoreactive cilia arranged in tufts which were located on the apical surface of epitheloid cells. Research data indicate that ependymal cells are spontaneously derived from NSCs [5].
The standard culture method for neural stem cells cannot prevent the attachment of activity, accelerated mineralization, and the replication of bone sialoprotein and osteocalcin genes was noneuro spheres, which eventually result in differentiation. Neural stem cells were said to be cultured in flasks coated with 1. 5% agarose gel. As a control, cells were cultured in plastic flasks. The 5-bromine-deoxyuridine incorporation assay was used to determine the S-phase labelling index of both groups. The methyl thiazolyl tetrazolium (MTT) colorimetric assay was said to be used to determine the total cell vitality. After a 3 month culture, the spontaneous differentiation of stem cells were said to be studied using immunocyto chemistry for neuroepithelial stem cell protein. It was found that neural stem cells grew rapidly in the anti attachment flasks. There was no statistically significant difference noticed between the two groups i. When cultured for 3 month in vitro, many more cells differentiated in the control than in the anti attachment group (32.05 vs. 0.64%, P <0.01). Moreover, the neural stem cells in the antiattachment group were reported that they remained multipotent. Therefore, it was said flasks coated with agarose gel are suitable for longterm neural stem cell culture [7].
A novel three-dimensional pellet cultivation system for periodontal ligament stem cells (PDLSCs) to recreate the biologically favorable microenvironment similar to that of a regenerative milieu was used. In this work mono dispersed human PDLSCs were cultured in a medium with ascorbic acid and conditioned medium i.e. with a-minimum essential medium (a-MEM; Gibco BRL, Gaithersburg, MD) supplemented with 10% fetal bovine serum (FBS), used along with medium from developing apical tooth germ cells and were harvested from culture plate as a contiguous cell sheet with abundant extracellular matrix present. The separated cell matrix membrane was seen to immediately contract to produce a single cell pellet. The PDLSCs embedded within this cell matrix complex was noted to exhibit several with the hypothesis that liver and pancreas arise from the common endo dermal progenitor cells. These results provide evidence that the hepatic stem/progenitor cells may adopt different cell lineages fate depending on aphenotypic characteristics of cement blast lineages, as up regulated alkaline phosphates ted. When this PDLSC pellets were transplanted into immune compromised mice, a regular aligned cemented i.e. PDL-like complex was formed. The results suggested that the combination of apical tooth germ cell conditioned medium and endogenous extracellular matrix could maximally mimic the microenvironment of root periodontal tissue development and enhance the rebuilding of physiological architecture of a cemented PDL-like complex in a tissue-mimicking way; on the other hand, such PDLSC pellet may also be a promising alternative to promote periodontal defect repair in the near future clinical applications [8].
To exclude the hematopoietic cells from ED 12-14 fetal mouse liver, FACS-sorted CD45 TER119, c-kit- fetal liver cells were collected. The collected cell subpopulation was said to contain hepatic stem/ progenitor cells. They were cultured in gelatin coated six well plates with the special culture medium. To assess these cells developmental changes resulting from specific modified culture conditions, insulin expression test was done using dithizone staining and immunocytochemical staining.Dithizone staining demonstrated the insulin-producing cells appeared at d 15 and exist at d 30 and 40 after the initiation of culture. To confirm in vitro insulin production by the cultured fetal liver stem cells, insulin examination was done by immune reactivity at d 15, 30 and 40 after isolation in culture. Some of the cells were said to show positive for insulin. Results were said to be consistent with the previous report. To demonstrate the function of the differentiated cells, the cells were said to be further transplanted under the left renal capsule of the alloxan induced diabetic mice. The non fasting blood glucose level was seen to be reduced till 14 d after post transplantation. At 21st day post transplant, the non fasting blood glucose was said to be slightly increased, but remained lower than the level before transplantation. This is consistent activation of the key developmental regulators. The insulin-producing cells could be used as a potential cell source for successful cell replacement therapy for diabetes [9].
One ofthe problems to be solved in attaining the full potentials of hematopoietic stem cell (HSC) applications is the availability is very limited of the cells. Growing HSCs in a bioreactor offers an alternative solution to this problem. In spite of this, the optimization of HSC cultivation in a bioreactor has been barely explored. The model was further modified to optimize the cultivation by proposing three alternative feeding strategies in order to prevent the occurrence of nutrient limitation in the bioreactor. The analysis of these strategies, periodic step change increase in the inlet oxygen concentration, the periodic step change increase in the media inflow, and the feedback control of media inflow, shows that these strategies can be successful in improving the cell yield of the bioreactor. The developed model was seen to be useful for the design and optimization of bioreactor operation [10]. Fibroblast growth factor18 (FGF18) belongs to the FGF family and it is an pleiotropic protein that stimulates proliferation in several tissues. FGF18 was investigated by its effects on rat BMSCs (rBMSCs). The proliferative effects of FGF18 on rBMSCs were examined using different assays to further validate the osteogenic differentiation effects of FGF18, FGF18 significantly enhanced rBMSCs proliferation (p<0.001) and induction of the osteogenic differentiation was done by elevating ALP and mineralization activity of rBMSCs (p<0.001). Furthermore, these osteogenic differentiation effects of FGF18 were said to be confirmed by increasing the mRNA levels of collagen type I (Col I), bone morphogenetic protein 4 (BMP4), and Runt-related transcription factor 2 (Runx2) at 3 and 7 days. These results show that FGF18 could be used in order to improve the bone repair and regeneration [11].
Embryonic stem (ES) cells are pluripotent cells that have the capacity to differentiate into all the somatic cell derivatives or themselves. Cells derived from ES cells cultured in the three dimensional (3D) culture system with exogenous growth factors and hormones can be differentiated into hepatic-like cells with morphologic characteristics of typical mature hepatocytes was reported. All the testes including reverse transcription polymerase chain reaction testing, Western blot testing, immunostaining, and flow cytometric analysis were reported to show these cells express liver specific genes. Differentiated cells further are said to exhibit morphologic traits and biomarkers characteristic of liver cells. When these stem cell bearing scaffolds are transplanted into severe combined immunodeficient mice, the 3D constructs are reported to show they remain viable, undergoing further differentiation and maturation of hepatic like cells in vivo. It is also reported that in all of the sim[mu] g experiments oxygen concentration modulates cell signaling and adhesion, in line with the knowledge that sim[mu]g affects cell shape and cyto-skeletal organization [12]. The growth and differentiation of ES cells in a biodegradable polymer scaffold and a rotating microgravity bioreactor is said to yield functional and organizational hepatocytes useful for research involving bio-artificial liver and engineered liver tissue [13].
Niño mechanics of Human Adipose-Derived Stem Cells: Small GTPases Impact Chondrogenic Differentiation Most recently, few attempts were successful in using easily available adipose- derived stem cells for enhancing chondrogenic differentiation, characterized by collagen type II (col II) production. At present, the main troubling issue is maturation into hypertrophic cells, which is characterized by over-expression of cells. The growth factor BMP-2 enhances the production of chondrogenic markers of ASCs; however, increased hypertrophy has been noted [14].
Different Routes of Administration of Stem Cells
Route of administration is an important factor to keep in mind for any kind of medicine or dosage form, stem cells are used mainly as personalized therapy these days, which makes it an important reason for improving our research strategies on therapeutic efficacy of stem cells and route of administration with its therapeutic outcome. Stem cell route of administration cannot work as pharmaceutical medicine on general population basis, stem cell therapy is called personalized medicine for a reason, as it can be used only on individual basis i.e. the owner of stem cells from whom it is collected. The genetic material, efficacy, the rate of differentiation and duration activity are important factors which differ from individual to other, here is the list on different routes of administration of stem cells. It was proven that mesenchymal stem cell have anti-inflammatory properties. Effect of administering MSC directly into the airspaces of the lung 4h after the intrapulmonary administration of Escherichia coli endotoxin (5 mg/kg) was tested. It was seen that MSC increased survival compared with PBS-treated control mice at 48 hrs. It was noted that there was a significant decrease in excess lung water. The beneficial effect of MSC was independent of the cells to engraft in the lung. MSC administration noted to mediate a down regulation of proinflammatory responses to endotoxin while increasing the anti-inflammatory cytokine IL-10. In vitro co culture studies of MSC with alveolar macrophages provided evidence that the anti-inflammatory effect was paracrine and was not cell contact dependent and treatment with intrapulmonary MSC markedly decreases the severity of endotoxin induced acute lung injury and improves survival [15].
Human MSC are located around blood vessels in the sub endoestal region of femurs. It was tested and proved that MSC can restore BM stromal function and also engraft when a higher stromal damage was done. The location of MSC were detected locally where they were administered but not in the contra lateral femur [16]. It is reported that stem cell administration in eight patients with SCI with 2 yrs of follow up via different routes of administration like: directly in to the spinal cord, directly in to the spinal canal, and intravenous route and was reported to be safe and had no tumor formations, and no cases of infections or increased pain, and only few cases with minor ADRs. It was also stated that bone marrow stem cells administered via different routes is safe and improve quality of life [17].
Therapeutic potential of intrathecal administration of human umbilical cord derived MSCs by lumbar puncture (LP), in to the lumbar spinal cord or intravenously into the tail vein in a rat model of stroke, and then investigated whether hUCBMSCs could enter the brain, survive, and improve post-stroke neurological functional recovery. It was seen that animals that received hUCBMSCs had significantly improved motor function than the others and reduced ischemic damage when compared with untreated control animals regardless the route of administration. MSCs showed better neurological recovery, without significant differences between the two treatment groups. The cells administered intrathecally were seen to survive and migrate into the ischemic area more extensively, and differentiate significantly into neurons and astrocytes. The final result stated that intrathecal administration of MSCs by LP may be useful and feasible for MSCs treatment of brain injuries, such as stroke, or neurodegenerative disorders and for further treatment [18]. Transplantation procedures using intra parenchymal injection of stem cells result in tissue injury in addition to associated surgical risks. IV injection of mesenchymal stem cells gives raise to lesions. In traumatic brain injuries (TBI), there is a transient breakdown of the blood-brain barrier and an inflammatory response, which increase migration of cells from blood to parenchyma. A technique using endovascular system was used to administer human mesenchymal stem cells in the ipsilateral internal carotid artery. The results obtained were compared to that of the intravenous administration of stem cells. Selective intra arterial administration of mesenchymal stem cells to the injured CNS is a minimally invasive method for transplantation. The method was said to be significantly more efficient than the intravenous route and causes no side effects. The technique can potentially be used for repeated transplantation to the CNS after TBI and in other diseases [19]. A report also suggested that intracoronary infusion of stem cells in mice is feasible and effective [20]. A report states that canine ASCs have proliferation and differentiation potential similar to that of ASCs from other species. Results also show that autologous SVF or allogeneic ASCs can be safely used in acupoint inj in the treatment of hip dysplasia [21]. A research in which ASCs labelled with PKH26 were reported to have been applied via four methods to the cranially based random pattern skin flaps:
I. Intravenous injection
II. Subcutaneous inj
III. Application with collagen sponge seeding and the last is
IV. Application with fibrin glue seeding
The reports showed that ASCs has led to a significant increase in flap viability in the subcutaneous injection group and the collagen sponge group. Coetaneous blood flow was noted to have been increased in the intravenous injection, subcutaneous injection and collagen sponge groups [22]. The appropriate route of cell administration to the damaged organ is an essential step for a successful of organ repair. Targeted and regional administration of cells should be preferred.
A. In regional heart muscle disease, as in myocardial infarction, selective cell delivery by intracoronary catheterization techniques leads to an effective accumulation and concentration of cells within the infracted zone. This can be realized in humans with bone marrow-derived cells. With intracoronary administration, all cells must pass the infarct and peri infarct tissue during the immediate first passage.
B. The transendocardial and transpericardial route of application has been used in large animal experiments and was also recently tested in patients. The main potential advantage of the surgical procedure is injection under visualization, which allows anatomic identification of the target area and even distribution of the injections. Intraventricular catheter manipulation, however, is known to injure the myocardium, inducing ventricular premature beats and short runs of ventricular tachycardia. In certain cases, this injection if given to more arrhythmogenic zones, it may extend the duration of the procedure and should always be carefully monitored. Transendocardial injection of autologous bone marrow cells has also been performed as part of several pilot and phase I studies.
C. The intravenous route of administration is easiest. The main disadvantage, however, is that approximately only minute percent i.e 3% of normal cardiac output will flow per minute through the left ventricle, and it is also very limited because of transpulmonary first-pass attenuation effect which is seen on the cells. There are some major cell types, such as skeletal my oblasts, which have the disadvantage of emboligenic potency when delivered systemically. Intramyocardial injection during open-heart surgery has been tested in humans. It is said that the therapeutic effect is limited because of severe arrhythmogenic complications. Another approach is implantation of autologous bone marrow cells during open heart surgery and there is a possibility that it shows improvement in myocardial perfusion in 3 out of 5 treated patients [23].
The red colored area represents apical lesion of the left ventricle by myocardial infarction. The balloon catheter is localized in the infarct-related artery and is placed above the border zone of the infarction. Blue and green arrows suggest the possible route of cell infusion and migration into the infarct. The 2 small figures depict the transendocardial and intramyocardial route of administration (Figure 1). RCA indicates right coronary artery; LAD, left anterior descending coronary artery; and CFX, circumflex artery [23].
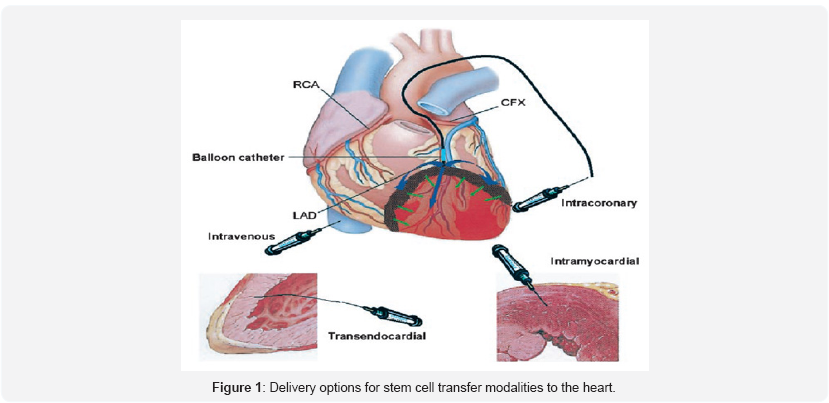
Detection of Transplanted Stem Cells The transplanted cell or cell population is a single unit in a complex biological network of other cells. Hence for both localization and fate mapping of stem cells within the target organ, specific cell markers are most desirable. Analysis of stem cell behavior will presume [1] in situ labeling of a single cell or a transplanted cell population or [2] transplantation of already in vitro labeled cells or cell populations [23].
Regulatory Requirements for Stem Cells
The US Food and Drug Administration defines somatic cell therapy as the administration or giving of autologous, allogeneic, or xenogeneic non-germ cells excluding blood products for transfusion to the patient. Cell therapy is an organ based treatment procedure and is done on individual basis considering the patients or donors condition and type of stem cells given. Cell therapy for diabetes is based on islet transplantation into the portal vein of the liver and results in an improved glucose. Liver diseases (congenital, acute, or chronic) may be treated by hepatocyte transplantation [24].
Nanotech in Stem Cell Therapy
Nanotech is used in different research methods for improving the delivery system or to further improve the efficacy of different methods. Using of stem cells and nanotech together can help in improving future therapeutic efficacy looking at its prospective. There are different programs like cell PROM which were conducted to test the efficacy of the stem cells with nanotechnology. The technology has been developed in the lab of Nick Melos in the Materials Science Department at Stanford. The Melos group is using arrays of nano-reservoirs on a chip to stimulate desired adult stem cell behavior. Both embryonic and adult stem cells have the ability to differentiate and help in treating the diseases such as neurological disorders, heart failure, and diabetes. Adult stem cells, however have less ethical debates from a political standpoint than embryonic stem cells, are less likely to generate immune reactions in patients and, therefore, could be a better option for tissue engineering than embryonic stem cells. The solution they were looking into pursuing is, to build a device that can interact with the stem cell at the micro and nano scale. Exposure to minute amounts of chemical at the appropriate time and place could be the key for guiding stem cells isolated from fat tissue to turn into cartilage or bone constructs. It is believed nanotechnology is the key to unleashing the potential of stem cells. There is a future scope for surgeons to use these cells in further management of failed heart by regenerative medicine [25,26].
Stem Cells and Alzheimer’s, Step Taking Forward
Neurodegenerative diseases are characterized by the neurodegenerative change of neurons, which are important for conduction and communication. With the increase in age, exposure to different chemicals in present world the incidence of neurodegenerative diseases also increases [27]. As known, it is difficult to diagnose for most of neurodegenerative diseases. In the present scenario, there are only few effective therapies. Advances in stem cell biology have raised the hope and possibility for the effective therapy of neurodegenerative diseases [28]. There is much progress and prospects of various stem cells, including embryonic stem cells, mesenchymal stem cell and neural stem cells, for the treatments of neurodegenerative diseases, such as Parkinson's disease, Alzheimer's disease, Huntington's disease and Amyotrophic lateral sclerosis disease [29].
References
- Ulloa-Montoya F, Verfaillie CM, Hu WS (2005) Stem cell research; Cell cultivation bioreactors can be readily adapted for stem cell cultivation and differentiation. Stem cell week Trade journals 100(1): 12-27.
- Rossana Domenis, Natascha Bergamin, Giuseppe Giangranceschi, Carlo Vascotto, Milena Romanello, et al. (2014) The redox function of APE1 is involved in the differentiation process of stem cells toward a neuronal cell fate. PLoS One 9(2): 1-12.
- Takenaka C, Miyajima H, Yoda Y, Imazato H, Yamoamoto T, et al. (2015) Controlled growth and the maintenance of human pluripotent stem cells by cultivation with defined medium on extracellular matrix- coated micropatterned dishes. PLoS one 26: 10(6).
- Peng HM (2006) Embryonic stem cell research; Serum-free medium improves efficacy of embryonic stem cell line cultivation. Trade Journals 21(1): 217-222.
- Mokry J, Karbanova J (2006) Foetal mouse neural stem cells give raise to ependymal cells in vitro. Folia Biol (Praha)52(5): 149-155.
- Rauch Ulrich, Hansgen Andrea, Hagl Comelia, Holland-Cunz, Stefan, et al. (2006) Isolation and cultivation of neuronal precursor cells from the developing human enteric nervous system as a tool for cell therapy in dysganglionosis. Int J Colorectal Dis 21(6): 554-559.
- Zheng XS, Yang XF, Liu WG, Shen G, Pan DS, et al. (2007) A novel method for culturing neural stem cells. In Vitro Cell Dev Biol Anim 43(5/6): 155-158.
- Zhenhua Yang, Fang Jin, Xiaojun Zhang, Dandan Ma, Chun Han, et al. (2009) Tissue engineering of cementrum/periodontal-ligament complex using a novel three-dimendional pellet cultivation system for human periodontal ligament stem cells. Tissue Eng Part C Methods 15(4): 571-581.
- Feng Ren Qing, Du Li Ying, Zhen Quan (2005) In vitro cultivation and differentiation of fetal liver stem cells from mice. Cell research 15(5): 401-405.
- Kresnowati MT, Forde GM, Chen XD (2011) Model-based analysis and optimization of bioreactor for hematopoietic stem cell cultivation. Bioprocess Biosyst Eng 34(1): 81-93.
- Jeon Eunyi, Yun Ye-Rang, Kang Wonmo, Lee Sujin, Koh Young-Hyag, et al. (2012) Investigating the role of FGF-18 in the cultivation and osteogenic differentiation of mesenchymal stem cells. PLOS One 7(8): e43982.
- Wang Yingjie, Zhang Yunping, Zhang Shichang, Peng Guangyong, Liu Tao, et al. (2012) Rotating microgravity-bioreactor cultivation enhances the hepatic differentiation of mouse embryonic stem cells on biodegradable polymer scaffolds. Tissue Engineering 18(21-22): 2376-2385.
- Versari Silvia, Klein-nulend, Jenneke, Van Loon Jack, Bradamante Silvia, et al. (2013) Influence of oxygen in the cultivation of human mesenchymal stem cells in stimulated microgravity: an explorative study. Spinger science & business media 25(1): 59-66.
- Jungmann Pia M, Mehlhom, Alexander T, Schmal Hangen, Schillers, et al. (2012) Nanomechanics of human adipose-derived stem cells: small GTPases impact chondrogenic differentiation. Tissue engineering 18(9-10): 1035-1044.
- Naveen Gupta, Xiao SU, Boris Popov, Jae Woo Lee, Vladimir Serikov, et al. (2007) Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin- induced acue lung injury in mice. J Immunol 179(3): 1855-1863.
- Carrancio Soraya, Blanco Blanco, Romo Carols, Muntion Sandra, Lopez- Holgado Natali, et al. (2011) Bone marrow mesenchymal stem cells for improving hematopoietic function: an in vitro and in vivo model. Part 2: effect on bone marrow microenvironment. PLoS One 6(10): e26241.
- Geffner LF, Santacruz P, Izurieta M, Flor L, Maldonado B, et al. (2008) Adm of inistrationautologous bone marrow stem cells into spinal cord injury patients via multiple routes is safe and improves their quality of life: comprehensive case study. Cell transplantation 17: 1277-1293.
- Jung Yeon Lim, Chang Hyun Jeong, Jin Ae Jun, Seong Muk Kim, Chung Heon Ryu, et al. (2011) Therapeutic effects of human umbilical cord blood-derived mesenchymal stem cells after intrathecal administration by lumbar puncture in a rat model of cerebral ischemia. Stem cell research & therapy 2(5): 38: 1-13.
- Lundberg Johan, Le Black Katarina, Sodeman Mikael, Andersson Tommy, Holmin Staffan, et al. (2009) Endovascular transplantation of stem cells to the injured rat CNS. Neuroradiology 51(10): 661-667.
- Li Qianhong, Guo Yiru, Ou Qinghui, Chen Ning, Wu Wen-jian, et al. (2011) Intracoronary administration of cardiac stem cells in mice: a new, improved techinique for cell therapy in murine models. Basic Res Cardiol 106(5): 849-864.
- Marx Camila, Maiele Dornelles Silveira, Selbach, Isabel Ariel Silveira da Silba, Luisa Maria Gomes de Macedo Braga, et al. (2014) Acupoint injection of autologous stromal vascular fraction and allogeneic adipose-derived stem cells to treat hip dysplasia in dogs. Stem Cells Int 2014: 391274.
- Samuel Golpanian, Ivonne H Schulman, Ray F Ebert, Alan W Heldman, Darcy L Di Fede, et al. (2016) and for the Cardiovascular Cell Therapy Research Network. Concise Review: Review and Perspective of Cell Dosage and Routes of Administration From Preclinical and Clinical Studies of Stem Cell Therapy for Heart Disease, Stem Cells Transl Med 5(2): 186-191.
- Bodo E Strauer, Ran Kornowski (2003) Stem cell therapy in perspective. Circulation 107(7): 929-934.
- Antonio Liras. (2010) Future research and therapeutic applications pf human stem cells: general, regulatory, and bioethical aspects. Journal of translational medicine 18: 131.
- MEDICAL devices & surgical technology week (2005) Stanford university: NAICS: 611310: SCI: 8220. Nanotechnology: Nanotech stem cell device licensed from Stanford University. Trade journals, pp. 369.
- HOU LingLing, HONG Tao (2008) Stem cells and neurodegenerative diseases. Sci china ser c-life sci 51(4): 287-294.
- Waldau B, Shetty AK (2008) Behaviour of neural stem cells in Alzheimer brain. Cell Mol Life sci 65(15): 2372-284.
- Asok Verma, Nipun Verma (2011) Induced pluripotent stem cells and promises of neuroregenerative medicine. Neurobiology India 59(4): 555-557.
- Carlos P Fitzsimons, Emma Van Bodegraven, Marjin Schouten, Roy Lardenoije, Konstantinos Kompotis, et al. (2014) Epigenetic regulation of adult neural stem cells: implications for Alzheimer's disease. Molecular degeneration 9(25): 1-21.






























